Lactobacillus Adhesion to Mucus
Abstract
:1. Introduction
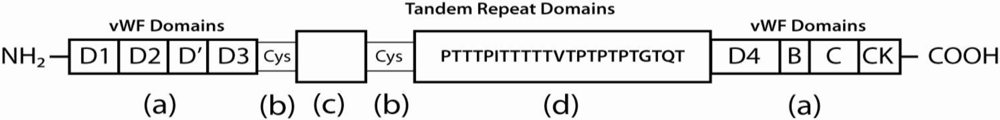
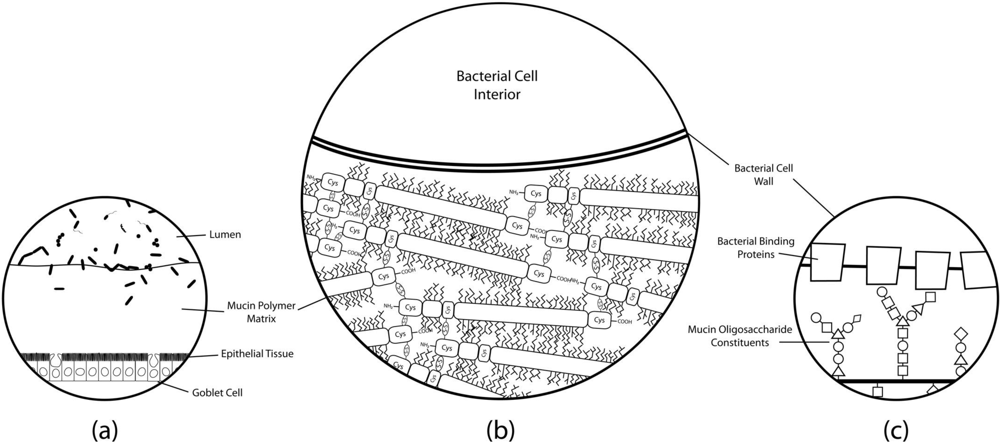
2. Intestinal Mucus
| Gene | Organisms with known homologues 1 | Function 2 | GeneAtlas location of highest expression 2 | Type | Selected references |
|---|---|---|---|---|---|
| MUC1 | Dog, cow, mouse, rat, rabbit | Cellular signal transduction, barrier activity | Lungs | Membrane | [13,14] |
| MUC2 | Chimpanzee, dog, chicken | Primary extracellular matrix constituent in colon, lubricant activity | Colon | Secretory | [14,15] |
| MUC3A | Rat, mouse | Involved in epithelial cell protection, adhesion modulation, and signaling | Various | Membrane | [16] |
| MUC3B | Rat, mouse | Unknown, possibly cellular signal transduction | Various | Membrane | [16] |
| MUC4 | Many mammals, chicken, frog, platypus | Involved in intestinal epithelial cell differentiation, renewal, lubrication | Colon | Membrane | [17,18] |
| MUC5B (MUC9) | Chimpanzee, zebrafish, mouse, chicken, more | Unknown, primarily lubricant | Various | Secretory | [19,20] |
| MUC5AC | Chimpanzee, rat, zebrafish | Major component of airway mucus involved in intestinal epithelial cell differentiation | Trachea, Lungs | Secretory | [21,22] |
| MUC6 | Chimpanzee, dog, mouse, chicken | Unknown, involved in renal morphogenesis processes | Pancreas, digestive and reproductive systems | Secretory | [22,23,24] |
| MUC7 | Chimpanzee, cow, rat | Facilitating the clearance of oral bacteria | Salivary Gland | Secretory | [25,26] |
| MUC8 | Unknown | Unknown | Trachea | Secretory | [27] |
| MUC12 (MUC11) | Cow, M.grisea, N. crassa, rice | May be involved in epithelial cell regulation | Colon | Membrane | [28] |
| MUC13 | Chimpanzee, dog, mouse, rat | Barrier function in epithelial tissues | Pancreas, small intestine, colon | Membrane | [29] |
| EMCN (MUC14) | Dog, cow, mouse, rat, chicken | Interferes with the assembly of focal adhesion complexes | Fetal lung, uterus, thyroid | Membrane | [30] |
| MUC15 | Chimpanzee, cow, mouse, rat | Barrier function in epithelial tissues | Testis leydig cell | Membrane | [31] |
| MUC16 (CA125) | Chimpanzee, dog, mouse, chicken | Unknown, plays a role in ovarian cancer | Lymph nodes, respiratory tract | Membrane | [32,33] |
| MUC17 (MUC3) | Chimpanzee, S. pombe, S. cerevisiae, and K. lactis | Extracellular matrix constituent, lubricant activity | Small intestine, stomach | Membrane | [34,35] |
| MCAM (MUC18, CD146) | Chimpanzee, dog, mouse, rat, zebrafish | AKA “melanoma cell adhesion molecule”, cell-cell adhesion | Various | Membrane | [36,37] |
| MUC19 | Chimpanzee, dog, mouse, rat, frog | Major gel-forming mucin in the human middle ear | Secretory cells of the ears and eyes | Secretory | [38] |
| MUC20 | Chimpanzee, dog, cow, mouse, rat | Cellular signal transduction | Intestine, respiratory and urinary tract | Membrane | [39] |
| MUC21 | Chimpanzee, cow, mosquito, and A. thaliana | Unknown, mediates cell adhesion | Unknown | Membrane | [40,41] |
| CD164 (MUC24) | Chimpanzee, dog, cow, mouse, rat, chicken, zebrafish | Regulates stem cell localization to the bone marrow | Thyroid, placenta, intestine, immune cells | Membrane | [42] |
Mucin Genes and Modifications
3. Adhesion
3.1. Mucus Binding Proteins
| Protein | Info. | Species | References |
|---|---|---|---|
| MUB | Demonstrates binding to mucus in vitro | L. reuteri | [95] |
| MucBP Domain Containing Proteins | Contain MucBP domains, implicated in mucus adhesion | 13 known Lactobacillus spp. | [98] |
| Pili | Pilin subunit SpaC binds to mucus in vitro | L. johnsonii, L. rhamnosus | [99,100,101] |
| 32-Mmubp | Demonstrates binding to mucus in vitro | L. fermentum | [102] |
| SlpA | Knockouts show diminished adhesion to mucus in vitro | L. acidophilus | [103] |
| Msa | Demonstrates binding of mannose in vitro | L. plantarum | [104] |
| MapA | Demonstrates binding to mucus in vitro | L. reuteri | [105,106] |
| EF-Tu | Expression upregulated in the presence of mucus | L. johnsonii | [107,108,109,110,111] |
| Currently available whole genomes | Accession# | Gene | # of domains | Size |
|---|---|---|---|---|
| Lactobacillus acidophilus NCFM | Q5FKK8 | LBA0909 | 1 | 508aa |
| Q5FKA8 | LBA1017 | 1 | 294aa | |
| Q5FKA7 | LBA1018 | 1 | 346aa | |
| Q5FKA6 | LBA1019 | 7 | 2650aa | |
| Q5FKA5 | LBA1020 | 5 | 2310aa | |
| Q5FJS1 | LBA1218 | 1 | 697aa | |
| Q5FJC2 | LBA1377 | 2 | 1017aa | |
| Q5FJA7 | LBA1392 | 17 | 4326aa | |
| Q5FJ43 | LBA1460 | 2 | 339aa | |
| Q5FIQ0 | LBA1609 | 2 | 643aa | |
| Q5FIL0 | LBA1652 | 3 | 1174aa | |
| Q5FIF3 | LBA1709 | 3 | 1208aa | |
| Lactobacillus brevis ATCC 367 | Q03U29 | LVIS_0122 | 2 | 912aa |
| Q03T21 | LVIS_0493 | 3 | 1519aa | |
| Q03P66 | LVIS_1947 | 1 | 1111aa | |
| Q03NB2 | LVIS_2262 | 1 | 422aa | |
| Lactobacillus crispatus ST1 | D5H0E1 | LCRIS_00029 | 3 | 1232aa |
| D5H2Y1 | LCRIS_00919 | 7 | 2935aa | |
| D5GXR1 | LCRIS_01123 | 1 | 304aa | |
| D5GZ92 | LCRIS_01654 | 2 | 3552aa | |
| Lactobacillus fermentum IFO 3956 | B2GFA4 | LAF_0157 | 1 | 208aa |
| B2GBH7 | LAF_0673 | 2 | 1059aa | |
| Lactobacillus gasseri ATCC 33323 | Q047B3 | LGAS_0044 | 4 | 873aa |
| Q047B2 | LGAS_0045 | 11 | 3692aa | |
| Q047B1 | LGAS_0046 | 4 | 985aa | |
| Q046R7 | LGAS_0143 | 6 | 2823aa | |
| Q045Q7 | LGAS_0410 | 5 | 2457aa | |
| Q043P5 | LGAS_0939 | 2 | 615aa | |
| Q043P2 | LGAS_0942 | 10 | 2833aa | |
| Q043P0 | LGAS_0944 | 1 | 524aa | |
| Q041C4 | LGAS_1655 | 2 | 1425aa | |
| Q041B7 | LGAS_1663 | 6 | 2449aa | |
| Q041A9 | LGAS_1671 | 4 | 2552aa | |
| Q040V9 | LGAS_1725 | 6 | 1993aa | |
| Lactobacillus helveticus DPC 4571 | A8YTV1 | lhv_0494 | 1 | 155aa |
| A8YTV2 | lhv_0495 | 1 | 178aa | |
| A8YUX0 | lhv_0973 | 1 | 278aa | |
| A8YUX3 | lhv_0979 | 1 | 858aa | |
| Lactobacillus johnsonii FI9785 | D0R4C3 | FI9785_1070 | 6 | 3401aa |
| D0R5H6 | FI9785_1482 | 5 | 1356aa | |
| Lactobacillus johnsonii NCC 533 | Q74LY7 | LJ_0046 | 4 | 870aa |
| Q74LY6 | LJ_0047 | 6 | 2139aa | |
| Q74LY5 | LJ_0048 | 4 | 983aa | |
| Q74L43 | LJ_0382 | 4 | 3619aa | |
| Q74KU3 | LJ_0484 | 4 | 4037aa | |
| Q74HP3 | LJ_0574 | 5 | 1571aa | |
| Q74HU0 | LJ_0621 | 5 | 2789aa | |
| Q74HW0 | LJ_0641 | 3 | 1563aa | |
| Q74HA8 | LJ_1839 | 7 | 1814aa | |
| Lactobacillus plantarum JDM1 | C6VP10 | JDM1_1038 | 4 | 1082aa |
| C6VQ03 | JDM1_1381 | 6 | 2219aa | |
| C6VKM3 | JDM1_2438 | 4 | 1345aa | |
| C6VL52 | JDM1_2491 | 4 | 2037aa | |
| C6VL55 | JDM1_2494 | 1 | 750aa | |
| Lactobacillus plantarum WCFS1 | Q88Y49 | lp_0946 | 1 | 1189aa |
| Q88XH5 | lp_1229 | 3 | 1010aa | |
| Q88WI9 | lp_1643 | 6 | 2219aa | |
| Q88UJ0 | lp_2486 | 2 | 917aa | |
| Q88TB8 | lp_3059 | 4 | 1356aa | |
| Q88T70 | lp_3114 | 4 | 2032aa | |
| Q88T67 | lp_3117 | 1 | 750aa | |
| Lactobacillus reuteri DSM 20016 | A5VKZ1 | Lreu_1258 | 1 | 745aa |
| Lactobacillus reuteri JCM 1112 | B2G8C6 | LAR_1192 | 1 | 745aa |
| Lactobacillus salivarius CECT 5713 | D8IM74 | HN6_01114 | 4 | 785aa |
| Lactobacillus salivarius UCC118 | Q1WSI9 | LSL_1335 | 4 | 785aa |
3.2. Factors that Influence Binding in Vivo and in Vitro
4. In Vitro Models
| Model | Description | Advantages | Disadvantages | References |
|---|---|---|---|---|
| Immobilized mucus | Mucus preparations immobilized, usually in microtitre wells | Fast, isolates mucus-microbe interactions from other in vivo conditions | Difficult to separate mucus-specific from hydrophobic interactions | [91,131,132,133] |
| Cell culture | Polar monolayer of enterocytes resembling intestinal tissue | Provides conditions more similar to in vivo environment | Derived from cancer cells, could differ from healthy tissue. Not representative of cell-type ratios in mucosal epithelial tissues | |
| Caco-2/HT29 | Caco-2 and HT29 carcinoma cell lines | Simple, well established in literature | Does not account for mucus presence | [134,135,136,137] |
| HT29-MTX/FU | HT29 culture treated with methotrexate or fluoruracil to secret mucus of different types | Accounts for presence of mucus | May not represent appropriate MUC gene expression | [138,139,140,141,142,143,144] |
| Co-cultures | Mixed culture of secreting and mucus-secreting cells | Better represents cell-type ratio of mucosal epithelial tissues | Little literature for use in adhesion studies | [145,146,147] |
| Whole tissue | Whole, intact or excised tissue | Provides in vitro conditions most similar to in vivo environment | Costly, difficult to obtain | |
| Resected tissue | Fragments of tissue excised from host | Mucus, epithelial tissue, and commensal organisms accounted for in model | Only small fragments at a time available from living hosts | [148,149] |
| Organ culture | Whole organs maintained in vitro | Better maintains the architecture of the tissue | Prohibitively expensive, may not function in same manner as in vivo | [150,151] |
4.1. Mucus Adhesion Models
4.2. Cell-Culture Models
4.3. Whole Tissue Models
5. Conclusion
Acknowledgements
References
- Ouwehand, A.C.; Salminen, S.; Isolauri, E. Probiotics: An overview of beneficial effects. Antonie van Leeuwenhoek 2002, 82, 279–289. [Google Scholar]
- Matsuo, K.; Ota, H.; Akamatsu, T.; Sugiyama, A.; Katsuyama, T. Histochemistry of the surface mucous gel layer of the human colon. Gut 1997, 40, 782–789. [Google Scholar]
- Swidsinski, A.; Loening-Baucke, V.; Theissig, F.; Engelhardt, H.; Bengmark, S.; Koch, S.; Lochs, H.; Dörffel, Y. Comparative study of the intestinal mucus barrier in normal and inflamed colon. Gut 2007, 56, 343–350. [Google Scholar]
- Maassen, C.B.; van Holten-Neelen, C.; Balk, F.; den Bak-Glashouwer, M.J.; Leer, R.J.; Laman, J.D.; Boersma, W.J.; Claassen, E. Strain-dependent induction of cytokine profiles in the gut by orally administered Lactobacillus strains. Vaccine 2000, 18, 2613–2623. [Google Scholar]
- Deplancke, B.; Gaskins, H.R. Microbial modulation of innate defense: Goblet cells and the intestinal mucus layer. Am. J. Clin. Nutr. 2001, 73, 1131S–1141S. [Google Scholar]
- Valeur, N.; Engel, P.; Carbajal, N.; Connolly, E.; Ladefoged, K. Colonization and immunomodulation by Lactobacillus reuteri ATCC 55730 in the human gastrointestinal tract. Appl. Environ. Microbiol. 2004, 70, 1176–1181. [Google Scholar]
- Weiss, G.; Jespersen, L. Transcriptional analysis of genes associated with stress and adhesion in Lactobacillus acidophilus NCFM during the passage through an in vitro gastrointestinal tract model. J. Mol. Microbiol. Biotechnol. 2010, 18, 206–214. [Google Scholar]
- Blum, S. Adhesion studies for probiotics: Need for validation and refinement. Trends Food Sci. Technol. 1999, 10, 405–410. [Google Scholar]
- Fuller, R. Probiotics in man and animals. J. Appl. Bacteriol. 1989, 66, 365–378. [Google Scholar]
- Laboisse, C.; Jarry, A.; Branka, J.E.; Merlin, D.; Bou-Hanna, C.; Vallette, G. Recent aspects of the regulation of intestinal mucus secretion. Proc. Nutr. Soc. 1996, 55, 259–264. [Google Scholar]
- Akiba, Y.; Guth, P.H.; Engel, E.; Nastaskin, I.; Kaunitz, J.D. Dynamic regulation of mucus gel thickness in rat duodenum. Am. J. Physiol. Gastrointest. Liver Physiol. 2000, 279, G437–G447. [Google Scholar]
- Byrd, J.C.; Bresalier, R.S. Mucins and mucin binding proteins in colorectal cancer. Cancer Metastasis Rev. 2004, 23, 77–99. [Google Scholar]
- Gendler, S.J. MUC1, the renaissance molecule. J. Mammary Gland Biol. Neoplasia 2001, 6, 339–353. [Google Scholar]
- Chambers, J.A.; Hollingsworth, M.A.; Trezise, A.E.; Harris, A. Developmental expression of mucin genes MUC1 and MUC2. J. Cell Sci. 1994, 107, 413–424. [Google Scholar]
- Allen, A.; Hutton, D.A.; Pearson, J.P. The MUC2 gene product: A human intestinal mucin. Int. J. Biochem. Cell Biol. 1998, 30, 797–801. [Google Scholar]
- Pratt, W.S.; Crawley, S.; Hicks, J.; Ho, J.; Nash, M.; Kim, Y.S.; Gum, J.R.; Swallow, D.M. Multiple transcripts of MUC3: Evidence for two genes, MUC3A and MUC3B. Biochem. Biophys. Res. Commun. 2000, 275, 916–923. [Google Scholar]
- Carraway, K.L.; Perez, A.; Idris, N.; Jepson, S.; Arango, M.; Komatsu, M.; Haq, B.; Price-Schiavi, S.A.; Zhang, J.; Carraway, C.A. Muc4/sialomucin complex, the intramembrane ErbB2 ligand, in cancer and epithelia: To protect and to surviv. Prog. Nucleic Acid Res. Mol. Biol. 2002, 71, 149–185. [Google Scholar]
- Porchet, N.; Nguyen, V.C.; Dufosse, J.; Audie, J.P.; Guyonnet-Duperat, V.; Gross, M.S.; Denis, C.; Degand, P.; Bernheim, A.; Aubert, J.P. Molecular cloning and chromosomal localization of a novel human tracheo-bronchial mucin cDNA containing tandemly repeated sequences of 48 base Pairs. Biochem. Biophys. Res. Commun. 1991, 175, 414–422. [Google Scholar]
- Desseyn, J.L.; Guyonnet-Dupérat, V.; Porchet, N.; Aubert, J.P.; Laine, A. Human mucin gene MUC5B, the 10.7-Kb large central exon encodes various alternate subdomains resulting in a super-repeat. Structural evidence for a 11p15.5 gene family. J. Biol. Chem. 1997, 272, 3168–3178. [Google Scholar] [PubMed]
- Nielsen, P.A.; Bennett, E.P.; Wandall, H.H.; Therkildsen, M.H.; Hannibal, J.; Clausen, H. Identification of a major human high molecular weight salivary mucin (MG1) as tracheobronchial mucin MUC5B. Glycobiology 1997, 7, 413–419. [Google Scholar]
- Escande, F.; Aubert, J.P.; Porchet, N.; Buisine, M.P. Human mucin gene MUC5AC: Organization of its 5′-region and central repetitive region. Biochem. J. 2001, 358, 763–772. [Google Scholar]
- Nordman, H.; Davies, J.R.; Lindell, G.; de Bolós, C.; Real, F.; Carlstedt, I. Gastric MUC5AC and MUC6 are large oligomeric mucins that differ in size, glycosylation and tissue distribution. Biochem. J. 2002, 364, 191–200. [Google Scholar]
- Toribara, N.W.; Ho, S.B.; Gum, E.; Gum, J.R.; Lau, P.; Kim, Y.S. The carboxyl-terminal sequence of the human secretory mucin, MUC6. Analysis of the primary amino acid sequence. J. Biol. Chem. 1997, 272, 16398–16403. [Google Scholar] [PubMed]
- Bartman, A.E.; Buisine, M.P.; Aubert, J.P.; Niehans, G.A.; Toribara, N.W.; Kim, Y.S.; Kelly, E.J.; Crabtree, J.E.; Ho, S.B. The MUC6 secretory mucin gene is expressed in a wide variety of epithelial tissues. J. Pathol. 1998, 186, 398–405. [Google Scholar]
- Bobek, L.A.; Tsai, H.; Biesbrock, A.R.; Levine, M.J. Molecular cloning, sequence, and specificity of expression of the gene encoding the low molecular weight human salivary mucin (MUC7). J. Biol. Chem. 1993, 268, 20563–20569. [Google Scholar] [PubMed]
- Bobek, L.A.; Liu, J.; Sait, S.N.; Shows, T.B.; Bobek, Y.A.; Levine, M.J. Structure and chromosomal localization of the human salivary mucin gene, MUC7. Genomics 1996, 31, 277–282. [Google Scholar]
- Shankar, V.; Pichan, P.; Eddy, R.L.; Tonk, V.; Nowak, N.; Sait, S.N.; Shows, T.B.; Schultz, R.E.; Gotway, G.; Elkins, R.C.; et al. Chromosomal localization of a human mucin gene (MUC8) and cloning of the cDNA corresponding to the carboxy terminus. Am. J. Respir. Cell Mol. Biol. 1997, 16, 232–241. [Google Scholar]
- Williams, S.J.; McGuckin, M.A.; Gotley, D.C.; Eyre, H.J.; Sutherland, G.R.; Antalis, T.M. Two novel mucin genes down-regulated in colorectal cancer identified by differential display. Cancer Res. 1999, 59, 4083–4089. [Google Scholar]
- Williams, S.J.; Wreschner, D.H.; Tran, M.; Eyre, H.J.; Sutherland, G.R.; McGuckin, M.A. Muc13, a novel human cell surface mucin expressed by epithelial and hemopoietic cells. J. Biol. Chem. 2001, 276, 18327–18336. [Google Scholar]
- Liu, C.; Shao, Z.M.; Zhang, L.; Beatty, P.; Sartippour, M.; Lane, T.; Livingston, E.; Nguyen, M. Human endomucin is an endothelial marker. Biochem. Biophys. Res. Commun. 2001, 288, 129–136. [Google Scholar]
- Pallesen, L.T.; Berglund, L.; Rasmussen, L.K.; Petersen, T.E.; Rasmussen, J.T. Isolation and characterization of MUC15, a novel cell membrane-associated mucin. Eur. J. Biochem. 2002, 269, 2755–2763. [Google Scholar]
- Yin, B.W.T.; Dnistrian, A.; Lloyd, K.O. Ovarian cancer antigen CA125 is encoded by the MUC16 mucin gene. Int. J. Cancer 2002, 98, 737–740. [Google Scholar]
- McLemore, M.R.; Aouizerat, B. Introducing the MUC16 gene: Implications for prevention and early detection in epithelial ovarian cancer. Biol. Res. Nurs. 2005, 6, 262–267. [Google Scholar]
- Gum, J.R.; Crawley, S.C.; Hicks, J.W.; Szymkowski, D.E.; Kim, Y.S. MUC17, a novel membrane-tethered mucin. Biochem. Biophys. Res. Commun. 2002, 291, 466–475. [Google Scholar]
- Moniaux, N.; Junker, W.M.; Singh, A.P.; Jones, A.M.; Batra, S.K. Characterization of human mucin muc17. Complete coding sequence and organization. J. Biol. Chem. 2006, 281, 23676–23685. [Google Scholar]
- Lehmann, J.M.; Riethmüller, G.; Johnson, J.P. MUC18, a marker of tumor progression in human melanoma, shows sequence similarity to the neural cell adhesion molecules of the immunoglobulin superfamily. Proc. Natl. Acad. Sci. USA 1989, 86, 9891–9895. [Google Scholar]
- Johnson, J.P.; Rothbächer, U.; Sers, C. The progression associated antigen MUC18: A unique member of the immunoglobulin supergene family. Melanoma Res. 1993, 3, 337–340. [Google Scholar]
- Chen, Y.; Zhao, Y.H.; Kalaslavadi, T.B.; Hamati, E.; Nehrke, K.; Le, A.D.; Ann, D.K.; Wu, R. Genome-wide search and identification of a novel gel-forming mucin muc19/muc19 in glandular tissues. Am. J. Respir. Cell Mol. Biol. 2004, 30, 155–165. [Google Scholar]
- Higuchi, T.; Orita, T.; Nakanishi, S.; Katsuya, K.; Watanabe, H.; Yamasaki, Y.; Waga, I.; Nanayama, T.; Yamamoto, Y.; Munger, W.; et al. Molecular cloning, genomic structure, and expression analysis of MUC20, a novel mucin protein, up-regulated in injured kidney. J. Biol. Chem. 2004, 279, 1968–1979. [Google Scholar] [PubMed]
- Yi, Y.; Kamata-Sakurai, M.; Denda-Nagai, K.; Itoh, T.; Okada, K.; Ishii-Schrade, K.; Iguchi, A.; Sugiura, D.; Irimura, T. Mucin 21/epiglycanin modulates cell adhesion. J. Biol. Chem. 2010, 285, 21233–21240. [Google Scholar]
- Itoh, Y.; Kamata-Sakurai, M.; Denda-Nagai, K.; Nagai, S.; Tsuiji, M.; Ishii-Schrade, K.; Okada, K.; Goto, A.; Fukayama, M.; Irimura, T. Identification and expression of human epiglycanin/muc21: A novel transmembrane mucin. Glycobiology 2008, 18, 74–83. [Google Scholar]
- Kurosawa, N.; Kanemitsu, Y.; Matsui, T.; Shimada, K.; Ishihama, H.; Muramatsu, T. Genomic analysis of a murine cell-surface sialomucin, MGC-24/CD164. Eur. J. Biochem. 1999, 265, 466–472. [Google Scholar]
- National Center for Biotechnology Information. HomoloGene Home Page. Available online: http://0-www-ncbi-nlm-nih-gov.brum.beds.ac.uk/homologene (accessed on 30 October 2010).
- Université René Descartes-Paris. GENATLAS. Available online: http://genatlas.medecine.univ-paris5.fr (accessed on 30 October 2010).
- Genomics Intstitute of the Nevartis Research Foundation. BioGPS. Available online: http://biogps.gnf.org (accessed on 30 October 2010).
- Hanisch, F.G. O-Glycosylation of the mucin type. Biol. Chem. 2001, 382, 143–149. [Google Scholar]
- Dekker, J.; Rossen, J.W.A.; Büller, H.A.; Einerhand, A.W.C. The MUC family: An obituary. Trends Biochem. Sci. 2002, 27, 126–131. [Google Scholar]
- Allen, A.; Pearson, J.P. Mucus glycoproteins of the normal gastrointestinal tract. Eur. J. Gastroenterol. Hepatol. 1993, 5, 193–199. [Google Scholar]
- Moncada, D.M.; Kammanadiminti, S.J.; Chadee, K. Mucin and toll-like receptors in host defense against intestinal parasites. Trends Parasitol. 2003, 19, 305–311. [Google Scholar]
- Liévin-Le Moal, V.; Servin, A.L.; Coconnier-Polter, M. The increase in mucin exocytosis and the upregulation of MUC genes encoding for membrane-bound mucins induced by the thiol-activated exotoxin listeriolysin O is a host cell defence response that inhibits the cell-entry of Listeria monocytogenes. Cell. Microbiol. 2005, 7, 1035–1048. [Google Scholar]
- Johansson, M.E.V.; Phillipson, M.; Petersson, J.; Velcich, A.; Holm, L.; Hansson, G.C. The inner of the two MUC2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl. Acad. Sci.USA 2008, 105, 15064–15069. [Google Scholar]
- Strous, G.J.; Dekker, J. Mucin-type glycoproteins. Crit. Rev. Biochem. Mol. Biol. 1992, 27, 57–92. [Google Scholar]
- Dharmani, P.; Srivastava, V.; Kissoon-Singh, V.; Chadee, K. Role of intestinal mucins in innate host defense mechanisms against pathogens. J. Innate Immun. 2009, 1, 123–135. [Google Scholar]
- Marcaurelle, L.A.; Bertozzi, C.R. Recent advances in the chemical synthesis of mucin-like glycoproteins. Glycobiology 2002, 12, 69R–77R. [Google Scholar]
- Brockhausen, I.; Schutzbach, J.; Kuhns, W. Glycoproteins and their relationship to human disease. Acta Anat. (Basel) 1998, 161, 36–78. [Google Scholar] [CrossRef] [PubMed]
- Slomiany, A.; Zdebska, E.; Slomiany, B.L. Structures of the neutral oligosaccharides isolated from A-active human gastric mucin. J. Biol. Chem. 1984, 259, 14743–14749. [Google Scholar]
- Corfield, A.P.; Myerscough, N.; Gough, M.; Brockhausen, I.; Schauer, R.; Paraskeva, C. Glycosylation patterns of mucins in colonic disease. Biochem. Soc. Trans. 1995, 23, 840–845. [Google Scholar]
- Sheahan, D.G.; Jervis, H.R. Comparative histochemistry of gastrointestinal mucosubstances. Am. J. Anat. 1976, 146, 103–131. [Google Scholar]
- Chance, D.L.; Mawhinney, T.P. Carbohydrate sulfation effects on growth of pseudomonas aeruginosa. Microbiology 2000, 146, 1717–1725. [Google Scholar]
- Fontaine, N.; Meslin, J.C.; Doré, J. Selective in vitro degradation of the sialylated fraction of germ-free rat mucins by the caecal flora of the rat. Reprod. Nutr. Dev. 1998, 38, 289–296. [Google Scholar]
- Roberto, A.M.; Wright, D.P. Bacterial glycosulphatases and sulphomucin degradation. Can. J. Gastroenterol. 1997, 11, 361–366. [Google Scholar]
- McCool, D.J.; Forstner, J.F.; Forstner, G.G. Synthesis and secretion of mucin by the human colonic tumour cell line LS180. Biochem. J. 1994, 302, 111–118. [Google Scholar]
- Sonnenburg, J.L.; Angenent, L.T.; Gordon, J.I. Getting a grip on things: How do communities of bacterial symbionts become established in our intestine? Nat. Immunol. 2004, 5, 569–573. [Google Scholar] [PubMed]
- Carrington, S.D.; Clyne, M.; Reid, C.J.; FitzPatrick, E.; Corfield, A.P. Microbial Interaction with Mucus and Mucins. In Microbial Glycobiology: Structures, Relevance and Applications; Moran, A., Holst, O., Brennan, P., von Itzstein, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 655–671. [Google Scholar]
- Varki, A. Biological roles of oligosaccharides: All of the theories are correct. Glycobiology 1993, 3, 97–130. [Google Scholar]
- Gagneux, P.; Varki, A. Evolutionary considerations in relating oligosaccharide diversity to biological function. Glycobiology 1999, 9, 747–755. [Google Scholar]
- Patsos, G.; Corfield, A. Management of the human mucosal defensive barrier: Evidence for glycan legislation. Biol. Chem. 2009, 390, 581–590. [Google Scholar]
- Robbe, C.; Capon, C.; Maes, E.; Rousset, M.; Zweibaum, A.; Zanetta, J.P.; Michalski, J.C. Evidence of regio-specific glycosylation in human intestinal mucins: Presence of an acidic gradient along the intestinal tract. J. Biol. Chem. 2003, 278, 46337–46348. [Google Scholar]
- Brockhausen, I. Pathways of O-glycan biosynthesis in cancer cells. Biochim. Biophys. Acta 1999, 1473, 67–95. [Google Scholar]
- Kelly, R.J.; Rouquier, S.; Giorgi, D.; Lennon, G.G.; Lowe, J.B. Sequence and expression of a candidate for the human secretor blood group Alpha(1,2)Fucosyltransferase gene (FUT2). homozygosity for an enzyme-inactivating nonsense mutation commonly correlates with the non-secretor phenotype. J. Biol. Chem. 1995, 270, 4640–4649. [Google Scholar] [PubMed]
- Hoskins, L.C.; Boulding, E.T. Degradation of blood group antigens in human colon ecosystems. II. A gene interaction in man that affects the fecal population density of certain enteric bacteria. J. Clin. Invest. 1976, 57, 74–82. [Google Scholar] [PubMed]
- Salyers, A.A.; Pajeau, M.; McCarthy, R.E. Importance of mucopolysaccharides as substrates for Bacteroides thetaiotaomicron growing in intestinal tracts of exgermfree mice. Appl. Environ. Microbiol. 1988, 54, 1970–1976. [Google Scholar]
- Hwa, V.; Salyers, A.A. Analysis of two chondroitin sulfate utilization mutants of Bacteroides thetaiotaomicron that differ in their abilities to compete with the wild type in the gastrointestinal tracts of germfree mice. Appl. Environ. Microbiol. 1992, 58, 869–876. [Google Scholar]
- Corfield, A.P.; Wagner, S.A.; Clamp, J.R.; Kriaris, M.S.; Hoskins, L.C. Mucin degradation in the human colon: Production of sialidase, sialate O-acetylesterase, N-acetylneuraminate lyase, arylesterase, and glycosulfatase activities by strains of fecal bacteria. Infect. Immun. 1992, 60, 3971–3978. [Google Scholar] [PubMed]
- Katayama, T.; Fujita, K.; Yamamoto, K. Novel bifidobacterial glycosidases acting on sugar chains of mucin glycoproteins. J. Biosci. Bioeng. 2005, 99, 457–465. [Google Scholar]
- Ruiz-Palacios, G.M.; Cervantes, L.E.; Ramos, P.; Chavez-Munguia, B.; Newburg, D.S. Campylobacter jejuni binds intestinal H(O) antigen (Fuc Alpha 1, 2Gal Beta 1, 4GlcNAc), and fucosyloligosaccharides of human milk inhibit its binding and infection. J. Biol. Chem. 2003, 278, 14112–14120. [Google Scholar] [PubMed]
- Oakey, H.J.; Harty, D.W.; Knox, K.W. Enzyme production by lactobacilli and the potential link with infective endocarditis. J. Appl. Bacteriol. 1995, 78, 142–148. [Google Scholar]
- Ashida, H.; Miyake, A.; Kiyohara, M.; Wada, J.; Yoshida, E.; Kumagai, H.; Katayama, T.; Yamamoto, K. Two distinct Alpha-l-Fucosidases from Bifidobacterium bifidum are essential for the utilization of fucosylated milk oligosaccharides and glycoconjugates. Glycobiology 2009, 19, 1010–1017. [Google Scholar]
- Lindén, S.K.; Sutton, P.; Karlsson, N.G.; Korolik, V.; McGuckin, M.A. Mucins in the mucosal barrier to infection. Mucosal Immunol. 2008, 1, 183–197. [Google Scholar]
- Hashimoto, K.; Goto, S.; Kawano, S.; Aoki-Kinoshita, K.F.; Ueda, N.; Hamajima, M.; Kawasaki, T.; Kanehisa, M. KEGG as a Glycome Informatics Resource. Glycobiology 2006, 16, 63R–70R. [Google Scholar]
- Raman, R.; Venkataraman, M.; Ramakrishnan, S.; Lang, W.; Raguram, S.; Sasisekharan, R. Advancing glycomics: Implementation strategies at the consortium for functional glycomics. Glycobiology 2006, 16, 82R–90R. [Google Scholar]
- Tannock, G.W.; Munro, K.; Harmsen, H.J.; Welling, G.W.; Smart, J.; Gopal, P.K. Analysis of the fecal microflora of human subjects consuming a probiotic product containing Lactobacillusrhamnosus DR20. Appl. Environ. Microbiol. 2000, 66, 2578–2588. [Google Scholar]
- Garrido, D.; Suau, A.; Pochart, P.; Cruchet, S.; Gotteland, M. Modulation of the fecal microbiota by the intake of a Lactobacillus johnsonii La1-Containing product in human volunteers. FEMS Microbiol. Lett. 2005, 248, 249–256. [Google Scholar]
- Wadström, T.; Andersson, K.; Sydow, M.; Axelsson, L.; Lindgren, S.; Gullmar, B. Surface properties of lactobacilli isolated from the small intestine of pigs. J. Appl. Bacteriol. 1987, 62, 513–520. [Google Scholar]
- Lichtenberger, L.M. The hydrophobic barrier properties of gastrointestinal mucus. Annu. Rev. Physiol. 1995, 57, 565–583. [Google Scholar]
- Ehrmann, M.A.; Kurzak, P.; Bauer, J.; Vogel, R.F. Characterization of lactobacilli towards their use as probiotic adjuncts in poultry. J. Appl. Microbiol. 2002, 92, 966–975. [Google Scholar]
- Kos, B.; Susković, J.; Vuković, S.; Simpraga, M.; Frece, J.; Matosić, S. Adhesion and Aggregation ability of probiotic strain Lactobacillus acidophilus M92. J. Appl. Microbiol. 2003, 94, 981–987. [Google Scholar]
- Ouwehand, A.C.; Kirjavainen, P.V.; Grönlund, M.M.; Isolauri, E.; Salminen, S.J. Adhesion of probiotic micro-organisms to intestinal mucus. Int. Dairy J. 1999, 9, 623–630. [Google Scholar]
- Muñoz-Provencio, D.; Llopis, M.; Antolín, M.; de Torres, I.; Guarner, F.; Pérez-Martínez, G.; Monedero, V. Adhesion properties of Lactobacillus casei strains to resected intestinal fragments and components of the extracellular matrix. Arch. Microbiol. 2009, 191, 153–161. [Google Scholar]
- Jacobsen, C.N.; Rosenfeldt, N.V.; Hayford, A.E.; Møller, P.L.; Michaelsen, K.F.; Paerregaard, A.; Sandström, B.; Tvede, M.; Jakobsen, M. Screening of probiotic activities of forty-seven strains of Lactobacillus spp. by in vitro techniques and evaluation of the colonization ability of five selected strains in humans. Appl. Environ. Microbiol. 1999, 65, 4949–4956. [Google Scholar] [PubMed]
- Jonsson, H.; Ström, E.; Roos, S. Addition of mucin to the growth medium triggers mucus-binding activity in different strains of Lactobacillus reuteri in vitro. FEMS Microbiol. Lett. 2001, 204, 19–22. [Google Scholar]
- MacKenzie, D.A.; Tailford, L.E.; Hemmings, A.M.; Juge, N. Crystal structure of a mucus-binding protein repeat reveals an unexpected functional immunoglobulin binding activity. J. Biol. Chem. 2009, 284, 32444–32453. [Google Scholar]
- Dam, T.K.; Brewer, C.F. Multivalent lectin-carbohydrate interactions energetics and mechanisms of binding. Adv. Carbohydr. Chem. Biochem. 2010, 63, 139–164. [Google Scholar]
- Roos, S.; Jonsson, H. A high-molecular-mass cell-surface protein from Lactobacillus reuteri 1063 adheres to mucus components. Microbiology 2002, 148, 433–442. [Google Scholar]
- Boekhorst, J.; Helmer, Q.; Kleerebezem, M.; Siezen, R.J. Comparative analysis of proteins with a mucus-binding domain found exclusively in lactic acid bacteria. Microbiology 2006, 152, 273–280. [Google Scholar] [Green Version]
- Altermann, E.; Russell, W.M.; Azcarate-Peril, M.A.; Barrangou, R.; Buck, B.L.; McAuliffe, O.; Souther, N.; Dobson, A.; Duong, T.; Callanan, M.; et al. Complete genome sequence of the probiotic lactic acid bacterium Lactobacillus acidophilus Ncfm. Proc. Natl. Acad. Sci. USA 2005, 102, 3906–3912. [Google Scholar]
- Kleerebezem, M.; Hols, P.; Bernard, E.; Rolain, T.; Zhou, M.; Siezen, R.J.; Bron, P.A. The extracellular biology of the lactobacilli. FEMS Microbiol. Rev. 2010, 34, 199–230. [Google Scholar]
- Mackenzie, D.A.; Jeffers, F.; Parker, M.L.; Vibert-Vallet, A.; Bongaerts, R.J.; Roos, S.; Walter, J.; Juge, N. Strain-specific diversity of mucus-binding proteins in the adhesion and aggregation properties of Lactobacillus reuteri. Microbiology 2010, 156, 3368–3378. [Google Scholar]
- Pridmore, R.D.; Berger, B.; Desiere, F.; Vilanova, D.; Barretto, C.; Pittet, A.C.; Zwahlen, M.C.; Rouvet, M.; Altermann, E.; Barrangou, R.; et al. The genome sequence of the probiotic intestinal bacterium Lactobacillus johnsonii NCC 533. Proc. Natl. Acad. Sci. USA 2004, 101, 2512–2517. [Google Scholar]
- Kankainen, M.; Paulin, L.; Tynkkynen, S.; von Ossowski, I.; Reunanen, J.; Partanen, P.; Satokari, R.; Vesterlund, S.; Hendrickx, A.P.A.; Lebeer, S. Comparative genomic analysis of Lactobacillus rhamnosus GG reveals pili containing a human-mucus binding protein. Proc. Natl. Acad. Sci. USA 2009, 106, 17193–17198. [Google Scholar]
- Connell, I.; Agace, W.; Klemm, P.; Schembri, M.; Mărild, S.; Svanborg, C. Type 1 fimbrial expression enhances Escherichia coli virulence for the urinary tract. Proc. Natl. Acad. Sci. USA 1996, 93, 9827–9832. [Google Scholar]
- Macías-Rodríguez, M.E.; Zagorec, M.; Ascencio, F.; Vázquez-Juárez, R.; Rojas, M. Lactobacillus fermentum BCS87 expresses mucus- and mucin-binding proteins on the cell surface. J. Appl. Microbiol. 2009, 107, 1866–1874. [Google Scholar]
- Buck, B.L.; Altermann, E.; Svingerud, T.; Klaenhammer, T.R. Functional analysis of putative adhesion factors in Lactobacillus acidophilus NCFM. Appl. Environ. Microbiol. 2005, 71, 8344–8351. [Google Scholar]
- Pretzer, G.; Snel, J.; Molenaar, D.; Wiersma, A.; Bron, P.A.; Lambert, J.; de Vos, W.M.; van der Meer, R.; Smits, M.A.; Kleerebezem, M. Biodiversity-based identification and functional characterization of the mannose-specific adhesin of Lactobacillus plantarum. J. Bacteriol. 2005, 187, 6128–6136. [Google Scholar]
- Rojas, M.; Ascencio, F.; Conway, P.L. Purification and characterization of a surface protein from Lactobacillus fermentum 104R that binds to porcine small intestinal mucus and gastric mucin. Appl. Environ. Microbiol. 2002, 68, 2330–2336. [Google Scholar]
- Miyoshi, Y.; Okada, S.; Uchimura, T.; Satoh, E. A mucus adhesion promoting protein, MapA, mediates the adhesion of Lactobacillus reuteri to Caco-2 human intestinal epithelial cell. Biosci. Biotechnol. Biochem. 2006, 70, 1622–1628. [Google Scholar]
- Jacobson, G.R.; Rosenbusch, J.P. Abundance and membrane association of elongation factor Tu in E. coli. Nature 1976, 261, 23–26. [Google Scholar] [PubMed]
- Dallo, S.F.; Kannan, T.R.; Blaylock, M.W.; Baseman, J.B. Elongation factor Tu and E1 beta subunit of pyruvate dehydrogenase complex act as fibronectin binding proteins in Mycoplasma pneumoniae. Mol. Microbiol. 2002, 46, 1041–1051. [Google Scholar]
- Granato, D.; Bergonzelli, G.E.; Pridmore, R.D.; Marvin, L.; Rouvet, M.; Corthésy-Theulaz, I.E. Cell surface-associated elongation factor Tu mediates the attachment of Lactobacillus johnsonii NCC533 (La1) to human intestinal cells and mucins. Infect. Immun. 2004, 72, 2160–2169. [Google Scholar]
- Nakamura, J.; Ito, D.; Nagai, K.; Umehara, Y.; Hamachi, M.; Kumagai, C. Rapid and sensitive detection of hiochi bacteria by amplification of hiochi bacterial common antigen gene by PCR method and characterization of the antigen. J. Ferment. Bioeng. 1997, 83, 161–167. [Google Scholar]
- Ramiah, K.; van Reenen, C.A.; Dicks, L.M.T. Expression of the mucus adhesion genes mub and MapA, adhesion-like factor EF-Tu and bacteriocin gene plaA of Lactobacillus plantarum 423, monitored with real-time PC. Int. J. Food Microbiol. 2007, 116, 405–409. [Google Scholar]
- Finn, R.D.; Mistry, J.; Tate, J.; Coggill, P.; Heger, A.; Pollington, J.E.; Gavin, O.L.; Gunesekaran, P.; Ceric, G.; Forslund, K.; et al. The Pfam protein families database. Nucleic Acids Res. 2010, 38, D211–D222. [Google Scholar]
- The UniProt Consortium. The universal protein resource (UniProt) in 2010. Nucleic Acids Res. 2010, 38, D142–D148. [CrossRef] [PubMed]
- Sleytr, U.B.; Bayley, H.; Sára, M.; Breitwieser, A.; Küpcü, S.; Mader, C.; Weigert, S.; Unger, F.M.; Messner, P.; Jahn-Schmid, B. Applications of s-layers. FEMS Microbiol. Rev. 1997, 20, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Xu, J.; Shuai, J.; Chen, J.; Zhang, Z.; Fang, W. The s-layer proteins of Lactobacillus crispatus strain ZJ001 is responsible for competitive exclusion against Escherichia coli O157:H7 and Salmonella typhimurium. Int. J. Food Microbiol. 2007, 115, 307–312. [Google Scholar]
- Sánchez, B.; Arias, S.; Chaignepain, S.; Denayrolles, M.; Schmitter, J.M.; Bressollier, P.; Urdaci, M.C. Identification of surface proteins involved in the adhesion of a probiotic Bacillus cereus strain to mucin and fibronectin. Microbiology 2009, 155, 1708–1716. [Google Scholar]
- Zhang, Y.C.; Zhang, L.W.; Tuo, Y.F.; Guo, C.F.; Yi, H.X.; Li, J.Y.; Han, X.; Du, M. Inhibition of Shigella sonnei adherence to HT-29 cells by lactobacilli from Chinese fermented food and preliminary characterization of s-layer protein involvement. Res. Microbiol. 2010, 161, 667–672. [Google Scholar]
- Bøhle, L.A.; Brede, D.A.; Diep, D.B.; Holo, H.; Nes, I.F. The mucus adhesion promoting protein (MapA) of Lactobacillus reuteri is specifically degraded to an antimicrobial peptide. Appl. Environ. Microbiol. 2010, 76, 7306–7309. [Google Scholar]
- Ouwehand, A.C.; Salminen, S. In vitro adhesion assays for probiotics and their in vivo relevance: A review. Microb. Ecol. Health Dis. 2003, 15, 175–184. [Google Scholar]
- Ouwehand, A.C.; Tuomola, E.M.; Tölkkö, S.; Salminen, S. Assessment of adhesion properties of novel probiotic strains to human intestinal mucus. Int. J. Food Microbiol. 2001, 64, 119–126. [Google Scholar]
- Kankaanpää, P.E.; Salminen, S.J.; Isolauri, E.; Lee, Y.K. The influence of polyunsaturated fatty acids on probiotic growth and adhesion. FEMS Microbiol. Lett. 2001, 194, 149–153. [Google Scholar]
- Dunne, C.; O’Mahony, L.; Murphy, L.; Thornton, G.; Morrissey, D.; O’Halloran, S.; Feeney, M.; Flynn, S.; Fitzgerald, G.; Daly, C.; et al. In vitro selection criteria for probiotic bacteria of human origin: Correlation with in vivo findings. Am. J. Clin. Nutr. 2001, 73, 386S–392S. [Google Scholar]
- Juntunen, M.; Kirjavainen, P.V.; Ouwehand, A.C.; Salminen, S.J.; Isolauri, E. Adherence of probiotic bacteria to human intestinal mucus in healthy infants and during rotavirus infection. Clin. Diagn. Lab. Immunol. 2001, 8, 293–296. [Google Scholar]
- Schiffrin, E.J.; Brassart, D.; Servin, A.L.; Rochat, F.; Donnet-Hughes, A. Immune modulation of blood leukocytes in humans by lactic acid bacteria: Criteria for strain selection. Am. J. Clin. Nutr. 1997, 66, 515S–520S. [Google Scholar]
- Vélez, M.P.; Petrova, M.I.; Lebeer, S.; Verhoeven, T.L.A.; Claes, I.; Lambrichts, I.; Tynkkynen, S.; Vanderleyden, J.; De Keersmaecker, S.C.J. Characterization of MabA, a modulator of Lactobacillus rhamnosus GG adhesion and biofilm formation. FEMS Immunol. Med. Microbiol. 2010, 59, 386–398. [Google Scholar]
- Lee, Y.; Puong, K. Competition for adhesion between probiotics and human gastrointestinal pathogens in the presence of carbohydrate. Br. J. Nutr. 2002, 88, S101–S108. [Google Scholar]
- Candela, M.; Perna, F.; Carnevali, P.; Vitali, B.; Ciati, R.; Gionchetti, P.; Rizzello, F.; Campieri, M.; Brigidi, P. Interaction of probiotic Lactobacillus and Bifidobacterium strains with human intestinal epithelial cells: Adhesion properties, competition against enteropathogens and modulation of IL-8 production. Int. J. Food Microbiol. 2008, 125, 286–292. [Google Scholar]
- Gueimonde, M.; Jalonen, L.; He, F.; Hiramatsu, M.; Salminen, S. Adhesion and competitive inhibition and displacement of human enteropathogens by selected lactobacilli. Food Res. Int. 2006, 39, 467–471. [Google Scholar]
- Majamaa, H.; Isolauri, E.; Saxelin, M.; Vesikari, T. Lactic acid bacteria in the treatment of acute rotavirus gastroenteritis. J. Pediatr. Gastroenterol. Nutr. 1995, 20, 333–338. [Google Scholar]
- O’Halloran, S.; Feeney, M.; Morrissey, D.; Murphy, L.; Thornton, G.; Shanahan, F.; O’Sullivan, G.C.; Collins, J.K. Adhesion of potential probiotic bacteria to human epithelial cell lines. Int. Dairy J. 1998, 8, 596. [Google Scholar]
- Laparra, J.M.; Sanz, Y. Comparison of in vitro models to study bacterial adhesion to the intestinal epithelium. Lett. Appl. Microbiol. 2009, 49, 695–701. [Google Scholar]
- Tallon, R.; Arias, S.; Bressollier, P.; Urdaci, M.C. Strain- and matrix-dependent adhesion of Lactobacillus plantarum is mediated by proteinaceous bacterial compounds. J. Appl. Microbiol. 2007, 102, 442–451. [Google Scholar]
- Li, X.J.; Yue, L.Y.; Guan, X.F.; Qiao, S.Y. The adhesion of putative probiotic lactobacilli to cultured epithelial cells and porcine intestinal mucus. J. Appl. Microbiol. 2008, 104, 1082–1091. [Google Scholar]
- Pinto, M.; Robine-Leon, S.; Appay, M.D. Enterocyte-like differentiation and polarization of the human colon Carcinoma Cell Line Caco-2 in culture. Biol. Cell 1983, 47, 323–330. [Google Scholar]
- Rousset, M. The human colon carcinoma cell lines HT-29 and Caco-2: Two in vitro models for the study of intestinal differentiation. Biochimie 1986, 68, 1035–1040. [Google Scholar]
- Lenaerts, K.; Bouwman, F.G.; Lamers, W.H.; Renes, J.; Mariman, E.C. Comparative proteomic analysis of cell lines and scrapings of the human intestinal epithelium. BMC Genomics 2007, 8, 91. [Google Scholar]
- Crociani, J.; Grill, J.P.; Huppert, M.; Ballongue, J. Adhesion of different Bifidobacteria strains to human enterocyte-like Caco-2 cells and comparison with in vivo study. Lett. Appl. Microbiol. 1995, 21, 146–148. [Google Scholar]
- Lesuffleur, T.; Barbat, A.; Dussaulx, E.; Zweibaum, A. Growth adaptation to methotrexate of HT-29 human colon carcinoma cells is associated with their ability to differentiate into columnar absorptive and mucus-secreting cells. Cancer Res. 1990, 50, 6334–6343. [Google Scholar]
- Leteurtre, E.; Gouyer, V.; Rousseau, K.; Moreau, O.; Barbat, A.; Swallow, D.; Huet, G.; Lesuffleur, T. Differential mucin expression in colon carcinoma HT-29 clones with variable resistance to 5-fluorouracil and methotrexate. Biol. Cell 2004, 96, 145–151. [Google Scholar]
- Bernet, M.F.; Brassart, D.; Neeser, J.R.; Servin, A.L. Lactobacillus acidophilus LA1 binds to cultured human intestinal cell lines and inhibits cell attachment and cell invasion by enterovirulent bacteria. Gut 1994, 35, 483–489. [Google Scholar]
- Gopal, P.K.; Prasad, J.; Smart, J.; Gill, H.S. In vitro adherence properties of Lactobacillus rhamnosus DR20 and Bifidobacterium lactis DR10 strains and their antagonistic activity against an enterotoxigenic Escherichiacoli. Int. J. Food Microbiol. 2001, 67, 207–216. [Google Scholar]
- Lesuffleur, T.; Porchet, N.; Aubert, J.P.; Swallow, D.; Gum, J.R.; Kim, Y.S.; Real, F.X.; Zweibaum, A. Differential expression of the human mucin genes MUC1 to MUC5 in relation to growth and differentiation of different mucus-secreting HT-29 cell subpopulations. J. Cell Sci. 1993, 106, 771–783. [Google Scholar]
- Kerneis, S.; Bernet, M.F.; Coconnier, M.H.; Servin, A.L. Adhesion of human enterotoxigenic Escherichia coli to human mucus secreting HT-29 cell subpopulations in culture. Gut 1994, 35, 1449–1454. [Google Scholar]
- Nutten, S.; Sansonetti, P.; Huet, G.; Bourdon-Bisiaux, C.; Meresse, B.; Colombel, J.F.; Desreumaux, P. Epithelial inflammation response induced by Shigella flexneri depends on mucin gene expression. Microbes Infect. 2002, 4, 1121–1124. [Google Scholar]
- Walter, E.; Janich, S.; Roessler, B.J.; Hilfinger, J.M.; Amidon, G.L. HT29-MTX/Caco-2 cocultures as an in vitro model for the intestinal epithelium: In vitro-in vivo correlation with permeability data from rats and humans. J. Pharm. Sci. 1996, 85, 1070–1076. [Google Scholar]
- Pontier, C.; Pachot, J.; Botham, R.; Lenfant, B.; Arnaud, P. HT29-MTX and Caco-2/TC7 monolayers as predictive models for human intestinal absorption: Role of the mucus layer. J. Pharm. Sci. 2001, 90, 1608–1619. [Google Scholar]
- Chen, X.M.; Elisia, I.; Kitts, D.D. Defining conditions for the co-culture of Caco-2 and HT29-MTX cells using taguchi design. J. Pharmacol. Toxicol. Methods 2010, 61, 334–342. [Google Scholar]
- Henriksson, A.; Szewzyk, R.; Conway, P.L. Characteristics of the adhesive determinants of Lactobacillus fermentum 104. Appl. Environ. Microbiol. 1991, 57, 499–502. [Google Scholar]
- Vesterlund, S.; Paltta, J.; Karp, M.; Ouwehand, A. Adhesion of bacteria to resected human colonic tissue: Quantitative analysis of bacterial adhesion and viability. Res. Microbiol. 2005, 156, 238–244. [Google Scholar]
- Phillips, A.D.; Navabpour, S.; Hicks, S.; Dougan, G.; Wallis, T.; Frankel, G. Enterohaemorrhagic Escherichia coli O157:H7 target Peyer’s patches in humans and cause attaching/effacing lesions in both human and bovine intestine. Gut 2000, 47, 377–381. [Google Scholar]
- Girard, F.; Dziva, F.; van Diemen, P.; Phillips, A.D.; Stevens, M.P.; Frankel, G. Adherence of enterohemorrhagic Escherichia coli O157, O26, and O111 strains to bovine intestinal explants ex viv. Appl. Environ. Microbiol. 2007, 73, 3084–3090. [Google Scholar]
- Day, C.J.; Tiralongo, J.; Hartnell, R.D.; Logue, C.; Wilson, J.C.; von Itzstein, M.; Korolik, V. Differential carbohydrate recognition by Campylobacter jejuni strain 11168: Influences of temperature and growth conditions. PLoS One 2009, 4, 781–790. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Van Tassell, M.L.; Miller, M.J. Lactobacillus Adhesion to Mucus. Nutrients 2011, 3, 613-636. https://0-doi-org.brum.beds.ac.uk/10.3390/nu3050613
Van Tassell ML, Miller MJ. Lactobacillus Adhesion to Mucus. Nutrients. 2011; 3(5):613-636. https://0-doi-org.brum.beds.ac.uk/10.3390/nu3050613
Chicago/Turabian StyleVan Tassell, Maxwell L., and Michael J. Miller. 2011. "Lactobacillus Adhesion to Mucus" Nutrients 3, no. 5: 613-636. https://0-doi-org.brum.beds.ac.uk/10.3390/nu3050613






