Discovery of the Gene Encoding a Novel Small Serum Protein (SSP) of Protobothrops flavoviridis and the Evolution of SSPs
Abstract
:1. Introduction
2. Results and Discussion
2.1. Discovery of the Gene Encoding a Novel SSP, PfSSP-6, Far Upstream of the Array of Five PfSSP Genes
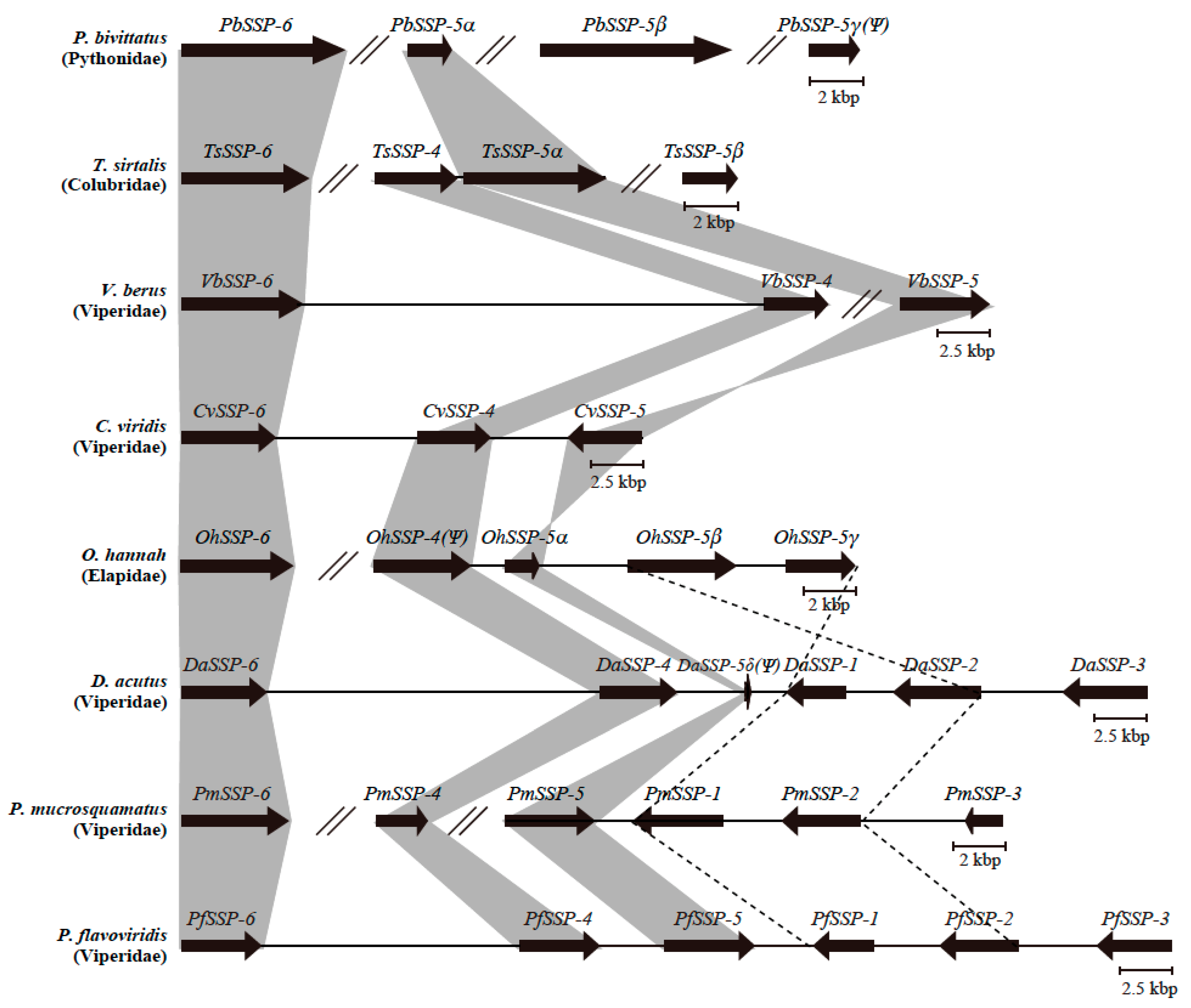
2.2. Sequence Configurations Classify the Six PfSSPs Into Three Subgroups
2.3. Configurations of SSP Paralogs Relevant to Snake Habitat Conditions
2.4. Diversified SSPs Acquired by Advanced Snakes Have More Complex Venom Compositions
3. Materials and Methods
3.1. Materials
3.2. Cloning and Sequencing of the Genome Segment Containing PfSSP-6
3.3. RepeatMasker Analysis of the Nucleotide Sequence of PfSSP-6
3.4. Determination of Nucleotide Sequences and Chromosomal Configurations of Genes Encoding Orthologs of PfSSPs from Seven Snake Taxa
3.5. Determining the Nucleotide Sequences of SSP Paralogs of P. mucrosquamatus and O. hannah
3.6. Expression Analysis of PfSSP-6 mRNA Using Semi-Quantitative RT-PCR
3.7. Mathematical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Aird, S.D.; Watanabe, Y.; Villar-Briones, A.; Roy, M.C.; Terada, K.; Mikheyev, A.S. Quantitative high-throughput profiling of snake venom gland transcriptomes and proteomes (Ovophis okinavensis and Protobothrops flavoviridis). BMC Genomics 2013, 14, 790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodrigues, R.S.; Boldrini-França, J.; Fonseca, F.P.P.; de la Torre, P.; Henrique-Silva, F.; Sanz, L.; Calvete, J.J.; Rodrigues, V.M. Combined snake venomics and venom gland transcriptomic analysis of Bothropoides pauloensis. J. Proteomics 2012, 75, 2707–2720. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; Margres, M.J. The genesis of an exceptionally lethal venom in the timber rattlesnake (Crotalus horridus) revealed through comparative venom-gland transcriptomics. BMC Genomics 2013, 14, 394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunie, S.; Bolin, J.; Gewirth, D.; Sigler, P.B. The refined crystal structure of dimeric phospholipase A2 at 2.5 Å Access to a shielded catalytic center. J. Biol. Chem. 1985, 260, 9742–9749. [Google Scholar] [PubMed]
- Holland, D.R.; Clancy, L.L.; Muchmore, S.W.; Ryde, T.J.; Einspahr, H.M.; Finzel, R.L.; Heinrikson, R.L.; Watenpaugh, K.D. The crystal structure of a lysine 49 phospholipase A2 from the venom of the cottonmouth snake at 2.0-Å resolution. J. Biol. Chem. 1990, 265, 17649–17656. [Google Scholar]
- Renetseder, R.; Brunie, S.; Dijkstra, B.W.; Drenth, J.; Sigler, P.B. A comparison of the crystal structures of phospholipase A2 from bovine pancreas and Crotalus atrox venom. J. Biol. Chem. 1985, 260, 11627–11634. [Google Scholar]
- Suzuki, A.; Matsueda, E.; Yamane, T.; Ashida, T.; Kihara, H.; Ohno, M. Crystal Structure Analysis of Phospholijase A2 from Trimeresurus flavoviridis (Habu Snake) Venom at 1.5 Å Resolution. J. Biochem. 1995, 117, 730–740. [Google Scholar] [CrossRef]
- Fox, J.W.; Serrano, S.M. Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon 2005, 45, 969–985. [Google Scholar] [CrossRef]
- Omori-Satoh, T.; Sadahiro, S. Resolution of the major hemorrhagic component of Trimeresurus flavoviridis venom into two parts. Biochim. Biophys. Acta 1979, 580, 392–404. [Google Scholar] [CrossRef]
- Kini, R.M. Excitement ahead: Structure, function and mechanism of snake venom phospholipase A2 enzymes. Toxicon 2003, 42, 827–840. [Google Scholar] [CrossRef]
- Masuda, S.; Hayashi, H.; Atobe, H.; Morita, T.; Araki, S. Purification, cDNA cloning and characterization of the vascular apoptosis-inducing protein, HV1, from Trimeresurus flavoviridis. J. Biochem. 2001, 268, 3339–3345. [Google Scholar] [CrossRef]
- Matsui, T.; Fujimura, Y.; Titani, K. Snake venom proteases affecting hemostasis and thrombosis. Biochim. Biophys. Acta 2000, 1447, 146–156. [Google Scholar] [CrossRef]
- Carone, S.E.I.; Menaldo, D.L.; Sartim, M.A.; Bernardes, C.P.; Caetano, R.C.; da Silva, R.R.; Cabral, H.; Barraviera, B.; Ferreira Junior, R.S.; Sampaio, S.V. BjSP, a novel serine protease from Bothrops jararaca snake venom that degrades fibrinogen without forming fibrin clots. Toxicol. Appl. Pharmacol. 2018, 357, 50–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kini, R.M. Venom Phospholipase A2 Enzymes: Structure, Function and Mechanism; Kini, R.M., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 1997. [Google Scholar]
- Kihara, H.; Uchikawa, R.; Hattori, S.; Ohno, M. Myotoxicity and physiological effects of three Trimeresurus flavoviridis phospholipases A2. Biochem. Int. 1992, 28, 895–903. [Google Scholar] [PubMed]
- Oda, N.; Ogawa, T.; Ohno, M.; Sasaki, H.; Sakaki, Y.; Kihara, H. Cloning and Sequence Analysis of cDNA for Trimeresurus flavoviridis Phospholipase A2, and Consequent Revision of the Amino Acid Sequence. J. Biochem. 1990, 108, 816–821. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Shimohigashi, Y.; Chijiwa, T.; Nakai, M.; Ogawa, T.; Hattori, S.; Ohno, M. Characterization, amino acid sequence and evolution of edema-inducing, basic phospholipase A2 from Trimeresurus flavoviridis venom. Toxicon 2001, 39, 1069–1076. [Google Scholar] [CrossRef]
- Posada Arias, S.; Rey-Suárez, P.; Pereáñez, J.A.; Acosta, C.; Rojas, M.; Delazari dos Santos, L.; Ferreira, R.S., Jr.; Núñez, V. Isolation and Functional Characterization of an Acidic Myotoxic Phospholipase A2 from Colombian Bothrops asper Venom. Toxins 2017, 9, 342. [Google Scholar] [CrossRef] [PubMed]
- Chijiwa, T.; Hamai, S.; Tsubouchi, S.; Ogawa, T.; Deshimaru, M.; Oda-Ueda, N.; Hattori, S.; Kihara, H.; Tsunasawa, S.; Ohno, M. Interisland Mutation of a Novel Phospholipase A2 from Trimeresurus flavoviridis Venom and Evolution of Crotalinae Group II Phospholipases A2. J. Mol. Evol. 2003, 57, 546–554. [Google Scholar] [CrossRef]
- Liu, S.Y.; Yoshizumi, K.; Oda, N.; Ohno, M.; Tokunaga, F.; Iwanaga, S.; Kihara, H. Purification and Amino Acid Sequence of Basic Protein II, a Lysine-49-Phospholipase A2 with Low Activity, from Trimeresurus flavoviridis Venom. J. Biochem. 1990, 107, 400–408. [Google Scholar] [CrossRef]
- Yoshizumi, K.; Liu, S.Y.; Miyata, T.; Saita, S.; Ohno, M.; Iwanaga, S.; Kihara, H. Purification and amino acid sequence of basic protein I, a lysine-49-phospholipase A2 with low activity, from the venom of Trimeresurus flavoviridis (Habu snake). Toxicon 1990, 28, 43–54. [Google Scholar] [CrossRef]
- Teixera, L.F.; de Carvalho, L.H.; de Castro, O.B.; Bastos, J.F.S.; Néry, N.M.; Oliveira, G.A.; Kayano, A.M.; Soares, A.M.; Zuliani, J.P. Local and systemic effects of BdipTX-I, a Lys-49 phospholipase A2 isolated from Bothrops diporus snake venom. Toxicon 2018, 141, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Nobuhisa, I.; Deshimaru, M.; Chijiwa, T.; Nakashima, K.; Ogawa, T.; Shimohigashi, Y.; Fukumaki, Y.; Hattori, S.; Kihara, H.; Ohno, M. Structures of genes encoding phospholipase A2 inhibitors from the serum of Trimeresurus flavoviridis snake. Gene 1997, 191, 31–37. [Google Scholar] [CrossRef]
- So, S.; Chijiwa, T.; Ikeda, N.; Nobuhisa, I.; Oda-Ueda, N.; Hattori, S.; Ohno, M. Identification of the B Subtype of γ-Phospholipase A2 Inhibitor from Protobothrops flavoviridis Serum and Molecular Evolution of Snake Serum Phospholipase A2 Inhibitors. J. Mol. Evol. 2008, 66, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Gimenes, S.N.C.; Ferreira, F.B.; Silveira, A.N.P.; Rodrigues, R.S.; Yoneyama, K.A.G.; Izabel dos Santos, J.; Fontes, M.R.d.M.; de Campos Brites, V.L.; Santos, A.L.Q.; Borges, M.H.; et al. Isolation and biochemical characterization of a γ-type phospholipase A2 inhibitor from Crotalus durissus collilineatus snake serum. Toxicon 2014, 81, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.Z.; Santos-Filho, N.A.; Menaldo, D.L.; Boldrini-Franca, J.; Giglio, J.R.; Calderon, L.A.; Stabeli, R.G.; Rodrigues, F.H.S.; Tasic, L.; da Silva, S.L.; et al. Structural and Functional Characterization of a γ-Type Phospholipase A2 Inhibitor from Bothrops jararacussu Snake Plasma. Curr. Top. Med. Chem. 2011, 11, 2509–2519. [Google Scholar] [CrossRef]
- Serino-Silva, C.; Morais-Zani, K.; Toyama, M.H.; Toyama, D.d.O.; Gaeta, H.H.; Rodrigues, C.F.B.; Aguiar, W.d.S.; Tashima, A.K.; Grego, K.F.; Tanaka-Azevedo, A.M. Purification and characterization of the first γ-phospholipase inhibitor (γPLI) from Bothrops jararaca snake serum. PLoS ONE 2018, 13, e0193105. [Google Scholar] [CrossRef] [Green Version]
- Yamakawa, Y.; Omori-Satoh, T. Primary Structure of the Antihemorrhagic Factor in Serum of the Japanese Habu: A Snake Venom Metalloproteinase Inhibitor with a Double-Headed Cystatin Domain. J. Biochem. 1992, 112, 583–589. [Google Scholar] [CrossRef]
- Deshimaru, M.; Tanaka, C.; Tokunaga, A.; Goto, M.; Terada, S. Efficient Purification of an Antihemorrhagic Factor (HSF) in Serum of Japanese Habu (Trimeresurus flavoviridis). Fukuoka Univ. Sci. Rep. 2003, 33, 45–53. [Google Scholar]
- Aoki, N.; Sakiyama, A.; Deshimaru, M.; Terada, S. Identification of novel serum proteins in a Japanese viper: Homologs of mammalian PSP94. Biochem. Biophys. Res. Commun. 2007, 359, 330–334. [Google Scholar] [CrossRef]
- Aoki, N.; Matsuo, H.; Deshimaru, M.; Terada, S. Accelerated evolution of small serum proteins (SSPs)-The PSP94 family proteins in a Japanese viper. Gene 2008, 426, 7–14. [Google Scholar] [CrossRef]
- Shioi, N.; Narazaki, M.; Terada, S. Novel function of antihemorrhagic factor HSF as an SSP-binding protein in Habu (Trimeresurus flavoviridis) serum. Fukuoka Univ. Sci. Rep. 2011, 41, 177–184. [Google Scholar]
- Aoki, N.; Sakiyama, A.; Kuroki, K.; Maenaka, K.; Kohda, D.; Deshimaru, M.; Terada, S. Serotriflin, a CRISP family protein with binding affinity for small serum protein-2 in snake serum. Biochim. Biophys. Acta 2008, 1784, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, Y.; Koike, H.; Sugiyama, Y.; Motoyoshi, K.; Wada, T.; Hishinuma, S.; Mita, M.; Morita, T. Cloning and characterization of novel snake venom proteins that block smooth muscle contraction. Eur. J. Biochem. 2002, 269, 2708–2715. [Google Scholar] [CrossRef] [PubMed]
- Shioi, N.; Ogawa, E.; Mizukami, Y.; Abe, S.; Hayashi, R.; Terada, S. Small serum protein-1 changes the susceptibility of an apoptosis-inducing metalloproteinase HV1 to a metalloproteinase inhibitor in habu snake (Trimeresurus flavoviridis). J. Biochem. 2013, 153, 121–129. [Google Scholar] [CrossRef] [Green Version]
- Shioi, N.; Nishijima, A.; Terada, S. Flavorase, a novel non-haemorrhagic metalloproteinase in Protobothrops flavoviridis venom, is a target molecule of small serum protein-3. J. Biochem. 2015, 158, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Chijiwa, T.; So, S.; Hattori, S.; Yoshida, A.; Oda-Ueda, N.; Ohno, M. Suppression of severe lesions, myonecrosis and hemorrhage, caused by Protobothrops flavoviridis venom with its serum proteins. Toxicon 2013, 76, 197–205. [Google Scholar] [CrossRef]
- Tanaka, Y.; Shioi, N.; Terada, S.; Deshimaru, M. Structural Organization and Evolution of a Cluster of Small Serum Protein Genes of Protobothrops Flavoviridis Snake. Fukuoka Univ. Sci. Rep. 2013, 43, 59–66. [Google Scholar]
- Chijiwa, T.; Inamaru, K.; Takeuchi, A.; Maeda, M.; Yamaguchi, K.; Shibata, H.; Hattori, S.; Oda-Ueda, N.; Ohno, M. Unique structure (construction and configuration) and evolution of the array of small serum protein genes of Protobothrops flavoviridis snake. Biosci. Rep. 2019, 39, BSR20190560. [Google Scholar] [CrossRef] [Green Version]
- Shibata, H.; Chijiwa, T.; Oda-Ueda, N.; Nakamura, H.; Yamaguchi, K.; Hattori, S.; Matsubara, K.; Matsuda, Y.; Yamashita, A.; Isomoto, A.; et al. The habu genome reveals accelerated evolution of venom protein genes. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Ito, Y.; Tsuda, R.; Kimura, H. Ultrastructural localizations of beta-microseminoprotein, a prostate-specific antigen, in human prostate and sperm: Comparison with gamma-seminoprotein, another prostate-specific antigen. J. Lab. Clin. Med. 1989, 114, 272–277. [Google Scholar]
- Haas, N.B.; Grabowski, J.M.; Sivitz, A.B.; Burch, J.B. Chicken repeat 1 (CR1) elements, which define an ancient family of vertebrate non-LTR retrotransposons, contain two closely spaced open reading frames. Gene 1997, 197, 305–309. [Google Scholar] [CrossRef]
- Kajikawa, M.; Ohshima, K.; Okada, N. Determination of the entire sequence of turtle CR1: The first open reading frame of the turtle CR1 element encodes a protein with a novel zinc finger motif. Mol. Biol. Evol. 1997, 14, 1206–1217. [Google Scholar] [CrossRef] [PubMed]
- Blin, N.; Stafford, D.W. A general method for isolation of high molecular weight DNA from eukaryotes. Nucleic Acids Res. 1976, 3, 2023–2308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smit, A.; Hubley, R.; Green, P. RepeatMasker-Open-3.0 [WWW Document] RepeatMasker-Open3.0. 1996. Available online: http://repeatmasker.org (accessed on 12 March 2018).
- Benon, G. Tandem repeats finder: A program to analyze DNA sequences. Nucleic Acids Res. 1999, 27, 573–580. [Google Scholar] [CrossRef] [Green Version]
- Pasquesi, G.I.M.; Adams, R.H.; Card, D.C.; Schield, D.R.; Corbin, A.B.; Perry, B.W.; Reyes-Velasco, J.; Ruggiero, R.P.; Vandewege, M.W.; Shortt, J.A.; et al. Squamate reptiles challenge paradigms of genomic repeat element evolution set by birds and mammals. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Yin, W.; Wang, Z.; Li, Q.; Lian, J.; Zhou, Y.; Lu, B.; Jin, L.; Qiu, P.; Zhang, P.; Zhu, W.; et al. Evolutionary trajectories of snake genes and genomes revealed by comparative analyses of five-pacer viper. Nat. Commun. 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Vonk, F.J.; Casewell, N.R.; Henkel, C.V.; Heimberg, A.M.; Jansen, H.J.; McCleary, R.J.; Kerkkamp, H.M.; Vos, R.A.; Guerreiro, I.; Calvete, J.J.; et al. The king cobra genome reveals dynamic gene evolution and adaptation in the snake venom system. Proc. Natl. Acad. Sci. USA 2013, 110, 20651–20656. [Google Scholar] [CrossRef] [Green Version]
- Castoe, T.A.; de Koning, A.P.; Hall, K.T.; Card, D.C.; Schield, D.R.; Fujita, M.K.; Ruggiero, R.P.; Degner, J.F.; Daza, J.M.; Gu, W.; et al. The Burmese python genome reveals the molecular basis for extreme adaptation in snakes. Proc. Natl. Acad. Sci. USA 2013, 110, 20645–20650. [Google Scholar] [CrossRef] [Green Version]
- Aird, S.D.; Arora, J.; Barua, A.; Qiu, L.; Terada, K.; Mikheyev, A.S. Population Genomic Analysis of a Pitviper Reveals Microevolutionary Forces Underlying Venom Chemistry. Genome Biol. Evol. 2017, 9, 2640–2649. [Google Scholar] [CrossRef] [Green Version]
- Warren, W.C.; Wilson, R.K. Direct Submission; Genome Institute; Washington University School of Medicine: St. Louis, MO, USA, 2015. [Google Scholar]
- Liu, Y.; Hughes, D.; Dinh, H.; Dugan, S.; Jhangiani, S.; Lee, S.; Okwuonu, G.; Santibanez, J.; Bandaranaike, D.; Chao, H.; et al. Direct Submission; Baylor College of Medicine: Houston, TX, USA, 2014. [Google Scholar]
- Chijiwa, T.; Nakasone, H.; Irie, S.; Ikeda, N.; Tomoda, K.; Oda-Ueda, N.; Hattori, S.; Ohno, M. Structural characteristics and evolution of the Protobothrops elegans pancreatic phospholipase A2 gene in contrast with those of Protobothrops genus venom phospholipase A2 genes. Biosci. Biotechnol. Biochem. 2013, 77, 97–102. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z. PAML 4: Phylogenetic Analysis by Maximum Likelihood. Mol. Biol. Evol. 2007, 24, 1586–1591. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| CvSSP-4 | CvSSP-5 | CvSSP-6 | |
|---|---|---|---|
| CvSSP-4 | 0.749 | 0.879 | |
| CvSSP-5 | 0.306 | ||
| CvSSP-6 |
| DaSSP-1 | DaSSP-2 | DaSSP-3 | DaSSP-4 | DaSSP-6 | |
|---|---|---|---|---|---|
| DaSSP-1 | 1.61 | 0.934 | 0.823 | 0.832 | |
| DaSSP-2 | 0.849 | 0.705 | 1.03 | ||
| DaSSP-3 | 1.77 | 0.878 | |||
| DaSSP-4 | 0.830 | ||||
| DaSSP-6 |
| OhSSP-4(Ψ) | OhSSP-5α | OhSSP-5β | OhSSP-5γ | OhSSP-6 | |
|---|---|---|---|---|---|
| OhSSP-4(Ψ) | 0.931 | 0.875 | 0.700 | 0.919 | |
| OhSSP-5α | 0.836 | 0.719 | 0.666 | ||
| OhSSP-5β | 0.832 | 0.685 | |||
| OhSSP-5γ | 0.509 | ||||
| OhSSP-6 |
| PfSSP-5α | PfSSP-5β | PbSSP-6 | |
|---|---|---|---|
| PbSSP-5α | 1.594 | 0.343 | |
| PbSSP-5β | 0.296 | ||
| PbSSP-6 |
| PmSSP-1 | PmSSP-2 | PmSSP-3 | PmSSP-4 | PmSSP-5 | PmSSP-6 | |
|---|---|---|---|---|---|---|
| PmSSP-1 | 1.49 | 0.891 | 0.821 | 0.639 | 0.694 | |
| PmSSP-2 | 0.825 | 0.662 | 0.476 | 0.620 | ||
| PmSSP-3 | 1.35 | 0.479 | 0.780 | |||
| PmSSP-4 | 0.586 | 0.889 | ||||
| PmSSP-5 | 0.370 | |||||
| PmSSP-6 |
| PfSSP-1 | PfSSP-2 | PfSSP-3 | PfSSP-4 | PfSSP-5 | PfSSP-6 | |
|---|---|---|---|---|---|---|
| PfSSP-1 | 1.80 | 0.660 | 0.790 | 0.597 | 0.792 | |
| PfSSP-2 | 0.808 | 0.781 | 0.504 | 0.891 | ||
| PfSSP-3 | 1.40 | 0.599 | 0.990 | |||
| PfSSP-4 | 0.670 | 1.07 | ||||
| PfSSP-5 | 0.626 | |||||
| PfSSP-6 |
| TsSSP-4 | TsSSP-5α | TsSSP-5β | TsSSP-6 | |
|---|---|---|---|---|
| TsSSP-4 | 1.06 | 0.603 | 0.651 | |
| TsSSP-5α | 0.675 | 0.447 | ||
| TsSSP-5β | 0.659 | |||
| TsSSP-6 |
| VbSSP-5 | VbSSP-6 | |
|---|---|---|
| VbSSP-5 | 0.604 | |
| VbSSP-6 |
| CvSSP-4 | CvSSP-5 | CvSSP-6 | |
|---|---|---|---|
| CvSSP-4 | 0.319 | 0.358 | |
| CvSSP-5 | 0.372 | ||
| CvSSP-6 |
| DaSSP-1 | DaSSP-2 | DaSSP-3 | DaSSP-4 | DaSSP-6 | |
|---|---|---|---|---|---|
| DaSSP-1 | 0.0227 | 0.248 | 0.253 | 0.249 | |
| DaSSP-2 | 0.247 | 0.251 | 0.246 | ||
| DaSSP-3 | 0.0050 | 0.285 | |||
| DaSSP-4 | 0.288 | ||||
| DaSSP-6 |
| OhSSP-4(Ψ) | OhSSP-5α | OhSSP-5β | OhSSP-5γ | OhSSP-6 | |
|---|---|---|---|---|---|
| OhSSP-4(Ψ) | 0.331 | 0.340 | 0.324 | 0.530 | |
| OhSSP-5α | 0.0615 | 0.0801 | 0.277 | ||
| OhSSP-5β | 0.0857 | 0.226 | |||
| OhSSP-5γ | 0.275 | ||||
| OhSSP-6 |
| PmSSP-1 | PmSSP-2 | PmSSP-3 | PmSSP-4 | PmSSP-5 | PmSSP-6 | |
|---|---|---|---|---|---|---|
| PmSSP-1 | 0.154 | 0.338 | 0.342 | 0.339 | 0.374 | |
| PmSSP-2 | 0.281 | 0.284 | 0.262 | 0.316 | ||
| PmSSP-3 | 0.0397 | 0.293 | 0.346 | |||
| PmSSP-4 | 0.296 | 0.349 | ||||
| PmSSP-5 | 0.295 | |||||
| PmSSP-6 |
| PfSSP-1 | PfSSP-2 | PfSSP-3 | PfSSP-4 | PfSSP-5 | PfSSP-6 | |
|---|---|---|---|---|---|---|
| PfSSP-1 | 0.0317 | 0.251 | 0.258 | 0.248 | 0.231 | |
| PfSSP-2 | 0.254 | 0.261 | 0.253 | 0.229 | ||
| PfSSP-3 | 0.0283 | 0.261 | 0.279 | |||
| PfSSP-4 | 0.270 | 0.287 | ||||
| PfSSP-5 | 0.267 | |||||
| PfSSP-6 |
| Name | Positions | Nucleotide Sequence (GC Content: %, Tm: °C) |
|---|---|---|
| SSP6-5UTR-1 | 1–20 (f) | 5′- ggC gTC CCT CCT TCT CCT Tg -3′ (65, 66) |
| SSP6ex3-2 | 1431–1453 (r) | 5′- CTC gCA TTC CAT ACA ATT ggC Tg-3′ (48, 68) |
| SSP6ex3-1 | 1365–1385 (f) | 5′- TgT ggC CAA CCA AAT gCg Tgg -3′ (57, 66) |
| SSP6-3UTR-2 | 3581–3604 (r) | 5′- ACA TgA gAg ATT TAT TCC AgT gTg-3′(38, 66) |
| SSP6-3flank-1 | 3705–3727 (r) | 5′- CAg CTA TgC ATg CCT TAT ATC AC -3′ (43, 66) |
| Ireg64-1 | 3254–3275 (f) | 5′- CTC CAT gCA AAg gAg gAT TTC C -3′ (50, 66) |
| Ireg64-6 | 10,950–10,970 (r) | 5′- TAg gCC TTg ACA CAT gAT ggC -3′ (52, 64) |
| Ireg64-5 | 8449–8469 (f) | 5′- CAT TgT TgA gCA ACC CTT ggC -3′ (52, 64) |
| Ireg64-8 | 13,709–13,731 (r) | 5′- ggA CTA TTA AgC AgT ggA ATg gC -3′ (48, 68) |
| Ireg64-9 | 13,277–13,297 (f) | 5′- ggC CCT CTT CCA Agg ACA AgC -3′ (62, 68) |
| Ireg64-10 | 16,228–16,247 (r) | 5′- ACC TCg TTC CTC CAg CCA CT -3′ (60, 64) |
| Name | Scaffold | Nucleotide Sequence (GC Content: %, Tm: °C) |
|---|---|---|
| PmSSP34-5UTR (f) | Pm Scaffold 462 | 5′- CAA ggg TTg gTC TTg gTT TTT g -3′ (45, 64) |
| PmSSP3ex2-R (r) | Pm Scaffold 462 | 5′- ggT AgA gAA AAg CCC CCA AAg -3′ (52, 64) |
| PmSSP3-F (f) | Pm Scaffold 462 | 5′- TgC TTT ggg ggC TTT TCT C -3′ (47, 56) |
| PmSSP34-R (r) | Pm Scaffold 462 | 5′- CTT gAC TgA GAC TgA AgT TCC -3′ (45, 62) |
| PmSSP4ex2-R (r) | Pm Scaffold 462 | 5′- CgT TTC Agg TAA Agg AAT ACT C -3′ (41, 62) |
| PmSSP4-F (f) | Pm Scaffold 21,362 | 5′- gAg TAT TCC TTT ACC TgA AAC g -3′ (41, 62) |
| OhSSPs-5UTR (f) | Oh Scaffold 4527 | 5′- ATA AAT Tgg Agg AgC RgA TTC CT -3′ (43, 66) |
| OhSSP5-ex2-R (r) | Oh Scaffold 4527 | 5′- CTC AgC TTC AAA gCC CCA gg -3′ (60, 64) |
| OhSSP5-F (f) | Oh Scaffold 4527 | 5′- gAg CAT gCT TTA CCT ggg gC -3′ (60, 64) |
| OhSSP5-R (r) | Oh Scaffold 47,978 | 5′- TCC ATg TgT AgA gAT CAA ACA Cg -3′ (43, 66) |
| OhSSP2-ex2-R (r) | Oh Scaffold 4527 | 5′- CTC AgC TTC AAA gAg CCC TCT -3′ (52, 64) |
| OhSSP2-F (f) | Oh Scaffold 4527 | 5′- gAg CAT gCT ATA gAg ggC TCT -3′ (52, 64) |
| OhSSP2-R (r) | Oh Scaffold 4527 | 5′- gAT CAA ACA TCA CAg CgC TgC -3′ (52, 64) |
| OhSSP1-ex2-R (r) | Oh Scaffold 4527 | 5′- TTA Agg AAC ACT CCA AAg CAC C -3′ (52, 64) |
| OhSSP1-F (f) | Oh Scaffold 4527 | 5′- gAg ggT gCT TTg gAg TgT TCC -3′ (45, 64) |
| OhSSP1-R (r) | Oh Scaffold 4527 | 5′- gAT CAg ACA CCA CAg CTg Tgg -3′ (57, 66) |
| OhSSP6-ex2-R (r) | Oh Scaffold 10,541 | 5′- TAA ACT gAg gTT TAA AgA gAT CCA -3′ (33, 64) |
| OhSSP6-F (f) | Oh Scaffold 10,541 | 5′- gCA gCA TgC TTC ATg gAT CTC -3′ (52, 64) |
| OhSSP6-R (r) | Oh Scaffold 12,359 | 5′- CCg TgT gAA AAg NTC AgA CAT C -3′ (50, 66) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inamaru, K.; Takeuchi, A.; Maeda, M.; Shibata, H.; Fukumaki, Y.; Oda-Ueda, N.; Hattori, S.; Ohno, M.; Chijiwa, T. Discovery of the Gene Encoding a Novel Small Serum Protein (SSP) of Protobothrops flavoviridis and the Evolution of SSPs. Toxins 2020, 12, 177. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12030177
Inamaru K, Takeuchi A, Maeda M, Shibata H, Fukumaki Y, Oda-Ueda N, Hattori S, Ohno M, Chijiwa T. Discovery of the Gene Encoding a Novel Small Serum Protein (SSP) of Protobothrops flavoviridis and the Evolution of SSPs. Toxins. 2020; 12(3):177. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12030177
Chicago/Turabian StyleInamaru, Kento, Ami Takeuchi, Marie Maeda, Hiroki Shibata, Yasuyuki Fukumaki, Naoko Oda-Ueda, Shosaku Hattori, Motonori Ohno, and Takahito Chijiwa. 2020. "Discovery of the Gene Encoding a Novel Small Serum Protein (SSP) of Protobothrops flavoviridis and the Evolution of SSPs" Toxins 12, no. 3: 177. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12030177




