Removal and Survival of Fecal Indicators in a Constructed Wetland after UASB Pre-Treatment
Abstract
:1. Introduction
- Universally present in large numbers in the feces of humans and warm-blooded animals;
- Readily detected by simple methods;
- Do not grow in natural waters;
- Persistence in water and removal by water treatment similar to waterborne pathogens.
2. Materials and Methods
- Verify the efficiency of the whole treatment process and also of individual stages with regard to FC and EN (as well as the common physico-chemical parameters, BOD5, COD, SS) through a statistical analysis of collected data;
- Verify the relationship among FIs and the most representative physico-chemical parameters in order to evaluate a possible equivalency correlation.
- Verify the sustainability of a statistical relationship between the concentrations of FC and EN by reconstructing their mathematical correlation with the relative coefficient of determination R2;
- Overall, verify the validity of the experimental treatment technology from the environmental and sanitary point of view.
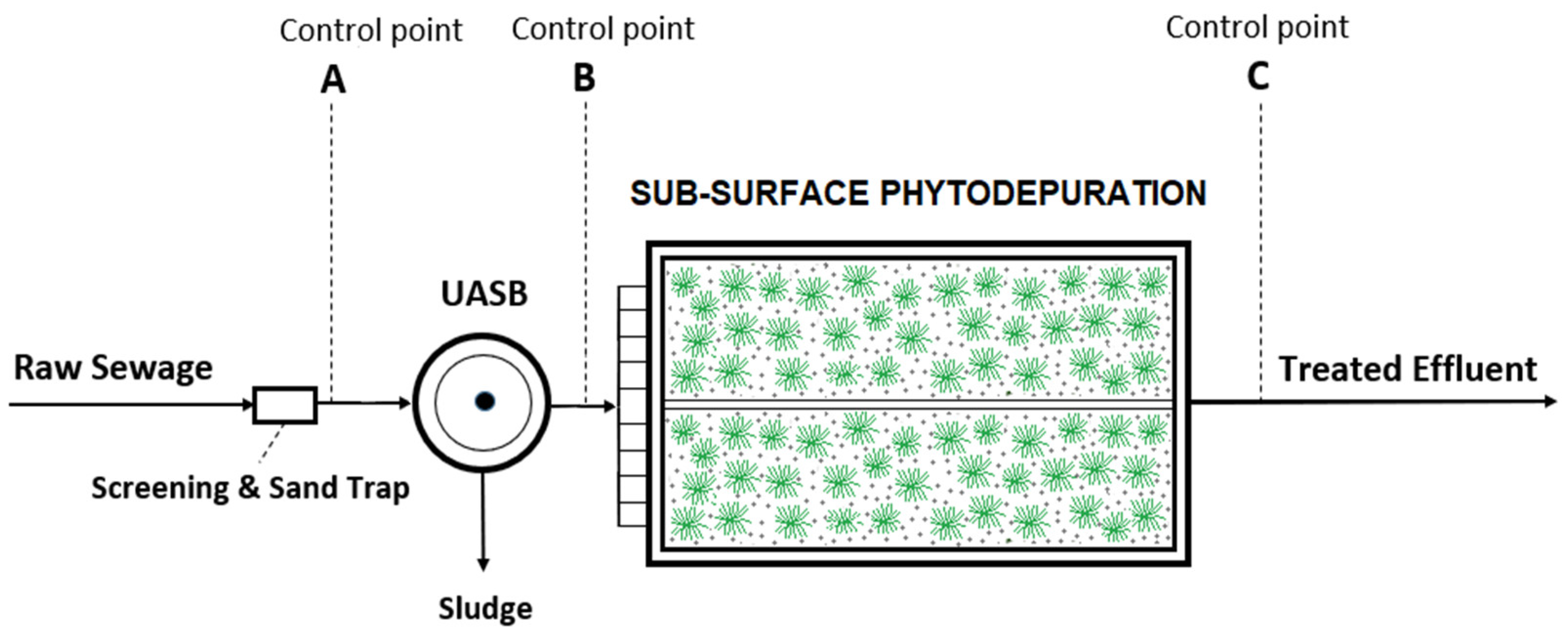
- Point A: raw sewage (after preliminary treatment);
- Point B: effluent of UASB reactor;
- Point C: effluent of SSHFP (whole plant effluent).
- Removal efficiency of the whole plant ηtot = (XA − XC)/XA;
- Removal efficiency of UASB reactor η1 = (XA − XB)/XA;
- Removal efficiency of phytodepuration unit η2 = (XB − XC)/XB;
- Share of total efficiency (ηtot) given by UASB reactor η1* = η1 = (XA − XB)/XA;
- Share of total efficiency (ηtot) given by phytodepuration unit η2* = (XB − XC)/XA;
- ηtot = η1* + η2*;
- XA is the value of the generic parameter at position A (raw sewage);
- XB at position B (UASB outlet = SSHFP inlet);
- XC at position C (SSHFP outlet = final effluent).
3. Results and Discussion
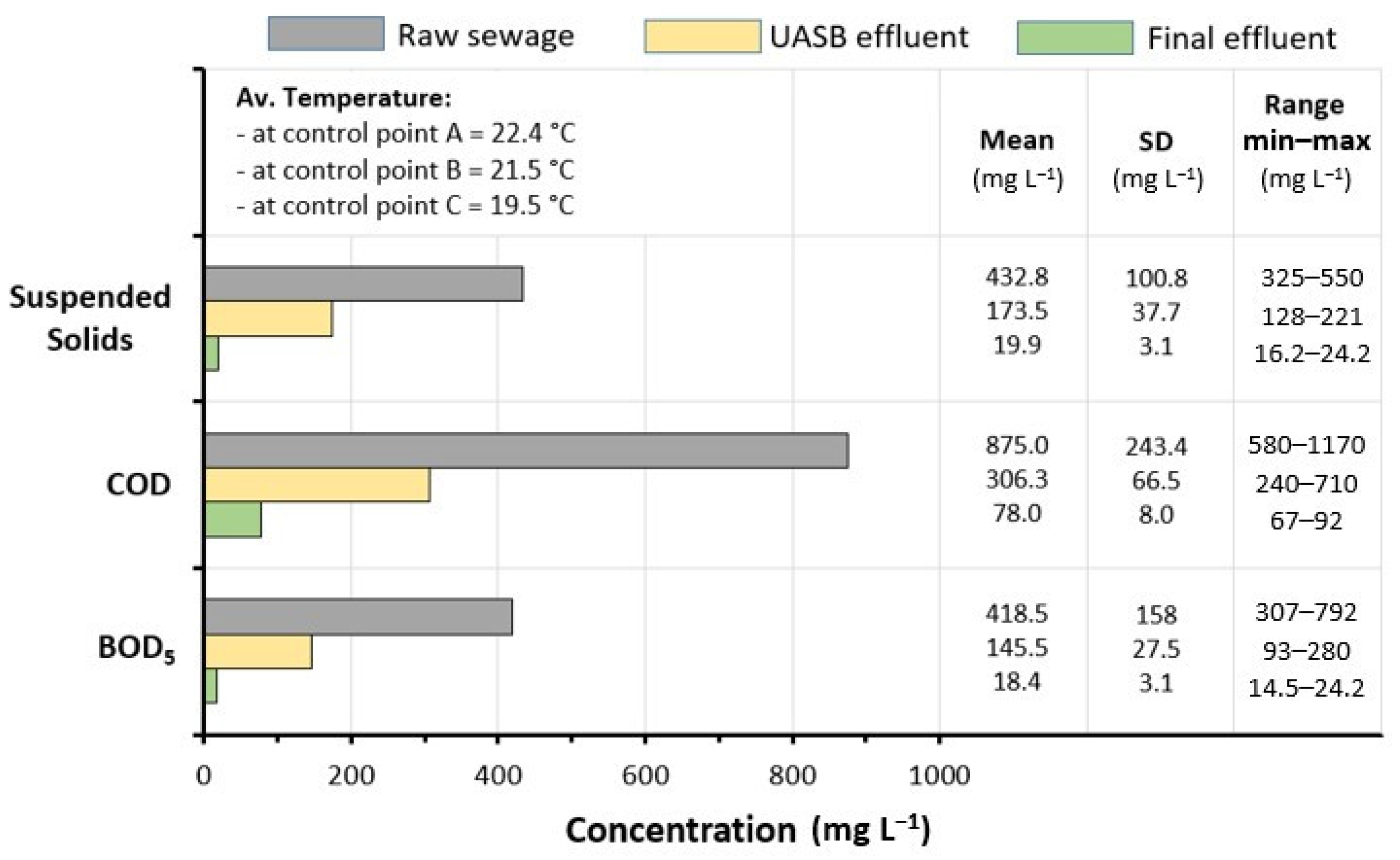
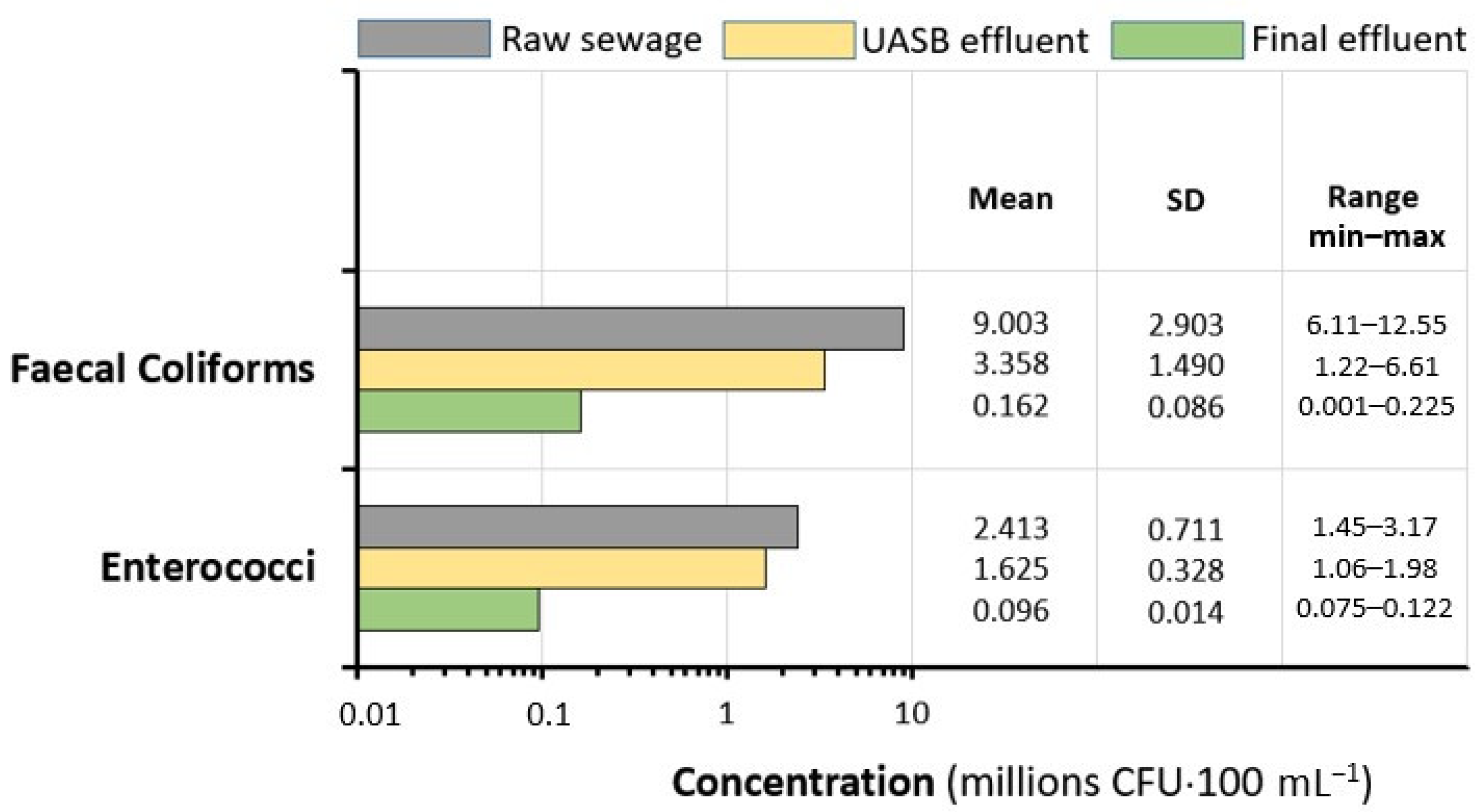
3.1. Raw Sewage Quality
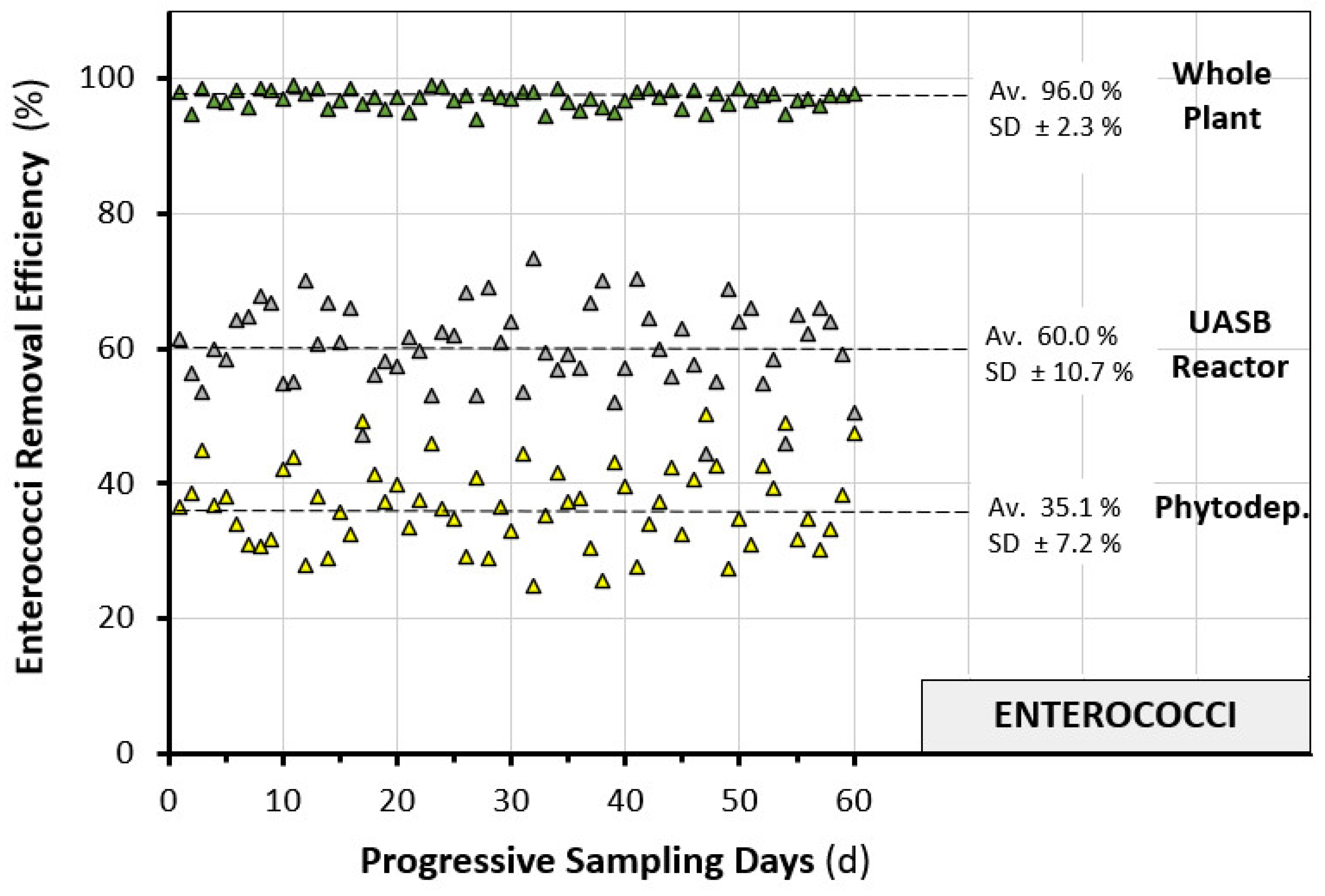
3.2. Plant Performances
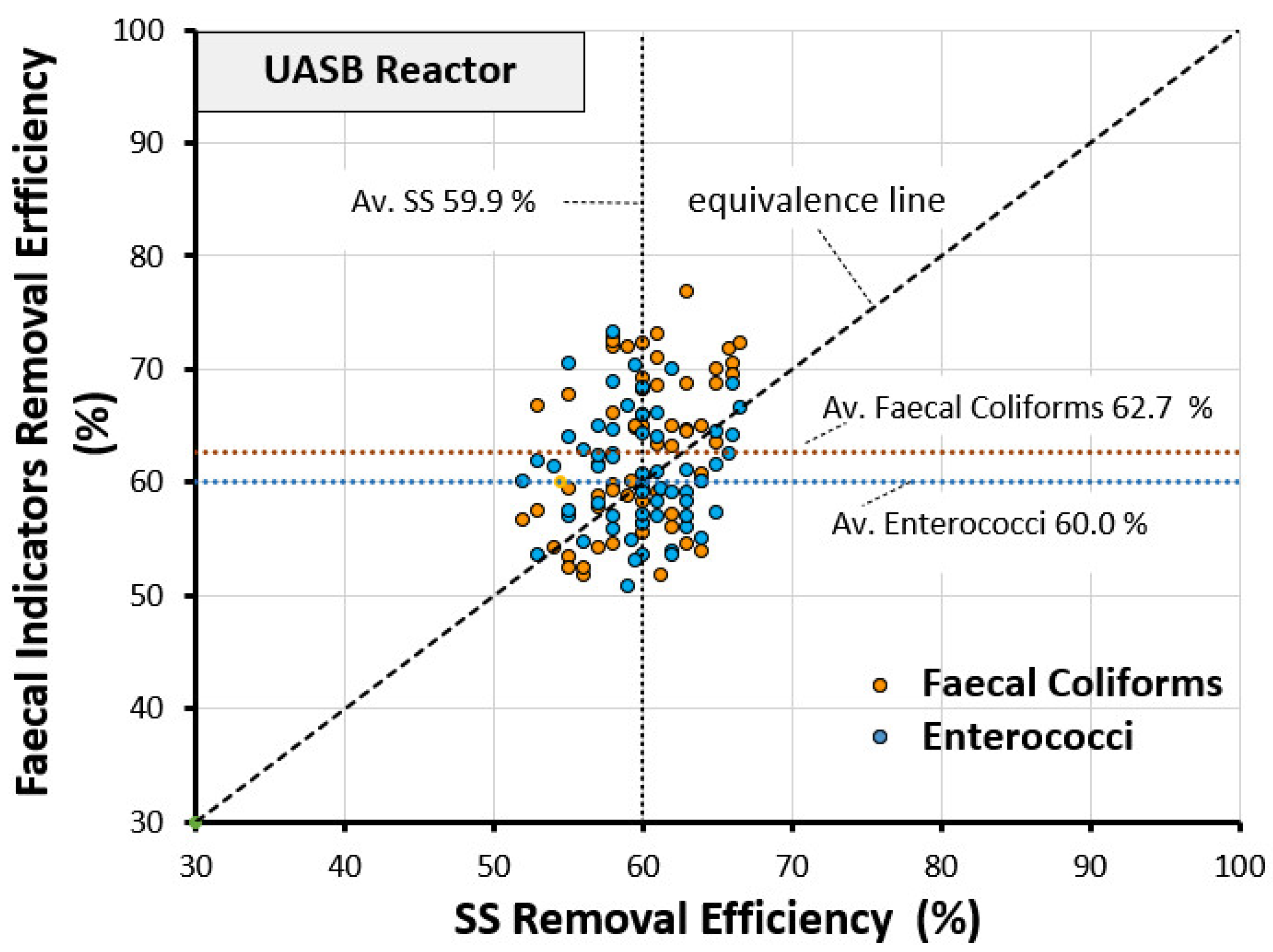
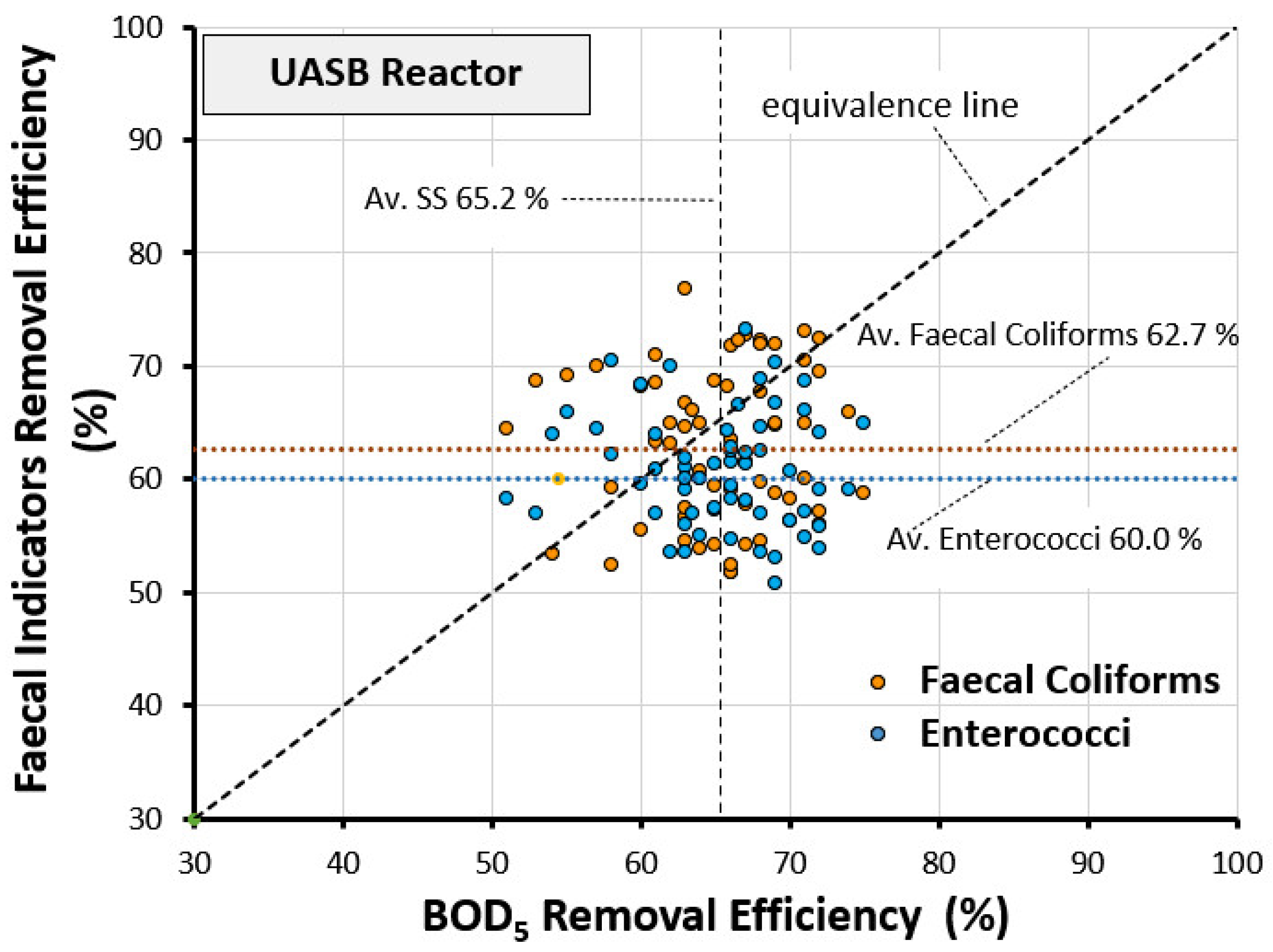
3.3. Comparison among Removal Efficiency of FIs with SS and BOD5
- Average FC removal efficiency = 62.7%.
- Average EN removal efficiency = 60.0%.
- Average SS removal efficiency = 59.9%.
- Average deviation of FC removal efficiency from the equivalence line = 4.11%.
- Average deviation of EN removal efficiency from the equivalence line = 0.83%.
- Average FC removal efficiency = 62.7%.
- Average EN removal efficiency = 60.0%.
- Average BOD5 removal efficiency = 65.2%.
- Average deviation of FC removal efficiency from the equivalence line = −4.66%.
- Average deviation of EN removal efficiency from the equivalence line = −8.08%.
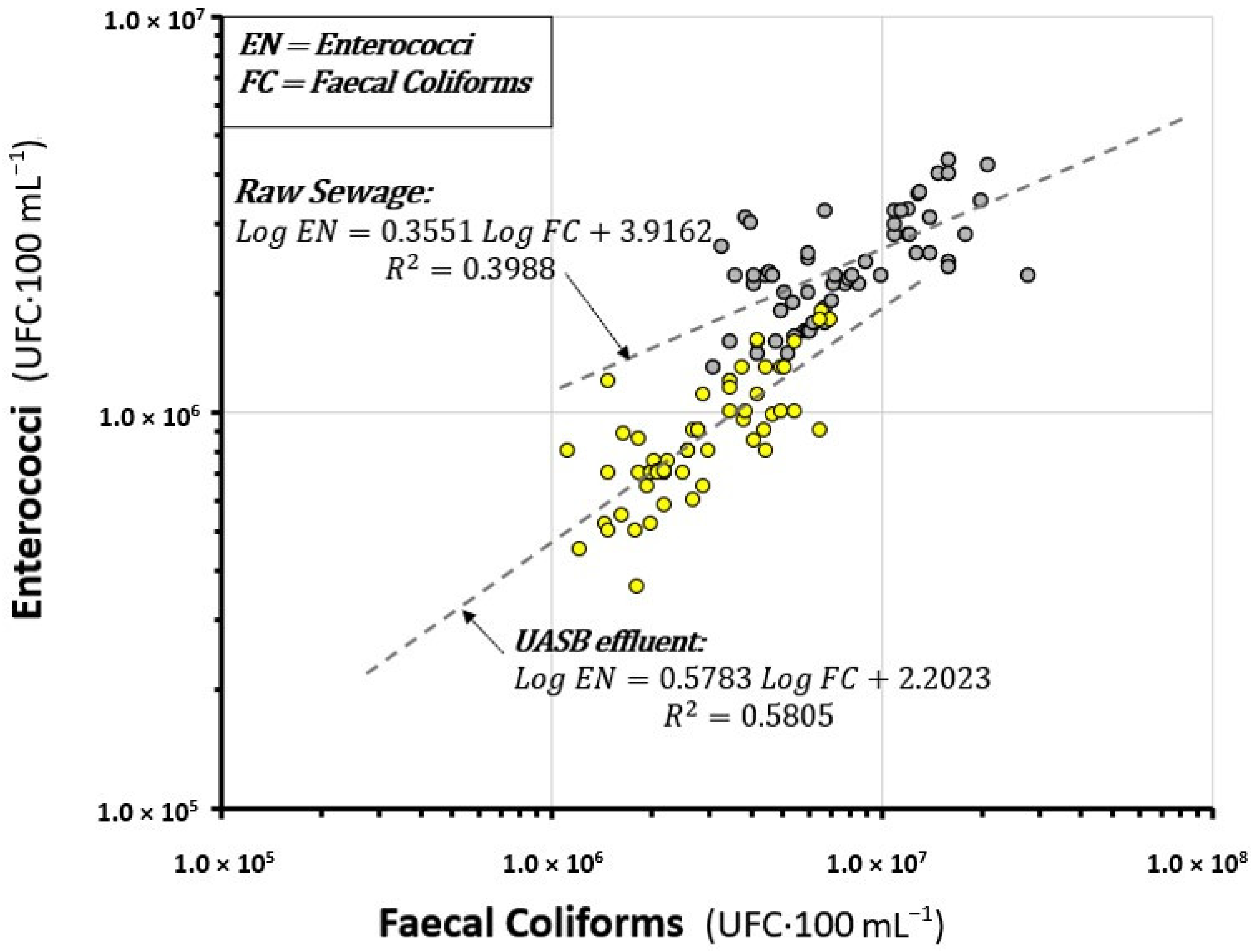
3.4. Correlation between the Concentrations of EN and FC
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Spence, S.D. Microorganisms-The Coliform Group Bacteria. Available online: https://www.scribd.com/presentation/425055000/microorg-3-ppt (accessed on 17 January 2021).
- WHO. Indicators of microbial water quality. In Water Quality: Guidelines, Standards and Health. Assessment of Risk and Risk Management for Water-Related Infectious Disease; Fewtrell, L., Bartram, J., Eds.; World Health Organization by IWA Publishing: London, UK, 2001; p. 424. [Google Scholar]
- Janahi, E.M.; Mustafa, S.; Parkar, S.F.D.; Naser, H.A.; Eisa, Z.M. Detection of Enteric Viruses and Bacterial Indicators in a Sewage Treatment Center and Shallow Water Bay. Int. J. Environ. Res. Public Health 2020, 17, 6483. [Google Scholar] [CrossRef]
- Solo-Gabriele, H.M.; Wolfert, M.A.; Desmarais, T.R.; Palmer, C.J. Sources of Escherichia coli in a coastal subtropical environment. Appl. Environ. Microbiol. 2000, 66, 230–237. [Google Scholar] [CrossRef] [Green Version]
- Yuan, T.; Vadde, K.K.; Tonkin, J.D.; Wang, J.; Lu, J.; Zhang, Z.; Zhang, Y.; McCarthy, A.J.; Sekar, R. Urbanization Impacts the Physicochemical Characteristics and Abundance of Fecal Markers and Bacterial Pathogens in Surface Water. Int. J. Environ. Res. Public Health 2019, 16, 1739. [Google Scholar] [CrossRef] [Green Version]
- Mahmud, Z.H.; Islam, M.S.; Imran, K.M.; Hakim, S.A.I.; Worth, M.; Ahmed, A.; Hossan, S.; Haider, M.; Islam, M.R.; Hossain, F.; et al. Occurrence of Escherichia coli and faecal coliforms in drinking water at source and household point-of-use in Rohingya camps, Bangladesh. Gut Pathog. 2019, 11, 52. [Google Scholar] [CrossRef] [Green Version]
- Qin, G.; Liu, J.; Xu, S.; Sun, Y. Pollution Source Apportionment and Water Quality Risk Evaluation of a Drinking Water Reservoir during Flood Seasons. Int. J. Environ. Res. Public Health 2021, 18, 1873. [Google Scholar] [CrossRef] [PubMed]
- Afolabi, E.; Quilliam, R.S.; Oliver, D.M. Impact of Freeze–Thaw Cycles on Die-Off of E. coli and Intestinal Enterococci in Deer and Dairy Faeces: Implications for Landscape Contamination of Watercourses. Int. J. Environ. Res. Public Health 2020, 17, 6999. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, W.; Schleifer, K.; Whitman, W.B. Family IV. Enterococcaceae fam. nov. In Bergey’s Manual of Systematic Bacteriology, 2nd ed.; De Vos, P., Garrity, G.M., Jones, D., Krieg, N.R., Ludwig, W., Rainey, F.A., Schleifer, K.H., Whitman, W.B., Eds.; Springer: New York, NY, USA, 2009; Volume 3, p. 594. [Google Scholar]
- Lebreton, F.; Willems, R.J.L.; Gilmore, M.S. Enterococcus Diversity, Origins in Nature, and Gut Colonization. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection; Gilmore, M.S., Clewell, D.B., Ike, Y., Shankar, N., Eds.; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014. Available online: https://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/books/NBK190427/ (accessed on 16 January 2021).
- Thamlikitkul, V.; Tiengrim, S.; Thamthaweechok, N.; Buranapakdee, P.; Chiemchaisri, W. Contamination by Antibiotic-Resistant Bacteria in Selected Environments in Thailand. Int. J. Environ. Res. Public Health 2019, 16, 3753. [Google Scholar] [CrossRef] [Green Version]
- Gotkowska-Płachta, A. The Prevalence of Virulent and Multidrug-Resistant Enterococci in River Water and in Treated and Untreated Municipal and Hospital Wastewater. Int. J. Environ. Res. Public Health 2021, 18, 563. [Google Scholar] [CrossRef]
- Tchobanoglous, G.; Burton, F.L.; Stensel, H.D.; Metcalf, E. Wastewater Engineering: Treatment and Reuse; McGraw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Capodaglio, A.G.; Hlavínek, P.; Raboni, M. Advances in wastewater nitrogen removal by biological processes: State of the art review. Rev. Ambiente Água 2013, 11, 250–267. [Google Scholar] [CrossRef] [Green Version]
- Capodaglio, A.G.; Hlavínek, P.; Raboni, M. Physico-chemical technologies for nitrogen removal from wastewaters: A review. Rev. Ambiente Água 2015, 10, 481–498. [Google Scholar]
- Copelli, S.; Raboni, M.; Urbini, G. Water pollution: Biological oxidation and natural control techniques. In Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Reedijk, J., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 1–28. [Google Scholar]
- George, I.; Crop, P.; Servais, P. Fecal coliform removal in wastewater treatment plants studied by plate counts and enzymatic methods. Water Res. 2002, 36, 2607–2617. [Google Scholar] [CrossRef]
- Raboni, M.; Gavasci, R.; Torretta, V. Assessment of the Fate of Escherichia coli in Different Stages of Wastewater Treatment Plants. Water Air Soil Pollut. 2016, 227, 455. [Google Scholar] [CrossRef]
- APAT-IRSA CNR. Metodi per la Determinazione di Microorganismi Indicatori Di Inquinamento e di Patogeni. Metod. Anal. Acque 2003, 3, 865. [Google Scholar]
- Cavallini, G.S.; De Campos, S.X.; De Souza, J.B.; De Souza Vidal, C.M. Evaluation of the Physical–Chemical Characteristics of Wastewater After Disinfection with Peracetic Acid. Water Air Soil Pollut. 2013, 224, 1752. [Google Scholar] [CrossRef]
- Raboni, M.; Gavasci, R.; Urbini, G. UASB followed by Sub-Surface Horizontal Flow Phytodepuration for the Treatment of the Sewage Generated by a Small Rural Community. Sustainability 2014, 6, 6998–7012. [Google Scholar] [CrossRef] [Green Version]
- Koivunen, J.; Sitonen, A.; Heinonen-Tanskia, H. Elimination of enteric bacteria in biological–chemical wastewater treatment and tertiary filtration units. Water Res. 2003, 37, 690–698. [Google Scholar] [CrossRef]
- Boni, M.R.; Copelli, S.; Raboni, M. Study of the performance of disinfection with sodium hypochlorite on a full-scale sewage treatment plant. Rev. Ambiente Água 2020, 15, e2652. [Google Scholar]
- Landa-Cansigno, O.; Durán-Álvarez, J.C.; Jiménez-Cisneros, B. Retention of Escherichia coli, Giardia lamblia cysts and Ascaris lumbricoides eggs in agricultural soils irrigated by untreated wastewater. J. Environ. Manag. 2013, 128, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Barrios-Hernandez, M.L.; Pronk, M.K.; Garcia, H.; Boersma, A.; Brdjanovic, D.; van Loosdrecht, M.C.M.; Hooijmans, C.M. Removal of bacterial and viral indicator organisms in full-scale aerobic granular sludge and conventional activated sludge systems. Water Res. 2020, 6, 100040. [Google Scholar] [CrossRef] [PubMed]
- Vilanova, X.; Manero, A.; Cerda-Cuellar, M.; Blanch, A.R. The composition and persistence of FC and enterococcal populations in sewage treatment plants. J. Appl. Microbiol. 2004, 96, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Dias, S.; Mucha, A.P.; Crespo, R.D.; Rodrigues, P.; Almeida, C.M.R. Livestock Wastewater Treatment in Constructed Wetlands for Agriculture Reuse. J. Environ. Res. Public Health 2020, 17, 8592. [Google Scholar] [CrossRef]
- Nawaz, N.; Ali, S.; Shabir, G.; Rizwan, M.; Shakoor, M.B.; Shahid, M.J.; Afzal, M.; Arslan, M.; Hashem, A.; Abd_Allah, E.F.; et al. Bacterial Augmented Floating Treatment Wetlands for Efficient Treatment of Synthetic Textile Dye Wastewater. Sustainability 2020, 12, 3731. [Google Scholar] [CrossRef]
- Raboni, M.; Torretta, V.; Viotti, P.; Urbini, G. Experimental plant for the physical-chemical treatment of groundwater polluted by Municipal Solid Waste (MSW) leachate, with ammonia recovery. Rev. Ambiente Água 2013, 8, 22–32. [Google Scholar]
- Trulli, E.; Torretta, V.; Rada, E.C. Water Restoration of an Urbanized Karst Stream by Free-Water-Surface Conducted Wetlands as Municipal Wastewater Posttreatment. UPB Sci. Bull 2016, 78, 163–174. [Google Scholar]
- Reinoso, R.; Torres, L.A.; Bécares, E. Efficiency of natural systems for removal of bacteria and pathogenic parasites from wastewater. Sci. Total Environ. 2008, 395, 80–86. [Google Scholar] [CrossRef]
- Pant, A.; Mittal, A.K. Monitoring of pathogenicity of effluents from the UASB based sewage treatment plant. Environ. Monit. Assess. 2007, 133, 43–51. [Google Scholar] [CrossRef]
- Tessele, F.; Monteggia, L.O.; Rubio, J. Treatment of municipal wastewater UASB reactor effluent by unconventional flotation and uv disinfection. Water Sci. Technol. 2005, 52, 315–322. [Google Scholar] [CrossRef]
- Antunes, S.; Dionisio, L.; Silva, M.C.; Valente, M.S.; Borrego, J.J. Coliforms as Indicators of Efficiency of Wastewater Treatment Plants. In Proceedings of the 3rd International Conference on Energy, Environment, Ecosystems and Sustainable Development, IASME/WSEAS, Agios Nikolaos, Greece, 24–26 July 2007; pp. 26–29. [Google Scholar]
- Vymazal, J. Removal of Enteric Bacteria in Constructed Treatment Wetlands with Emergent Macrophytes: A Review. J. Environ. Sci. Health 2005, 40, 1355–1367. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.E.; Halmi, M.I.E.B.; Samad, M.Y.B.A.; Uddin, M.K.; Mahmud, K.; Shukor, M.Y.A.; Abdullah, S.R.S.; Shamsuzzaman, S.M. Design, Operation and Optimization of Constructed Wetland for Removal of Pollutant. Int. J. Environ. Res. Public Health 2020, 17, 8339. [Google Scholar] [CrossRef]
- De La Mora-Orozco, C.; Gonzales-Acuna, I.J.; Saucedo-Teran, R.A.; Flores-Lopez, H.E.; Rubio-Arias, H.O.; Ochoa_riviero, J.M. Removing Organic Matter and Nutrients from Pig Farm Wastewater with a Constructed Wetland System. Int. J. Environ. Res. Public Health 2018, 15, 1031. [Google Scholar] [CrossRef] [Green Version]
- Herrera-Meliàn, J.A. Sustainable Wastewater Treatment Systems (2018–2019). Sustainability 2020, 12, 1940. [Google Scholar] [CrossRef] [Green Version]
- Raboni, M.; Torretta, V.; Urbini, G. Influence of strong diurnal variations in sewage quality on the performance of biological denitrification in small community wastewater treatment plants (WWTPs). Sustainability 2013, 5, 3679–3689. [Google Scholar] [CrossRef] [Green Version]
- Raboni, M.; Torretta, V.; Viotti, P.; Urbini, G. Pilot experimentation with complete mixing anoxic reactors to improve sewage denitrification in treatment plants in small communities. Sustainability 2014, 6, 112–122. [Google Scholar] [CrossRef] [Green Version]
- Sandoval-Herazo, L.C.; Alvarado-Lassman, A.; Marin-Muniz, J.L.; Mendez-Contreras, J.M.; Zamora-Castro, S.A. Effects of the Use of Ornamental Plants and Different Substrates in the Removal of Wastewater Pollutants through Microcosms of Constructed Wetlands. Sustainability 2018, 10, 1594. [Google Scholar] [CrossRef] [Green Version]
- Kochi, L.Y.; Freitas, P.L.; Maranho, L.T.; Juneau, P.; Gomes, M.P. Aquatic Macrophytes in Constructed Wetlands: A Fight against Water Pollution. Sustainability 2020, 12, 9202. [Google Scholar] [CrossRef]
- Yasar, A.; Tabinda, A.B. Anaerobic treatment of industrial wastewater by UASB reactor integrated with chemical oxidation processes: An overview. Pol. J. Environ. Stud. 2010, 19, 1051–1061. [Google Scholar]
- Powar, A.A.; Kore, V.S.; Kore, S.V.; Kulkarni, G.S. Review on application of UASB technologyfor wastewater treatment. Int. J. Adv. Sci. Eng. Technol. 2013, 2, 125–133. [Google Scholar]
- Musa, M.A.; Syazwani, I.; Hasfalina, C.M.; Norsyahariati, N.; Daud, N. Effect of Organic Loading Rate on Anaerobic Digestion Performance of Mesophilic (UASB) Reactor Using Cattle Slaughterhouse Wastewater as Substrate. Int. J. Environ. Res. Public Health 2018, 15, 2220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Musa, M.A.; Syazwani, I.; Harun, M.R.; Malek, A.; Wahab, A. A Comparative Study of Biogas Production from Cattle Slaughterhouse Wastewater Using Conventional and Modified Upflow Anaerobic Sludge Blanket (UASB) Reactors. Int. J. Environ. Res. Public Health 2020, 17, 283. [Google Scholar] [CrossRef] [Green Version]
- Van Lier, J.B.; Vashi, A.; van der Lubbe, J.; Heffernan, B. Anaerobic Sewage Treatment Using UASB Reactors: Engineering and Operational Aspects. In Environmental Anaerobic Technology: Applications and New Developments; Fang, H.H.P., Ed.; Imperial College Press: London, UK, 2010; p. 420. [Google Scholar]
- Hampannavar, U.S.; Shivayogimath, C.B. Anaerobic treatment of sugar industry wastewater by upflow anaerobic sludge blanket reactor at ambient temperature. Int. J. Environ. Sci. 2010, 1, 631–639. [Google Scholar]
- Vieira, S.M.M.; Souza, M.E. Development of technology for the use of the UASB reactor in domestic sewage treatment. Water Sci. Technol. 1986, 18, 109–121. [Google Scholar] [CrossRef]
- Chernicharo, C.A.L.; Almeida, P.G.S.; Lobato, L.C.S.; Couto, T.C.; Borges, J.M.; Lacerda, Y.S. Experience with the design and start up of two full-scale UASB plants in Brazil: Enhancements and drawbacks. Water Sci. Technol. 2009, 60, 507–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cosoli, P.; Sandri, A.; Paolazzi, F.; Rada, E.C.; Ragazzi, M. Wastewater treatment and disposal in Mahajanga, Madagascar—Scientific, Multidisciplinar cooperation. UPB Sci. Bull 2015, 77, 301–310. [Google Scholar]
- Ragazzi, M.; Catellani, R.; Rada, E.C.; Torretta, V.; Salazar-Valenzuela, X. Management of urban wastewater on one of the Galapagos Islands. Sustainability 2016, 8, 208. [Google Scholar] [CrossRef] [Green Version]
- Khalil, N.; Sinha, R.; Raghav, A.K.; Mittal, A.K. UASB Technology for Sewage Treatment in India: Experience, Economic Evaluation and Its Potential in Other Developing Countries. In Proceedings of the 12th International Water Technology Conference, IWTC12, Alexandria, Egypt, 27–30 March 2008; pp. 1411–1427. [Google Scholar]
- Investigación y Desarrollo. Common Weed Revealed to Diminish Water Pollution. Available online: www.sciencedaily.com/releases/2015/03/150313083400.htm (accessed on 24 November 2020).
- APHA-American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005. [Google Scholar]
- EPA. Waste Water Treatment Manuals Primary, Secondary and Tertiary Treatment; Environmental Protection Agency Ardcavan: Wexford, Ireland, 1997; ISBN 1 899965 46 7.
- Lavrnic, S.; Pereyra, M.Z.; Cristino, A.; Cupido, D.; Lucchese, G.; Pascale, M.R.; Toscano, A.; Mancini, M. The Potential Role of Hybrid Constructed Wetlands Treating University Wastewater—Experience from Northern Italy. Sustainability 2020, 12, 10604. [Google Scholar] [CrossRef]
- Herrera-Meliàn, J.A.; Mendoza-Aguiar, M.; Guedes-Alfonso, R.; Garcia-Jimenez, P.; Carrasco-Acosta, M.; Ranieri, E. Multistage Horizontal Subsurface Flow vs. Hybrid Constructed Wetlands for the Treatment of Raw Urban Wastewater. Sustainability 2020, 12, 5102. [Google Scholar] [CrossRef]




| Bacteria | Protozoa | Viruses |
|---|---|---|
| Vibrio cholera—cholera | Entamoeba histolytica—amoebic dysentery | Polio virus—paralytic poliomyelitis |
| Yersinia enterocolitica—gastroenteritis | Giardia lamblia—intestinal disease | Rotaviruses—gastroenteritis |
| Escherichia coli—gastroenteritis | Cryptosporidium—intestinal disease | Norwalk viruses—gastroenteritis |
| Shigella sp.—gastroenteritis | Hepatitis Type A and E—liver inflammation | |
| Listeria monocytogenes—flu-like symptoms | ||
| Salmonella sp.—gastroenteritis, typhoid | ||
| Campylobacter jejuni—gastroenteritis (animal sources) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conti, F.; Rada, E.C.; Viotti, P.; Raboni, M. Removal and Survival of Fecal Indicators in a Constructed Wetland after UASB Pre-Treatment. Sustainability 2021, 13, 9302. https://0-doi-org.brum.beds.ac.uk/10.3390/su13169302
Conti F, Rada EC, Viotti P, Raboni M. Removal and Survival of Fecal Indicators in a Constructed Wetland after UASB Pre-Treatment. Sustainability. 2021; 13(16):9302. https://0-doi-org.brum.beds.ac.uk/10.3390/su13169302
Chicago/Turabian StyleConti, Fabio, Elena Cristina Rada, Paolo Viotti, and Massimo Raboni. 2021. "Removal and Survival of Fecal Indicators in a Constructed Wetland after UASB Pre-Treatment" Sustainability 13, no. 16: 9302. https://0-doi-org.brum.beds.ac.uk/10.3390/su13169302








