Classical and Alternative Activation of Rat Microglia Treated with Ultrapure Porphyromonas gingivalis Lipopolysaccharide In Vitro
Abstract
:1. Introduction
2. Results
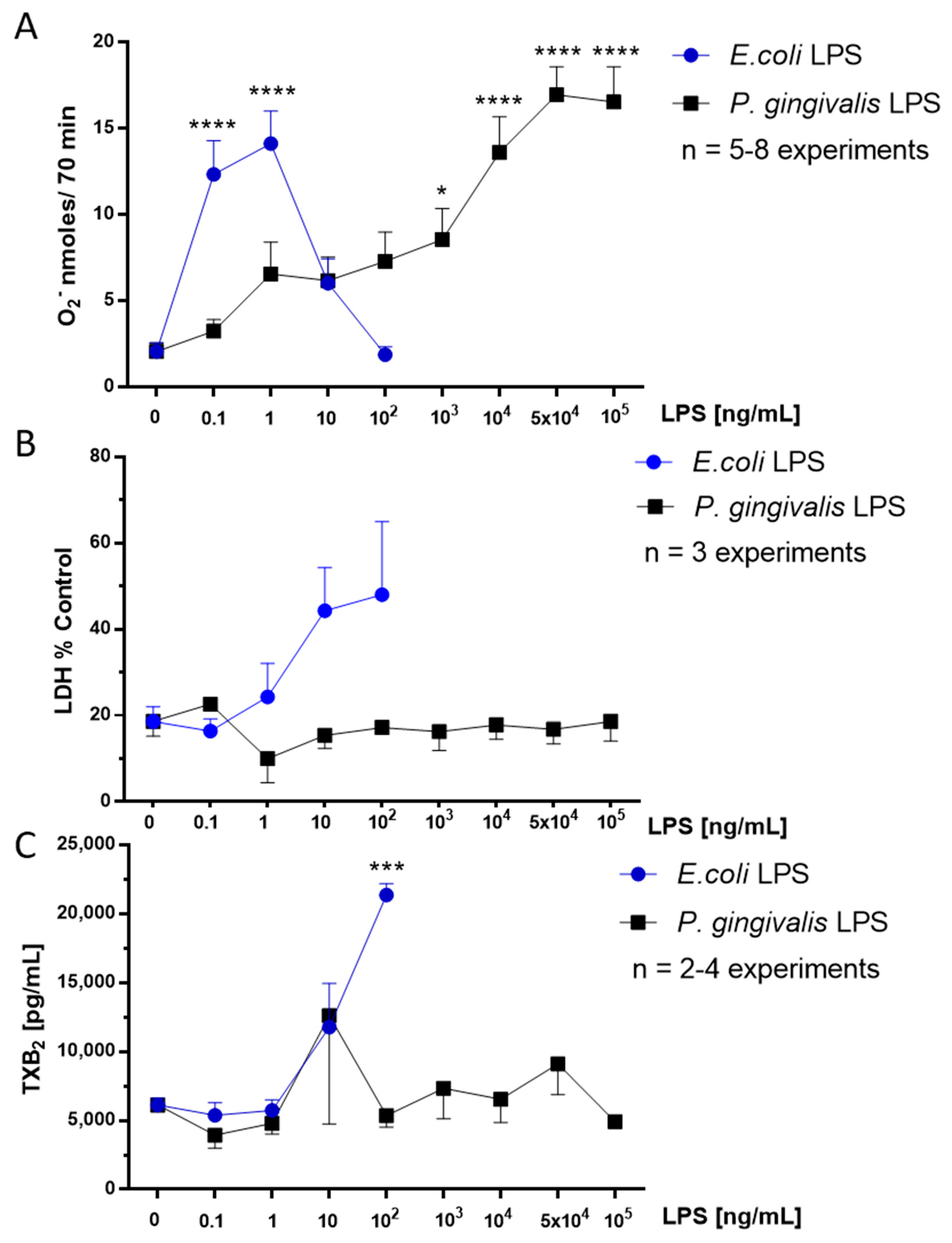
2.1. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia O2− Generation
2.2. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia LDH Generation
2.3. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia TXB2 Generation
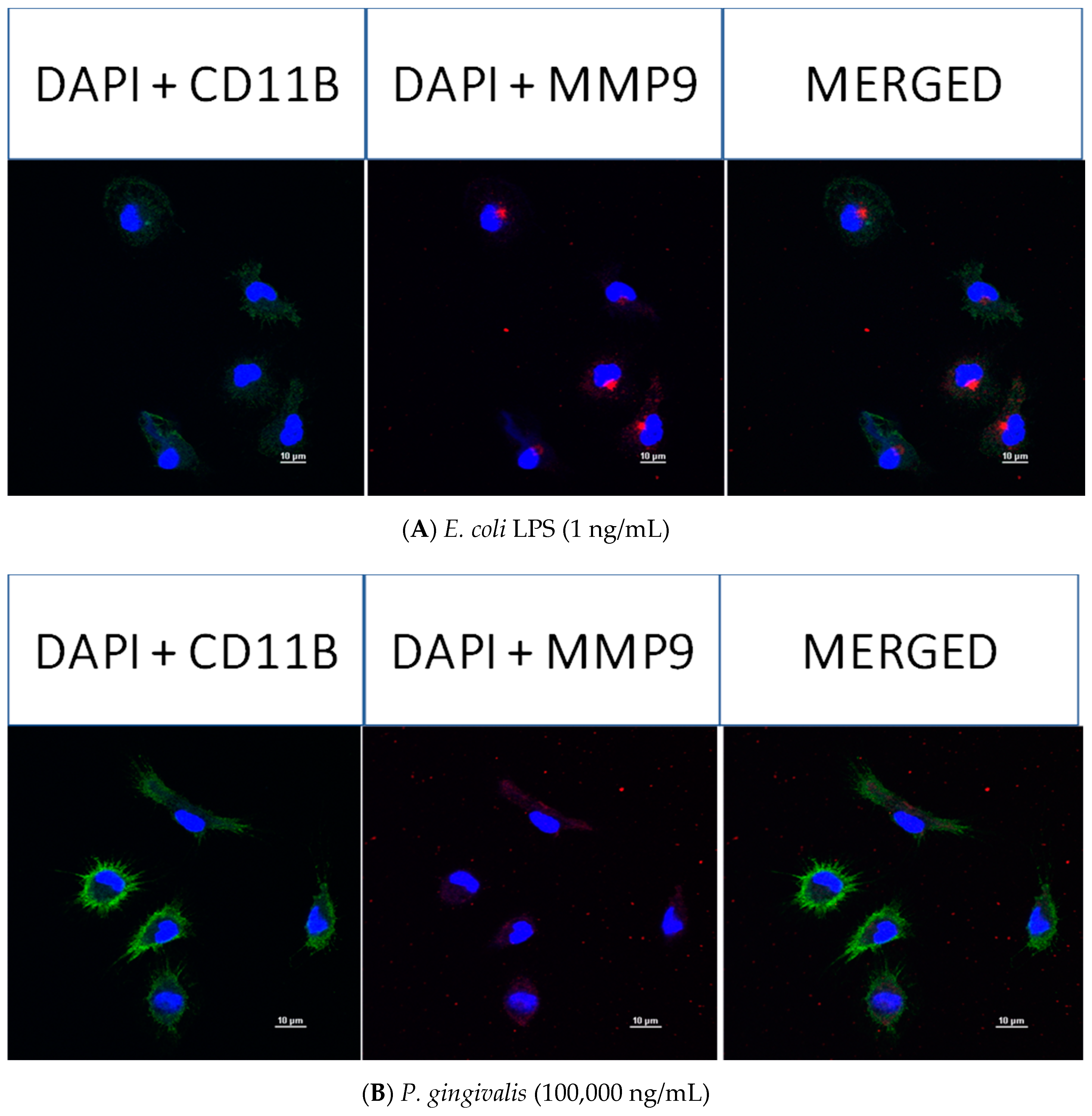
2.4. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia MMP-9 Generation
2.5. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia TNF-α and IL-6 Generation
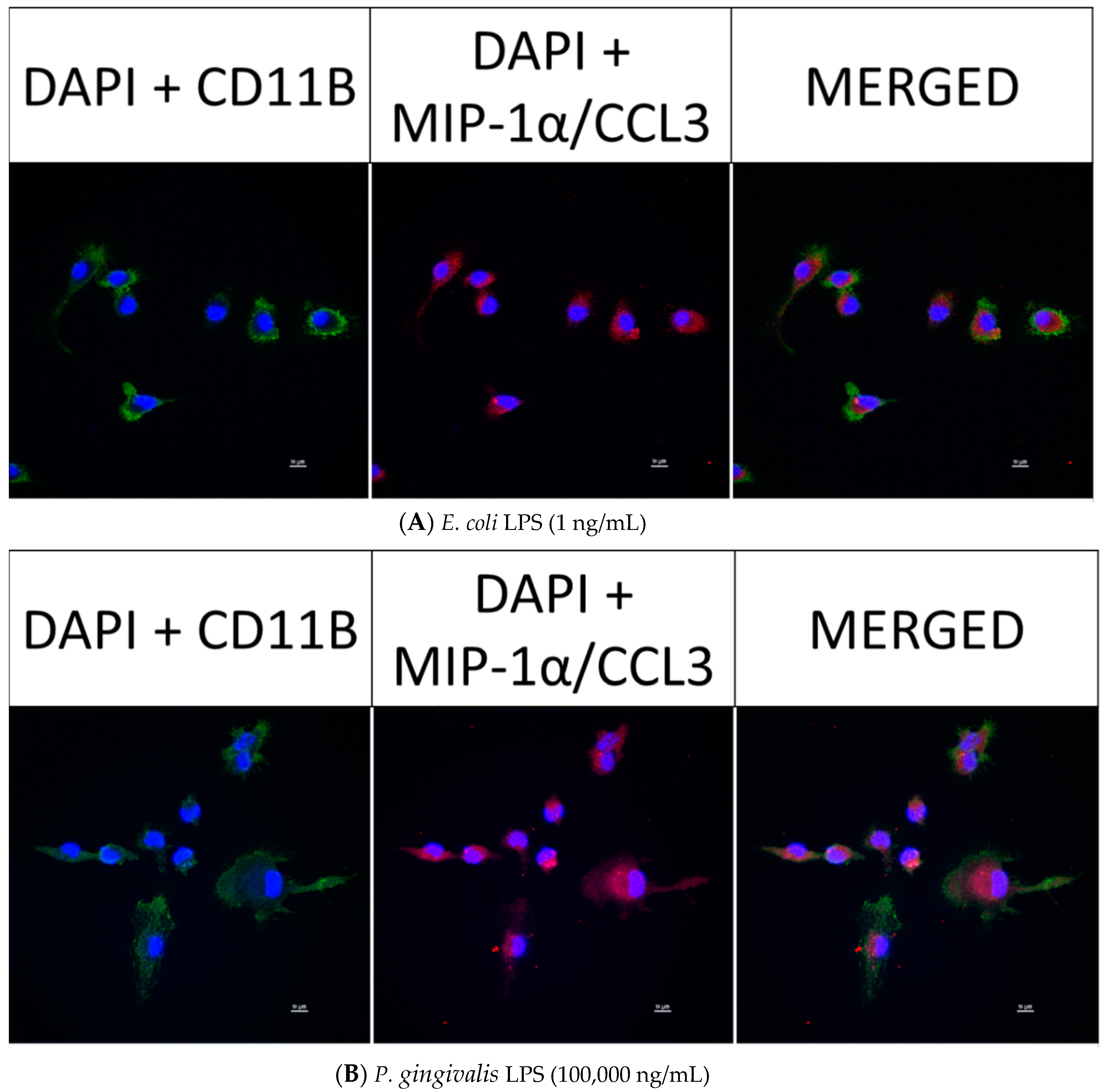
2.6. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia MIP-1α/CCL3, MIP-2/CXCL-2 and CINC-1/CXCL-1 Generation
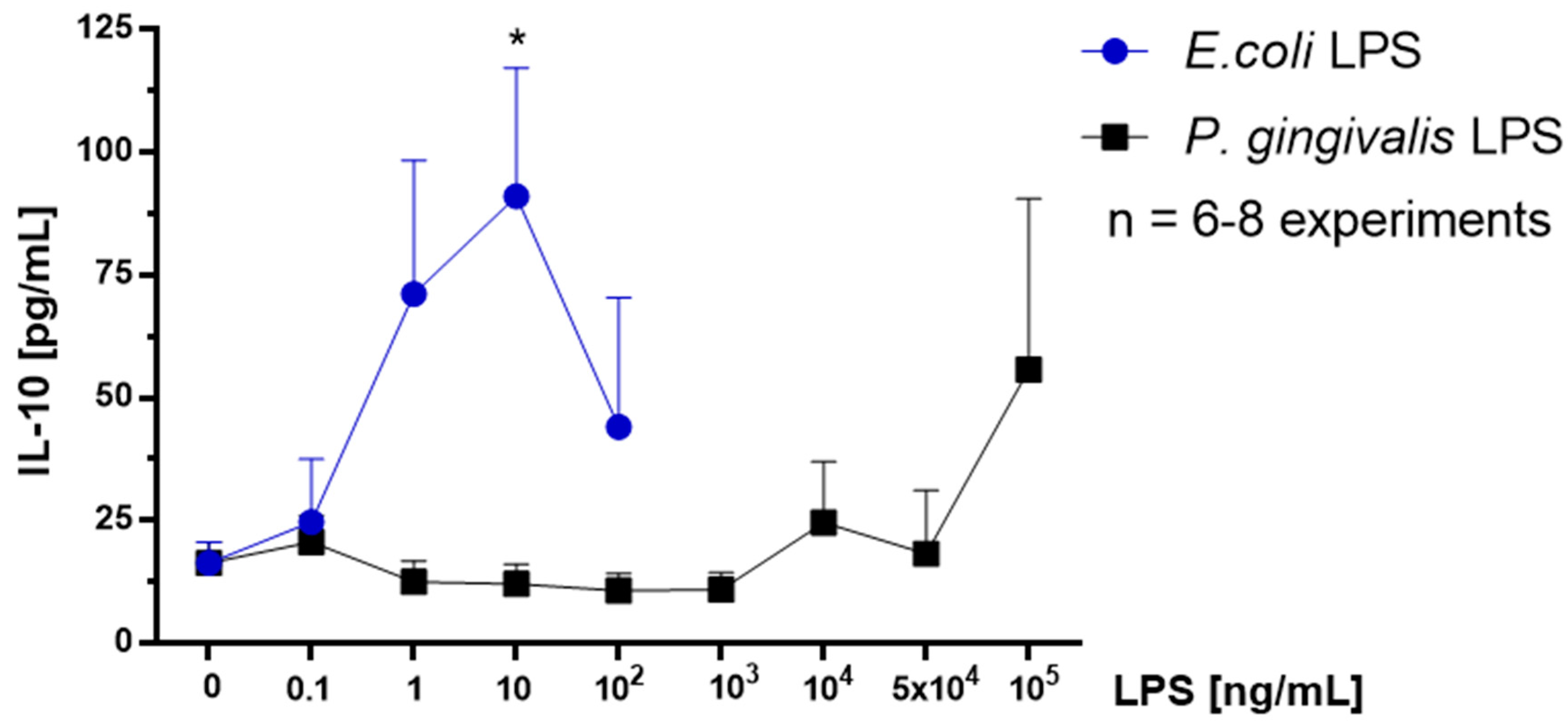
2.7. Effect of P. gingivalis LPS on Neonatal Rat Brain Microglia IL-10 Generation
2.8. Confocal Fluorescence Imaging of MMP-9, IL-6, MIP-1α/CCL3, MIP-2/CXCL-2 and CINC-1/CXCL-1 in Neonatal Rat Brain Microglia Treated with P. gingivalis and E. coli LPS
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Chemicals
5.2. LPS Decontamination
5.3. Isolation of Rat Neonatal Microglia
5.4. Activation of Microglia with LPS (Experimental Protocol)
5.5. Lactate Dehydrogenase (LDH) Assay
5.6. Assay for Microglia O2− Generation
5.7. Assay for Microglia TXB2 Generation
5.8. Assay for Microglia MMP-9 Generation
5.9. Milliplex MagPix Multiplex Array
5.10. Confocal Fluorescence Imaging
5.11. Statistical Analysis of the Data
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yu, Y.-H.; Kuo, H.-K. Association Between Cognitive Function and Periodontal Disease in Older Adults. J. Am. Geriatr. Soc. 2008, 56, 1693–1697. [Google Scholar] [CrossRef] [PubMed]
- Noble, J.M.; Borrell, L.N.; Papapanou, P.N.; Elkind, M.S.V.; Scarmeas, N.; Wright, C.B. Periodontitis is associated with cognitive impairment among older adults: Analysis of NHANES-III. J. Neurol. Neurosurg. Psychiatry 2009, 80, 1206–1211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stein, P.S.; Desrosiers, M.; Donegan, S.J.; Yepes, J.F.; Kryscio, R.J. Tooth loss, dementia and neuropathology in the Nun Study. J. Am. Dent. Assoc. 2007, 138, 1314–1322. [Google Scholar] [CrossRef] [PubMed]
- Ide, M.; Harris, M.; Stevens, A.; Sussams, R.; Hopkins, V.; Culliford, D.; Fuller, J.; Ibbett, P.; Raybould, R.; Thomas, R.; et al. Periodontitis and Cognitive Decline in Alzheimer’s Disease. PLoS ONE 2016, 11, e0151081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ilievski, V.; Zuchowska, P.K.; Green, S.J.; Toth, P.T.; Ragozzino, M.E.; Le, K.; Aljewari, H.; O’Brien-Simpson, N.M.; Reynolds, E.C.; Watanabe, K. Chronic oral application of a periodontal pathogen results in brain inflammation, neurodegeneration and amyloid beta production in wild type mice. PLoS ONE 2018, 13, e0204941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dominy, S.S.; Lynch, C.; Ermini, F.; Benedyk, M.; Marczyk, A.; Konradi, A.; Nguyen, M.; Haditsch, U.; Raha, D.; Griffin, C.; et al. Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 2019, 5, eaau3333. [Google Scholar] [CrossRef] [Green Version]
- Poole, S.; Singhrao, S.K.; Kesavalu, L.; A Curtis, M.; Crean, S. Determining the Presence of Periodontopathic Virulence Factors in Short-Term Postmortem Alzheimer’s Disease Brain Tissue. J. Alzheimer’s Dis. 2013, 36, 665–677. [Google Scholar] [CrossRef]
- Caroff, M.; Karibian, D. Structure of bacterial lipopolysaccharides. Carbohydr. Res. 2003, 338, 2431–2447. [Google Scholar] [CrossRef]
- Herath, T.D.; Darveau, R.P.; Seneviratne, C.J.; Wang, C.Y.; Wang, Y.; Jin, L. Tetra- and penta-acylated lipid A structures of Porphyromonas gingivalis LPS differentially activate TLR4-mediated NF-kappaB signal transduction cascade and immuno-inflammatory response in human gingival fibroblasts. PLoS ONE 2013, 8, e58496. [Google Scholar] [CrossRef] [Green Version]
- Ogawa, T. Chemical structure and immunobiological activity of Porphyromonas gingivalis lipid A. Front. Biosci. 2007, 12, 3795. [Google Scholar] [CrossRef]
- Nativel, B.; Couret, D.; Giraud, P.; Meilhac, O.; D’Hellencourt, C.L.; Viranaïcken, W.; Da Silva, C.R. Porphyromonas gingivalis lipopolysaccharides act exclusively through TLR4 with a resilience between mouse and human. Sci. Rep. 2017, 7, 15789. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Nakanishi, H. Connection Between Periodontitis and Alzheimer’s Disease: Possible Roles of Microglia and Leptomeningeal Cells. J. Pharmacol. Sci. 2014, 126, 8–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Wu, Z.; Zhang, X.; Ni, J.; Yu, W.; Zhou, Y.; Nakanishi, H. Leptomeningeal Cells Transduce Peripheral Macrophages Inflammatory Signal to Microglia in Reponse to Porphyromonas gingivalis LPS. Mediat. Inflamm. 2013, 2013, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Colton, C.A. Heterogeneity of Microglial Activation in the Innate Immune Response in the Brain. J. Neuroimmune Pharmacol. 2009, 4, 399–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colton, C.A.; Wilcock, D.M. Assessing activation states in microglia. CNS Neurol. Disord. Drug Targets 2010, 9, 174–191. [Google Scholar] [CrossRef]
- Tang, Y.; Le, W. Differential Roles of M1 and M2 Microglia in Neurodegenerative Diseases. Mol. Neurobiol. 2015, 53, 1181–1194. [Google Scholar] [CrossRef]
- Mayer, A.M.S.; Oh, S.; Ramsey, K.H.; Jacobson, P.B.; Glaser, K.B.; Romanic, A.M. Escherichia coli lipopolysaccharide potentiation and inhibition of rat neonatal microglia superoxide anion generation: Correlation with prior lactic dehydrogenase, nitric oxide, tumor necrosis factor-alpha, thromboxane B2, and metalloprotease release. Shock 1999, 11, 180–186. [Google Scholar] [CrossRef]
- Rock, R.B.; Gekker, G.; Hu, S.; Sheng, W.S.; Cheeran, M.C.-J.; Lokensgard, J.R.; Peterson, P.K. Role of Microglia in Central Nervous System Infections. Clin. Microbiol. Rev. 2004, 17, 942–964. [Google Scholar] [CrossRef] [Green Version]
- Mayer, A.M.S.; Murphy, J.; Macadam, D.; Osterbauer, C.; Baseer, I.; Hall, M.L.; Feher, D.; Williams, P. Classical and Alternative Activation of Cyanobacterium Oscillatoria sp. Lipopolysaccharide-Treated Rat Microglia in vitro. Toxicol. Sci. 2015, 149, 484–495. [Google Scholar] [CrossRef] [Green Version]
- Block, M.L.; Zecca, L.; Hong, J.-S. Microglia-mediated neurotoxicity: Uncovering the molecular mechanisms. Nat. Rev. Neurosci. 2007, 8, 57–69. [Google Scholar] [CrossRef]
- Choi, S.-H.; Aïd, S.; Bosetti, F. The distinct roles of cyclooxygenase-1 and -2 in neuroinflammation: Implications for translational research. Trends Pharmacol. Sci. 2009, 30, 174–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayer, A.M.S.; Hall, M.L.; Holland, M.; De Castro, C.; Molinaro, A.; Aldulescu, M.; Frenkel, J.; Ottenhoff, L.; Rowley, D.C.; Powell, J. Vibrio vulnificus MO6-24/O Lipopolysaccharide Stimulates Superoxide Anion, Thromboxane B2, Matrix Metalloproteinase-9, Cytokine and Chemokine Release by Rat Brain Microglia in Vitro. Mar. Drugs 2014, 12, 1732–1756. [Google Scholar] [CrossRef] [PubMed]
- Häusler, K.G.; Prinz, M.; Nolte, C.; Weber, J.R.; Schumann, R.R.; Kettenmann, H.; Hanisch, U.-K. Interferon-gamma differentially modulates the release of cytokines and chemokines in lipopolysaccharide- and pneumococcal cell wall-stimulated mouse microglia and macrophages. Eur. J. Neurosci. 2002, 16, 2113–2122. [Google Scholar] [CrossRef] [PubMed]
- Mayer, A.M.S.; Clifford, J.A.; Aldulescu, M.; Frenkel, J.A.; Holland, M.A.; Hall, M.L.; Glaser, K.B.; Berry, J. Cyanobacterial Microcystis aeruginosa Lipopolysaccharide Elicits Release of Superoxide Anion, Thromboxane B2, Cytokines, Chemokines, and Matrix Metalloproteinase-9 by Rat Microglia. Toxicol. Sci. 2011, 121, 63–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, Y.; Si, Q.; Kataoka, K. Lipopolysaccharide-induced microglial activation in culture: Temporal profiles of morphological change and release of cytokines and nitric oxide. Neurosci. Res. 1999, 35, 95–100. [Google Scholar] [CrossRef]
- Suuronen, T.; Huuskonen, J.; Pihlaja, R.; Kyrylenko, S.; Salminen, A. Regulation of microglial inflammatory response by histone deacetylase inhibitors. J. Neurochem. 2003, 87, 407–416. [Google Scholar] [CrossRef] [Green Version]
- Horvath, R.J.; Nutile-McMenemy, N.; Alkaitis, M.S.; DeLeo, J.A. Differential migration, LPS-induced cytokine, chemokine, and NO expression in immortalized BV-2 and HAPI cell lines and primary microglial cultures. J. Neurochem. 2008, 107, 557–569. [Google Scholar] [CrossRef] [Green Version]
- Johnson, E.A.; Dao, T.L.; Guignet, M.A.; Geddes, C.E.; Koemeter-Cox, A.I.; Kan, R.K. Increased expression of the chemokines CXCL1 and MIP-1α by resident brain cells precedes neutrophil infiltration in the brain following prolonged soman-induced status epilepticus in rats. J. Neuroinflamm. 2011, 8, 41. [Google Scholar] [CrossRef] [Green Version]
- Peterson, P.K.; Hu, S.; Salak-Johnson, J.; Molitor, T.W.; Chao, C.C. Differential production of and migratory response to beta chemokines by human microglia and astrocytes. J. Infect. Dis. 1997, 175, 478–481. [Google Scholar] [CrossRef] [Green Version]
- Sun, D.; Hu, X.; Liu, X.; Whitaker, J.N.; Walker, W.S. Expression of chemokine genes in rat glial cells: The effect of myelin basic protein-reactive encephalitogenic T cells. J. Neurosci. Res. 1997, 48, 192–200. [Google Scholar] [CrossRef]
- Diab, A.; Abdalla, H.; Li, H.L.; Shi, F.D.; Zhu, J.; Höjberg, B.; Lindquist, L.; Wretlind, B.; Bakhiet, M.; Link, H. Neutralization of Macrophage Inflammatory Protein 2 (MIP-2) and MIP-1α Attenuates Neutrophil Recruitment in the Central Nervous System during Experimental Bacterial Meningitis. Infect. Immun. 1999, 67, 2590–2601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lafrance, V.; Inoue, W.; Kan, B.; Luheshi, G.N. Leptin modulates cell morphology and cytokine release in microglia. Brain Behav. Immun. 2010, 24, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Seino, Y. Expression of cytokine-induced neutrophil chemoattractant in rat cardiac myocytes. J. Mol. Cell. Cardiol. 1995, 27, 2043–2051. [Google Scholar] [CrossRef]
- Campbell, L.R.; Pang, Y.; Ojeda, N.; Zheng, B.; Rhodes, P.G.; Alexander, B.T. Intracerebral lipopolysaccharide induces neuroinflammatory change and augmented brain injury in growth-restricted neonatal rats. Pediatr. Res. 2012, 71, 645–652. [Google Scholar] [CrossRef]
- Ransohoff, R.M.; Perry, V.H. Microglial Physiology: Unique Stimuli, Specialized Responses. Annu. Rev. Immunol. 2009, 27, 119–145. [Google Scholar] [CrossRef]
- Aloisi, F.; De Simone, R.; Columba-Cabezas, S.; Levi, G. Opposite effects of interferon-gamma and prostaglandin E2 on tumor necrosis factor and interleukin-10 production in microglia: A regulatory loop controlling microglia pro- and anti-inflammatory activities. J. Neurosci. Res. 1999, 56, 571–580. [Google Scholar] [CrossRef]
- Park, K.W.; Lee, H.G.; Jin, B.K.; Lee, Y.B. Interleukin-10 endogenously expressed in microglia prevents lipopolysaccharide-induced neurodegeneration in the rat cerebral cortex in vivo. Exp. Mol. Med. 2007, 39, 812–819. [Google Scholar] [CrossRef] [Green Version]
- Williams, K.; Dooley, N.; Ulvestad, E.; Becher, B.; Antel, J.P. IL-10 production by adult human derived microglial cells. Neurochem. Int. 1996, 29, 55–64. [Google Scholar] [CrossRef]
- Xia, C.-Y.; Zhang, S.; Gao, Y.; Wang, Z.-Z.; Chen, N.-H. Selective modulation of microglia polarization to M2 phenotype for stroke treatment. Int. Immunopharmacol. 2015, 25, 377–382. [Google Scholar] [CrossRef]
- Igboin, C.O.; Griffen, A.L.; Leys, E.J. Porphyromonas gingivalis Strain Diversity. J. Clin. Microbiol. 2009, 47, 3073–3081. [Google Scholar] [CrossRef] [Green Version]
- Slepko, N.; Minghetti, L.; Polazzi, E.; Nicolini, A.; Levi, G. Reorientation of prostanoid production accompanies “activation” of adult microglial cells in culture. J. Neurosci. Res. 1997, 49, 292–300. [Google Scholar] [CrossRef]
- Mayer, A.M.S.; Hall, M.L.; Walker, D.L. Inhibition of LPS-primed human brain microglia superoxide and thromboxanes B2 generation by the marine Manzamines. Inflamm. Res. 2004, 53, S217. [Google Scholar]
- Sheng, W.S.; Hu, S.; Kravitz, F.H.; Peterson, P.K.; Chao, C.C. Tumor necrosis factor alpha upregulates human microglial cell production of interleukin-10 in vitro. Clin. Diagn. Lab. Immunol. 1995, 2, 604–608. [Google Scholar] [CrossRef] [Green Version]
- Hoogland, I.C.; Houbolt, C.; Van Westerloo, D.J.; Van Gool, W.; Van De Beek, D. Systemic inflammation and microglial activation: Systematic review of animal experiments. J. Neuroinflamm. 2015, 12, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, L.; Wu, X.; Block, M.L.; Liu, Y.; Breese, G.R.; Hong, J.-S.; Knapp, D.J.; Crews, F.T. Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 2007, 55, 453–462. [Google Scholar] [CrossRef] [Green Version]
- Fonken, L.K.; Frank, M.G.; Kitt, M.M.; Barrientos, R.M.; Watkins, L.R.; Maier, S.F. Microglia inflammatory responses are controlled by an intrinsic circadian clock. Brain Behav. Immun. 2014, 45, 171–179. [Google Scholar] [CrossRef] [Green Version]
- Morgenstern, S.; Flor, R.; Kessler, G.; Klein, B. The Automated Determination of NAD-Coupled Enzymes. Clin. Chem. 1966, 12, 274–281. [Google Scholar] [CrossRef]





© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Memedovski, Z.; Czerwonka, E.; Han, J.; Mayer, J.; Luce, M.; Klemm, L.C.; Hall, M.L.; Mayer, A.M.S. Classical and Alternative Activation of Rat Microglia Treated with Ultrapure Porphyromonas gingivalis Lipopolysaccharide In Vitro. Toxins 2020, 12, 333. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12050333
Memedovski Z, Czerwonka E, Han J, Mayer J, Luce M, Klemm LC, Hall ML, Mayer AMS. Classical and Alternative Activation of Rat Microglia Treated with Ultrapure Porphyromonas gingivalis Lipopolysaccharide In Vitro. Toxins. 2020; 12(5):333. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12050333
Chicago/Turabian StyleMemedovski, Zylfi, Evan Czerwonka, Jin Han, Joshua Mayer, Margaret Luce, Lucas C. Klemm, Mary L. Hall, and Alejandro M. S. Mayer. 2020. "Classical and Alternative Activation of Rat Microglia Treated with Ultrapure Porphyromonas gingivalis Lipopolysaccharide In Vitro" Toxins 12, no. 5: 333. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12050333




