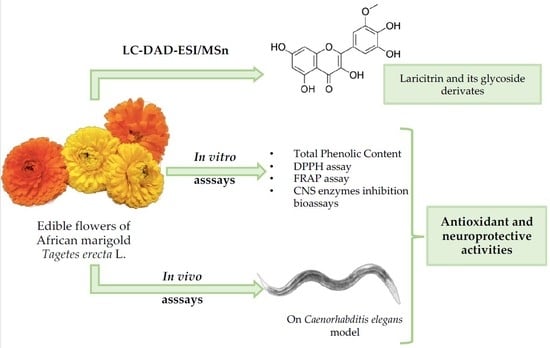
Edible Flowers of Tagetes erecta L. as Functional Ingredients: Phenolic Composition, Antioxidant and Protective Effects on Caenorhabditis elegans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Standards and Reagents
2.2. Plant Extracts
2.3. Analysis of Phenolic Compounds
2.4. In Vitro Antioxidant Activity
2.4.1. Determination of Folin-Ciocalteu Reducing Capacity
2.4.2. Radical Scavenging Activity (RSA)
2.4.3. Ferric Reducing Antioxidant Power (FRAP) Assay
2.5. Neuroprotective Potential
2.5.1. Inhibition of the Acetylcholinesterase Enzyme (AChE)
2.5.2. Inhibition of Monoamine Oxidase A (MAO-A) Enzyme
2.6. C. elegans Assays
2.6.1. C. elegans Strains and Maintenance Conditions
2.6.2. Assessment of Acute Toxicology
2.6.3. Evaluation of Resistance to Oxidative Stress
2.6.4. Lifespan Assay.
2.6.5. Paralysis Assay
2.7. Statistical Analysis
3. Results and Discussion
3.1. Identification and Quantification of Phenolic Compounds of T. erecta Extracts
3.2. In vitro Antioxidant Activity
3.3. Neuroprotective Potential
3.4. Evaluation of T. erecta Extracts Acute Toxicity
3.5. T. erecta Exerts Protective Effects on C. elegans under Lethal Oxidative Stress
3.6. T. erecta Flower Extracts Enhance the Lifespan of C. elegans
3.7. Extracts of T. erecta Flower Delayed the Paralysis of β-Amyloid (Aβ) Transgenic C. elegans
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mlcek, J.; Rop, O. Fresh edible flowers of ornamental plants—A new source of nutraceutical foods. Trends Food Sci. Technol. 2011, 22, 561–569. [Google Scholar] [CrossRef]
- Lu, B.; Li, M.; Yin, R. Phytochemical Content, Health Benefits, and Toxicology of Common Edible Flowers: A Review. Crit. Rev. Food Sci. Nutr. 2016, 56, S130–S148. [Google Scholar] [CrossRef] [PubMed]
- Egebjerg, M.M.; Olesen, P.T.; Eriksen, F.D.; Ravn-Haren, G.; Bredsdorff, L.; Pilegaard, K. Are wild and cultivated flowers served in restaurants or sold by local producers in Denmark safe for the consumer? Food Chem. Toxicol. 2018, 120, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Breithaupt, D.E. Modern application of xanthophylls in animal feeding—A review. Trends Food Sci. Technol. 2007, 18, 501–506. [Google Scholar] [CrossRef]
- Kaisoon, O.; Konczak, I.; Siriamornpun, S. Potential health enhancing properties of edible flowers from Thailand. Food Res. Int. 2012, 46, 563–571. [Google Scholar] [CrossRef]
- Navarro-González, I.; González-Barrio, R.; García-Valverde, V.; Bautista-Ortín, A.B.; Periago, M.J. Nutritional composition and antioxidant capacity in edible flowers: Characterisation of phenolic compounds by HPLC-DAD-ESI/MSn. Int. J. Mol. Sci. 2015, 16, 805–822. [Google Scholar] [CrossRef] [PubMed]
- Nwachukwu, I.D.; Udenigwe, C.C.; Aluko, R.E. Lutein and zeaxanthin: Production technology, bioavailability, mechanisms of action, visual function, and health claim status. Trends Food Sci. Technol. 2015, 49, 74–84. [Google Scholar] [CrossRef]
- Calvo, D.R.; Martorell, P.; Genovés, S.; Gosálbez, L. Development of novel functional ingredients: Need for testing systems and solutions with Caenorhabditis elegans. Trends Food Sci. Technol. 2016, 54, 197–203. [Google Scholar] [CrossRef]
- Gruber, J.; Fang, L.; Kumar, S.; Halliwell, B. Deceptively simple but simply deceptive–Caenorhabditis elegans lifespan studies: Considerations for aging and antioxidant effects. FEBS Lett. 2009, 583, 3377–3387. [Google Scholar] [CrossRef] [PubMed]
- Bessada, S.M.F.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R.; Oliveira, M.B.P.P. Phenolic profile and antioxidant activity of Coleostephus myconis (L.) Rchb.f.: An underexploited and highly disseminated species. Ind. Crop. Prod. 2016, 89, 45–51. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, J.; Shen, J.; Silva, A.; Dennis, D.A.; Barrow, C.J. A simple 96-well microplate method for estimation of total polyphenol content in seaweeds. J. Appl. Phycol. 2006, 18, 445–450. [Google Scholar] [CrossRef]
- López, V.; Akerreta, S.; Casanova, E.; García-mina, J.; Cavero, R.; Calvo, M.; López, V.; Akerreta, S.; Casanova, E.; García-mina, J. Screening of Spanish Medicinal Plants for Antioxidant and Antifungal Activities Screening of Spanish Medicinal Plants for Antioxidant and. Pharm. Biol. 2008, 46, 602–609. [Google Scholar] [CrossRef]
- Pulido, R.; Bravo, L.; Saura-calixto, F. Antioxidant Activity of Dietary Polyphenols As Determined by a Modified Ferric Reducing/Antioxidant Power Assay. J. Agric. Food Chem. 2000, 48, 3396–3402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rhee, I.K.; Van De Meent, M.; Ingkaninan, K.; Verpoorte, R. Screening for acetylcholinesterase inhibitors from Amaryllidaceae using silica gel thin-layer chromatography in combination with bioactivity staining. J. Chromatogr. A 2001, 915, 217–223. [Google Scholar] [CrossRef]
- Stafford, G.I.; Pedersen, P.D.; Jäger, A.K.; Staden, J. Van Monoamine oxidase inhibition by southern African traditional medicinal plants. South Afr. J. Bot. 2007, 73, 384–390. [Google Scholar] [CrossRef]
- Stiernagle, T. Maintenance of C. elegans. WormBook 1999, 2, 51–67. [Google Scholar] [CrossRef] [PubMed]
- Donkin, S.G.; Williams, P.L. Influence of developmental stage, salts and food presence on various end points using Caenorhabditis Elegans for aquatic toxicity testing. Environ. Toxicol. Chem. 1995, 14, 2139–2147. [Google Scholar] [CrossRef]
- Surco-Laos, F.; Cabello, J.; Gómez-Orte, E.; González-Manzano, S.; González-Paramás, A.M.; Santos-Buelga, C.; Dueñas, M. Effects of O-methylated metabolites of quercetin on oxidative stress, thermotolerance, lifespan and bioavailability on Caenorhabditis elegans. Food Funct. 2011, 2, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Solis, G.M.; Petrascheck, M. Measuring Caenorhabditis elegans life span in 96 well microtiter plates. J. Vis. Exp. 2011, e2496. [Google Scholar] [CrossRef] [PubMed]
- Dostal, V.; Link, C.D. Assaying β-amyloid Toxicity using a Transgenic C. elegans Model. J. Vis. Exp. 2010, 3791, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Everette, J.D.; Bryant, Q.M.; Green, A.M.; Abbey, Y.A.; Wangila, G.W.; Walker, R.B. A thorough study of reactivity of various compound classes towards the Folin-Ciocalteu. J. Agric. Food Chem. 2014, 58, 8139–8144. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Gao, Y.; Zhao, J.; Qi, W. Phenolic, flavonoid, and lutein ester content and antioxidant activity of 11 cultivars of Chinese marigold. J. Agric. Food Chem. 2007, 55, 8478–8484. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Deng, M.; Lv, Z.; Peng, Y. Evaluation of antioxidant activities of extracts from 19 Chinese edible flowers. Springerplus 2014, 3, 315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingkasupart, P.; Manochai, B.; Song, W.T.; Hong, J.H. Antioxidant activities and lutein content of 11 marigold cultivars (Tagetes spp.) grown in Thailand. Food Sci. Technol. 2015, 35, 380–385. [Google Scholar] [CrossRef]
- Kaisoon, O.; Siriamornpun, S.; Weerapreeyakul, N.; Meeso, N. Phenolic compounds and antioxidant activities of edible flowers from Thailand. J. Funct. Foods 2011, 3, 88–99. [Google Scholar] [CrossRef]
- Siriamornpun, S.; Kaisoon, O.; Meeso, N. Changes in colour, antioxidant activities and carotenoids (lycopene, b-carotene, lutein) of marigold flower (Tagetes erecta L.) resulting from different drying processes. J. Funct. Foods 2012, 4, 757–766. [Google Scholar] [CrossRef]
- Müller, L.; Fröhlich, K.; Böhm, V. Comparative antioxidant activities of carotenoids measured by ferric reducing antioxidant power (FRAP), ABTS bleaching assay (αTEAC), DPPH assay and peroxyl radical scavenging assay. Food Chem. 2011, 129, 139–148. [Google Scholar] [CrossRef]
- Tan, C.; Xue, J.; Abbas, S.; Feng, B.; Zhang, X.; Xia, S. Liposome as a delivery system for carotenoids: Comparative antioxidant activity of carotenoids as measured by ferric reducing antioxidant power, DPPH assay and lipid peroxidation. J. Agric. Food Chem. 2014, 62, 6726–6735. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ortega, G.; Angeles-López, G.E.; Argueta-Villamar, A.; González-Trujano, M.E. Preclinical evidence of the anxiolytic and sedative-like activities of Tagetes erecta L. reinforces its ethnobotanical approach. Biomed. Pharmacother. 2017, 93, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Sprando, R.L.; Olejnik, N.; Cinar, H.N.; Ferguson, M. A method to rank order water soluble compounds according to their toxicity using Caenorhabditis elegans, a Complex Object Parametric Analyzer and Sorter, and axenic liquid media. Food Chem. Toxicol. 2009, 47, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Büchter, C.; Ackermann, D.; Honnen, S.; Arnold, N.; Havermann, S.; Koch, K.; Wätjen, W. Methylated derivatives of myricetin enhance life span in Caenorhabditis elegans dependent on the transcription factor DAF-16. Food Funct. 2015, 6, 3383–3392. [Google Scholar] [CrossRef] [PubMed]
- Grünz, G.; Haas, K.; Soukup, S.; Klingenspor, M.; Kulling, S.E.; Daniel, H.; Spanier, B. Structural features and bioavailability of four flavonoids and their implications for lifespan-extending and antioxidant actions in C. elegans. Mech. Ageing Dev. 2012, 133, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Augusti, P.R.; Brasil, A.V.S.; Souto, C.; Göethel, G.; de Oliveira Rios, A.; Emanuelli, T.; Bürger, M.E.; Garcia, S.C. Microcystin-LR exposure induces oxidative damage in Caenorhabditis elegans: Protective effect of lutein extracted from marigold flowers. Food Chem. Toxicol. 2017, 109, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Kenyon, C. The first long-lived mutants: Discovery of the insulin/IGF-1 pathway for ageing. Philos. Trans. R. Soc. B-Biol. Sci. 2011, 366, 9–16. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, C.N.; Broihier, H.T. Keeping Neurons Young and Foxy: FoxOs Promote Neuronal Plasticity. Trends Genet. 2018, 34, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Barclay, J.W.; Burgoyne, R.D.; Morgan, A. Using C. elegans to discover therapeutic compounds for ageing-associated neurodegenerative diseases. Chem. Cent. J. 2015, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Khanam, H.; Ali, A.; Asif, M. Shamsuzzaman Neurodegenerative diseases linked to misfolded proteins and their therapeutic approaches: A review. Eur. J. Med. Chem. 2016, 124, 1121–1141. [Google Scholar] [CrossRef] [PubMed]
- Rand, J.B.; Duerr, J.S.; Frisby, D.L. Neurogenetics of vesicular transporters in C. elegans. FASEB J. 2000, 14, 2414–2422. [Google Scholar] [CrossRef] [PubMed]
- Bonda, D.J.; Wang, X.; Perry, G.; Nunomura, A.; Tabaton, M.; Zhu, X.; Smith, M.A. Oxidative stress in Alzheimer disease: A possibility for prevention. Neuropharmacology 2010, 59, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Drake, J.; Link, C.D.; Butterfield, D.A. Oxidative stress precedes fibrillar deposition of Alzheimer’ s disease amyloid beta-peptide (1–42) in a transgenic Caenorhabditis elegans model. Neurobiol. Aging 2003, 24, 415–420. [Google Scholar] [CrossRef]
- Shimmyo, Y.; Kihara, T.; Akaike, A.; Niidome, T.; Sugimoto, H. Multifunction of Myricetin on Aβ: Neuroprotection Via to Conformational Change of Aβ and Reduction of Aβ Via the Interference of Secretases. J. Neurosci. Res. 2008, 86, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Caligiuri, S.P.B.; Brown, D.; Pierce, G.N. Clinical trials using functional foods provide unique challenges. J. Funct. Foods 2018, 45, 233–238. [Google Scholar] [CrossRef]




| Peak | Rt (min) | λmax (nm) | Molecular ion [M-H]− (m/z) | MS2 (m/z) | Tentative Identification | Quantification | t-Students Test p-Value | |
|---|---|---|---|---|---|---|---|---|
| T. erecta Orange | T. erecta Yellow | |||||||
| 1 | 5.81 | 273 | 321 | 169 (100) | Digallic acid A | 1.233 ± 0.004 | 1.25 ± 0.02 | 0.007 |
| 2 | 13.83 | 360 | 655 | 493 (100), 331 (11) | Laricitrin-di-hexoside B | 1.669 ± 0.001 | 1.477 ± 0.003 | 0.110 |
| 3 | 14.80 | 359 | 479 | 317 (100) | Myricetin-hexoside C | 4.42 ± 0.03 | 3.6 ± 0.1 | 0.008 |
| 4 | 15.64 | 354 | 655 | 493 (45), 331 (100) | Laricitrin-di-hexoside B | 2.53 ± 0.01 | 1.859 ± 0.003 | 0.016 |
| 5 | 17.67 | 356 | 645 | 493 (100), 331 (16) | Laricitrin-galloyl-hexoside B | 1.444 ±0.001 | 1.466 ± 0.001 | 0.116 |
| 6 | 18.63 | 370 | 493 | 331 (100) | Laricitrin-hexoside B | 28.9 ± 0.1 | 31.5 ± 0.4 | 0.40 |
| 7 | 23.33 | 362 | 493 | 331 (100) | Laricitrin-hexoside B | 3.69 ± 0.03 | 3.20 ± 0.04 | 0.261 |
| 8 | 25.67 | 365 | 493 | 331 (100) | Laricitrin-hexoside B | 3.02 ± 0.03 | 2.328 ± 0.001 | 0.01 |
| 9 | 32.06 | 368 | 331 | 316 (100), 287 (5), 271 (5) | Laricitrin B | 4.1 ± 0.1 | 8.08 ± 0.01 | 0.02 |
| Total Phenolic Acids | 1.23 ± 0.04 | 1.25 ± 0.02 | 0.07 | |||||
| Total Flavonoids | 49.8 ± 0.2 | 53.5 ± 0.5 | 0.131 | |||||
| Total Phenolic Compounds | 51.1 ± 0.2 | 54.8 ± 0.4 | 0.145 | |||||
| Folin-Ciocalteu mg PE/g Extract | DPPH· IC50 (µg/mL) | FRAP µmol Fe2+/g Extract | AchE IC50 (mg/ML) | MAO-A IC50 (mg/mL) | |
|---|---|---|---|---|---|
| Orange cultivar | 77 ± 3 | 21.3 ± 0.9 | 112 ± 3 | 1.13 ± 0.05 | 0.023 ± 0.001 |
| Yellow cultivar | 81 ± 3 | 22.0 ± 1.0 | 78 ± 7 | 1.25 ± 0.07 | 0.024 ± 0.003 |
| t-Studens test p-value | >0.05 | >0.05 | ≤0.05 | >0.05 | >0.05 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moliner, C.; Barros, L.; Dias, M.I.; López, V.; Langa, E.; Ferreira, I.C.F.R.; Gómez-Rincón, C. Edible Flowers of Tagetes erecta L. as Functional Ingredients: Phenolic Composition, Antioxidant and Protective Effects on Caenorhabditis elegans. Nutrients 2018, 10, 2002. https://0-doi-org.brum.beds.ac.uk/10.3390/nu10122002
Moliner C, Barros L, Dias MI, López V, Langa E, Ferreira ICFR, Gómez-Rincón C. Edible Flowers of Tagetes erecta L. as Functional Ingredients: Phenolic Composition, Antioxidant and Protective Effects on Caenorhabditis elegans. Nutrients. 2018; 10(12):2002. https://0-doi-org.brum.beds.ac.uk/10.3390/nu10122002
Chicago/Turabian StyleMoliner, Cristina, Lillian Barros, Maria Inês Dias, Víctor López, Elisa Langa, Isabel C.F.R. Ferreira, and Carlota Gómez-Rincón. 2018. "Edible Flowers of Tagetes erecta L. as Functional Ingredients: Phenolic Composition, Antioxidant and Protective Effects on Caenorhabditis elegans" Nutrients 10, no. 12: 2002. https://0-doi-org.brum.beds.ac.uk/10.3390/nu10122002