Solid Phase Adsorption Toxin Tracking (SPATT) Technology for the Monitoring of Aquatic Toxins: A Review
Abstract
:1. Introduction
2. Application of SPATT Technology for the Monitoring of Aquatic Toxins
2.1. Lipophilic Toxins
2.1.1. Diarrheic Shellfish Poisoning (DSP) Toxins and Pectenotoxins (PTXs)
2.1.2. Yessotoxins (YTXs)
2.1.3. Azaspiracid Shellfish Poisoning (AZP) Toxins
2.1.4. Cyclic Imines (CI)
2.1.5. Ciguatoxins (CTXs)
2.2. Hydrophilic Toxins
2.2.1. Amnesic Shellfish Poisoning (ASP) Toxins
2.2.2. Paralytic Shellfish Poisoning (PSP) Toxins
2.2.3. Microcystins (MCs) and Nodularins (NODs)
2.2.4. Anatoxin (ANTX)
2.2.5. Maitotoxins (MTXs)
3. Implications in Terms of Monitoring of Emerging Toxic Risks and Reinforcement of Risk Assessment Programs
Conflicts of Interest
References
- Berdalet, E.; Fleming, L.E.; Gowen, R.; Davidson, K.; Hess, P.; Backer, L.; Moore, S.K.; Hoagmand, P.; Enevoldsen, H. Marine harmful algal blooms, human health and wellbeing: Challenges and opportunities in the 21st century. J. Mar. Biol. Assoc. UK 2016, 96, 61–91. [Google Scholar] [CrossRef] [PubMed]
- Bernard, C.; Ballot, A.; Thomazeau, S.; Maloufi, S.; Furey, A.; Mankiewicz Boczek, J.; Pawlik-Skowronska, B.; Capelli, C.; Salmaso, N. Appendix 2: Cyanobacteria Associated with the Production of Cyanotoxins. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Cood, G.A., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017; ISBN 978-1-119-06868-6. [Google Scholar]
- Merel, S.; Walker, D.; Chicana, R.; Snyder, S.; Baurès, E.; Thomas, O. State of knowledge and concerns on cyanobacterial blooms and cyanotoxins. Environ. Int. 2013, 59, 303–327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Botana, L.M. Toxicological Perspective on Climate Change: Aquatic Toxins. Chem. Res. Toxicol. 2016, 29, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Andersen, P.; Bricelj, V.M.; Cullen, J.J.; Rensel, J.E.J. Monitoring and Management Strategies for Harmful Algal Blooms in Coastal Waters; Asia Pacific Economic Program, Singapore, and Intergovernmental Oceanographic Commission Technical Series No. 59; UNESCO: Paris, France, 2001. [Google Scholar]
- Roué, M.; Gugger, M.; Golubic, S.; Amzil, Z.; Araoz, R.; Turquet, J.; Chinain, M.; Laurent, D. Marine cyanotoxins potentially harmful to human health. In Outstanding Marine Molecules: Chemistry, Biology, Analysis; La Barre, S., Kornprobst, J.-M., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; pp. 3–22. [Google Scholar]
- Vasconcelos, V.; Leao, P. Marine Cyanobacterial Toxins: Source, Chemistry, Toxicology, Pharmacology, and Detection. In Seafood and Freshwater Toxins; CRC Press: Boca Raton, FL, USA, 2014; pp. 1073–1090. ISBN 978-1-4665-0514-8. [Google Scholar]
- Stewart, I.; Seawright, A.A.; Shaw, G.R. Cyanobacterial poisoning in livestock, wild mammals and birds—An overview. Adv. Exp. Med. Biol. 2008, 619, 613–637. [Google Scholar] [CrossRef] [PubMed]
- Dorantes-Aranda, J.J.; Seger, A.; Mardones, J.I.; Nichols, P.D.; Hallegraeff, G.M. Progress in understanding algal bloom-mediated fish kills: The role of superoxide radicals, phycotoxins and fatty acids. PLoS ONE 2015, 10, e0133549. [Google Scholar] [CrossRef] [PubMed]
- Fleming, L.E.; Kirkpatrick, B.; Backer, L.C.; Bean, J.A.; Wanner, A.; Reich, A.; Zaias, J.; Cheng, Y.S.; Pierce, R.; Naar, J.; et al. Aerosolized red-tide toxins (brevetoxins) and asthma. Chest 2007, 131, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Werner, K.A.; Marquart, L.; Norton, S.A. Lyngbya dermatitis (toxic seaweed dermatitis). Int. J. Dermatol. 2012, 51, 59–62. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Liu, Y.L.; Conklin, A.; Westrick, J.; Weavers, L.K.; Dionysiou, D.D.; Lenhart, J.J.; Mouser, P.J.; Szlag, D.; Walker, H.W. Toxic cyanobacteria and drinking water: Impacts, detection, and treatment. Harmful Algae 2016, 54, 174–193. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Chorus, I. Accumulation of cyanobacterial toxins in freshwater “seafood” and its consequences for public health: A review. Environ. Pollut. 2007, 150, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Gerssen, A.; Pol-Hofstad, I.E.; Poelman, M.; Mulder, P.P.J.; van den Top, H.J.; Dde Boer, J. Marine toxins: Chemistry, toxicity, occurrence and detection, with special reference to the dutch situation. Toxins 2010, 2, 878–904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valério, E.; Chaves, S.; Tenreiro, R. Diversity and impact of prokaryotic toxins on aquatic environments: A review. Toxins 2010, 2, 2359–2410. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Reguera, B.; Pitcher, G.C.; Enevoldsen, H.O. The IOC International Harmful Algal Bloom Program: History and science impacts. Oceanography 2011, 23, 72–85. [Google Scholar] [CrossRef]
- De Figueiredo, D.R.; Azeiteiro, U.M.; Esteves, S.M.; Gonçalves, F.J.M.; Pereira, M.J. Microcystin-producing blooms—A serious global public health issue. Ecotoxicol. Environ. Saf. 2004, 59, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.; Suárez, F.; Ramírez, A.; Acosta, F. Ciguatera, an Emerging Human Poisoning in Europe. J. Aquac. Mar. Biol. 2015, 3, 00053. [Google Scholar] [CrossRef]
- Sanseverino, I.; Conduto, D.; Pozzoli, L.; Dobricic, S.; Lettieri, T. Algal Bloom and Its Economic Impact; JRC Technical Reports; EUR 27905 EN; European Commission: Brussels, Belgium, 2016. [Google Scholar] [CrossRef]
- Pitcher, G.C. Harmful algae—The requirement for species-specific information. Harmful Algae 2012, 14, 1–4. [Google Scholar] [CrossRef]
- MacKenzie, L.; Beuzenberg, V.; Holland, P.; McNabb, P.; Selwood, A. Solid phase adsorption toxin tracking (SPATT): A new monitoring tool that simulates the biotoxin contamination of filter feeding bivalves. Toxicon 2004, 44, 901–918. [Google Scholar] [CrossRef] [PubMed]
- Litaker, R.W.; Vandersea, M.W.; Faust, M.A.; Kibler, S.R.; Nau, A.W.; Holland, W.C.; Chinain, M.; Holmes, M.J.; Tester, P.A. Global distribution of ciguatera causing dinoflagellates in the genus Gambierdiscus. Toxicon 2010, 56, 711–730. [Google Scholar] [CrossRef] [PubMed]
- Zendong, Z.; Bertrand, S.; Herrenknecht, C.; Abadie, E.; Jauzein, C.; Lemée, R.; Gouriou, J.; Amzil, Z.; Hess, P. Passive Sampling and High Resolution Mass Spectrometry for Chemical Profiling of French Coastal Areas with a Focus on Marine Biotoxins. Environ. Sci. Technol. 2016, 50, 8522–8529. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, L.A. In situ passive solid-phase adsorption of micro-algal biotoxins as a monitoring tool. Curr. Opin. Biotechnol. 2010, 21, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Kudela, R.M. Passive Sampling for Freshwater and Marine Algal Toxins. Compr. Anal. Chem. 2017, 78, 379–409. [Google Scholar] [CrossRef]
- Gibble, C.M.; Peacock, M.B.; Kudela, R.M. Evidence of freshwater algal toxins in marine shellfish: Implications for human and aquatic health. Harmful Algae 2016, 59, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Bakke, M.J.; Horsberg, T.E. Kinetic properties of saxitoxin in Atlantic salmon (Salmo salar) and Atlantic cod (Gadus morhua). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2010, 152, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Cazenave, J.; Wunderlin, D.A.; Bistoni, M.D.L.Á.; Amé, M.V.; Krause, E.; Pflugmacher, S.; Wiegand, C. Uptake, tissue distribution and accumulation of microcystin-RR in Corydoras paleatus, Jenynsia multidentata and Odontesthes bonariensis: A field and laboratory study. Aquat. Toxicol. 2005, 75, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Jauffrais, T.; Kilcoyne, J.; Herrenknecht, C.; Truquet, P.; Séchet, V.; Miles, C.O.; Hess, P. Dissolved azaspiracids are absorbed and metabolized by blue mussels (Mytilus edulis). Toxicon 2013, 65, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Caillaud, A.; de la Iglesia, P.; Barber, E.; Eixarch, H.; Mohammad-Noor, N.; Yasumoto, T.; Diogène, J. Monitoring of dissolved ciguatoxin and maitotoxin using solid-phase adsorption toxin tracking devices: Application to Gambierdiscus pacificus in culture. Harmful Algae 2011, 10, 433–446. [Google Scholar] [CrossRef]
- García-Altares, M.; Casanova, A.; Bane, V.; Diogeǹe, J.; Furey, A.; De La Iglesia, P. Confirmation of pinnatoxins and spirolides in shellfish and passive samplers from Catalonia (Spain) by liquid chromatography coupled with triple quadrupole and high-resolution hybrid tandem mass spectrometry. Mar. Drugs 2014, 12, 3706–3732. [Google Scholar] [CrossRef] [PubMed]
- Roué, M.; Darius, H.T.; Viallon, J.; Ung, A.; Gatti, C.; Harwood, D.T.; Chinain, M. Application of solid phase adsorption toxin tracking (SPATT) devices for the field detection of Gambierdiscus toxins. Harmful Algae 2018, 71, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Fux, E.; Marcaillou, C.; Mondeguer, F.; Bire, R.; Hess, P. Field and mesocosm trials on passive sampling for the study of adsorption and desorption behaviour of lipophilic toxins with a focus on OA and DTX1. Harmful Algae 2008, 7, 574–583. [Google Scholar] [CrossRef]
- Zendong, Z.; Herrenknecht, C.; Abadie, E.; Brissard, C.; Tixier, C.; Mondeguer, F.; Séchet, V.; Amzil, Z.; Hess, P. Extended evaluation of polymeric and lipophilic sorbents for passive sampling of marine toxins. Toxicon 2014, 91, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Fux, E.; Bire, R.; Hess, P. Comparative accumulation and composition of lipophilic marine biotoxins in passive samplers and in mussels (M. edulis) on the West Coast of Ireland. Harmful Algae 2009, 8, 523–537. [Google Scholar] [CrossRef]
- Rundberget, T.; Gustad, E.; Samdal, I.A.; Sandvik, M.; Miles, C.O. A convenient and cost-effective method for monitoring marine algal toxins with passive samplers. Toxicon 2009, 53, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Fux, E.; Gonzalez-Gil, S.; Lunven, M.; Gentien, P.; Hess, P. Production of diarrhetic shellfish poisoning toxins and pectenotoxins at depths within and below the euphotic zone. Toxicon 2010, 56, 1487–1496. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Staedler, M.; Miller, W.A.; Toy-Choutka, S.; Dominik, C.; et al. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from land to sea otters. PLoS ONE 2010, 5, e12576. [Google Scholar] [CrossRef] [PubMed]
- Rundberget, T.; Aasen, J.A.B.; Selwood, A.I.; Miles, C.O. Pinnatoxins and spirolides in Norwegian blue mussels and seawater. Toxicon 2011, 58, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, G.; Moroño, Á.; Paz, B.; Franco, J.M.; Pazos, Y.; Reguera, B. Evaluation of passive samplers as a monitoring tool for early warning of dinophysis toxins in shellfish. Mar. Drugs 2013, 11, 3823–3845. [Google Scholar] [CrossRef] [PubMed]
- Gibble, C.M.; Kudela, R.M. Detection of persistent microcystin toxins at the land-sea interface in Monterey Bay, California. Harmful Algae 2014, 39, 146–153. [Google Scholar] [CrossRef]
- Zendong, Z.; Kadiri, M.; Herrenknecht, C.; Nézan, E.; Mazzeo, A.; Hess, P. Algal toxin profiles in Nigerian coastal waters (Gulf of Guinea) using passive sampling and liquid chromatography coupled to mass spectrometry. Toxicon 2016, 114, 16–27. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.; Van Pelt, F.N.A.M.; Bane, V.; O’Halloran, J.; Furey, A. Application of passive (SPATT) and active sampling methods in the profiling and monitoring of marine biotoxins. Toxicon 2014, 89, 77–86. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.; Bane, V.; García-Altares, M.; Van Pelt, F.N.A.M.; Furey, A.; O’Halloran, J. Assessment of emerging biotoxins (pinnatoxin G and spirolides) at Europe’s first marine reserve: Lough Hyne. Toxicon 2015, 108, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.Q.; Roddam, C.M.; Langlois, G.W.; Kudela, R.M. Application of Solid Phase Adsorption Toxin Tracking (SPATT) for field detection of the hydrophilic phycotoxins domoic acid and saxitoxin in coastal California. Limnol. Oceanogr. Methods 2010, 8, 645–660. [Google Scholar] [CrossRef]
- Kudela, R.M. Characterization and deployment of Solid Phase Adsorption Toxin Tracking (SPATT) resin for monitoring of microcystins in fresh and saltwater. Harmful Algae 2011, 11, 117–125. [Google Scholar] [CrossRef]
- Wood, S.A.; Holland, P.T.; MacKenzie, L. Development of solid phase adsorption toxin tracking (SPATT) for monitoring anatoxin-a and homoanatoxin-a in river water. Chemosphere 2011, 82, 888–894. [Google Scholar] [CrossRef] [PubMed]
- Zendong, Z.; McCarron, P.; Herrenknecht, C.; Sibat, M.; Amzil, Z.; Cole, R.B.; Hess, P. High resolution mass spectrometry for quantitative analysis and untargeted screening of algal toxins in mussels and passive samplers. J. Chromatogr. A 2015, 1416, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Li, F.-L.; Li, Z.-X.; Guo, M.-M.; Wu, H.-Y.; Zhang, T.-T.; Song, C.-H. Investigation of diarrhetic shellfish toxins in Lingshan Bay, Yellow Sea, China, using solid-phase adsorption toxin tracking (SPATT). Food Addit. Contam. Part A 2016, 33, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, G.; Paz, B.; Franco, J.M.; Suzuki, T.; Reguera, B. First detection of Pectenotoxin-11 and confirmation of OA-D8 diol-ester in Dinophysis acuta from European waters by LC-MS/MS. Toxicon 2008, 52, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Pizarro, G.; Paz, B.; González-Gil, S.; Franco, J.M.; Reguera, B. Seasonal variability of lipophilic toxins during a Dinophysis acuta bloom in Western Iberia: Differences between picked cells and plankton concentrates. Harmful Algae 2009, 8, 926–937. [Google Scholar] [CrossRef]
- Li, Z.; Mengmeng, G.; Shouguo, Y.; Qingyin, W.; Zhijun, T. Investigation of pectenotoxin profiles in the Yellow Sea (China) using a passive sampling technique. Mar. Drugs 2010, 8, 1263–1272. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, L.A.; Selwood, A.I.; McNabb, P.; Rhodes, L. Benthic dinoflagellate toxins in two warm-temperate estuaries: Rangaunu and Parengarenga Harbours, Northland, New Zealand. Harmful Algae 2011, 10, 559–566. [Google Scholar] [CrossRef]
- Li, A.; Ma, F.; Song, X.; Yu, R. Dynamic adsorption of diarrhetic shellfish poisoning (DSP) toxins in passive sampling relates to pore size distribution of aromatic adsorbent. J. Chromatogr. A 2011, 1218, 1437–1442. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Sun, G.; Qiu, J.; Ma, Q.; Hess, P.; Li, A. Effect of seawater salinity on pore-size distribution on a poly(styrene)-based HP20 resin and its adsorption of diarrhetic shellfish toxins. J. Chromatogr. A 2014, 1373, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Rundberget, T.; Sandvik, M.; Larsen, K.; Pizarro, G.M.; Reguera, B.; Castberg, T.; Gustad, E.; Loader, J.I.; Rise, F.; Wilkins, A.L.; et al. Extraction of microalgal toxins by large-scale pumping of seawater in Spain and Norway, and isolation of okadaic acid and dinophysistoxin-2. Toxicon 2007, 50, 960–970. [Google Scholar] [CrossRef] [PubMed]
- Qu, P.; Xu, Y.; Yang, J.; Gao, H.; Tong, M. Application of solid phase adsorption toxin tracking (SPATT) in the East China. In Proceedings of the 16th International Conference on Harmful Algae, Wellington, New Zealand, 27–31 October 2014; pp. 206–209. [Google Scholar]
- Touzet, N.; Lacaze, J.P.; Maher, M.; Turrell, E.; Raine, R. Summer dynamics of Alexandrium ostenfeldii (Dinophyceae) and spirolide toxins in Cork Harbour, Ireland. Mar. Ecol. Prog. Ser. 2011, 425, 21–33. [Google Scholar] [CrossRef]
- Kim, J.H.; Tillmann, U.; Adams, N.G.; Krock, B.; Stutts, W.L.; Deeds, J.R.; Han, M.S.; Trainer, V.L. Identification of Azadinium species and a new azaspiracid from Azadinium poporum in Puget Sound, Washington State, USA. Harmful Algae 2017, 68, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Valdiglesias, V.; Prego-Faraldo, M.V.; Paśaro, E.; Meńdez, J.; Laffon, B. Okadaic Acid: More than a diarrheic toxin. Mar. Drugs 2013, 11, 4328–4349. [Google Scholar] [CrossRef] [PubMed]
- Reguera, B.; Riobó, P.; Rodríguez, F.; Díaz, P.A.; Pizarro, G.; Paz, B.; Franco, J.M.; Blanco, J. Dinophysis toxins: Causative organisms, distribution and fate in shellfish. Mar. Drugs 2014, 12, 394–461. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Benford, D.; Cockburn, A.; Cravedi, J.P.; Dogliotti, E.; Di Domenico, A.; Galli, C. Scientific Opinion of the Panel on Contaminants in the Food Chain on a request from the European Commission on marine biotoxins in shellfish—Pectenotoxin group. EFSA J. 2009, 1109, 1–52. [Google Scholar] [CrossRef]
- Vale, C.; Botana, L.M. Marine toxins and the cytoskeleton: Okadaic acid and dinophysistoxins. FEBS J. 2008, 275, 6060–6066. [Google Scholar] [CrossRef] [PubMed]
- García, C.; Truan, D.; Lagos, M.; Santelices, J.P.; Díaz, J.C.; Lagos, N. Metabolic Transformation of Dinophysistoxin-3 into Dinophysistoxin-1 Causes Human Intoxication by Consumption of O-Acyl-Derivatives Dinophysistoxins Contaminated Shellfish. J. Toxicol. Sci. 2005, 30, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Espiña, B.; Rubiolo, J.A. Marine toxins and the cytoskeleton: Pectenotoxins, unusual macrolides that disrupt actin. FEBS J. 2008, 275, 6082–6088. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Wilkins, A.L.; Munday, R.; Dines, M.H.; Hawkes, A.D.; Briggs, L.R.; Sandvik, M.; Jensen, D.J.; Cooney, J.M.; Holland, P.T.; et al. Isolation of pectenotoxin-2 from Dinophysis acuta and its conversion to pectenotoxin-2 seco acid, and preliminary assessment of their acute toxicities. Toxicon 2004, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Satake, M.; MacKenzie, L.; Yasumoto, T. Identification of Protoceratium reticulatum as the biogenetic origin of yessotoxin. Nat. Toxins 1997, 5, 164–167. [Google Scholar] [CrossRef] [PubMed]
- Paz, B.; Riobó, P.; Luisa Fernández, M.; Fraga, S.; Franco, J.M. Production and release of yessotoxins by the dinoflagellates Protoceratium reticulatum and Lingulodinium polyedrum in culture. Toxicon 2004, 44, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.; McNabb, P.; De Salas, M.; Briggs, L.; Beuzenberg, V.; Gladstone, M. Yessotoxin production by Gonyaulax spinifera. Harmful Algae 2006, 5, 148–155. [Google Scholar] [CrossRef]
- Álvarez, G.; Uribe, E.; Regueiro, J.; Blanco, J.; Fraga, S. Gonyaulax taylorii, a new yessotoxins-producer dinoflagellate species from Chilean waters. Harmful Algae 2016, 58, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Tubaro, A.; Dell’Ovo, V.; Sosa, S.; Florio, C. Yessotoxins: A toxicological overview. Toxicon 2010, 56, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Aune, T.; Sorby, R.; Yasumoto, T.; Ramstad, H.; Landsverk, T. Comparison of oral and intraperitoneal toxicity of yessotoxins towards mice. Toxicon 2002, 40, 77–82. [Google Scholar] [CrossRef]
- De Wit, P.; Rogers-Bennett, L.; Kudela, R.M.; Palumbi, S.R. Forensic genomics as a novel tool for identifying the causes of mass mortality events. Nat. Commun. 2014, 5, 3652. [Google Scholar] [CrossRef] [PubMed]
- Satake, M.; Ofuji, K.; Naoki, H.; James, K.; Furey, A.; McMahon, T.; Silke, J.; Yasumoto, T. New toxic event caused by Irish mussels. In Harmful Algae, Proceedings of the VIII International Conference on Harmful Algae, Vigo, Spain, 25–29 June 1997; Reguera, B., Blanco, J., Fernandez, M., Wyatt, T., Eds.; Xunta de Galicia and IOC of UNESCO: Santiago de Compostela, Spain, 1997; pp. 468–469. [Google Scholar]
- Satake, M.; Ofuji, K.; Naoki, H.; James, K.J.; Furey, A.; McMahon, T.; Silke, J.; Yasumoto, T. Azaspiracid, a New Marine Toxin Having Unique Spiro Ring Assemblies, Isolated from Irish Mussels, Mytilus edulis. J. Am. Chem. Soc. 1998, 120, 9967–9968. [Google Scholar] [CrossRef]
- Tillmann, U.; Jaén, D.; Fernández, L.; Gottschling, M.; Witt, M.; Blanco, J.; Krock, B. Amphidoma languida (Amphidomatacea, Dinophyceae) with a novel azaspiracid toxin profile identified as the cause of molluscan contamination at the Atlantic coast of southern Spain. Harmful Algae 2017, 62, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Furey, A.; O’Doherty, S.; O’Callaghan, K.; Lehane, M.; James, K.J. Azaspiracid poisoning (AZP) toxins in shellfish: Toxicological and health considerations. Toxicon 2010, 56, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Chevallier, O.P.; Graham, S.F.; Alonso, E.; Duffy, C.; Silke, J.; Campbell, K.; Botana, L.M.; Elliott, C.T. New insights into the causes of human illness due to consumption of azaspiracid contaminated shellfish. Sci. Rep. 2015, 5, 9818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vilariño, N. Marine toxins and the cytoskeleton: Azaspiracids. FEBS J. 2008, 275, 6075–6081. [Google Scholar] [CrossRef] [PubMed]
- James, K.J.; Fidalgo Sáez, M.J.; Furey, A.; Lehane, M. Azaspiracid poisoning, the food-borne illness associated with shellfish consumption. Food Addit. Contam. 2004, 21, 879–892. [Google Scholar] [CrossRef] [PubMed]
- Colman, J.R.; Twiner, M.J.; Hess, P.; McMahon, T.; Satake, M.; Yasumoto, T.; Doucette, G.J.; Ramsdell, J.S. Teratogenic effects of azaspiracid-1 identified by microinjection of Japanese medaka (Oryzias latipes) embryos. Toxicon 2005, 45, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Jauffrais, T.; Contreras, A.; Herrenknecht, C.; Truquet, P.; Séchet, V.; Tillmann, U.; Hess, P. Effect of Azadinium spinosum on the feeding behaviour and azaspiracid accumulation of Mytilus edulis. Aquat. Toxicol. 2012, 124–125, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Stivala, C.E.; Benoit, E.; Araoz, R.; Servent, D.; Novikov, A.; Molgó, J.; Zakarian, A. Synthesis and Biology of Cyclic Imine Toxins, An Emerging Class of Potent, Globally Distributed Marine Toxins. Nat. Prod. Rep. 2015, 32, 411–435. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.; Smith, K.; Selwood, A.; McNabb, P.; Munday, R.; Suda, S.; Molenaar, S.; Hallegraeff, G. Dinoflagellate Vulcanodinium rugosum identified as the causative organism of pinnatoxins in Australia, New Zealand and Japan. Phycologia 2011, 50, 624–628. [Google Scholar] [CrossRef]
- Seki, T.; Satake, M.; Mackenzie, L.; Kaspar, H.F.; Yasumoto, T. Gymnodimine, a new marine toxin of unprecedented structure isolated from New Zealand oysters and the dinoflagellate, Gymnodinium sp. Tetrahedron Lett. 1995, 36, 7093–7096. [Google Scholar] [CrossRef]
- Miles, C.O.; Wilkins, A.L.; Stirling, D.J.; MacKenzie, A.L. Gymnodimine C, an Isomer of Gymnodimine B, from Karenia selliformis. J. Agric. Food Chem. 2003, 51, 4838–4840. [Google Scholar] [CrossRef] [PubMed]
- Van Wagoner, R.M.; Misner, I.; Tomas, C.R.; Wright, J.L.C. Occurrence of 12-methylgymnodimine in a spirolide-producing dinoflagellate Alexandrium peruvianum and the biogenetic implications. Tetrahedron Lett. 2011, 52, 4243–4246. [Google Scholar] [CrossRef]
- Harju, K.; Koskela, H.; Kremp, A.; Suikkanen, S.; De La Iglesia, P.; Miles, C.O.; Krock, B.; Vanninen, P. Identification of gymnodimine D and presence of gymnodimine variants in the dinoflagellate Alexandrium ostenfeldii from the Baltic Sea. Toxicon 2016, 112, 68–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cembella, A.D.; Lewis, N.I.; Quilliam, M.A. The marine dinoflagellate Alexandrium ostenfeldii (Dinophyceae) as the causative organism of spirolide shellfish toxins. Phycologia 2000, 39, 67–74. [Google Scholar] [CrossRef]
- Touzet, N.; Franco, J.M.; Raine, R. Morphogenetic diversity and biotoxin composition of Alexandrium (Dinophyceae) in Irish coastal waters. Harmful Algae 2008, 7, 782–797. [Google Scholar] [CrossRef]
- Munday, R.; Towers, N.R.; Mackenzie, L.; Beuzenberg, V.; Holland, P.T.; Miles, C.O. Acute toxicity of gymnodimine to mice. Toxicon 2004, 44, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Munday, R.; Quilliam, M.A.; LeBlanc, P.; Lewis, N.; Gallant, P.; Sperker, S.A.; Stephen Ewart, H.; MacKinnon, S.L. Investigations into the toxicology of spirolides, a group of marine phycotoxins. Toxins 2012, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.; Smith, K.; Munday, R.; Hallegraeff, G.M.; Selwood, A.; Molenaar, S.; McNabb, P.; Adamson, J.; Wilkinson, C. Potency of pinnatoxins produced by dinoflagellates isolated from New Zealand and South Australia. In Proceedings of the 14th International Conference on Harmful Algae, Hersonissos, Greece, 1–5 November 2010; pp. 209–211. [Google Scholar]
- Litaker, R.W.; Holland, W.C.; Hardison, D.R.; Pisapia, F.; Hess, P.; Kibler, S.R.; Tester, P.A. Ciguatoxicity of Gambierdiscus and Fukuyoa species from the Caribbean and Gulf of Mexico. PLoS ONE 2017, 12, e0185776. [Google Scholar] [CrossRef] [PubMed]
- Caillaud, A.; De La Iglesia, P.; Darius, H.T.; Pauillac, S.; Aligizaki, K.; Fraga, S.; Chinain, M.; Diogène, J. Update on methodologies available for ciguatoxin determination: Perspectives to confront the onset of ciguatera fish poisoning in Europe. Mar. Drugs 2010, 8, 1838–1907. [Google Scholar] [CrossRef] [PubMed]
- Dickey, R.W.; Plakas, S.M. Ciguatera: A public health perspective. Toxicon 2010, 56, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Davin, W.T.; Kohler, C.C.; Tindall, D.R. Ciguatera Toxins Adversely Affect Piscivorous Fishes. Trans. Am. Fish. Soc. 1988, 117, 374–384. [Google Scholar] [CrossRef]
- Ledreux, A.; Brand, H.; Chinain, M.; Bottein, M.Y.D.; Ramsdell, J.S. Dynamics of ciguatoxins from Gambierdiscus polynesiensis in the benthic herbivore Mugil cephalus: Trophic transfer implications. Harmful Algae 2014, 39, 165–174. [Google Scholar] [CrossRef]
- Lewis, R.J. Ciguatoxins are potent ichthyotoxins. Toxicon 1992, 30, 207–211. [Google Scholar] [CrossRef]
- Colman, J.R.; Dechraoui, M.Y.B.; Dickey, R.W.; Ramsdell, J.S. Characterization of the developmental toxicity of Caribbean ciguatoxins in finfish embryos. Toxicon 2004, 44, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Mak, Y.L.; Li, J.; Liu, C.N.; Cheng, S.H.; Lam, P.K.S.; Cheng, J.; Chan, L.L. Physiological and behavioural impacts of Pacific ciguatoxin-1 (P-CTX-1) on marine medaka (Oryzias melastigma). J. Hazard. Mater. 2017, 321, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Darius, H.T.; Ponton, D.; Revel, T.; Cruchet, P.; Ung, A.; Tchou Fouc, M.; Chinain, M. Ciguatera risk assessment in two toxic sites of French Polynesia using the receptor-binding assay. Toxicon 2007, 50, 612–626. [Google Scholar] [CrossRef] [PubMed]
- Darius, H.T.; Roué, M.; Sibat, M.; Viallon, J.; Gatti, C.M.I.; Vandersea, M.W.; Tester, P.A.; Litaker, R.W.; Amzil, Z.; Hess, P.; et al. Tectus niloticus (Tegulidae, Gastropod) as a Novel Vector of Ciguatera Poisoning: Detection of Pacific Ciguatoxins in Toxic Samples from Nuku Hiva Island (French Polynesia). Toxins 2018, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Hermann, G.; Lacaze, J.; Morley, L.; Drago, G.; Keady, E.; Cabado, A.; Gubbins, M.; Turrell, E. An investigation into solid phase adsorption toxin tracking (SPATT) for the detection of domoic acid, the amnesic shellfish poison, in seawater. In Proceedings of the 14th International Conference on Harmful Algae, Hersonissos, Greece, 1–5 November 2010; pp. 200–202. [Google Scholar]
- Peacock, M.B.; Gibble, C.M.; Senn, D.B.; Cloern, J.E.; Kudela, R.M. Blurred lines: Multiple freshwater and marine algal toxins at the land-sea interface of San Francisco Bay, California. Harmful Algae 2018, 73, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, P.; Alfonso, A.; Turrell, E.; Lacaze, J.P.; Botana, L.M. Study of solid phase adsorption of paralytic shellfish poisoning toxins (PSP) onto different resins. Harmful Algae 2011, 10, 447–455. [Google Scholar] [CrossRef]
- Howard, M.D.A.; Nagoda, C.; Kudela, R.M.; Hayashi, K.; Tatters, A.; Caron, D.A.; Busse, L.; Brown, J.; Sutula, M.; Stein, E.D. Microcystin prevalence throughout lentic waterbodies in coastal southern California. Toxins 2017, 9, 231. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Qiu, J.; Fan, H.; Li, A. Mechanism and application of solid phase adsorption toxin tracking for monitoring microcystins. J. Chromatogr. A 2013, 1300, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.A.; Kuhajek, J.M.; de Winton, M.; Phillips, N.R. Species composition and cyanotoxin production in periphyton mats from three lakes of varying trophic status. FEMS Microbiol. Ecol. 2012, 79, 312–326. [Google Scholar] [CrossRef] [PubMed]
- Bouma-Gregson, K.; Higgins, P. Cyanobacteria and Cyanotoxins in the Eel River, 2013–2014; University of California: Berkeley, CA, USA, 2015. [Google Scholar]
- Lelong, A.; Hégaret, H.; Soudant, P.; Bates, S.S. Pseudo-nitzschia (Bacillariophyceae) species, domoic acid and amnesic shellfish poisoning: Revisiting previous paradigms. Phycologia 2012, 51, 168–216. [Google Scholar] [CrossRef]
- Lefebvre, K.A.; Robertson, A. Domoic acid and human exposure risks: A review. Toxicon 2010, 56, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Hampson, D.R.; Manalo, J.L. The activation of glutamate receptors by kainic acid and domoic acid. Nat. Toxins 1998, 6, 153–158. [Google Scholar] [CrossRef]
- Jones, T.O.; Whyte, J.N.C.; Townsend, L.D.; Ginther, N.G.; Iwama, G.K. Effects of domoic acid on haemolymph pH, PCO2 and PO2 in the Pacific oyster, Crassostrea gigas and the California mussel, Mytilus californianus. Aquat. Toxicol. 1995, 31, 43–55. [Google Scholar] [CrossRef]
- Wiese, M.; D’Agostino, P.M.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic alkaloids: Saxitoxin and its analogs. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef] [PubMed]
- Lagos, N.W.; Andrinolo, D. Paralytic shellfish poisoning (PSP): Toxicology and kinetics. In Seafood and Freshwater Toxins: Pharmacology, Physiology and Detection; Botana, L., Ed.; Marcel Dekker: New York, NY, USA, 2000; pp. 203–215. [Google Scholar]
- Lassus, P.; Chomérat, N.; Hess, P.; Elisabeth, N. Toxic and Harmful Microalgae of the World Ocean; Denmark, International Society for the Study of Harmful Algae/Intergovernmental Oceanographic Commission of UNESCO; IOC Manuals and Guides 68; UNESCO: Paris, France, 2016; ISBN 978-87-990827-6-6. [Google Scholar]
- Gehringer, M.M. Microcystin-LR and okadaic acid-induced cellular effects: A dualistic response. FEBS Lett. 2004, 557, 1–8. [Google Scholar] [CrossRef]
- Van Dolah, F.M. Diversity of Marine and Freshwater Algal Toxins. In Seafood and Freshwater Toxins; CRC Press: Boca Raton, FL, USA, 2000; ISBN 978-0-8247-8956-5. [Google Scholar]
- Azevedo, S.M.F.O.; Carmichael, W.W.; Jochimsen, E.M.; Rinehart, K.L.; Lau, S.; Shaw, G.R.; Eaglesham, G.K. Human intoxication by microcystins during renal dialysis treatment in Caruaru—Brazil. Toxicology 2002, 181–182, 441–446. [Google Scholar] [CrossRef]
- Vidal, F.; Sedan, D.; D’Agostino, D.; Cavalieri, M.L.; Mullen, E.; Parot Varela, M.M.; Flores, C.; Caixach, J.; Andrinolo, D. Recreational exposure during algal bloom in Carrasco Beach, Uruguay: A liver failure case report. Toxins (Basel) 2017, 9, 267. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Jiang, X.; Manubolu, M.; Riedl, K.; Ludsin, S.A.; Martin, J.F.; Lee, J. Fresh produce and their soils accumulate cyanotoxins from irrigation water: Implications for public health and food security. Food Res. Int. 2017, 102, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, W.W.; Eschedor, J.T.; Patterson, G.M.; Moore, R.E. Toxicity and partial structure of a hepatotoxic peptide produced by the cyanobacterium Nodularia spumigena Mertens emend. L575 from New Zealand. Appl. Environ. Microbiol. 1988, 54, 2257–2263. [Google Scholar] [PubMed]
- Ohta, T.; Sueoka, E.; Iida, N.; Komori, A.; Suganuma, M.; Nishiwaki, R.; Tatematsu, M.; Kim, S.J.; Carmichael, W.W.; Fujiki, H. Nodularin, a potent inhibitor of protein phosphatase-1 and phosphatase-2A, is a new environmental carcinogen in male F344 rat-liver. Cancer Res. 1994, 54, 6402–6406. [Google Scholar] [PubMed]
- Harding, W.R.; Rowe, N.; Wessels, J.C.; Beattie, K.A.; Codd, G.A. Death of a dog attributed to the cyanobacterial (blue-green algal) hepatotoxin nodularin in South Africa. J. S. Afr. Vet. Assoc. 1995, 66, 256–259. [Google Scholar] [PubMed]
- Mazur-Marzec, H.; Tyminska, A.; Szafranek, J.; Plinski, M. Accumulation of nodularin in sediments, mussels, and fish from the Gulf of Gdansk, southern Baltic Sea. Environ. Toxicol. 2007, 22, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Méjean, A.; Peyraud-Thomas, C.; Kerbrat, A.S.; Golubic, S.; Pauillac, S.; Chinain, M.; Laurent, D. First identification of the neurotoxin homoanatoxin-a from mats of Hydrocoleum lyngbyaceum (marine cyanobacterium) possibly linked to giant clam poisoning in New Caledonia. Toxicon 2010, 56, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Osswald, J.; Rellán, S.; Gago, A.; Vasconcelos, V. Toxicology and detection methods of the alkaloid neurotoxin produced by cyanobacteria, anatoxin-a. Environ. Int. 2007, 33, 1070–1089. [Google Scholar] [CrossRef] [PubMed]
- Mez, K.; Beattie, K.; Codd, G.; Hanselmann, K.; Hauser, B.; Naegeli, H.; Preisig, H. Identification of a microcystin in benthic cyanobacteria linked to cattle deaths on alpine pastures in Switzerland. Eur. J. Phycol. 1997, 32, 111–117. [Google Scholar] [CrossRef]
- Wood, S.A.; Selwood, A.I.; Rueckert, A.; Holland, P.T.; Milne, J.R.; Smith, K.F.; Smits, B.; Watts, L.F.; Cary, C.S. First report of homoanatoxin-a and associated dog neurotoxicosis in New Zealand. Toxicon 2007, 50, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, T.; Aritake, S.; Yasumoto, T. Mechanisms underlying the hemolytic and ichthyotoxic activities of maitotoxin. Nat. Toxins 1999, 7, 71–79. [Google Scholar] [CrossRef]
- Holmes, M.J.; Lewis, R.J. Purification and characterisation of large and small maitotoxins from cultured Gambierdiscus toxicus. Nat. Toxins 1994, 2, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Pisapia, F.; Sibat, M.; Herrenknecht, C.; Lhaute, K.; Gaiani, G.; Ferron, P.-J.; Fessard, V.; Fraga, S.; Nascimento, S.M.; Litaker, R.W.; et al. Maitotoxin-4, a Novel MTX Analog Produced by Gambierdiscus excentricus. Mar. Drugs 2017, 15, 220. [Google Scholar] [CrossRef] [PubMed]
- Kohli, G.S.; Papiol, G.G.; Rhodes, L.L.; Harwood, D.T.; Selwood, A.; Jerrett, A.; Murray, S.A.; Neilan, B.A. A feeding study to probe the uptake of Maitotoxin by snapper (Pagrus auratus). Harmful Algae 2014, 37, 125–132. [Google Scholar] [CrossRef]
- Smith, K.F.; Rhodes, L.; Verma, A.; Curley, B.G.; Harwood, D.T.; Kohli, G.S.; Solomona, D.; Rongo, T.; Munday, R.; Murray, S.A. A new Gambierdiscus species (Dinophyceae) from Rarotonga, Cook Islands: Gambierdiscus cheloniae sp. nov. Harmful Algae 2016, 60, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.; Smith, K.F.; Verma, A.; Curley, B.G.; Harwood, D.T.; Murray, S.; Kohli, G.S.; Solomona, D.; Rongo, T.; Munday, R.; et al. A new species of Gambierdiscus (Dinophyceae) from the south-west Pacific: Gambierdiscus honu sp. nov. Harmful Algae 2017, 65, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W.; Hall, N.S.; Calandrino, E.S. Controlling harmful cyanobacterial blooms in a world experiencing anthropogenic and climatic-induced change. Sci. Total Environ. 2011, 409, 1739–1745. [Google Scholar] [CrossRef] [PubMed]
- Hinder, S.L.; Hays, G.C.; Edwards, M.; Roberts, E.C.; Walne, A.W.; Gravenor, M.B. Changes in marine dinoflagellate and diatom abundance under climate change. Nat. Clim. Chang. 2012, 2, 271–275. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Harmful Cyanobacterial Blooms: Causes, Consequences, and Controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef] [PubMed]

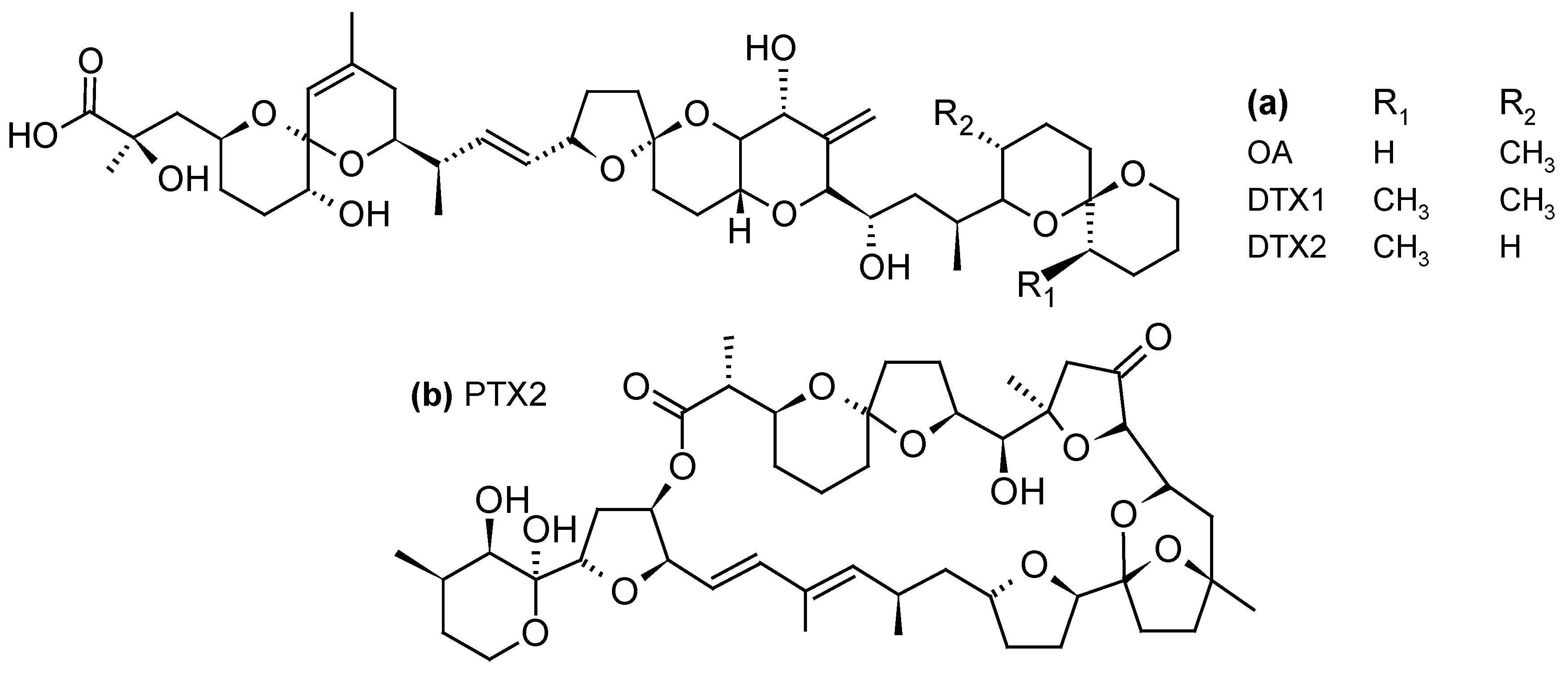
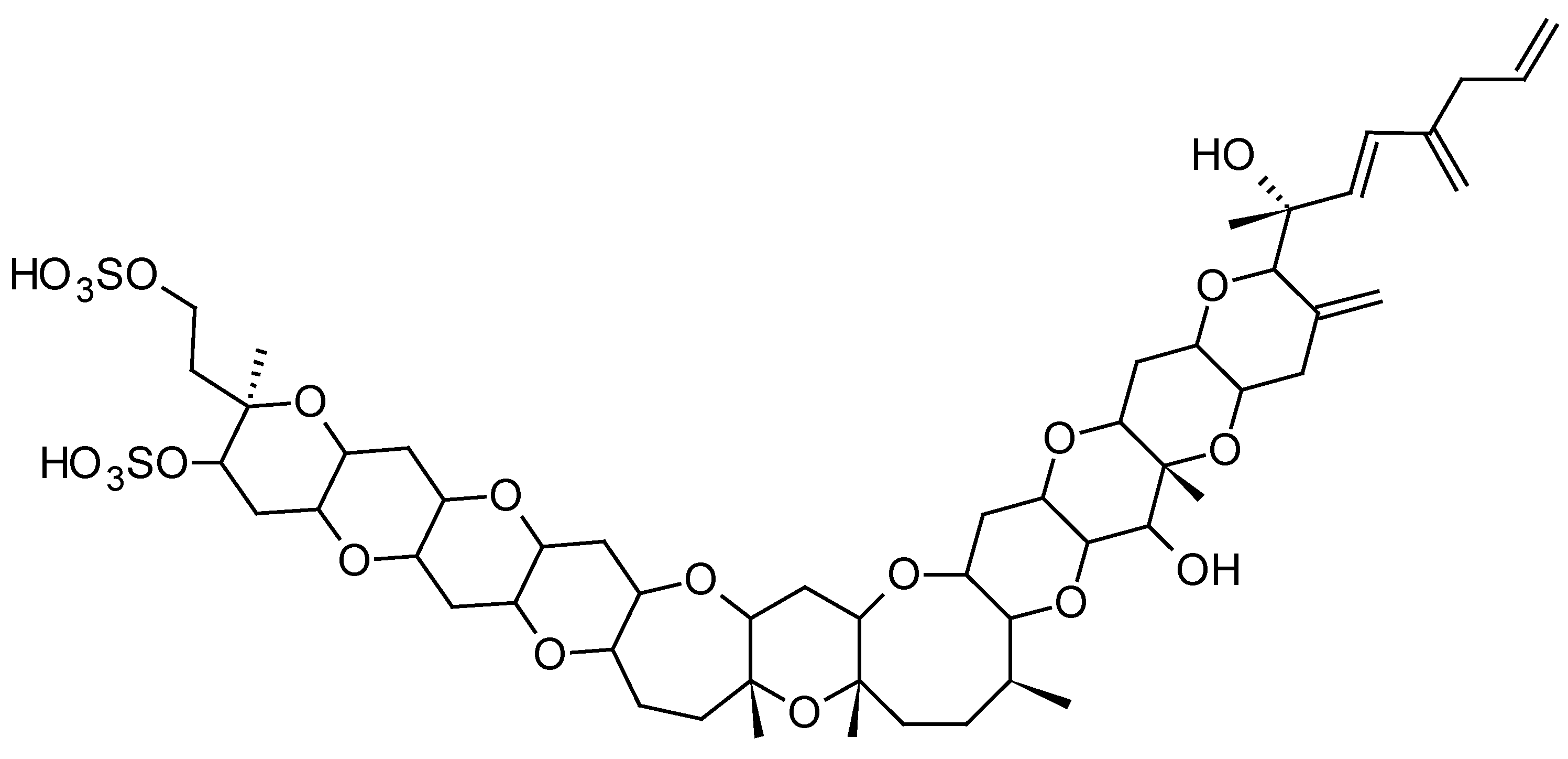
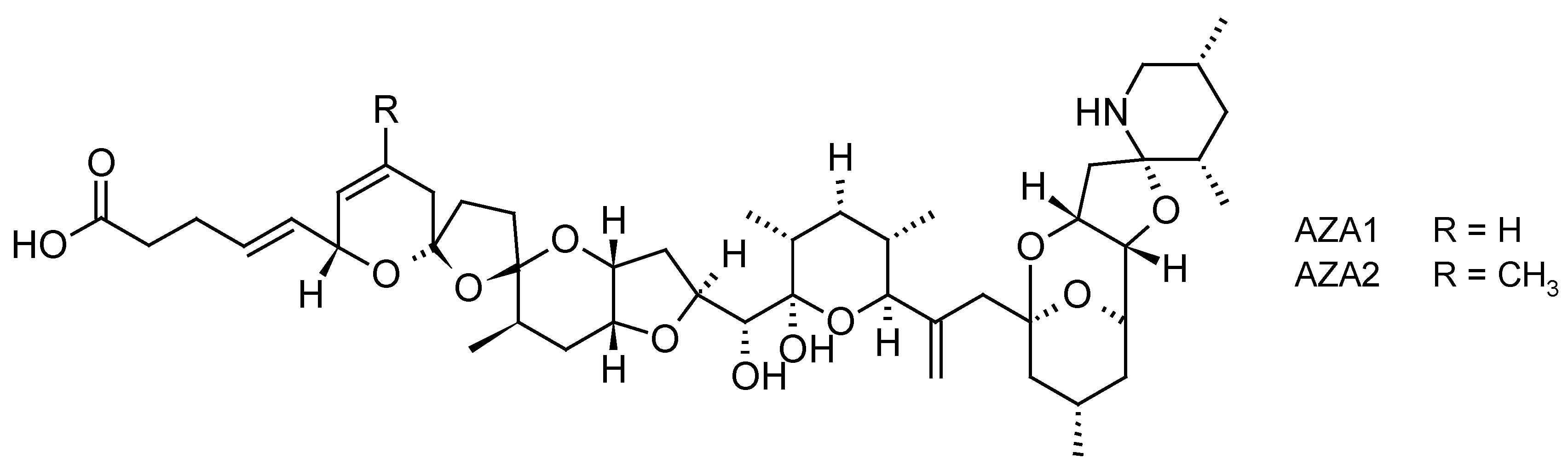
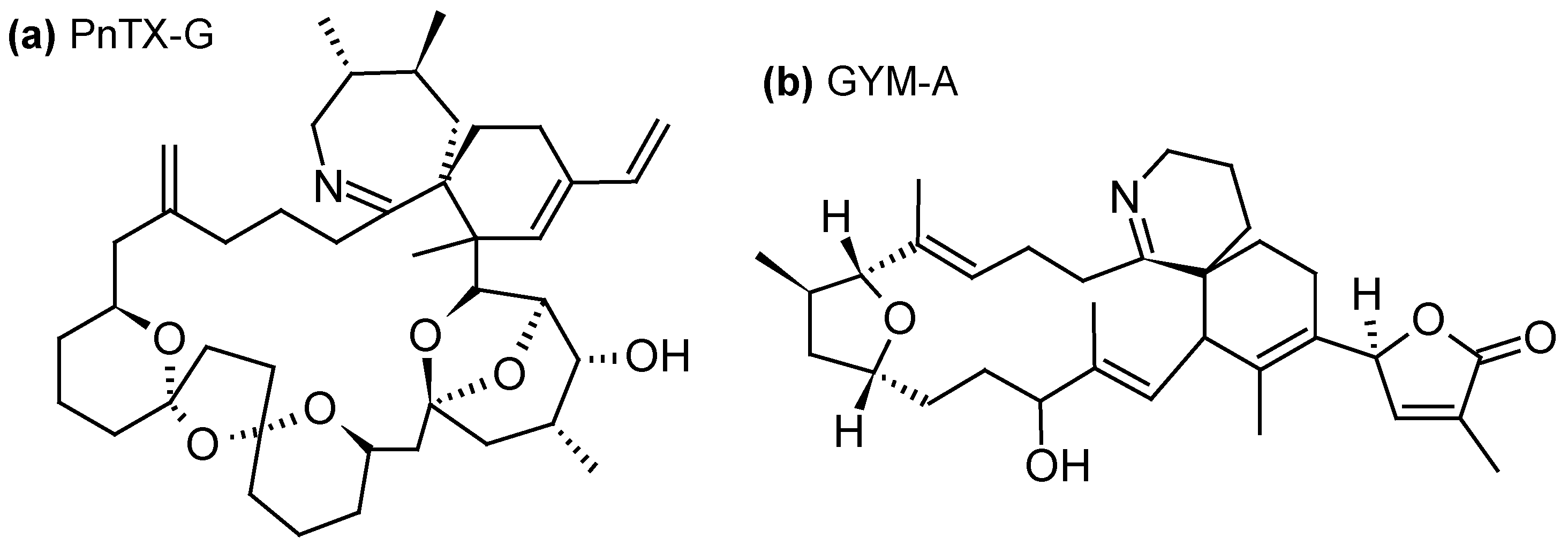


| Toxins Detected | Adsorbent Resins Tested | References |
|---|---|---|
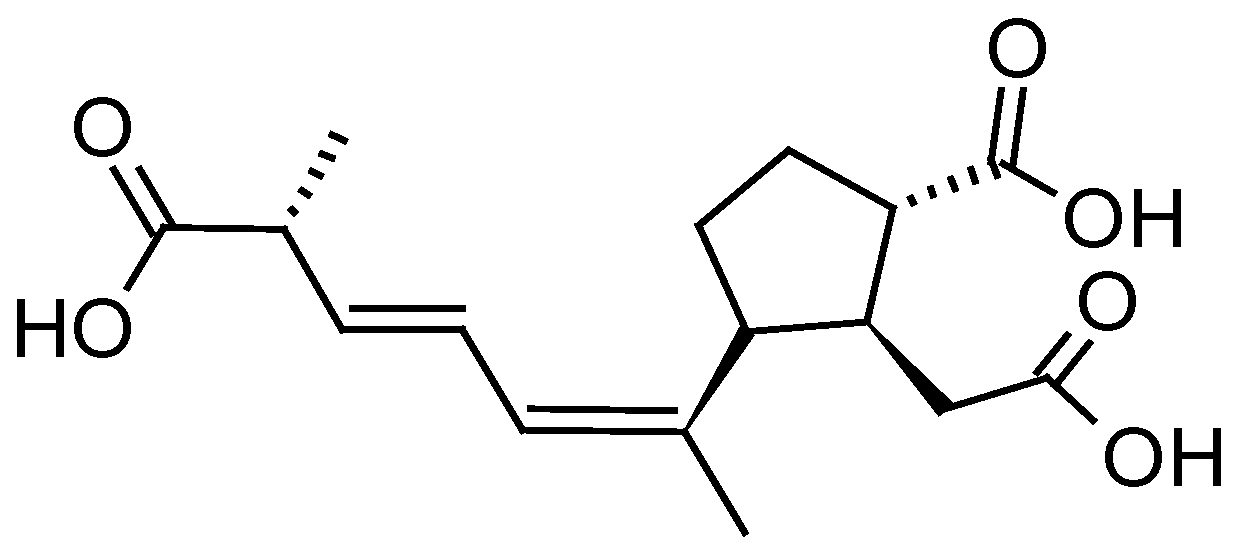
| DSP toxins and PTXs -OA, OA-D8 -DTX1, DTX2 -PTX2, PTX2sa, 7-epi-PTX2sa, PTX12, a PTX1 isomer, a PTX3 isomer, a PTX-11 isomer | -Diaion® HP20, HP2MG -Sepabeads® SP207, SP700, SP850, SP825L -Amberlite® XAD4, XAD761 -Dowex® Optipore® L-493 -Oasis® HLB -Strata-X® | [21,23,33,34,35,36,37,40,42,43,48,49,50,51,52,53,54,55,56,57,58] |
| YTXs YTX, homoYTX | -Diaion® HP20, HP2MG -Sepabeads® SP207 | [21,35,48,49,57] |
| AZP toxins AZA1, AZA2, AZA3, AZA-59 | -Diaion® HP20 -Sepabeads® SP700 -Oasis® HLB -Strata-X® | [33,34,35,36,48,58,59] |
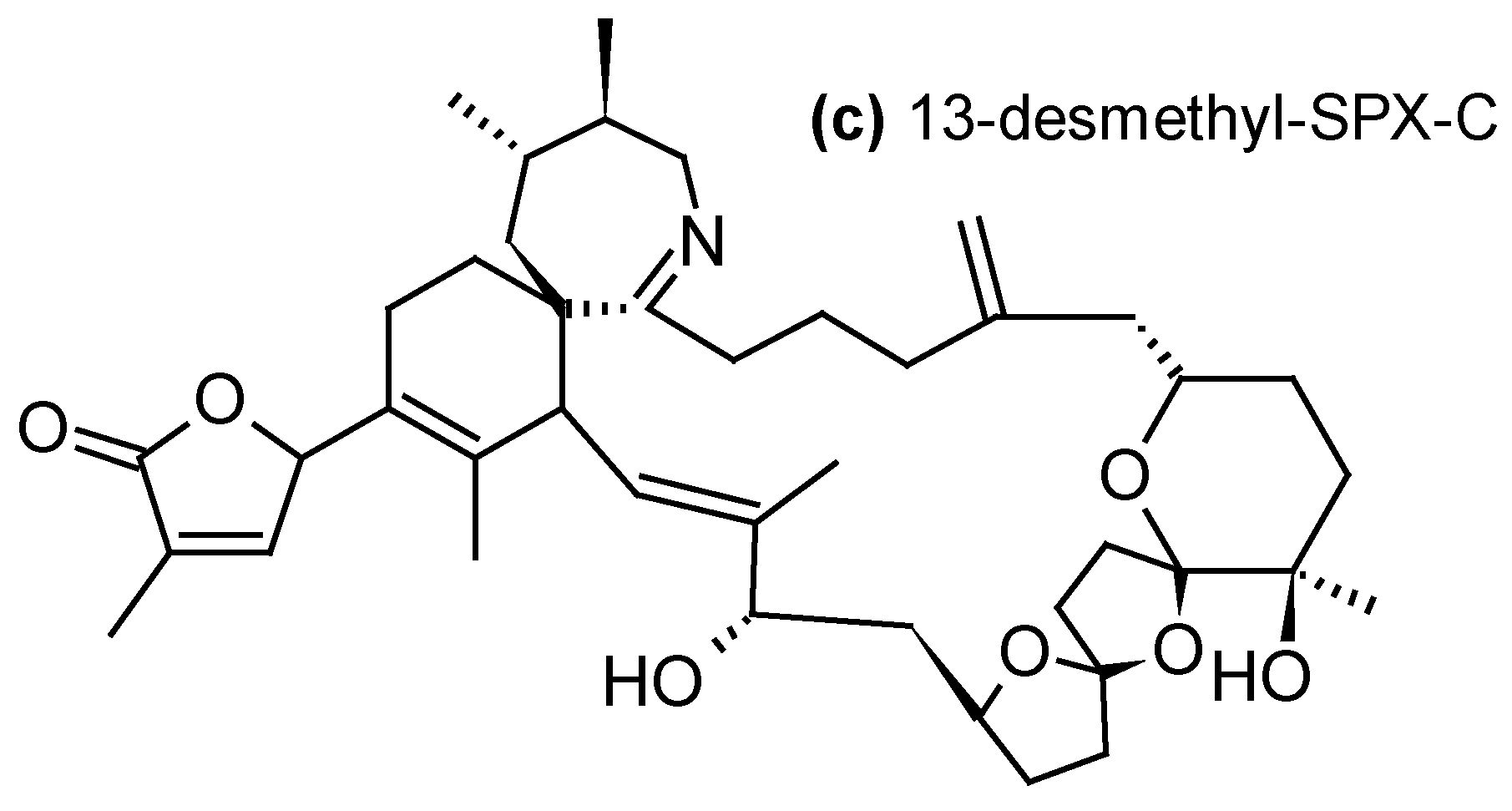
| Cyclic imines -13-desmethyl-SPX-C, 13,19-didesmethyl-SPX-C, iso-SPX-C, 20-methyl-SPX-G- PnTX-E, PnTX-F, PnTX-G-GYM-A | -Diaion® HP20 -Sepabeads® SP700 -Amberlite® XAD761 -Oasis® HLB -Strata-X® | [23,31,34,35,36,39,44,48,53,57,58] |
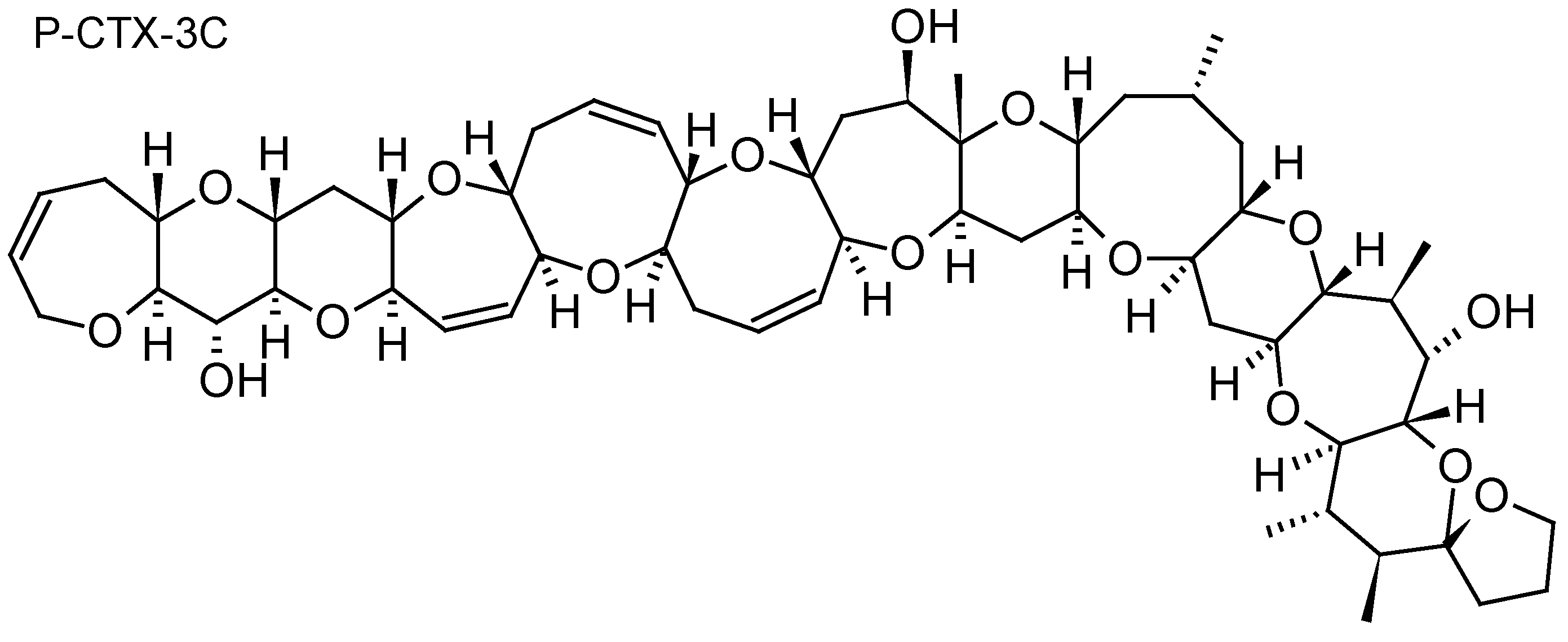
| Ciguatoxins P-CTX-1B, P-CTX-3C, P-CTX-3B, iso-P-CTX-3B/C | Diaion® HP20 | [30,32] |
| Toxins Detected | Adsorbent Resins Tested | References |
|---|---|---|
| ASP toxin DA | -Diaion® HP20 -Sepabeads® SP207, SP207SS, SP700 | [45,48,104,105] |
| PSP toxins -STX, neo-STX -GTX1, GTX2, GTX3, GTX4 -C1, C2 | -Diaion® HP20 -Sepabeads® SP700 -Computationally designed polymer | [45,106] |
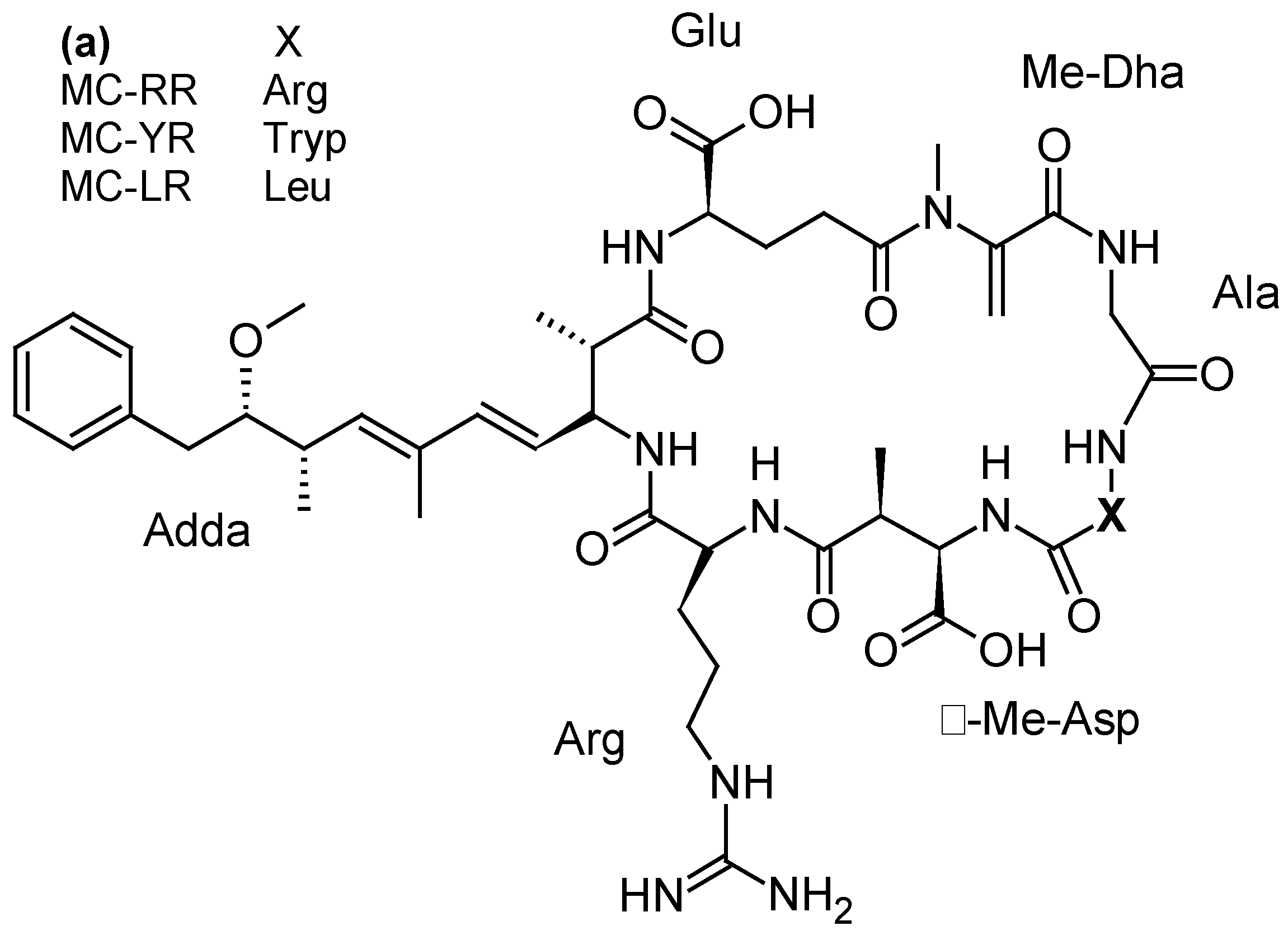
| MCs and NODs MC-LR, MC-YR, MC-RR, MC-LA, MC-LF, MC-Desmethyl-LR, [Dha7] MC-LR, NOD | -Diaion® HP20 -Sepabeads® SP700 | [38,41,46,105,107,108,109] |
| ANTX | -(Norit® GAC -830, -1020, -1240) -Darco® PAC G-60 -(Carbograph) -WCX -Strata –X®, -X-C, -X-CW -Oasis® MCX -AG® 50W-X4, (501-X8) -(Diaion® HP20) -Amberlite® IRP-64 | [47,110] |
| CFP toxins MTX-1, putative MTX-3 | Diaion® HP20 | [30,32] |
| Into brackets: resins that were not effective for the targeted toxins | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roué, M.; Darius, H.T.; Chinain, M. Solid Phase Adsorption Toxin Tracking (SPATT) Technology for the Monitoring of Aquatic Toxins: A Review. Toxins 2018, 10, 167. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins10040167
Roué M, Darius HT, Chinain M. Solid Phase Adsorption Toxin Tracking (SPATT) Technology for the Monitoring of Aquatic Toxins: A Review. Toxins. 2018; 10(4):167. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins10040167
Chicago/Turabian StyleRoué, Mélanie, Hélène Taiana Darius, and Mireille Chinain. 2018. "Solid Phase Adsorption Toxin Tracking (SPATT) Technology for the Monitoring of Aquatic Toxins: A Review" Toxins 10, no. 4: 167. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins10040167







