Strongly and Loosely Bound Water in Ambient Particulate Matter—Qualitative and Quantitative Determination by Karl Fischer Coulometric Method
Abstract
:1. Introduction
2. Materials and Methods
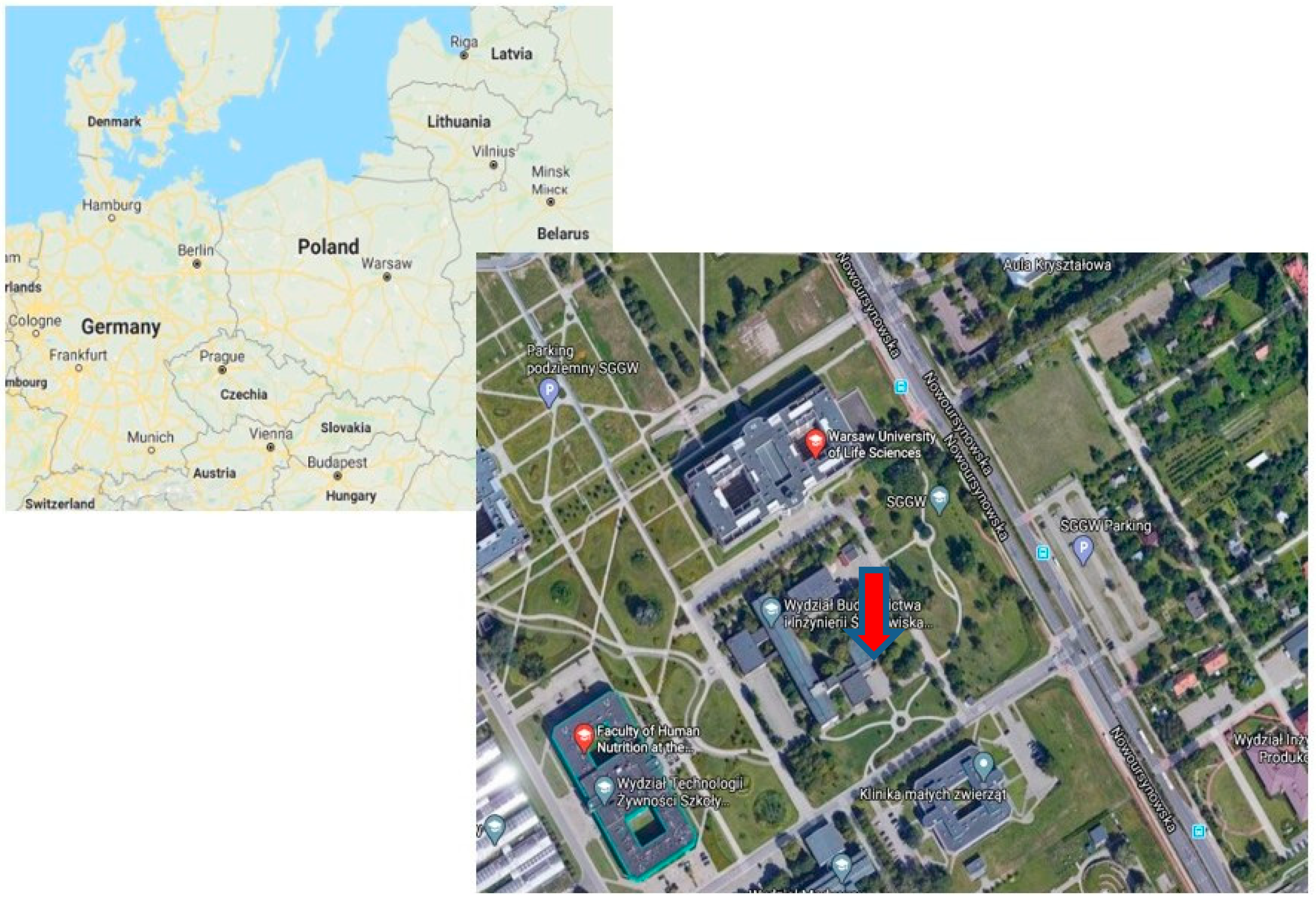
2.1. Sampling Area and PM Samples Collection
2.2. Meteorological Parameters during Measurement Campaign
2.3. Apparatus
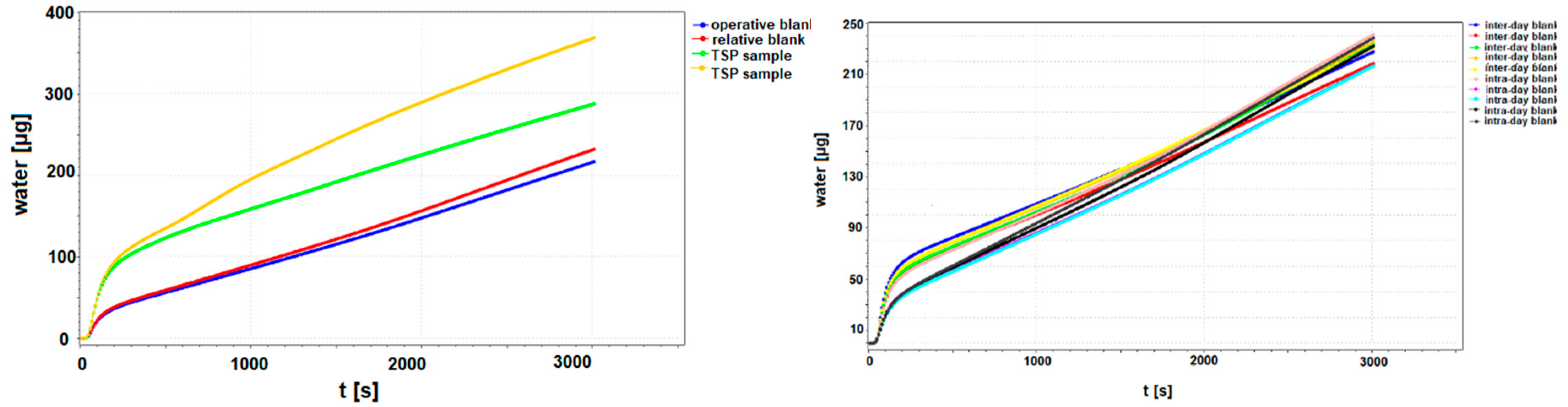
2.4. Method Validation and Quality Control
2.5. Statistical Analysis
3. Results and Discussion
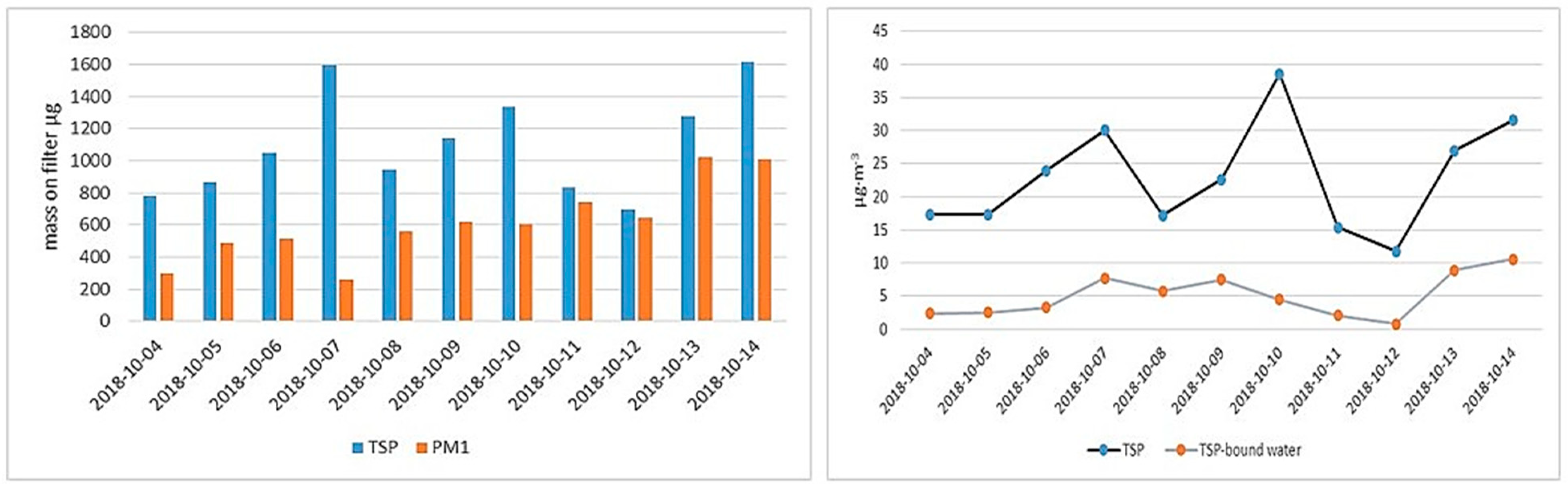
3.1. Relationship between PM (TSP; PM1) Concentrations and Particulate Water Contents, Including Total Water and Different Temperature-Dependent Water Contributions
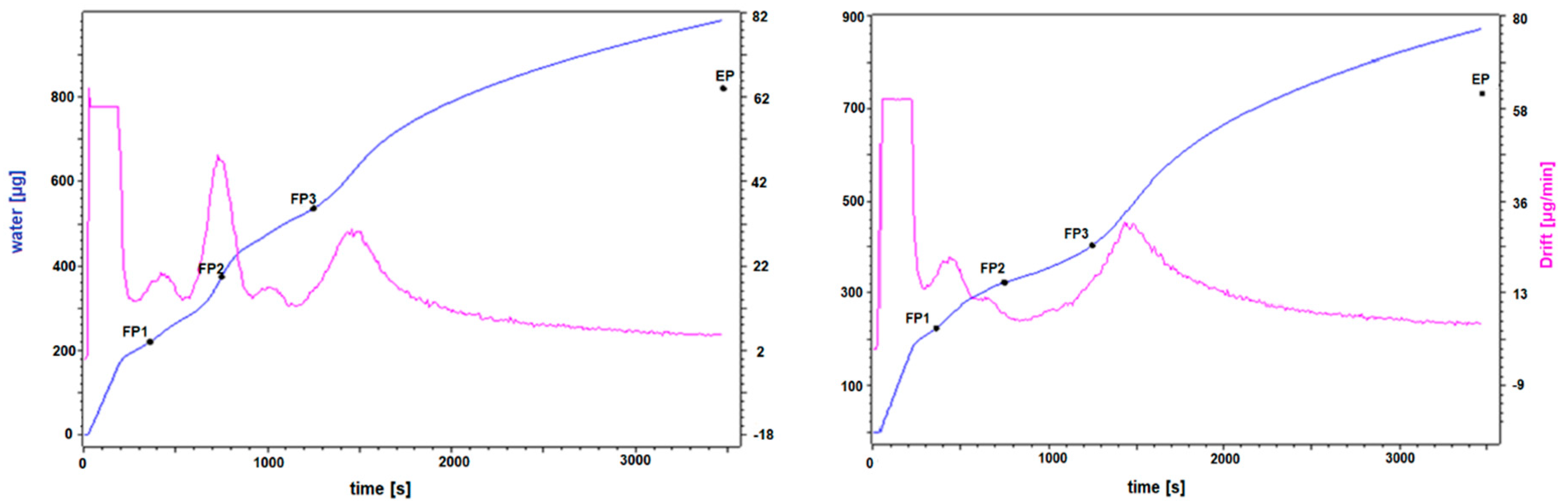
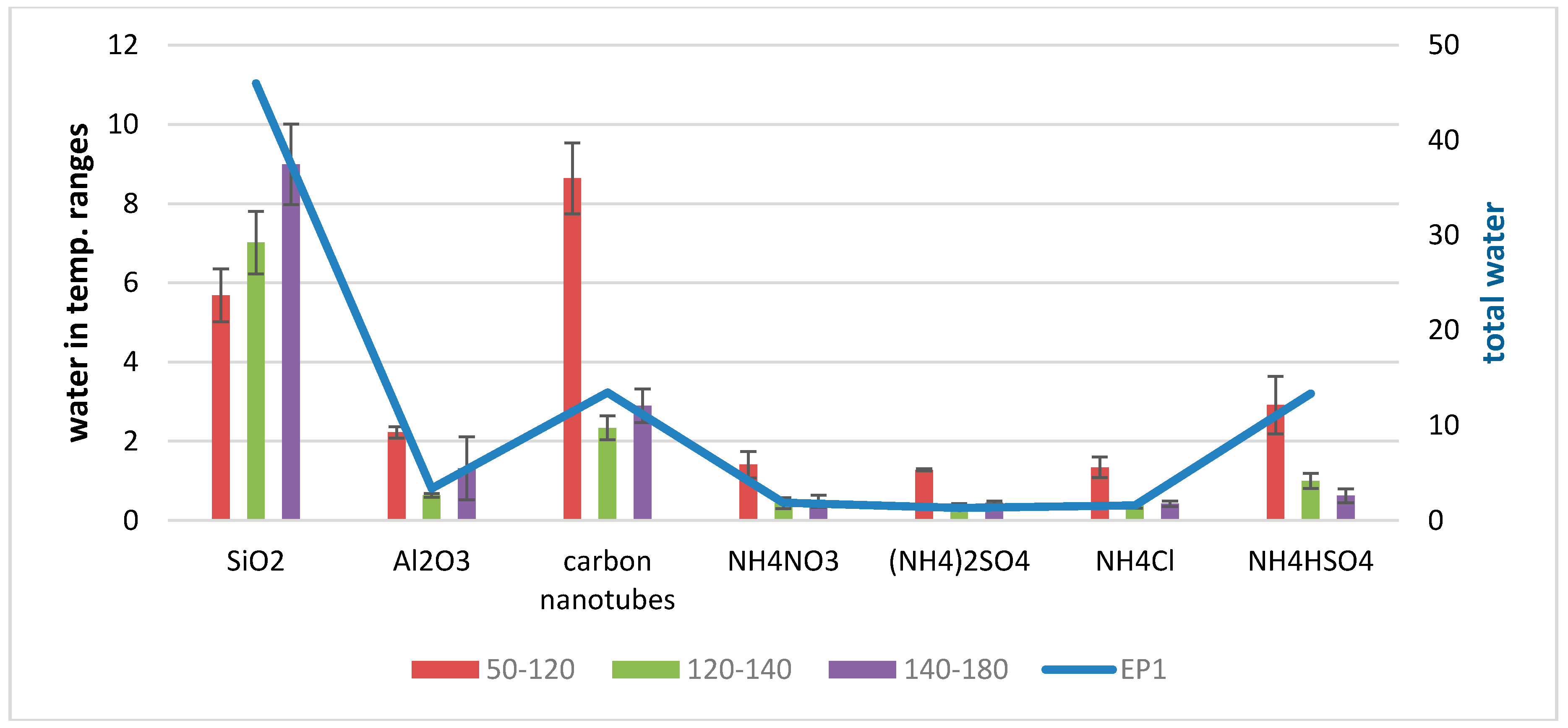
3.2. Qualitative Determination of Water Bound to TSP and to Selected Pure TSP Compounds
3.3. Relationship between 24-h Measurements of TSP, TSP-Bound Water and Meteorological Factors
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Variable | n | Mean | Minimum | Maximum | SD |
|---|---|---|---|---|---|
| Temperature [°C] | 11 | 11.9 | 7.9 | 15.3 | 2.2 |
| Humidity [%] | 11 | 74.1 | 65.4 | 81.5 | 6.0 |
| Pressure [hPa] | 11 | 1012.0 | 1000.4 | 1017.1 | 4.9 |
| Tmin [°C] | 11 | 6.2 | 2.3 | 11.1 | 2.4 |
| Tmax [°C] | 11 | 18.9 | 13.2 | 21.8 | 3.2 |
| Precipitation [mm] | 11 | 0.0 | 0.0 | 0.1 | 0.0 |
| TSP [µg/m3] | 11 | 23.0 | 11.8 | 38.6 | 8.1 |
| PM1 [µg/m3] | 11 | 16.7 | 6.8 | 25.6 | 6.4 |
| EP1 total water [µg] | 11 | 250.8 | 42.5 | 543.1 | 166.9 |
| FP1 water at 50–120 °C [µg] | 11 | 130.6 | 25.4 | 265.3 | 102.6 |
| FP2 water at 120–140 °C [µg] | 11 | 171.2 | 9.1 | 345.3 | 143.6 |
| FP3 water at 140–180 °C [µg] | 11 | 190.3 | bdl | 379.2 | 153.5 |
| TSP mass [µg] | 11 | 1094.3 | 597.0 | 1620.0 | 333.5 |
| PM1 mass [µg] | 11 | 615.3 | 262.0 | 1020.0 | 243.0 |
| TSP/PM1 mass ratio | 11 | 1.6 | 0.5 | 4.4 | 1.0 |
| WSO | SRM Mass µg | Water Content µg | Recovery % |
|---|---|---|---|
| WSO1 | 2520 | 21.5 | 84 |
| WSO2 | 2792 | 25.3 | 90 |
| WSO3 | 3201 | 29.6 | 92 |
References
- Hueglin, C.; Gehrig, R.; Baltensperger, U.; Gysel, M.; Monn, C.; Vonmont, H. Chemical characterization of PM2.5, PM10 and coarse particles at urban, near-city and rural sites in Switzerland. Atmos. Environ. 2005, 39, 637–651. [Google Scholar] [CrossRef]
- Ho, K.F.; Lee, S.C.; Cao, J.J.; Chow, J.C.; Watson, J.G.; Chan, C.C. Seasonal variations and mass closure analysis of particulate matter in Hong Kong. Sci. Total Environ. 2006, 355, 276–287. [Google Scholar] [CrossRef] [PubMed]
- Perrino, C.; Canaperi, S.; Catrambone, M. Comparing the performance of teflon and quartz membrane filters collecting atmospheric PM: Influence of atmospheric water. Aerosol Air Qual. Res. 2013, 13, 137–147. [Google Scholar] [CrossRef] [Green Version]
- Perrino, C.; Catrambone, M.; Torre, S.D.; Rantica, E.; Sargolini, T.; Canaperi, S. Seasonal variations in the chemical composition of particulate matter: A case study in the Po Valley. Part I: Macro-components and mass closure. Environ. Sci. Pollut. Res. 2014, 21, 3999–4009. [Google Scholar] [CrossRef] [PubMed]
- Rogula-Kozlowska, W. Size-segregated urban particulate matter: Mass closure, chemical composition, and primary and secondary matter content. Air Qual. Atmos. Health 2016, 9, 533–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taiwo, A.M. Source apportionment of urban background particulate matter in Birmingham, United Kingdom using a mass closure model. Aerosol Air Qual. Res. 2016, 16, 1244–1252. [Google Scholar] [CrossRef] [Green Version]
- Rogula-Kozłowska, W.; Majewski, G.; Widziewicz, K.; Rogula-Kopiec, P.; Tytła, M.; Mathews, B.; Ciuka-Witrylak, M. Seasonal variations of PM1-bound water concentration in urban areas in Poland. Atmos. Pollut. Res. 2019, 10, 267–273. [Google Scholar] [CrossRef]
- Canepari, S.; Simonetti, G.; Perrino, C. Mass size distribution of particle-bound water. Atmos. Environ. 2017, 165, 46–56. [Google Scholar] [CrossRef]
- Rogula-Kozłowska, W.; Widziewicz, K.; Majewski, G. A simple method for determination of total water in PM1 collected on quartz fiber filters. Microchem. J. 2017, 132, 327–332. [Google Scholar] [CrossRef]
- Su, J.; Zhao, P.; Dong, Q. Chemical compositions and liquid water content of size-resolved aerosol in Beijing. Aerosol Air Qual. Res. 2017, 18, 680–692. [Google Scholar] [CrossRef]
- Canepari, S.; Farao, C.; Marconi, E.; Giovannelli, C.; Perrino, C. Qualitative and quantitative determination of water in airborne particulate matter. Atmos. Chem. Phys. 2013, 13, 1193–1202. [Google Scholar] [CrossRef] [Green Version]
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics from Air Pollution to Climate Change; Wiley: New York, NY, USA, 1998. [Google Scholar]
- Saxena, P.; Hildemann, L.M.; McMurry, P.H.; Seinfeld, J.H. Organics alter hygroscopic behavior of atmospheric particles. J. Geophys. Res. 1995, 100, 18755–18770. [Google Scholar] [CrossRef]
- Pye, H.O.T.; Murphy, B.N.; Xu, L.; Ng, N.L.; Carlton, A.G.; Guo, H.; Weber, R.; Vasilakos, P.; Appel, K.W.; Budisulistiorini, S.H.; et al. On the implications of aerosol liquid water and phase separation for organic aerosol mass. Atmos. Chem. Phys. 2017, 17, 343–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- You, Y.; Renbaum-Wolff, L.; Bertram, A.K. Liquid-liquid phase separation in particles containing organics mixed with ammonium sulfate, ammonium bisulfate, ammonium nitrate or sodium chloride. Atmos. Chem. Phys. 2013, 13, 11723–11734. [Google Scholar] [CrossRef] [Green Version]
- Veschi, R.; Valli, G.; Fermo, P.; D'Alessandro, A.; Piazzalunga, A.; Bernardoni, V. Organic and inorganic sampling artefacts assessment. Atmos. Environ. 2009, 43, 1713–1720. [Google Scholar] [CrossRef]
- Tang, I.N. Deliquescence Properties and Particle-Size Change of Hygroscopic Aerosols; BNL-27094; Brookhaven National Lab: Upton, NY, USA, 1979. [Google Scholar]
- Tsyro, S.G. To what extent can aerosol water explain the discrepancy between model calculated and gravimetric PM10 and PM2.5? Atmos. Chem. Phys. 2005, 5, 515–532. [Google Scholar] [CrossRef] [Green Version]
- Ohta, S.; Horial, M.; Yamagata, S.; Murao, N. Chemical characterization of atmospheric fine particles in Sapporo with determination of water content. Atmos. Environ. 1998, 32, 1021–1025. [Google Scholar] [CrossRef]
- Putaud, J.P.; Putaud, J.-P.; Van Dingenen, R.; Alastuey, A.; Bauer, H.; Birmili, W.; Cyrys, J.; Flentje, H.; Fuzzi, S.; Gehrig, R.; et al. A European aerosol phenomenology—3: Physical and chemical characteristics of particulate matter from 60 rural, urban, and kerbside sites across Europe. Atmos. Environ. 2010, 44, 1308–1320. [Google Scholar] [CrossRef]
- Chow, J.C. methods to determine compliance with ambient air quality standards for suspended particles. J. Air Waste Manag. Assoc. 1995, 45, 320–382. [Google Scholar] [CrossRef] [Green Version]
- Widziewicz, K.; Rogula-Kozłowska, W.; Rogula-Kopiec, P.; Majewski, G.; Loska, K. PM1 and PM1 -bound metals during dry and wet periods: Ambient concentration and health effects. Environ. Eng. Sci. 2016, 34, 312–320. [Google Scholar] [CrossRef]
- Majewski, G.; Rogula-Kozłowska, W.; Czechowski, P.O.; Badyda, A.J.; Brandyk, A. The impact of selected parameters on visibility: First results from a long-term campaign in Warsaw, Poland. Atmosphere 2015, 6, 1154–1174. [Google Scholar] [CrossRef] [Green Version]
- Majewski, G.; Rogula-Kozłowska, W.; Rozbicka, K.; Rogula-Kopiec, P.; Mathews, B.; Brandyk, A. concentration, chemical composition and origin of PM1: Results from the first long-term measurement campaign in Warsaw (Poland). Aerosol Air Qual. Res. 2018, 18, 636–654. [Google Scholar] [CrossRef]
- Rogula-Kozłowska, W.; Majewski, G.; Rogula-Kopiec, P.; Mathews, B. Mass concentration and chemical composition of submicron particulate matter (PM1) in the Polish urban areas. IOP Conf. Ser. Earth Environ. Sci. 2019, 214, 12092. [Google Scholar] [CrossRef]
- European Committee for Standardization (CEN). EN 12341:2014 Ambient Air. Standard Gravimetric Measurement Method for the Determination of the PM10 or PM2.5 Mass Concentration of Suspended Particulate Matter; European Committee for Standardization: Brussels, Belgium, 2014. [Google Scholar]
- Zapp, J.A.; Limperos, G.; Brinker, K.C. Toxicity of pyrolysis products of “Teflon” tetrafluoroethylene resin. In Proceedings of the American Industrial Hygiene Association Annual Meeting, New York, NY, USA, 3–9 September 1955. [Google Scholar]
- Conesa, J.A.; Font, R. Polytetrafluoroethylene decomposition in air and nitrogen. Polym. Eng. Sci. 2001, 41, 2137–2147. [Google Scholar] [CrossRef]
- Perrino, C.; Marconi, E.; Tofful, L.; Farao, C.; Materazzi, S.; Canaperi, S. Thermal stability of inorganic and organic compounds in atmospheric particulate matter. Atmos. Environ. 2012, 54, 36–43. [Google Scholar] [CrossRef]
- HYDRANAL®-Technical Center. Eugen Scholz Reagents for Karl Fischer Titration. In Hydranal Manual; Honeywell: Charlotte, NC, USA, 2017. [Google Scholar]
- Widziewicz-Rzońca, K.; Tytła, M. Studies on water sorption by different types of filter media used for PM collection under varying temperature and humidity conditions. Int. J. Environ. Res. Public Health 2020, 17, 5180. [Google Scholar]
- Rogula-Kozlowska, W.; Klejnowsi, K.; Rogila-Kopiec, P.; Ośródka, L.; Krajny, E.; Błaszczak, B.; Mathews, B. Spatial and seasonal variability of the mass concentration and chemical composition of PM2.5 in Poland. Air Qual. Atmos. Health 2014, 7, 41–58. [Google Scholar] [CrossRef] [Green Version]
- Rogula-Kozlowska, W.; Klejnowski, K.; Rogula-Kopiec, P.; Mathews, B.; Szopa, S. A study on the seasonal mass closure of ambient fine and coarse dusts in Zabrze, Poland. Bull. Environ. Contamin. Toxicol. 2012, 88, 722–729. [Google Scholar] [CrossRef]
- Rogula-Kozłowska, W.; Rogula-Kopiec, P.; Klejnowski, K.; Błaszczak, B. Wpływ emisji komunikacyjnej na stężenie dwóch form węgla i rozkład ich masy względem wielkości cząstek w aerozolu atmosferycznym obszaru miejskiego. Rocz. Ochr. Śr. 2013, 15, 1623–1644. [Google Scholar]
- Rogula-Kozłowska, W. Chemical composition and mass closure of ambient particulate matter at a crossroads and a highway in Katowice, Poland. Environ. Prot. Eng. 2015, 41, 15–29. [Google Scholar] [CrossRef]
- Sówka, I.; Chlebowska-Styś, A.; Pachurka, Ł.; Rogula-Kozłowska, W.; Mathews, B. Analysis of particulate matter concentration variability and origin in selected urban areas in Poland. Sustainability 2019, 11, 5725. [Google Scholar] [CrossRef] [Green Version]
- Coker, E.N. The Oxidation of Aluminum at High Temperature Studied by Thermogravimetric Analysis and Differential Scanning Calorimetry; SAND2013-8424; Sandia National Laboratories: Albuquerque, NM, USA, 2013. [Google Scholar] [CrossRef] [Green Version]
- Popovicheva, O.; Persiantseva, N.M.; Shonija, N.K.; DeMott, P.; Koehler, K.; Petters, M.; Kreidenweis, S.; Tishkova, V.; Demirdjian, B.; Suzanne, J. Water interaction with hydrophobic and hydrophilic soot particles. Phys. Chem. Chem. Phys. 2008, 10, 2332–2344. [Google Scholar] [CrossRef] [PubMed]
- Zotov, R.; Meshcheryakov, E.; Livanova, A.; Minakova, T.; Megaev, O.; Isupova, L.; Kurzina, I. Influence of the composition, structure, and physical and chemical properties of aluminium-oxide-based sorbents on water adsorption ability. Materials 2018, 11, 132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Widziewicz-Rzońca, K.; Tytła, M. First systematic review on PM-bound water: Exploring the existing knowledge domain using the CiteSpace software. Scientometrics 2020. [Google Scholar] [CrossRef]
- Meng, Z.; Seinfeld, J.H.; Saxena, P.; Kim, Y.P. Contribution of Water to Particulate Mass in the South Coast Air Basin. Aerosol Sci. Technol. 1995, 22, 111–123. [Google Scholar] [CrossRef] [Green Version]
- Metzger, S.; Abdelkader, M.; Steil, B.; Klingmüller, K. Aerosol water parameterization: Long-term evaluation and importance for climate studies, Atmos. Chem. Phys. 2018, 18, 16747–16774. [Google Scholar] [CrossRef] [Green Version]
- Choi, M.Y.; Chan, C.K. The Effects of Organic Species on the Hygroscopic Behaviors of Inorganic Aerosols. Environ. Sci. Technol. 2002, 36, 2422–2428. [Google Scholar] [CrossRef]



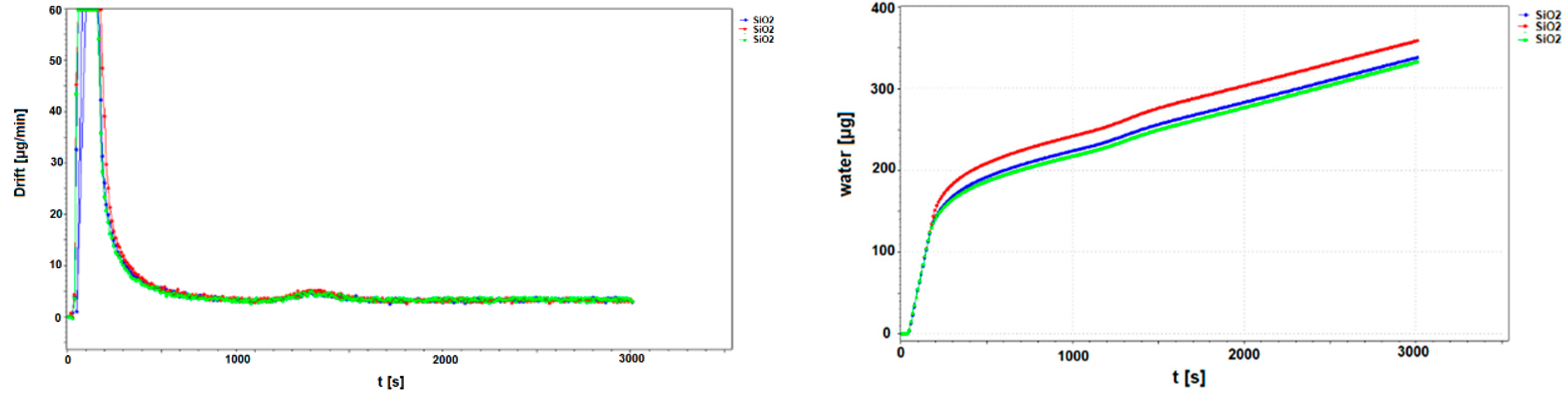

| WSO | SRM Mass µg | Water Content µg | Recovery % |
|---|---|---|---|
| WSO1 | 92110 | 879.6 | 95 |
| WSO2 | 84844 | 804 | 94 |
| WSO3 | 97286 | 922.5 | 94 |
| HYD1 | 205260 | 11419 | 101 |
| HYD2 | 208954 | 11603 | 100 |
| HYD3 | 170004 | 9419.7 | 99 |
| Variable | Temerature [°C] | Humidity [%] | Pressure [hPa] | Tmin * [°C] | Tmax ** [°C] | Precipitation [mm] |
|---|---|---|---|---|---|---|
| TSP [µg/m3] | 0.05 | −0.06 | −0.24 | 0.00 | −0.04 | −0.10 |
| EP1 total water [µg] | −0.08 | −0.05 | −0.23 | −0.23 | 0.05 | −0.40 |
| FP1 water at 50–120 °C [µg] | −0.07 | 0.02 | −0.05 | −0.23 | 0.15 | −0.40 |
| FP2 water at 120–140 °C [µg] | −0.07 | 0.02 | −0.05 | −0.23 | 0.15 | −0.40 |
| FP3 water at 140–180 °C [µg] | −0.01 | −0.05 | −0.06 | −0.15 | 0.18 | −0.40 |
| TSP [µg] | 0.20 | −0.07 | −0.29 | −0.17 | 0.07 | −0.40 |
| PM1 [µg] | 0.41 | 0.05 | 0.70 | 0.45 | 0.54 | −0.40 |
| TSP/PM1 [µg] | −0.33 | 0.01 | −0.54 | −0.29 | −0.53 | 0.40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Widziewicz-Rzońca, K.; Tytła, M.; Majewski, G.; Rogula-Kopiec, P.; Loska, K.; Rogula-Kozłowska, W. Strongly and Loosely Bound Water in Ambient Particulate Matter—Qualitative and Quantitative Determination by Karl Fischer Coulometric Method. Sustainability 2020, 12, 6196. https://0-doi-org.brum.beds.ac.uk/10.3390/su12156196
Widziewicz-Rzońca K, Tytła M, Majewski G, Rogula-Kopiec P, Loska K, Rogula-Kozłowska W. Strongly and Loosely Bound Water in Ambient Particulate Matter—Qualitative and Quantitative Determination by Karl Fischer Coulometric Method. Sustainability. 2020; 12(15):6196. https://0-doi-org.brum.beds.ac.uk/10.3390/su12156196
Chicago/Turabian StyleWidziewicz-Rzońca, Kamila, Malwina Tytła, Grzegorz Majewski, Patrycja Rogula-Kopiec, Krzysztof Loska, and Wioletta Rogula-Kozłowska. 2020. "Strongly and Loosely Bound Water in Ambient Particulate Matter—Qualitative and Quantitative Determination by Karl Fischer Coulometric Method" Sustainability 12, no. 15: 6196. https://0-doi-org.brum.beds.ac.uk/10.3390/su12156196






