Effect of Free Fatty Acids on Inflammatory Gene Expression and Hydrogen Peroxide Production by Ex Vivo Blood Mononuclear Cells
Abstract
:1. Introduction
2. Methods
2.1. Experimental Procedure
2.2. Anthropometric and Clinical Measurements
2.3. PBMCs Purification
2.4. PBMCs Incubation with FA and/or LPS
2.5. RNA Extraction and Real-Time PCR Assay
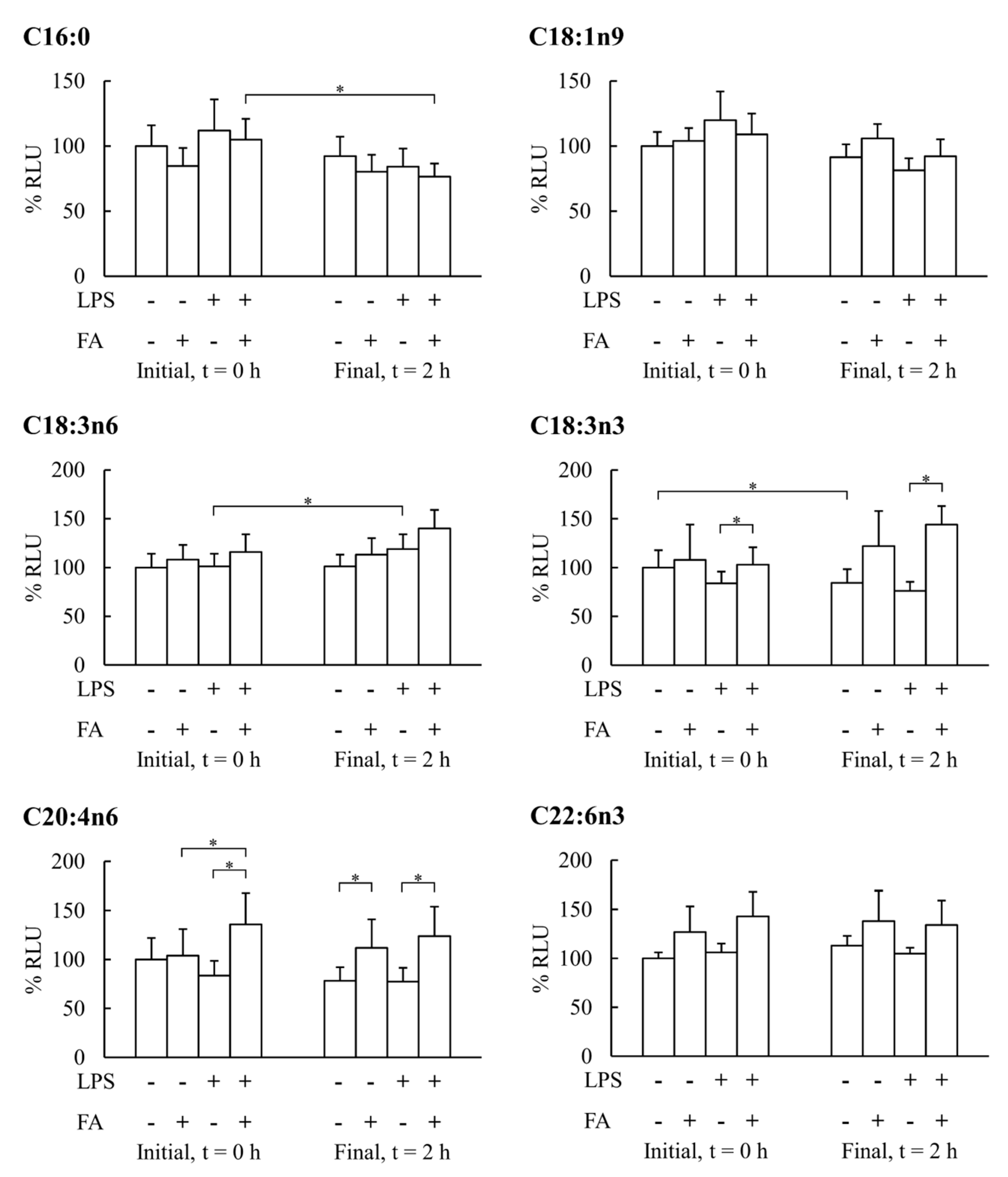
2.6. Hydrogen Peroxide Production
2.7. Statistical Analysis
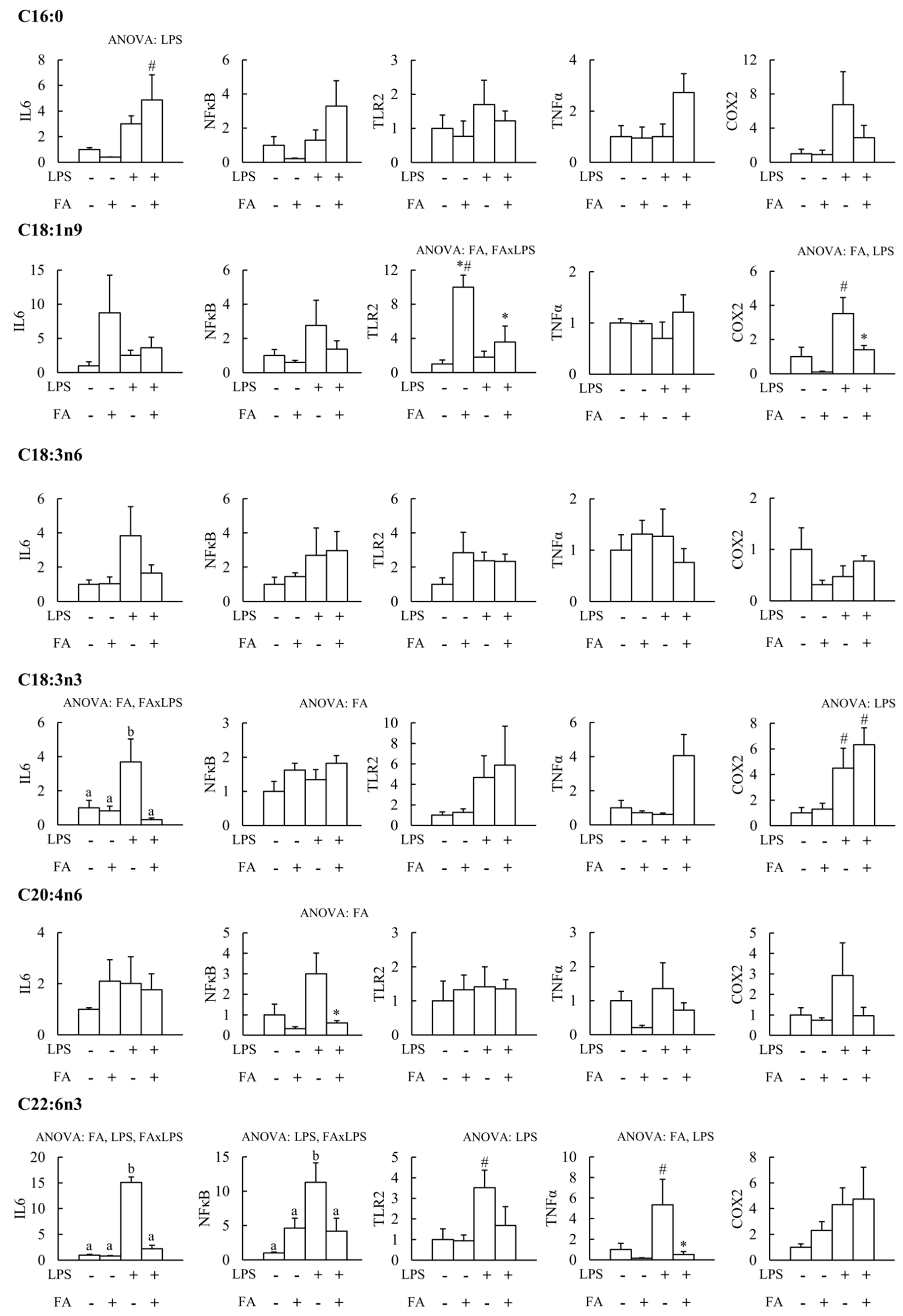
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| COX2 | Cyclooxygenase 2 |
| DAMP | Damage-associated molecular patterns |
| DHA | Docosahexaenoic acid |
| FA | Fatty acids |
| IL | Interleukin |
| LPS | Lipopolysaccharide |
| NFκB | Nuclear factor kappa beta |
| PBMC | Peripheral mononuclear blood cells |
| PPAR | Peroxisome proliferator activated receptors |
| ROS | Reactive oxygen species |
| SPSS | Statistical Package for Social Sciences |
| TLR | Toll-like receptor |
| TNFα | Tumor necrosis factor alpha |
References
- Hofmann, T.; Klenow, S.; Borowicki, A.; Gill, C.I.R.; Pool-Zobel, B.L.; Glei, M. Gene expression profiles in human peripheral blood mononuclear cells as biomarkers for nutritional in vitro and in vivo investigations. Genes Nutr. 2010, 5, 309–319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Mello, V.D.F.; Kolehmanien, M.; Schwab, U.; Pulkkinen, L.; Uusitupa, M. Gene expression of peripheral blood mononuclear cells as a tool in dietary intervention studies: What do we know so far? Mol. Nutr. Food Res. 2012, 56, 1160–1172. [Google Scholar] [CrossRef] [PubMed]
- Liew, C.C.; Ma, J.; Tang, H.C.; Zheng, R.; Dempsey, A.A. The peripheral blood transcriptome dynamically reflects system wide biology: A potential diagnostic tool. J. Lab. Clin. Med. 2006, 147, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Bouwens, M.; Afman, L.A.; Muller, M. Fasting induces changes in peripheral blood mononuclear cell gene expression profiles related to increases in fatty acid beta-oxidation: Functional role of peroxisome proliferator-activated receptor alpha in human peripheral blood mononuclear cells. Am. J. Clin. Nutr. 2007, 86, 1515–1523. [Google Scholar] [CrossRef]
- Riordan, S.M.; Skinner, N.; Nagree, A.; McCallum, H.; McIver, C.J.; Kurtovic, J.; Hamilton, J.A.; Bengmark, S.; Williams, R.; Visvanathan, K. Peripheral blood mononuclear cell expression of toll-like receptors and relation to cytokine levels in cirrhosis. Hepatology 2003, 37, 1154–1164. [Google Scholar] [CrossRef]
- Capo, X.; Martorell, M.; Sureda, A.; Tur, J.A.; Pons, A. Effects of Docosahexaenoic Supplementation and In Vitro Vitamin C on the Oxidative and Inflammatory Neutrophil Response to Activation. Oxidative Med. Cell. Longev. 2015, 2015, 187849. [Google Scholar] [CrossRef] [Green Version]
- Busquets-Cortés, C.; Capó, X.; Martorell, M.; Tur, J.A.; Sureda, A.; Pons, A. Training Enhances Immune Cells Mitochondrial Biosynthesis, Fission, Fusion, and Their Antioxidant Capabilities Synergistically with Dietary Docosahexaenoic Supplementation. Oxid. Medative Cell. Longev. 2016, 2016, 8950384. [Google Scholar] [CrossRef] [Green Version]
- Wallace, F.A.; Miles, E.A.; Calder, P.C. Comparison of the effects of linseed oil and different doses of fish oil on mononuclear cell function in healthy human subjects. Br. J. Nutr. 2003, 89, 679–689. [Google Scholar] [CrossRef]
- Patterson, E.; Wall, R.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. Health implications of high dietary omega-6 polyunsaturated Fatty acids. J. Nutr. Metab. 2012, 2012, 539426. [Google Scholar] [CrossRef]
- Capó, X.; Martorell, M.; Llompart, I.; Sureda, A.; Tur, J.A.; Pons, A. Docosahexanoic acid diet supplementation attenuates the peripheral mononuclear cell inflammatory response to exercise following LPS activation. Cytokine 2014, 69, 155–164. [Google Scholar] [CrossRef]
- Capó, X.; Martorell, M.; Sureda, A.; Tur, J.A.; Pons, A. Effects of dietary docosahexaenoic, training and acute exercise on lipid mediators. J. Int. Soc. Sports Nutr. 2016, 13, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapkin, R.S.; Kim, W.; Lupton, J.R.; McMurray, D.N. Dietary docosahexaenoic and eicosapentaenoic acid: Emerging mediators of inflammation. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 187–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandez-Gonzalo, R.; De Paz, J.A.; Rodriguez-Miguelez, P.; Cuevas, M.J.; González-Gallego, J. Effects of eccentric exercise on toll-like receptor 4 signaling pathway in peripheral blood mononuclear cells. J. Appl. Physiol. 2012, 112, 2011–2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, H.; Lee, S.; Cho, I.H.; Lee, S.J. Toll-Like Receptors: Sensor Molecules for Detecting Damage to the Nervous System. Curr. Protein Pept. Sci. 2013, 14, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Dowling, J.K.; O’Neill, L.A.J. Biochemical regulation of the inflammasome. Crit. Rev. Biochem. Mol. Biol. 2012, 47, 424–443. [Google Scholar] [CrossRef]
- Capó, X.; Martorell, M.; Sureda, A.; Llompart, I.; Tur, J.A.; Pons, A. Diet supplementation with DHA-enriched food in football players during training season enhances the mitochondrial antioxidant capabilities in blood mononuclear cells. Eur. J. Nutr. 2015, 54, 35–49. [Google Scholar] [CrossRef]
- Capó, X.; Martorell, M.; Sureda, A.; Batle, J.M.; Tur, J.A.; Pons, A. Docosahexaenoic diet supplementation, exercise and temperature affect cytokine production by lipopolysaccharide-stimulated mononuclear cells. J. Physiol. Biochem. 2016, 72, 421–434. [Google Scholar] [CrossRef]
- Endres, S.; Ghorbani, R.; Kelley, V.E.; Georgilis, K.; Lonnemann, G.; Van Der Meer, J.W.M.; Cannon, J.G.; Rogers, T.S.; Klempner, M.S.; Weber, P.C.; et al. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 1989, 320, 265–271. [Google Scholar] [CrossRef] [Green Version]
- Hassan, A.; Ibrahim, A.; Mbodji, K.; Coeffier, M.; Ziegler, F.; Bounoure, F.; Chardigny, J.-M.; Skiba, M.; Savoye, G.; D’echelotte, P.; et al. An alpha-linolenic acid-rich formula reduces oxidative stress and inflammation by regulating NF-kappaB in rats with TNBS-induced colitis. J. Nutr. 2010, 140, 1714–1721. [Google Scholar] [CrossRef] [Green Version]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2015, 1851, 469–484. [Google Scholar] [CrossRef]
- Cifre, M.; Díaz-Rúa, R.; Varela-Calviño, R.; Reynés, B.; Pericás-Beltrán, J.; Palou, A.; Oliver, P. Human peripheral blood mononuclear cell in vitro system to test the efficacy of food bioactive compounds: Effects of polyunsaturated fatty acids and their relation with BMI. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Yaqub, S.; Solhaug, V.; Vang, T.; Solberg, R.; Aasen, A.; Taskén, K.; Wang, J.E. A human whole blood model of LPS-mediated suppression of T cell activation. Med. Sci. Monit. 2003, 9, BR120–BR126. [Google Scholar] [PubMed]
- Jialal, I.; Kaur, H.; Devaraj, S. Toll-like receptor status in obesity and metabolic syndrome: A translational perspective. J. Clin. Endocrinol. Metab. 2014, 99, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Farinha, J.B.; Steckling, F.M.; Stefanello, S.T.; Cardoso, M.S.; Nunes, L.S.; Barcelos, R.P.; Duarte, T.; Kretzmann, N.A.; Mota, C.B.; Bresciani, G.; et al. Response of oxidative stress and inflammatory biomarkers to a 12-week aerobic exercise training in women with metabolic syndrome. Sports Med. Open 2015, 1, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyum, A. Separation of White Blood Cells. Nature 1964, 204, 793–794. [Google Scholar] [CrossRef] [PubMed]
- De Pablo, M.A.; De Cienfuegos, G.A. Modulatory effects of dietary lipids on immune system functions. Immunol. Cell Biol. 2000, 78, 31–39. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 polyunsaturated fatty acids and inflammation: From molecular biology to the clinic. Lipids 2003, 38, 343–352. [Google Scholar] [CrossRef]
- Sales-Campos, H.; De Souza, P.R.; Crema Peghini, B.; Santana da Silva, J.; Ribeiro Cardoso, C. An Overview of the Modulatory Effects of Oleic Acid in Health and Disease. Mini-Rev. Med. Chem. 2013, 13, 201–210. [Google Scholar]
- Passos, M.E.; Alves, H.H.; Momesso, C.M.; Faria, F.G.; Murata, G.; Cury-Boaventura, M.F.; Hatanaka, E.; Massao-Hirabara, S.; Gorjao, R. Differential effects of palmitoleic acid on human lymphocyte proliferation and function. Lipids Health Dis. 2016, 15, 217. [Google Scholar] [CrossRef] [Green Version]
- Radler, U.; Stangl, H.; Lechner, S.; Lienbacher, G.; Krepp, R.; Zeller, E.; Brachinger, M.; Eller-Berndl, D.; Fischer, A.; Anzur, C.; et al. A combination of (ω-3) polyunsaturated fatty acids, polyphenols and L-carnitine reduces the plasma lipid levels and increases the expression of genes involved in fatty acid oxidation in human peripheral blood mononuclear cells and HepG2 cells. Ann. Nutr. Metab 2011, 58, 133–140. [Google Scholar] [CrossRef]
- Capó, X.; Martorell, M.; Busquets-Cortés, C.; Sureda, A.; Riera, J.; Drobnic, F.; Tur, J.A.; Pons, A. Effects of dietary almond- and olive oil-based docosahexaenoic acid- and vitamin E-enriched beverage supplementation on athletic performance and oxidative stress markers. Food Funct. 2016, 7, 4920–4934. [Google Scholar] [CrossRef] [PubMed]
- Capó, X.; Martorell, M.; Sureda, A.; Riera, J.; Drobnic, F.; Tur, J.A.; Pons, A. Effects of Almond- and Olive Oil-Based Docosahexaenoic- and Vitamin E-Enriched Beverage Dietary Supplementation on Inflammation Associated to Exercise and Age. Nutrients 2016, 8, 619. [Google Scholar] [CrossRef] [PubMed]
- Polus, A.; Zapala, B.; Razny, U.; Gielicz, A.; Kiec-Wilk, B.; Malczewska-Malec, M.; Sanak, M.; Childs, C.E.; Calder, P.C.; Dembinska-Kiec, A. Omega 3 fatty acid supplementation influences the whole blood transcriptome in women with obesity, associated with pro-resolving lipid mediator production. Biochim. Biophys. Acta 2016, 1861, 1746–1755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocha, D.M.; Caldas, A.P.; Oliveira, L.L.; Bressan, J.; Hermsdorff, H.H. Saturated fatty acids trigger TLR4-mediated inflammatory response. Atherosclerosis 2016, 244, 211–215. [Google Scholar] [CrossRef]
- Guo, X.; Li, H.; Xu, H.; Halim, V.; Zhang, W.; Wang, H.; Ong, K.T.; Woo, S.-L.; Walzem, R.L.; Mashek, D.G.; et al. Palmitoleate induces hepatic steatosis but suppresses liver inflammatory response in mice. PLoS ONE 2012, 7, e39286. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Pahl, H.L. Activators and target genes of Rel/NF-kappa B transcription factors. Oncogene 1999, 18, 6853–6866. [Google Scholar] [CrossRef] [Green Version]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [Green Version]
- Scheller, J.; Rose-John, S. Interleukin-6 and its receptor: From bench to bedside. Med. Microbiol. Immunol. 2006, 195, 173–183. [Google Scholar] [CrossRef]
- Baggiolini, M. Chemokines in pathology and medicine. J. Int. Med. 2001, 250, 91–104. [Google Scholar] [CrossRef]
- Alonso, M.; Collado, P.S.; González-Gallego, J. Melatonin inhibits the expression of the inducible isoform of nitric oxide synthase and nuclear factor kappa B activation in rat skeletal muscle. J. Pineal Res. 2006, 41, 8–14. [Google Scholar] [CrossRef]
- Akira, S.; Uematsu, S.; Takeuchi, O. Pathogen recognition and innate immunity. Cell 2006, 124, 783–801. [Google Scholar] [CrossRef] [Green Version]
- Nahid, M.A.; Yao, B.; Dominguez-Gutierrez, P.R.; Kesavalu, L.; Satoh, M.; Chan, E.K. Regulation of TLR2-mediated tolerance and cross-tolerance through IRAK4 modulation by miR-132 and miR-212. J. Immunol. 2013, 19, 1250–1263. [Google Scholar] [CrossRef] [Green Version]
- Cook, D.N.; Pisetsky, D.S.; Schwartz, D.A. Toll-like receptors in the pathogenesis of human disease. Nat. Immunol. 2004, 5, 975–979. [Google Scholar] [CrossRef]
- Adkins, Y.; Kelley, D.S. Mechanisms underlying the cardioprotective effects of omega-3 polyunsaturated fatty acids. J. Nutr. Biochem. 2010, 21, 781–792. [Google Scholar] [CrossRef]
- Hernández, J.C.; Arteaga, J.; Paul, S.; Kumar, A.; Latz, E.; Urcuqui-Inchima, S. Up-regulation of TLR2 and TLR4 in dendritic cells in response to HIV type 1 and coinfection with opportunistic pathogens. Aids Res. Hum. Retrovir. 2011, 27, 1099–1109. [Google Scholar] [CrossRef] [Green Version]
- Mortazavi, S.H.; Amin, R.; Alyasin, S.; Kashef, S.; Karimi, M.H.; Babaei, M.; Younesi, V. Down-regulation of TLR2, 3, 9 and Signaling Mediators, MyD88 and TRIF, Gene Transcript Levels in Patients with Kawasaki Disease Treated with IVIG. Iran. J. Allergy Asthma Immunol. 2015, 14, 188–197. [Google Scholar]
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and inflammation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 986–1000. [Google Scholar] [CrossRef]
- Gorjao, R.; Cury-Boaventura, M.F.; De Lima, T.M.; Curi, R. Regulation of human lymphocyte proliferation by fatty acids. Cell. Biochem. Funct. 2007, 25, 305–315. [Google Scholar] [CrossRef]
- Rudolph, V.; Schopfer, F.J.; Khoo, N.K.H.; Rudolph, T.K.; Cole, M.P.; Woodcock, S.R.; Bonacci, G.; Groeger, A.L.; Golin-Bisello, F.; Chen, C.-S.; et al. Nitro-fatty Acid Metabolome: Saturation, Desaturation, beta-Oxidation, and Protein Adduction. J. Biol. Chem. 2009, 284, 1461–1473. [Google Scholar] [CrossRef] [Green Version]
- Cui, T.; Schopfer, F.J.; Zhang, J.; Chen, K.; Ichikawa, T.; Baker, P.R.S.; Batthyany, C.; Chacko, B.K.; Feng, X.; Patel, R.P.; et al. Nitrated fatty acids: Endogenous anti-inflammatory signaling mediators. J. Biol. Chem. 2006, 281, 35686–35698. [Google Scholar] [CrossRef] [Green Version]
- Dubuquoy, L.; Rousseaux, C.; Thuru, X.; Peyrin, L.; Romano, O.; Chavatte, P.; Chamaillard, M.; Desreumaux, P. PPAR gamma as a new therapeutic in inflammatory bowel diseases. Gut 2006, 55, 1341–1349. [Google Scholar] [CrossRef] [Green Version]
- Gong, J.; Campos, H.; McGarvey, S.; Wu, Z.; Goldberg, R.; Baylin, A. Adipose tissue palmitoleic acid and obesity in humans: Does it behave as a lipokine? Am. J. Clin. Nutr. 2011, 93, 186–191. [Google Scholar] [CrossRef] [Green Version]
- Emre, Y.; Hurtaud, C.; Nübel, T.; Criscuolo, F.; Ricquier, D.; Cassard-Doulcier, A.-M. Mitochondria contribute to LPS-induced MAPK activation via uncoupling protein UCP2 in macrophages. Biochem. J. 2007, 402, 271–278. [Google Scholar] [CrossRef] [Green Version]
- Asehnoune, K.; Strassheim, D.; Mitra, S.; Kim, J.Y.; Abraham, E. Involvement of reactive oxygen species in Toll-like receptor 4-dependent activation of NF-kappa B. J. Immunol. 2004, 172, 2522–2529. [Google Scholar] [CrossRef] [Green Version]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef] [Green Version]
- Busquets-Cortés, C.; Capó, X.; Argelich, E.; Ferrer, M.D.; Mateos, D.; Bouzas, C.; Abbate, M.; Tur, J.A.; Sureda, A.; Pons, A. Effects of Millimolar Steady-State Hydrogen Peroxide Exposure on Inflammatory and Redox Gene Expression in Immune Cells from Humans with Metabolic Syndrome. Nutrients 2018, 10, 1920. [Google Scholar] [CrossRef] [Green Version]
- Martorell, M.; Capó, X.; Sureda, A.; Batle, J.M.; Llompart, I.; Argelich, E.; Tur, J.A.; Pons, A. Effect of DHA on plasma fatty acid availability and oxidative stress during training season and football exercise. Food Funct. 2014, 5, 1920–1931. [Google Scholar] [CrossRef]
- Martorell, M.; Capó, X.; Sureda, A.; Tur, J.A.; Pons, A. Chromatographic and Enzymatic Method to Quantify Individual Plasma Free and Triacylglycerol Fatty Acids. Chromatographica 2015, 78, 259–266. [Google Scholar] [CrossRef]
- Villa, P.M.; Laivuori, H.; Kajantie, E.; Kaaja, R. Free fatty acid profiles in preeclampsia. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 17–21. [Google Scholar] [CrossRef]
- Lv, W.; Yang, T. Identification of possible biomarkers for breast cancer from free fatty acid profiles determined by GC-MS and multivariate statistical analysis. Clin. Biochem. 2012, 45, 127–133. [Google Scholar] [CrossRef]
- Huber, A.H.; Kleinfeld, A.M. Unbound free fatty acid profiles in human plasma and the unexpected absence of unbound palmitoleate. J. Lipid Res. 2017, 58, 578–585. [Google Scholar] [CrossRef] [Green Version]
| Gene | Primer | Conditions | ||
|---|---|---|---|---|
| 18S | Fw: | 5′-ATGTGAAGTCACTGTGCCAG-3′ | 95 °C | 10 s |
| Rv: | 5′-GTGTAATCCGTCTCCACAGA-3′ | 60 °C | 10 s | |
| 72 °C | 12 s | |||
| IL6 | Fw: | 5′-TACATCCTCGACGGCATCTC-3′ | 95 °C | 10 s |
| Rv: | 5′-ACTCATCTGCACAGCTCTGG-3′ | 63 °C | 10 s | |
| 72 °C | 12 s | |||
| NFκβ | Fw: | 5′-AAACACTGTGAGGATGGGATCTG-3′ | 95 °C | 10 s |
| Rv: | 5′-CGAAGCCGACCACCATGT-3′ | 60 °C | 10 s | |
| 72 °C | 15 s | |||
| TLR2 | Fw: | 5′-GGGTTGAAGCACTGGACAAT-3′ | 95 °C | 10 s |
| Rv: | 5′-TTCTTCCTTGGAGAGGCTGA-3′ | 60 °C | 10 s | |
| 72 °C | 15 s | |||
| TNFα | Fw: | 5′-CCCAGGCAGTCAGATCATCTTCTCGGAA-3′ | 94 °C | 10 s |
| Rv: | 5′-CTGGTTATCTCTCAGCTCCACGCCATT-3′ | 63 °C | 10 s | |
| 72 °C | 15 s | |||
| COX2 | Fw: | 5′-TTGCTGGCAGGGTTGCTGGTGGTA-3′ | 95 °C | 10 s |
| Rv: | 5′-CATCTGCCTGCTCTGGTCAATGGA A-3′ | 67 °C | 10 s | |
| 72 °C | 15 s | |||
| Weight (kg) | 92.8 ± 2.8 |
| Height (cm) | 165 ± 2 |
| Body Mass Index (BMI, kg/m2) | 34.0 ± 0.6 |
| Waist circumference (cm) | 114 ± 2 |
| Hip circumference (cm) | 116 ± 2 |
| Systolic blood pressure (mmHg) | 147 ± 3 |
| Diastolic blood pressure (mmHg) | 82.5 ± 1.9 |
| Glucose (mg/dL) | 127 ± 11 |
| Total cholesterol (mg/dL) | 186 ± 8 |
| HDL cholesterol (mg/dL) | 41.5 ± 2.2 |
| LDL cholesterol (mg/dL) | 110 ± 7 |
| Triglycerides (mg/dL) | 185 ± 20 |
| Erythrocytes (106 cells/µL) | 4.85 ± 0.11 |
| Haemoglobin (Hb, g/dL) | 14.5 ± 0.3 |
| Haematocrit (%) | 43.4 ± 0.9 |
| Mean corpuscular volume (MCV, fL) | 89.8 ± 1.3 |
| Leukocytes (103 cells/µL) | 7.56 ± 0.34 |
| Neutrophils (103 cells/µL) | 4.26 ± 0.29 |
| Lymphocytes (103 cells/µL) | 2.41 ± 0.16 |
| Monocytes (103 cells/µL) | 0.643 ± 0.031 |
| Eosinophils (103 cells/µL) | 0.220 ± 0.020 |
| Basophils (103 cells/µL) | 0.051 ± 0.005 |
| PBMCs (103 cells/µL) | 3.32 ± 0.18 |
| Platelet count (103 cells/µL) | 236 ± 10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sureda, A.; Martorell, M.; Bibiloni, M.d.M.; Bouzas, C.; Gallardo-Alfaro, L.; Mateos, D.; Capó, X.; Tur, J.A.; Pons, A. Effect of Free Fatty Acids on Inflammatory Gene Expression and Hydrogen Peroxide Production by Ex Vivo Blood Mononuclear Cells. Nutrients 2020, 12, 146. https://0-doi-org.brum.beds.ac.uk/10.3390/nu12010146
Sureda A, Martorell M, Bibiloni MdM, Bouzas C, Gallardo-Alfaro L, Mateos D, Capó X, Tur JA, Pons A. Effect of Free Fatty Acids on Inflammatory Gene Expression and Hydrogen Peroxide Production by Ex Vivo Blood Mononuclear Cells. Nutrients. 2020; 12(1):146. https://0-doi-org.brum.beds.ac.uk/10.3390/nu12010146
Chicago/Turabian StyleSureda, Antoni, Miquel Martorell, Maria del Mar Bibiloni, Cristina Bouzas, Laura Gallardo-Alfaro, David Mateos, Xavier Capó, Josep A. Tur, and Antoni Pons. 2020. "Effect of Free Fatty Acids on Inflammatory Gene Expression and Hydrogen Peroxide Production by Ex Vivo Blood Mononuclear Cells" Nutrients 12, no. 1: 146. https://0-doi-org.brum.beds.ac.uk/10.3390/nu12010146





