Zearalenone (ZEN) in Livestock and Poultry: Dose, Toxicokinetics, Toxicity and Estrogenicity
Abstract
:1. Introduction
2. ZEN Guidance and Concentration in Animal Feed
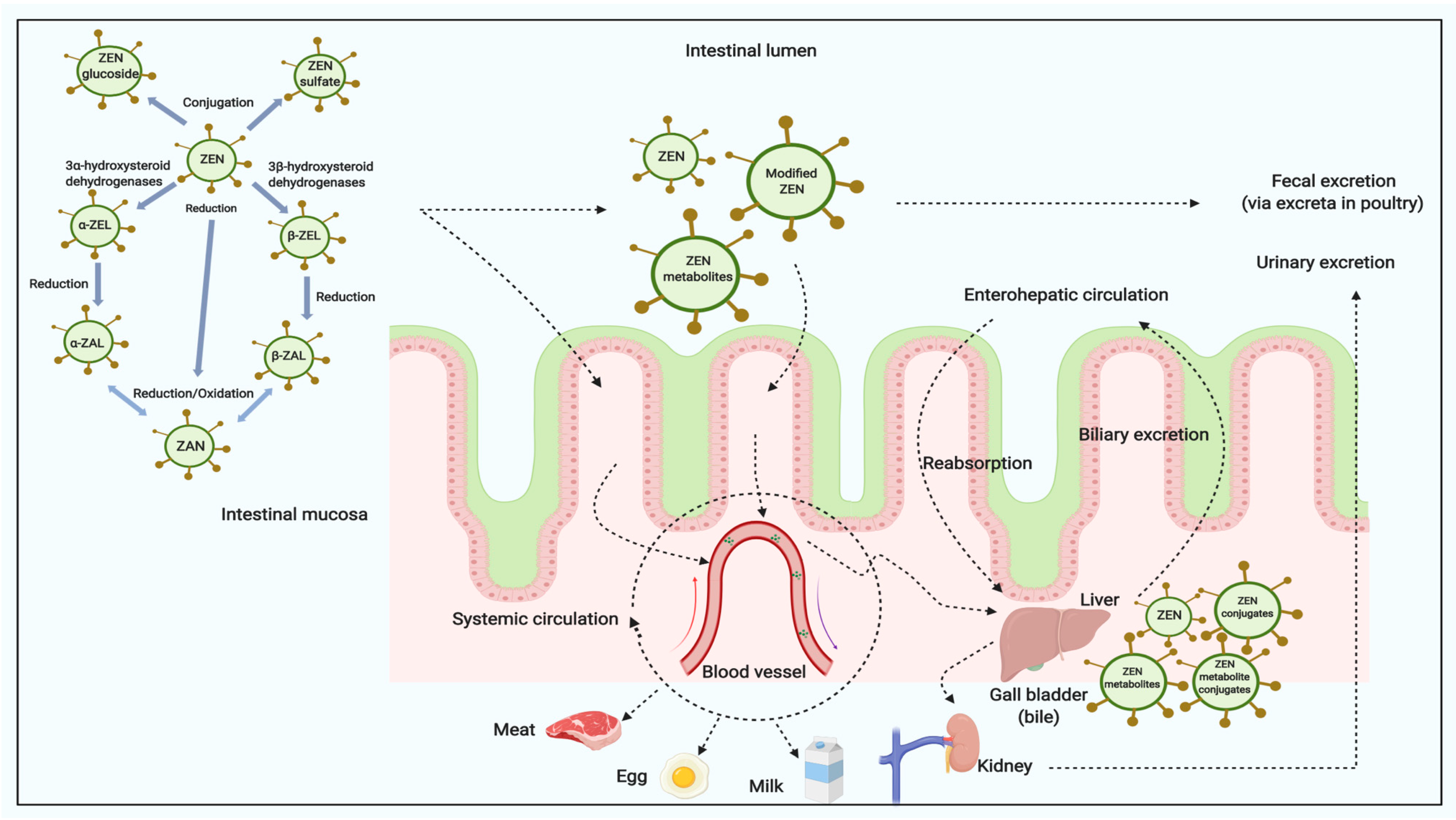
3. ZEN Absorption Rate, Distribution (Carry-Over), Metabolites and Excretion in Livestock Animals
| Article Type | Feed for Species | ZEN Concentration (µg/kg) | Analysis Method | ZEN Derivatives (α-ZEL, β-ZEL, ZAN, α-ZAL, β-ZAL) | Year |
|---|---|---|---|---|---|
| Survey | Poultry; swine | 44 to 797; 86 to 629 | LC | N.A. | 1997 [60] |
| Study | Poultry | 327 to 5850 | TLC | N.A. | 1998 [61] |
| Research | Starter pig | 200, 400 and 500 (also detected with other mycotoxins) | GC-MS | α, β-ZEL < d.l. (set at 0.2 mg/kg) | 2003 [62] |
| Study | Poultry | 0.53 | HPLC | N.A. | 2004 [63] |
| Research | Broiler | 400, 500, 600 and 700 (also detected with other mycotoxins) | GC-MS | α, β-ZEL < d.l. (set at 0.2 mg/kg) | 2004 [64] |
| Survey | Poultry | 3 to 86 | HPLC | N.A. | 2005 [65] |
| Research | Weaned pig | 300 to 710 (also detected with other mycotoxins) | HPLC | N.A | 2005 [66] |
| Research | Dairy cows | 22 and 59 (µg/kg DM) | HPLC | Mean recoveries for α-ZEL and β-ZEL were 81 and 74%; concentration N.A. | 2005 [67] |
| Survey | Laying hen | 7.4 to 61.4 | HPLC | N.A | 2006 [68] |
| Research | Broiler | 70, 3360 and 8280 | ELISA | N.A | 2008 [69] |
| Survey | Animal feed | 10 to 189 | HPLC | N.A | 2010 [70] |
| Research | Post-weaning gilt | 100 and 1300 (also detected with other mycotoxins) | ELISA | N.A | 2010 [43] |
| Research | Post-weaning gilt | 100 and 1300 (also detected with other mycotoxins) | ELISA | N.A | 2010 [44] |
| Research | Post-weaning gilt | 1100, 2000 and 3200 | ELISA | N.A | 2011 [71] |
| Survey | Animal feed | Up to 5791 | ELISA+HPLC | N.A | 2012 [72] |
| Research | Sow (during gestation and lactation) | Appr. 200, 500 and 1000 | Not mentioned | N.A | 2012 [73] |
| Research | Post-weaning piglet | 1050 (also detected with another mycotoxin) | ELISA | N.A | 2012 [74] |
| Research | Gilt | 200, 400 and 800 | GC-MS | N.A | 2012 [75] |
| Survey | Poultry; swine; ruminants | Appr. 12 to 109; 13 to 200; 57 to 194 | HPLC and LC-MS/MS | N.A | 2014 [76] |
| Research | Broiler | Appr. 18 and 280 | ELISA | N.A | 2014 [77] |
| Research | Dairy cow | 24.4 to 112.7 (µg/kg DM; also detected with another mycotoxin) | HPLC | Average recoveries for α-ZEL and β-ZEL were 85% and 86% | 2014 [78] |
| Survey | Broiler | 2.22 to 263.51 | LC-MS/MS | N.A | 2015 [79] |
| Study | Layer | 5.17 to 147.53 | HPLC | N.A | 2016 [80] |
| Survey | Swine | 36 to 219 | HPLC | α-ZEL: <15 to 529; β-ZEL: <11 | 2016 [81] |
| Research | Laying hen | 400 (also detected with another mycotoxin) | HPLC | N.A | 2017 [82] |
| Case study | Pig (hay pellet) | 479 | ELISA and LC-MS/MS | α-ZEL: 11.7; β-ZEL: 16.9; ZEN-sulfate: 530; ZEN-glucoside: <d.l. | 2018 [83] |
| Study | Cattle | 88.2 | UPLC-MS/MS and UPLC-QTOF-MS | N.A | 2018 [46] |
| Study | Duck; pig | 39.08 to 47.61; 124.78 | HPLC, and LC-MS/MS | For duck feed: α-ZEL:4.19 For pig feed: α-ZAL: 2.31 to 2.48; β-ZAL: 3.11; ZAN: 4.17 to 6.69 | 2018 [45] |
| Research | Pig | Appr. 800 | UPLC | N.A | 2018 [84] |
| Research | Turkey | 470 (also detected with other mycotoxins) | HPLC-MS/MS | N.A | 2019 [85] |
| Research | Broiler | Appr. 2000 (also detected with other mycotoxins) | ELISA | N.A | 2019 [86] |
| Research | Broiler | 280 to 520 | SIDA-UHPLC-MS/MS | N.A | 2020 [87] |
3.1. Poultry
3.2. Swine
3.3. Ruminants
| Species | ZEN Concentration (mg/kg of Diet, Fed-Basis) | Duration (Days) | Metabolites and Carry-Over into Tissues (ug/kg or ug/L) | Remarks | References |
|---|---|---|---|---|---|
| Laying hen | 14C-ZEN: 10 mg/kg b.w. | Single bolus | Leg, wing, breast muscle: very low radioactivity; Yolk: appr. 2000 ug/kg after 72 h | 94% of 14C radioactivity eliminated via excreta within 72 h of dosing; ZEN was readily conjugated with glucuronic acid | [93] |
| Broiler | 3H-ZEN: 5 mg/kg b.w. (appr. 50 mg/kg diet) | Single bolus | Muscle: relatively low, ZEN max. 111 at 24 h after dosing (α-ZEL, β-ZEL n.d.); Liver: total ZEN α-ZEL and β-ZEL 17-2543 within 24 h; Rapid clearance | Conjugated n.d. | [88] |
| Turkey | 800 | 14 | Liver: ZEN 282; α-ZEL 2720; Kidney: ZEN 120, α-ZEL 480; β-ZEL traces in liver and kidney (after incubation with β-glucuronidase and sulfatase) | ZEN and α-ZEL mainly conjugated in plasma and conjugates consisted of both glucuronides and sulfate conjugates | [105] |
| Chicken | 10 mg/kg b.w. | 20 | Liver: ZEN 207; Kidney: ZEN 416; Muscle: ZEN 170 | Metabolites and conjugated n.d. | [106] |
| Laying hen | 1.58 | 112 | Liver: α-ZEL 3.5-3.8 (36% free, 28% conjugated with glucuronic acid, and 36% with sulphate); ZEN<1-3.2 (46% free, 54% conjugated with glucuronic acid, and <5% with sulfate); n.d. residues in egg yolk, albumen, breast muscle, abdominal fat, ovary and follicles, magnum | [92] | |
| Pekin duck | Up to 0.06 | 49 | Liver: ZEN, α-ZEL and β-ZEL<d.l. kidney (after incubation with β-glucuronidase and sulfatase) | In bile, dose-response related increase in ZEN, α-ZEL and β-ZEL-concentrations; the mean proportions of ZEN, α-ZEL and β-ZEL of the sum of all three metabolites were 80, 16 and 4% respectively | [91] |
| Turkey | Up to appr. 0.04 | 35 | Plasma, liver or breast meat: ZEN or its metabolites n.d. | In bile, concentrations of ZEN and α-ZEL increased with dietary ZEN concentration; the mean proportions of ZEN, α-ZEL and β-ZEL of the sum of all three metabolites were 19, 77 and 4% respectively | [107] |
| Broiler | 0.3 mg/kg b.w. | Single bolus (intravenously and orally) | Plasma: ZEN and its metabolites n.d. | [108] | |
| Broiler (Female) | 1.2 mg/kg b.w. | Single bolus (orally) | Liver: ZEN 3.52; α-ZEL 7.84-105.2, β-ZEL 24.4-30.9; Kidney: ZEN 3.55; α-ZEL 1.63-77.99, β-ZEL 4.8-36.6; Muscle: α-ZEL 2.55, β-ZEL 2.40; (after incubation with glucuronidase/arylsulfatase) | Time-response decrease in ZEN, α-ZEL and β-ZEL concentration | [89] |
| Broilers (male; slow-growing breed) | 0.4 | 56 | Liver: ZEN n.d.; α-ZAL n.d.; α-ZEL 0.4-0.8 (5 out of 8 samples) | [87] |
| Species | ZEN Concentration (mg/kg of Diet, Fed-Basis) | Duration (Days) | Metabolites and Carry-Over into Tissues (ug/kg or ug/L) | Remarks | References |
|---|---|---|---|---|---|
| Pig (female, 8-11 kg b.w.) | 40 | 28 | Liver: ZEN 128; α-ZEL 94 and β-ZEL <d.l. | Conjugates n.d. | [109] |
| Piglet (appr. 18kg b.w.) | 0.5 mg/kg b.w. | Single bolus | Liver, kidney, muscle: ZEN, α-ZEL and β-ZEL<d.l. (after incubation with glucuronidase) | ZEN: d.l., α-ZEL and β-ZEL: 0.8-9.2 ug/kg | [110] |
| Pig (appr. 50 kg b.w.) | 1)ZEN: 0.25 2)ZEN: 0.25+OTA 0.1 | 90 | 1) liver, kidney, muscle, adipose tissue: ZEN and α-ZEL < d.l. 2) liver, kidney: α-ZEL-traces (max. 4 ug/kg after incubation with glucuronidase), ZEN<d.l.; muscle and adipose tissue: ZEN and ZAN<d.l. | [111] | |
| Pig (appr. 70 kg b.w.) | 0.7 | 18 | Liver: ZEN<d.l. -3.1; α-ZEL 3.6-12; β-ZEL 1.9 -4.8; Muscle: α-ZAL up to 13.3; α-ZEL up to 14.5; traces of ZEN and β-ZAL; ZEN and ZAN <d.l. | [112] | |
| Piglet (appr. 33 kg b.w.) | 0.01; 0.06; 0.15; 0.22; 0.42 | 35 | Liver (after incubation with β-glucuronidase and sulfatase) 1.8 ZEN + 0.3 α-ZEL; 0.2 ZEN + 0.1 α-ZEL; 2.1 ZEN + 1.1 α-ZEL; 2.9 ZEN + 1.7 α-ZEL; 5.3 ZEN + 2.8 α-ZEL | [113] | |
| Piglet (appr. 33 kg b.w.) | 1 mg/kg b.w. | Single bolus | Liver (14 days after the bolus, after incubation with β-glucuronidase and sulfatase): ZEN, α-ZEL and β-ZEL<d.l. | [94] | |
| Pig (female and barrows) | 0.056 | 84 | Liver: only α-ZEL was detected with mean carry-over factors (averaged over all group) of 0.0094; ZEN, α-ZEL and β-ZEL n.d. in serum | Residues of ZEN + α-ZEL + β-ZEL was positively correlated in liver and bile (Liver showed 0.9% carry-over ratio) | [99] |
| Species | ZEN Concentration (mg/kg of Diet) | Duration (Days) | Metabolites and Carry-Over into Tissues (ug/kg or ug/L) | Remarks | References |
|---|---|---|---|---|---|
| Lactating cow | 0.39-1.93 mg/kg concentrate | 49 | Muscle, liver, kidney, milk: ZEN<4 | [114] | |
| Lactating cow | 5000 mg/animal | Single bolus | Milk: ZEN and β-ZEL: traces (<1) | incubation with β-glucuronidase | [115] |
| Lactating cow | 1800 mg/animal | Single bolus | Milk: ZEN and β-ZEL: 1-2 | incubation with β-glucuronidase | [115] |
| Lactating cow | 25 | 7 | Milk: 1360 ug/l total residues of ZEN, α-ZEL, β-ZEL, free and conjugated | 0.7% of consumed ZEN recovered with milk | [52] |
| Lactating cow | 50-165 mg/day; 545 mg/day; 1800 or 6000 mg/animal | 21; 21 and single bolus | Milk: ZEN, α-ZEL and β-ZEL and conjugates < d.l.; Milk: ZEN max. 2.5; α-ZEL max. 3.0 (only as conjugates, incubation with β-glucuronidase/aryl sulfatase); Milk: ZEN max. 4.0 or 6.1; α-ZEL max. 1.5 or 4.0; β-ZEL max 4.1 or 6.64 | [116] | |
| Lactating cow | 25 or 100 mg/day | 6 | Milk: ZEN-equivalents max. 0.4 or 1.2 (by ELISA after incubation with β-glucuronidase) | [117] | |
| Lactating cow | 0.02-0.05 mg/kg dry matter | 63 | Milk: ZEN and α-ZEL<0.5 (after incubation with β-glucuronidase) | [118] | |
| Dairy cow | 0.05 mg/kg dry matter | 28 | Milk: ZEN, α-ZEL, β-ZEL, ZAN, α-ZAL, β-ZAL < d.l. | [67] | |
| Goat | 2.4 and 1.2 mg/kg b.w. | Single bolus (intravenously) | Liver: α-ZEL 5.2 and β-ZEL 4.5 at 48 h poste administration | The proportion of conjugated α-ZEL and β-ZEL were appr. 29 and 41% respectively | [104] |
| Dairy cow | 0.02 to 0.11 mg/kg dry matter | 28 wks | Milk: ZEN, α-ZEL, β-ZEL, ZAN, α-ZAL, β-ZAL < d.l | Bile: ZEN, α-ZEL, and β-ZEL were detectable (bile can be regarded as an indicator for dietary ZEN-exposure) | [78] |
4. ZEN Toxicity and Estrogenic Effect in Livestock Animals
4.1. Toxicity
4.2. Estrogenicity
5. Conclusions
- (1)
- The occurrence of ZEN and co-occurrence of ZEN with other mycotoxins in grain and complete feed is still relatively high;
- (2)
- The amount of ZEN that carries over into final animal products (meat, egg, milk) is very limited under normal farming systems. However, ZEN and its modified metabolites can be detected in blood, liver, gut, urine and feces;
- (3)
- The special structure of ZEN mimics the effects of estrogen, which closely relates to the reproductive functionality of livestock, with swine being the most sensitive species.
- (4)
- Modified and masked forms of ZEN should be included and taken into consideration for the risk assessment of ZEN for farm animals.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Diekman, M.A.; Green, M.L. Mycotoxins and reproduction in domestic livestock. J. Anim. Sci. 1992, 70, 1615–1627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Streit, E.; Schwab, C.; Sulyok, M.; Naehrer, K.; Krska, R.; Schatzmayr, G. Multi-mycotoxin screening reveals the occurrence of 139 different secondary metabolites in feed and feed ingredients. Toxins 2013, 5, 504–523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bennett, J.W. Mycotoxins, mycotoxicoses, mycotoxicology and mycopathologia. Mycopathologia 1987, 100, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Murugesan, G.R.; Ledoux, D.R.; Naehrer, K.; Berthiller, F.; Applegate, T.J.; Grenier, B.; Phillips, T.D.; Schatzmayr, G. Prevalence and effects of mycotoxins on poultry health and performance, and recent development in mycotoxin counteracting strategies. Poult. Sci. 2015, 94, 1298–1315. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.S.; Cunha, S.C.; Fernandes, J.O. Prevalent Mycotoxins in Animal Feed: Occurrence and Analytical Methods. Toxins 2019, 11, 290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tiemann, U.; Dänicke, S. In vivo and in vitro effects of the mycotoxins zearalenone and deoxynivalenol on different non-reproductive and reproductive organs in female pigs: A review. Food Addit. Contam. 2007, 24, 306–314. [Google Scholar] [CrossRef] [Green Version]
- Hasunuma, H.; Takagi, M.; Kawamura, O.; Taniguchi, C.; Nakamura, M.; Chuma, T.; Uno, S.; Kokushi, E.; Matsumoto, D.; Tshering, C.; et al. Natural contamination of dietary rice straw with zearalenone and urinary zearalenone concentrations in a cattle herd1. J. Anim. Sci. 2012, 90, 1610–1616. [Google Scholar] [CrossRef] [Green Version]
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013, 60, 218–237. [Google Scholar] [CrossRef]
- Link, H.F. Observationes in ordines plantarum naturals, Dissetatio I. Mag. Ges. Naturf. Freunde Berlin 1809, 3, 3–42. [Google Scholar]
- Stob, M.; Baldwin, R.S.; Tuite, J.; Andrews, F.N.; Gillette, K.G. Isolation of an anabolic, uterotrophic compound from corn infected with Gibberella zeae. Nature 1962, 196, 1318. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zinedine, A.; Soriano, J.M.; Molto, J.C.; Mañes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An estrogenic mycotoxin. Food Chem. Toxicol. 2007, 25, 1–18. [Google Scholar] [CrossRef]
- Urry, W.H.; Wehrmeister, H.L.; Hodge, E.B.; Hidy, P.H. The structure of zearalenone. Terahedron Lett. 1966, 7, 3109–3114. [Google Scholar] [CrossRef]
- Christensen, C.M.; Nelson, G.H.; Mirocha, C.J. Effect on the white rat uterus of a toxic substance isolated from Fusarium. Appl. Microbial. 1965, 13, 653. [Google Scholar] [CrossRef] [Green Version]
- Mirocha, C.J.; Christensen, C.M.; Nelson, G.H. Estrogenic metabolite produced by Fusarium graminearum in stored corn. Appl. Environ. Microbiol. 1967, 15, 497–503. [Google Scholar] [CrossRef] [Green Version]
- EFSA CONTAM Panel. Scientific opinion on risk for animal health related to the presence of zearalenone and its modified forms in feed. EFSA J. 2017, 15, 4851. [Google Scholar]
- Ji, F.; He, D.; Olaniran, A.O.; Mokoena, M.P.; Xu, J.; Shi, J. Occurrence, toxicity, production and detection of Fusarium mycotoxin: A review. Food Prod. Process. Nutr. 2019, 1, 6. [Google Scholar] [CrossRef]
- Kuiper-Goodman, T.; Scott, P.M.; Watanabe, H. Risk assessment of the mycotoxin zearalenone. Regul. Toxicol. Pharm. 1987, 7, 253–306. [Google Scholar] [CrossRef]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Global mycotoxin occurrence in feed: A ten-year survey. Toxins 2019, 11, 375. [Google Scholar] [CrossRef] [Green Version]
- Minervini, F.; Dell’Aquila, M.E. Zearalenone and reproductive function in farm animals. Int. J. Mol. Sci. 2008, 9, 2570–2584. [Google Scholar] [CrossRef] [Green Version]
- Zhang, G.-L.; Feng, Y.-L.; Song, J.-L.; Zhou, X.-S. Zearalenone: A mycotoxin with different toxic effect in domestic and laboratory animals’ granulosa cells. Front. Genet. 2018, 9, 667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bottalico, A.; Visconti, A.; Logrieco, A.; Solfrizzo, M.; Mirocha, C.J. Occurrence of zearalenols (diastereomeric mixture) in corn stalk rot and their production by associated Fusarium species. Appl. Environ. Microbiol. 1985, 49, 547–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urraca, J.L.; Marazuela, M.D.; Moreno-Bondi, M.C. Analysis for zearalenone and α-zearalenol in cereals and swine feed using accelerated solvent extraction and liquid chromatography with fluorescence detection. Anal. Chim. Acta 2004, 524, 175. [Google Scholar] [CrossRef]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Winkler, J. Invited review: Diagnosis of zearalenone (ZEN) exposure of farm animals and transfer of its residues into edible tissues (carry over). Food Chem. Toxicol. 2015, 84, 225–249. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, N.; Dänicke, S.; Edler, L.; Gottschalk, C.; Lassek, E.; Marko, D.; Rychlik, M.; Mally, A. A critical evaluation of health risk assessment of modified mycotoxins with a special focus on zearalenone. Mycotoxin Res. 2019, 35, 27–46. [Google Scholar] [CrossRef] [Green Version]
- Drzymala, S.S.; Binder, J.; Brodehl, A.; Penkert, M.; Rosowski, M.; Garbe, L.A.; Koch, M. Estrogenicity of novel phase I and phase II metabolites of zearalenone and cis-zearalenone. Toxicon 2015, 105, 10–12. [Google Scholar] [CrossRef]
- Gareis, M.; Bauer, J.; Thiem, J.; Plank, G.; Grabley, S.; Gedek, B. Cleavage of zearalenone-glycoside, a masked mycotoxin, during digestion in swine. J. Vet. Med. B 1990, 37, 236–240. [Google Scholar] [CrossRef]
- Plasencia, J.; Mirocha, C.J. Isolation and characterization of zearalenone sulfate produced by Fusarium spp. Appl. Environ. Microbiol. 1991, 57, 146–150. [Google Scholar] [CrossRef] [Green Version]
- Paris, M.P.K.; Schweiger, W.; Hametner, C.; Stuckler, R.; Muehlbauer, G.J.; Varga, E.; Krska, R.; Berthiller, F.; Adam, G. Zearalenone-16-O-glucoside: A new masked mycotoxin. J. Agric. Food Chem. 2014, 62, 1181–1189. [Google Scholar] [CrossRef]
- Grenier, B.; Applegate, T.J. Modulation of Intestinal Function Following Mycotoxin Ingestion: Meta-Analysis of Published Experiments in Animals. Toxins 2013, 5, 396–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patience, J.F.; Ensley, S.M.; Hoyer, S. Mycotoxin Contamination of Corn: What It Is, What It Does to Pigs and What Can Be Done about It; Iowa Pork Industry Center Fact Sheets: Ames, IA, USA, 2010; p. 18. [Google Scholar]
- Martins, H.M.; Guerra, M.M.M.; Bernardo, F.M.D.A. Occurrence of aflatoxin B1 in dairy cow’s feed over 10 years in Portugal. Rev. Iberoam Micol. 2007, 24, 69–71. [Google Scholar] [CrossRef]
- Rohweder, D.; Valenta, H.; Sondermann, S.; Schollenberger, M.; Drochner, W.; Pahlow, G.; Döll, S.; Dänicke, S. Effect of different storage conditions on the mycotoxin contamination of Fusarium culmorum-infected and non-infected wheat straw. Mycotoxin Res. 2011, 27, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, I.; Handl, J.; Binder, E.M. Mycotoxin occurrence in commodities, feeds and feed ingredients sourced in the Middle East and Africa. Food Addit. Contam. Part B 2011, 4, 168–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pittet, A. Natural occurrence of mycotoxins in food and feeds: A decade in review. In Mycotoxins and Phytotoxins in Perspective at the Turn of the Millennium, Proceedings of the Xth International IUPAC Symposium on Mycotoxins and Phytotoxins, Guarujá, São Paulo, Brazil, 21–25 May 2000; De Koe, W.J., Samson, R.A., van Egmond, H.P., Gilbert, J., Sabino, M., Eds.; Ponsen & Looyen: Wageninger, The Netherlands, 2001; pp. 153–172. [Google Scholar]
- Kovalsky, P.; Kos, G.; Nährer, K.; Schwab, C.; Jenkins, T.; Schatzmayr, G.; Sulyok, M.; Krska, R. Co-occurrence of regulated, masked and emerging mycotoxins and secondary metabolites in finished feed and maize—An extensive survey. Toxins 2016, 8, 363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrencein animal feed-focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef] [Green Version]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide: Long-term analysis with special focus on Europe and Asia. J. Sci. Food Agri. 2013, 93, 2892–2899. [Google Scholar] [CrossRef]
- Franco, L.T.; Petta, T.; Rottinghaus, G.E.; Bordin, K.; Gomes, G.A.; Oliveira, C.A. Co-occurrence of mycotoxins in maize food and maize-based feed from small-scale farms in Brazil: A pilot study. Mycotoxin Res. 2019, 35, 65–73. [Google Scholar] [CrossRef]
- Liu, J.D.; Doupovec, B.; Schatzmayr, D.; Murugesan, G.R.; Bortoluzzi, C.; Villegas, A.M.; Applegate, T.J. The impact of deoxynivalenol, fumonisins, and their combination on performance, nutrient, and energy digestibility in broiler chickens. Poult. Sci. 2020, 99, 272–279. [Google Scholar] [CrossRef]
- Nathanail, A.V.; Syvahuoko, J.; Malachova, A.; Jestoi, M.; Varga, E.; Michlmayr, H.; Adam, G.; Sievilainen, E.; Berthiller, F.; Peltonen, K. Simultaneous determination of major type A and B trichothecenes, zearalenone and certain modified metabolites in Finnish cereal grains with a novel liquid chromatography-tandem mass spectrometric method. Anal. Bioanal. Chem. 2015, 407, 4745–4755. [Google Scholar] [CrossRef] [Green Version]
- Jiang, S.Z.; Yang, Z.B.; Yang, W.R.; Gao, J.; Liu, F.X.; Chen, C.C.; Chi, F. Physiopathological effects of zearalenone in post-weaning female piglets with or without montmorillonite clay adsorbent. Livest. Sci. 2010, 131, 130–136. [Google Scholar] [CrossRef]
- Jiang, S.Z.; Yang, Z.B.; Yang, W.R.; Yao, B.Q.; Zhao, H.; Liu, F.X.; Chen, C.C.; Chi, F. Effects of feeding purified zearalenone contaminated diets with or without clay enterosorbent on growth, nutrient availability, and genital organs in post-weaning female pigs. Asian-Austral. J. Anim. Sci. 2010, 23, 74–81. [Google Scholar] [CrossRef]
- Lee, M.J.; Kim, H.J. Development of an immunoaffinity chromatography and LC-MS/MS method for the determination of 6 zearalenones in animal feed. PLoS ONE 2018, 13, e0193584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romera, D.; Mateo, E.M.; Mateo-Castro, R.; Gómez, J.V.; Gimeno-Adelantado, J.V.; Jiménez, M. Determination of multiple mycotoxins in feedstuffs by combined use of UPLC–MS/MS and UPLC–QTOF–MS. Food Chem. 2018, 267, 140–148. [Google Scholar] [CrossRef]
- EFSA CONTAM Panel. Scientific Opinion on the risks for human and animal health related to the presence of modified forms of certain mycotoxins in food and feed. EFSA J. 2014, 12, 3916. [Google Scholar]
- EFSA CONTAM Panel. Scientific Opinion on the appropriateness to set a group health-based guidance value for zearalenone and its modified forms. EFSA J. 2016, 14, 4425. [Google Scholar]
- Bennett, G.A.; Shotwell, O.L.; Hesseltine, C.W. Destruction of zearalenone in contaminated corn. J. Am. Oil Chem. Soc. 1980, 57, 245–247. [Google Scholar] [CrossRef]
- Ryu, D.; Hanna, M.A.; Bullerman, L.B. Stability of zearalenone during extrusion of corn grits. J. Food Protect. 1999, 62, 1482–1484. [Google Scholar] [CrossRef]
- Rai, A.; Das, M.; Tripathi, A. Occurrence and toxicity of a fusarium mycotoxin, zearalenone. Crit. Rev. Food Sci. Nutr. 2019, 1–20. [Google Scholar] [CrossRef]
- Mirocha, C.J.; Pathre, S.V.; Robison, T.S. Comparative metabolism of zearalenone and transmission into bovine milk. Food Cosmet. Toxicol. 1981, 19, 25–30. [Google Scholar] [CrossRef]
- Bauer, J.; Heinritzi, K.; Gareis, M.; Gedek, B. Changes in the genital tract of female swine after feeding with practice-relevant amounts of zearalenone. Tierarztl. Prax. 1987, 15, 33–36. [Google Scholar] [PubMed]
- Biehl, M.L.; Prelusky, D.B.; Koritz, G.D.; Hartin, K.; Buck, W.B.; Trenholm, H.L. Biliary excretion and enterohepatic cycling of zearalenone in immature pigs. Toxicol. Appl. Pharmacol. 1993, 121, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Olsen, M. Metabolism of zearalenone in farm animals. In Fusarium Mycotoxins, Taxonomy and Pathogenicity, 1st ed.; Chelkowsi, J., Ed.; Elsevier: Amsterdam, The Netherlands, 1989; pp. 167–177. [Google Scholar]
- Metzler, M.; Pfeiffer, E.; Hildebrand, A.A. Zearalenone and its metabolites as endocrine disrupting chemicals. World Mycotoxin J. 2010, 3, 385–401. [Google Scholar] [CrossRef]
- Binder, S.B.; Schwartz-Zimmermann, H.E.; Varga, E.; Bichl, G.; Michlmayr, H.; Adam, G.; Berthiller, F. Metabolism of zearalenone and its major modified forms in pigs. Toxins 2017, 9, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faisal, Z.; Lemli, B.; Szerencsés, D.; Kunsági-Máté, S.; Bálint, M.; Hetényi, C.; Kuzma, M.; Mayer, M.; Poór, M. Interactions of zearalenone and its reduced metabolites α-zearalenol and β-zearalenol with serum albumins: Species differences, binding sites, and thermodynamics. Mycotoxin Res. 2018, 34, 269–278. [Google Scholar] [CrossRef]
- Ma, L.; Maragos, C.M.; Zhang, Y. Interaction of zearalenone with bovine serum albumin as determined by fluorescence quenching. Mycotoxin Res. 2018, 34, 39–48. [Google Scholar] [CrossRef]
- Diaz, G.J.; Cespedes, A.E. Natural occurrence of zearalenone in feeds and feedstuffs used in poultry and pig nutrition in Colombia. Mycotoxin Res. 1997, 13, 81–87. [Google Scholar] [CrossRef]
- Dalcero, A.; Magnoli, C.; Luna, M.; Ancasi, G.; Reynoso, M.M.; Chiacchiera, S.; Miazzo, R.; Palacio, G. Mycoflora and naturally occurring mycotoxins in poultry feeds in Argentina. Mycopathologia 1998, 141, 37–43. [Google Scholar] [CrossRef]
- Swamy, H.V.L.N.; Smith, T.K.; MacDonald, E.J.; Karrow, N.A.; Woodward, B.; Boermans, H.J. Effects of feeding a blend of grains naturally contaminated with Fusarium mycotoxins on growth and immunological measurements of starter pigs, and the efficacy of a polymeric glucomannan mycotoxin adsorbent. J. Anim. Sci. 2003, 81, 2792–2803. [Google Scholar] [CrossRef]
- Jaimez, J.; Fente, C.A.; Franco, C.M.; Cepeda, A.; Vázquez, B.I. A survey of the fungal contamination and presence of ochratoxin A and zearalenone on Spanish feed and raw materials. J. Sci. Food Agric. 2004, 84, 832–840. [Google Scholar] [CrossRef]
- Swamy, H.V.L.N.; Smith, T.K.; Karrow, N.A.; Boermans, H.J. Effects of feeding blends of grains naturally contaminated with Fusarium mycotoxins on growth and immunological parameters of broiler chickens. Poult. Sci. 2004, 83, 533–543. [Google Scholar] [CrossRef] [PubMed]
- Labuda, R.; Parich, A.; Berthiller, F.; Tančinová, D. Incidence of trichothecenes and zearalenone in poultry feed mixtures from Slovakia. Int. J. Food Microbiol. 2005, 105, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Döll, S.; Gericke, S.; Dänicke, S.; Raila, J.; Ueberschär, K.H.; Valenta, H.; Schnurrbusch, U.; Schweigert, F.J.; Flachowsky, G. The efficacy of a modified aluminosilicate as a detoxifying agent in Fusarium toxin contaminated maize containing diets for piglets. J. Anim. Physiol. Anim. Nutr. 2005, 89, 342–358. [Google Scholar] [CrossRef] [PubMed]
- Seeling, K.; Dänicke, S.; Ueberschär, K.H.; Lebzien, P.; Flachowsky, G. On the effects of Fusarium toxin-contaminated wheat and the feed intake level on the metabolism and carry over of zearalenone in dairy cows. Food Addit. Contam. 2005, 22, 847–855. [Google Scholar] [CrossRef]
- Martins, H.M.; Guerra, M.M.; Bernardo, F. Zearalenone, deoxynivalenol and fumonisins in mixed-feed for laying hens. Mycotoxin Res. 2006, 22, 206–210. [Google Scholar] [CrossRef]
- Borutova, R.; Faix, S.; Placha, I.; Gresakova, L.; Cobanova, K.; Leng, L. Effects of deoxynivalenol and zearalenone on oxidative stress and blood phagocytic activity in broilers. Arch. Anim. Nutr. 2008, 62, 303–312. [Google Scholar] [CrossRef]
- Griessler, K.; Rodrigues, I.; Handl, J.; Hofstetter, U. Occurrence of mycotoxins in Southern Europe. World Mycotoxin J. 2010, 3, 301–309. [Google Scholar] [CrossRef]
- Jiang, S.Z.; Yang, Z.B.; Yang, W.R.; Gao, J.; Liu, F.X.; Broomhead, J.; Chi, F. Effects of purified zearalenone on growth performance, organ size, serum metabolites, and oxidative stress in postweaning gilts. J. Anim. Sci. 2011, 89, 3008–3015. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, I.; Naehrer, K. A three-year survey on the worldwide occurrence of mycotoxins in feedstuffs and feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef]
- Schoevers, E.J.; Santos, R.R.; Colenbrander, B.; Fink-Gremmels, J.; Roelen, B.A. Transgenerational toxicity of Zearalenone in pigs. Reprod. Toxicol. 2012, 34, 110–119. [Google Scholar] [CrossRef]
- Jiang, S.Z.; Yang, Z.B.; Yang, W.R.; Wang, S.J.; Wang, Y.; Broomhead, J.; Johnston, S.L.; Chi, F. Effect on hepatonephric organs, serum metabolites and oxidative stress in post-weaning piglets fed purified zearalenone-contaminated diets with or without Calibrin-Z. Anim. Physiol. Anim. Nutr. 2012, 96, 1147–1156. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.P.; Chi, F.; Kim, I.H. Effects of montmorillonite clay on growth performance, nutrient digestibility, vulva size, faecal microflora, and oxidative stress in weaning gilts challenged with zearalenone. Anim. Feed Sci. Technol. 2012, 178, 158–166. [Google Scholar] [CrossRef]
- Kim, D.H.; Lee, I.H.; Do, W.H.; Nam, W.S.; Li, H.; Jang, H.S.; Lee, C. Incidence and levels of deoxynivalenol, fumonisins and zearalenone contaminants in animal feeds used in Korea in 2012. Toxins 2014, 6, 20–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, S.Z.; Li, Z.; Wang, G.Y.; Yang, Z.B.; Yang, W.R.; Zhang, G.G.; Wu, Y.B. Effects of Fusarium mycotoxins with yeast cell wall absorbent on hematology, serum biochemistry, and oxidative stress in broiler chickens. J. Appl. Poult. Res. 2014, 23, 165–173. [Google Scholar] [CrossRef]
- Dänicke, S.; Keese, C.; Meyer, U.; Starke, A.; Kinoshita, A.; Rehage, J. Zearalenone (ZEN) metabolism and residue concentrations in physiological specimens of dairy cows exposed long-term to ZEN-contaminated diets differing in concentrate feed proportions. Arch. Anim. Nutr. 2014, 68, 492–506. [Google Scholar] [CrossRef] [PubMed]
- Kongkapan, J.; Poapolathep, S.; Isariyodom, S.; Kumagai, S.; Poapolathep, A. Simultaneous detection of multiple mycotoxins in broiler feeds using a liquid chromatography tandem-mass spectrometry. J. Vet. Med. Sci. 2016, 78, 259–264. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.; Seo, D.J.; Jeon, S.B.; Ok, H.E.; Jung, H.; Choi, C.; Chun, H.S. Detection of foodborne pathogens and mycotoxins in eggs and chicken feeds from farms to retail markets. Korean J. Food Sci. Anim. Resour. 2016, 36, 463. [Google Scholar] [CrossRef] [Green Version]
- Bernhoft, A.; Christensen, E.; Sandvik, M. The Surveillance Programme for Mycotoxins and Fungi in Feed Materials, and Complete and Complementary Feed in Norway 2015; Norwegian Veterinary Institute: Oslo, Norway, 2016. [Google Scholar]
- Cheng, Q.; Jiang, S.Z.; Li, S.Q.; Wang, Y.X.; Zhang, C.Y.; Yang, W.R. Effects of low-dose zearalenone-contaminated diets with or without montmorillonite clay adsorbent on nutrient metabolic rates, serum enzyme activities, and genital organs of growing-laying hens. J. Appl. Poult. Res. 2017, 26, 367–375. [Google Scholar] [CrossRef]
- Hennig-Pauka, I.; Koch, F.J.; Schaumberger, S.; Woechtl, B.; Novak, J.; Sulyok, M.; Nagl, V. Current challenges in the diagnosis of zearalenone toxicosis as illustrated by a field case of hyperestrogenism in suckling piglets. Porc. Health Manag. 2018, 4, 18. [Google Scholar] [CrossRef]
- Reddy, K.E.; young Jeong, J.; Lee, Y.; Lee, H.J.; Kim, M.S.; Kim, D.W.; Jung, H.J.; Choe, C.; Oh, Y.K.; Lee, S.D. Deoxynivalenol-and zearalenone-contaminated feeds alter gene expression profiles in the livers of piglets. Asian-Austral. J. Anim. Sci. 2018, 31, 595. [Google Scholar] [CrossRef] [Green Version]
- Travel, A.; Metayer, J.P.; Mika, A.; Bailly, J.D.; Cleva, D.; Boissieu, C.; Le Guennec, J.; Albaric, O.; Labrut, S.; Lepivert, G.; et al. Toxicity of fumonisins, deoxynivalenol, and zearalenone alone and in combination in turkeys fed with the maximum European Union–tolerated level. Avian Dis. 2019, 63, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cheng, Y.; Wen, C.; Wang, W.; Kang, Y.; Wang, A.; Zhou, Y. The protective effects of modified palygorskite on the broilers fed a purified zearalenone-contaminated diet. Poult. Sci. 2019, 98, 3802–3810. [Google Scholar] [CrossRef] [PubMed]
- Hort, V.; Nicolas, M.; Travel, A.; Jondreville, C.; Maleix, C.; Baéza, E.; Engel, E.; Guérin, T. Carry-over assessment of fumonisins and zearalenone to poultry tissues after exposure of chickens to a contaminated diet-A study implementing stable-isotope dilution assay and UHPLC-MS/MS. Food Control 2020, 107, 106789. [Google Scholar] [CrossRef]
- Mirocha, C.J.; Robison, T.S.; Pawlosky, R.J.; Allen, N.K. Distribution and residue determination of (3H)-zearalenone in broilers. Toxicol. Appl. Pharm. 1982, 66, 77–87. [Google Scholar] [CrossRef]
- Buranatragool, K.; Poapolathep, S.; Isariyodom, S.; Imsilp, K.; Klangkaew, N.; Poapolathep, A. Dispositions and tissue residue of zearalenone and its metabolites α-zearalenol and β-zearalenol in broilers. Toxicol. Rep. 2015, 2, 351–356. [Google Scholar] [CrossRef] [Green Version]
- Devreese, M.; Antonissen, G.; Broekaert, N.; De Baere, S.; Vanhaecke, L.; De Backer, P.; Croubels, S. Comparative toxicokinetics, absolute oral bioavailability, and biotransformation of zearalenone in different poultry species. J. Agric. Food Chem. 2015, 63, 5092–5098. [Google Scholar] [CrossRef]
- Dänicke, S.; Ueberschär, K.H.; Valenta, H.; Matthes, S.; Matthäus, K.; Halle, I. Effects of graded levels of Fusarium toxin-contaminated wheat in Pekin duck diets on performance, health and metabolism of deoxynivalenol and zearalenone. Br. Poult. Sci. 2004, 45, 264–272. [Google Scholar] [CrossRef]
- Dänicke, S.; Ueberschär, K.H.; Halle, I.; Matthes, S.; Valenta, H.; Flachowsky, G. Effect of addition of a detoxifying agent to laying hen diets containing uncontaminated or Fusarium toxin-contaminated maize on performance of hens and on carryover of zearalenone. Poult. Sci. 2002, 81, 1671–1680. [Google Scholar] [CrossRef]
- Dailey, R.E.; Reese, R.E.; Brouwer, E.A. Metabolism of (14C) zearalenone in laying hens. J. Agric. Food Chem. 1980, 28, 286–291. [Google Scholar] [CrossRef]
- Dänicke, S.; Swiech, E.; Buraczewska, L.; Ueberschär, K.H. Kinetics and metabolism of zearalenone in young female pigs. J. Anim. Physiol. Anim. Nutr. 2005, 89, 268–276. [Google Scholar] [CrossRef]
- Gajęcka, M.; Zielonka, Ł.; Gajęcki, M. Activity of zearalenone in the porcine intestinal tract. Molecules 2017, 22, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsen, M.; Pettersson, H.; Sandholm, K.; Visconti, A.; Kiessling, K.H. Metabolism of zearalenone by sow intestinal mucosa in vitro. Food Chem. Toxicol. 1987, 25, 681–683. [Google Scholar] [CrossRef]
- Dänicke, S.; Brüssow, K.P.; Goyarts, T.; Valenta, H.; Ueberschär, K.H.; Tiemann, U. On the transfer of the Fusarium toxins deoxynivalenol (DON) and zearalenone (ZON) from the sow to the full-term piglet during the last third of gestation. Food Chem. Toxicol. 2007, 45, 1565–1574. [Google Scholar] [CrossRef] [PubMed]
- Döll, S.; Dänicke, S.; Ueberschär, K.H.; Valenta, H.; Schnurrbusch, U.; Ganter, M.; Klobasa, F.; Flachowsky, G. Effects of graded levels of Fusarium toxin contaminated maize in diets for female weaned piglets. Arch. Anim. Nutr. 2003, 57, 311–334. [Google Scholar] [CrossRef] [PubMed]
- Goyarts, T.; Dänicke, S.; Valenta, H.; Ueberschär, K.H. Carry-over of Fusarium toxins (deoxynivalenol and zearalenone) from naturally contaminated wheat to pigs. Food Addit. Contam. 2007, 24, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, E. Mykotoxine im Grundfutter und ihre Bedeutung im Carry-over-Geschehen. In Kreisläufe erwün-schter und unerwünschter Stoffe–ihre Bedeutung in der Nahrungskette, Informationsveranstaltung der Arbeitsgruppe “Carry over unerwünschter Stoffe in Futtermitteln’’, Braunschweig, Germany, October 1998; Forsten, L., Ed.; Bundesministerium für Ernährung, Landwirtschaftsverlag: Münster, Germany, 1998; pp. 19–31. [Google Scholar]
- Völkel, I.; Schröer-Merker, E.; Czerny, C.P. The carry-over of mycotoxins in products of animal origin with special regard to its implications for the European food safety legislation. Food Nutr. Sci. 2011, 2, 852–867. [Google Scholar] [CrossRef] [Green Version]
- Fink-Gremmels, J. Mycotoxins in cattle feeds and carry-over to dairy milk: A review. Food Addit. Contam: Part A. Chem. Anal. Control Expo. Risk Assess. 2008, 25, 172–180. [Google Scholar]
- Miles, C.O.; Erasmuson, A.F.; Wilkins, A.L.; Towers, N.R.; Smith, B.L.; Garthwaite, I.; Scahill, B.G.; Hansen, R.P. Ovine metabolism of zearalenone to alpha-zearalanol (zeranol). J. Agric. Food Chem. 1996, 44, 3244–3250. [Google Scholar] [CrossRef]
- Dong, M.; He, X.J.; Tulayakul, P.; Li, J.Y.; Dong, K.S.; Manabe, N.; Nakayama, H.; Kumagai, S. The toxic effects and fate of intravenously administered zearalenone in goats. Toxicon 2010, 55, 523–530. [Google Scholar] [CrossRef]
- Olsen, M.; Mirocha, C.J.; Abbas, H.K.; Johansson, B. Metabolism of high concentrations of dietary zearalenone by young male turkey poults. Poult. Sci. 1986, 65, 1905–1910. [Google Scholar] [CrossRef]
- Maryamma, K.I.; Manomohan, C.B.; Nair, M.G.; Ismail, P.K.; Sreekumaran, T.; Rajan, A. Pathology of zearalenone toxicosis in chicken and evaluation of zearalenone residues in tissues. Indian J. Anim. Sci. 1992, 62, 105–107. [Google Scholar]
- Dänicke, S.; Valenta, H.; Ueberschär, K.H.; Matthes, S. On the interactions between Fusarium toxin-contaminated wheat and non-starch-polysaccharide hydrolysing enzymes in turkey diets on performance, health and carry-over of deoxynivalenol and zearalenone. Br. Poult. Sci. 2007, 48, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Osselaere, A.; Devreese, M.; Goossens, J.; Vandenbroucke, V.; De Baere, S.; De Backer, P.; Croubels, S. Toxicokinetic study and absolute oral bioavailability of deoxynivalenol, T-2 toxin and zearalenone in broiler chickens. Food Chem. Toxicol. 2013, 51, 350–355. [Google Scholar] [CrossRef] [PubMed]
- James, L.J.; Smith, T.K. Effect of dietary alfalfa on zearalenone toxicity and metabolism in rats and swine. J. Anim. Sci. 1982, 55, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Enders, C. Zur Analytik von Zearalenon, α- und β- Zearalenol in Material pflanzlichen und tierischen Ursprungs. Ph.D. Thesis, LMU München, München, Germany, 1984. [Google Scholar]
- Lusky, K.; Tesch, D.; Göbel, R.; Haider, W. Gleichzeitige Verabreichung der Mykotoxine Ochratoxin A und Zearalenon über das Futter an Schweine—Einfluss auf Tiergesundheit und Rückstandsverhalten. Tierärztl. Umschau 1997, 52, 212–221. [Google Scholar]
- Zöllner, P.; Jodlbauer, J.; Kleinova, M.; Kahlbacher, H.; Kuhn, T.; Hochsteiner, W.; Lindner, W. Concentration levels of zearalenone and its metabolites in urine, muscle tissue, and liver samples of pigs fed with mycotoxin-contaminated oats. J. Agric. Food Chem. 2002, 50, 2494–2501. [Google Scholar] [CrossRef] [PubMed]
- Döll, S.; Dänicke, S.; Schnurrbusch, U. The effect of increasing concentrations of Fusarium toxins in the diets for piglets on histological parameters of the uterus. Mycotoxin Res. 2003, 19, 73–76. [Google Scholar] [CrossRef]
- Shreeve, B.J.; Patterson, D.S.P.; Roberts, B.A. The ‘carry-over’of aflatoxin, ochratoxin and zearalenone from naturally contaminated feed to tissues, urine and milk of dairy cows. Food Cosmet. Toxicol. 1979, 17, 151–152. [Google Scholar] [CrossRef]
- Hagler, W.M.; Danko, G.; Horvath, L.; Palyusik, M.; Mirocha, C.J. Transmission of zearalenone and its metabolite into ruminant milk. Acta Vet. Acad. Sci. H. 1980, 28, 209–216. [Google Scholar]
- Prelusky, D.B.; Scott, P.M.; Trenholm, H.L.; Lawrence, G.A. Minimal transmission of Zearalenone to milk of dairy cows. J. Einviron. Sci. Health. B 1990, 25, 87–103. [Google Scholar] [CrossRef]
- Usleber, E.; Renz, V.; Märtlbauer, E.; Terplan, G. Studies on the application of enzyme immunoassays for the Fusarium mycotoxins deoxynivalenol, 3-acetyldeoxynivalenol, and zearalenone. J. Vet. Med. B. 1992, 39, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Goll, M.; Valenta, H.; Oldenburg, E. Übergang von Zearalenon in die Milch von Kühen nach Langzeitfütterung. In Proceedings of the 17. Mykotoxin-Workshop, Braunschweig, Germany, 15–17 May 1995; FAL: Braunschweig, Germany, 1995; pp. 131–134. [Google Scholar]
- D’Mello, J.P.F.; Placinta, C.M.; MacDonald, A.M.C. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed Sci. Technol. 1999, 80, 183–205. [Google Scholar] [CrossRef] [Green Version]
- FAO/WHO. Zearalenone. Prepared by the fifty-third meeting of the joint FAO/WHO Expert Committee on Food Additives (JECFA). In Safety Evaluation of Certain Food Additives and Contaminants; WHO Food Additives Series 44; WHO: Geneva, Switzerland, 2000. [Google Scholar]
- Allen, N.K.; Mirocha, C.J.; Weaver, G.; Aakhus-Allen, S.; Bates, F. Effects of dietary zearalenone on finishing broiler chickens and young turkey poults. Poultry Sci. 1981, 60, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Weaver, G.A.; Kurtz, H.J.; Behrens, J.C.; Robison, T.S.; Seguin, B.E.; Bates, F.Y.; Mirocha, C.J. Effects of zearalenone on the fertility of virgin dairy heifers. Am. J. Vet. Res. 1986, 47, 1395–1397. [Google Scholar]
- Berthiller, F.; Schuhmacher, R.; Adam, G.; Krska, R. Formation, determination and significance of masked and other conjugated mycotoxins. Anal. Bioanal. Chem. 2009, 395, 1243–1252. [Google Scholar] [CrossRef]
- Catteuw, A.; Broekaert, N.; De Baere, S.; Lauwers, M.; Gasthuys, E.; Huybrechts, B.; Callebaut, A.; Ivanova, L.; Uhlig, S.; De Boevre, M.; et al. Insights into in vivo absolute oral bioavailability, biotransformation, and toxicokinetics of zearalenone, α-zearalenol, β-zearalenol, zearalenone-14-glucoside, and zearalenone-14-sulfate in pigs. J. Agric. Food Chem. 2019, 67, 3448–3458. [Google Scholar] [CrossRef]
- McNutt, S.H.; Purwin, P.; Murray, C. Vulvovaginitis in swine. J. Am. Vet. Med. Assoc. 1928, 73, 484. [Google Scholar]
- Koen, J.S.; Smith, H.C. An unusual case of genital involvement in swine associated with eating moldy corn. Vet. Med. 1945, 40, 131. [Google Scholar]
- Ciegler, A. Mycotoxins, Occurrence, chemistry, biological activity. Lloydia 1975, 38, 21–35. [Google Scholar]
- da Rocha, M.E.B.; Freire, F.D.C.O.; Maia, F.E.F.; Guedes, M.I.F.; Rondina, D. Mycotoxins and their effects on human and animal health. Food Control 2014, 36, 159–165. [Google Scholar] [CrossRef]
- Massart, F.; Saggese, G. Oestrogenic Mycotoxin Exposures and precocious pubertal development. Int. J. Androl. 2010, 33, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Mirocha, C.J.; Christensen, C.M.; Nelson, G.H. F-2 (zearalenone) estrogenic mycotoxin from Fusarium. Microbial toxins 2013, 7, 107–138. [Google Scholar]
- Le Guevel, R.; Pakdel, F. Assessment of oestrogenic potency of chemi-cals used as growth promoter by in vitro methods. Hum. Reprod. 2001, 16, 1030–1036. [Google Scholar]
- Fruhauf, S.; Novak, B.; Nagl, V.; Hackl, M.; Hartinger, D.; Rainer, V.; Labudova, S.; Adam, G.; Aleschko, M.; Moll, W.D.; et al. Biotransformation of the mycotoxin zearalenone to its metabolites hydrolyzed zearalenone (HZEN) and decarboxylated hydrolyzed zearaleneone (DHEN) diminishes its estrogenicity in vitro and in vivo. Toxins 2019, 11, 481. [Google Scholar] [CrossRef] [Green Version]
- Fink-Gremmels, J.; Malekinejad, H. Clinical effects and biochemical mechanisms associated with exposure to the mycoestrogen zearalenone. Anim. Feed Sci. Technol. 2007, 137, 326–341. [Google Scholar] [CrossRef]
- National Research Council of Canada. Mycotoxins: A Canadian Perspective; Scott, P.M., Trenholm, H.L., Sutton, M.D., Eds.; NRCC: Ottawa, QC, Canada, 1985. [Google Scholar]
- Marasas, W.F.O.; Nelson, P.E. Mycotoxicology-Introduction to Mycology, Plant Pathology, Chemistry, Toxicology and Pathology of Naturally Occurring Mycotoxicoses in Animals and Man; Pennsylvania State University Press: University Park, PA, USA, 1987. [Google Scholar]


| Item | Species | ZEN (µg/kg) | |
|---|---|---|---|
| EU | Poultry | - | - |
| Swine | Sows and fattening pigs | 250 | |
| Piglets and gilts | 100 | ||
| Ruminants | - | 500 | |
| FDA | Poultry | - | No guidance levels |
| Swine | Sows and fattening pigs | ||
| Piglets and gilts | |||
| Ruminants | - | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Applegate, T. Zearalenone (ZEN) in Livestock and Poultry: Dose, Toxicokinetics, Toxicity and Estrogenicity. Toxins 2020, 12, 377. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12060377
Liu J, Applegate T. Zearalenone (ZEN) in Livestock and Poultry: Dose, Toxicokinetics, Toxicity and Estrogenicity. Toxins. 2020; 12(6):377. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12060377
Chicago/Turabian StyleLiu, Jundi, and Todd Applegate. 2020. "Zearalenone (ZEN) in Livestock and Poultry: Dose, Toxicokinetics, Toxicity and Estrogenicity" Toxins 12, no. 6: 377. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins12060377




