The Effectiveness of Dietary Byproduct Antioxidants on Induced CYP Genes Expression and Histological Alteration in Piglets Liver and Kidney Fed with Aflatoxin B1 and Ochratoxin A
Abstract
:1. Introduction
2. Results
2.1. Diet Composition
2.2. Animal Performance
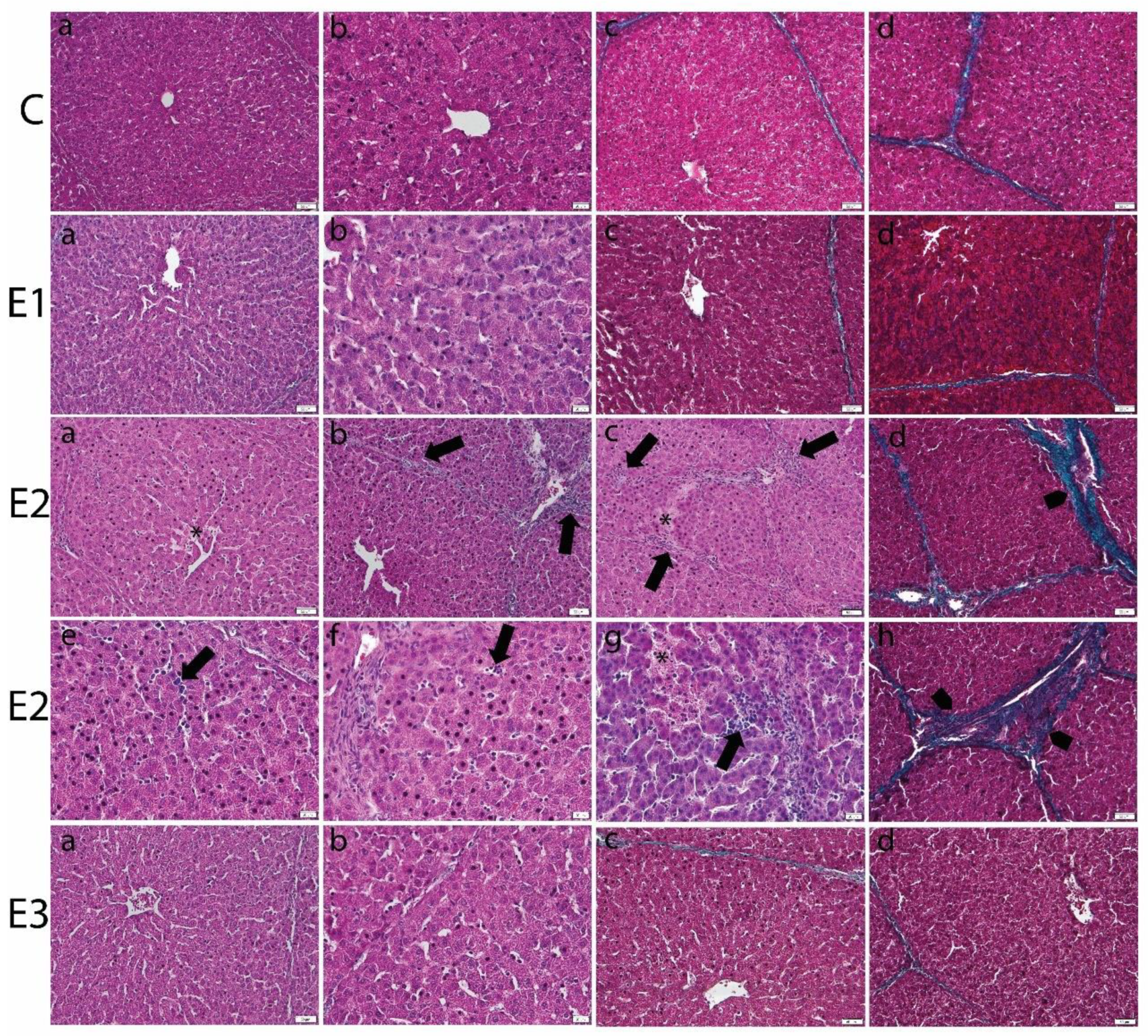
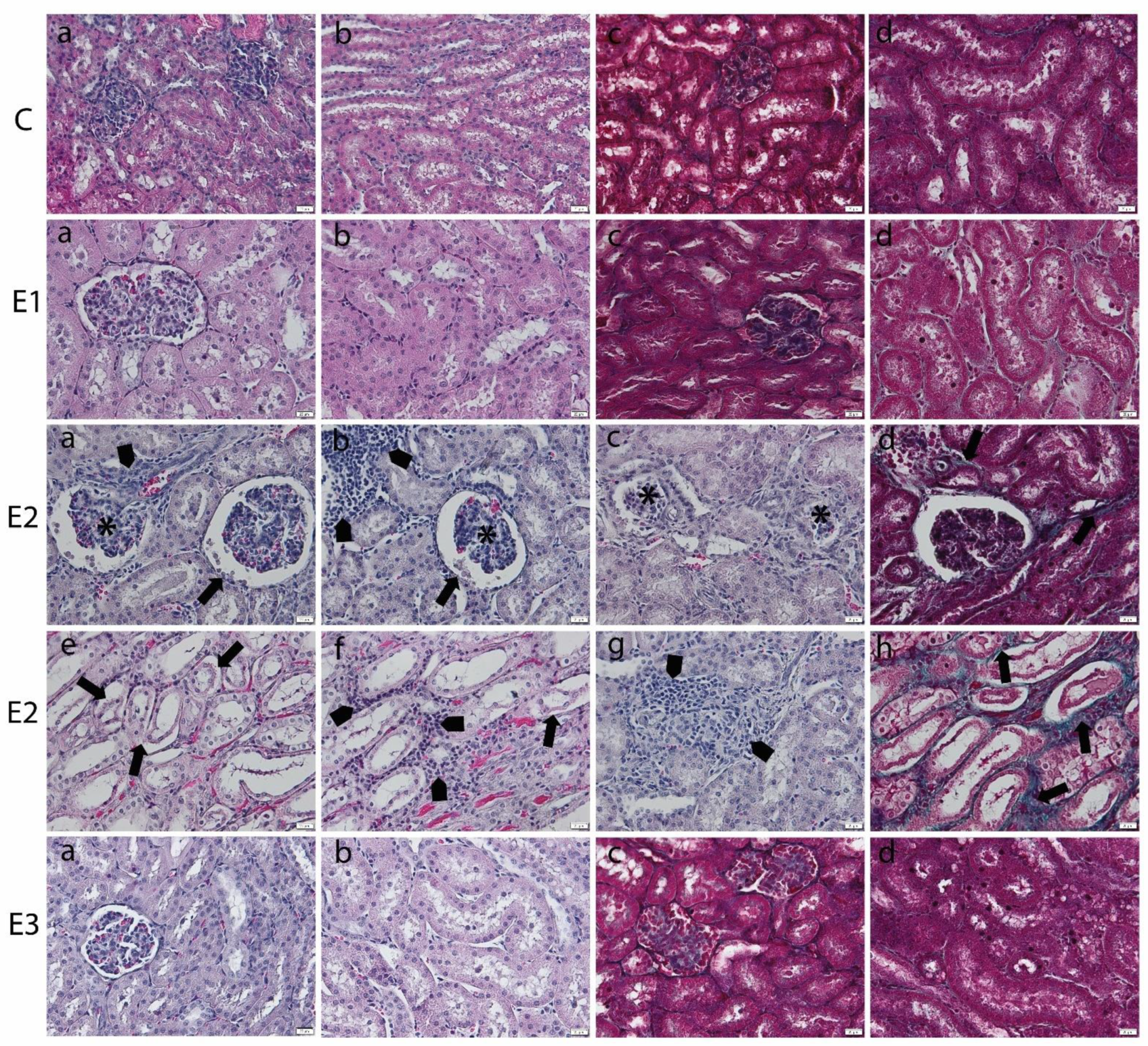
2.3. Histology of Liver and Kidney
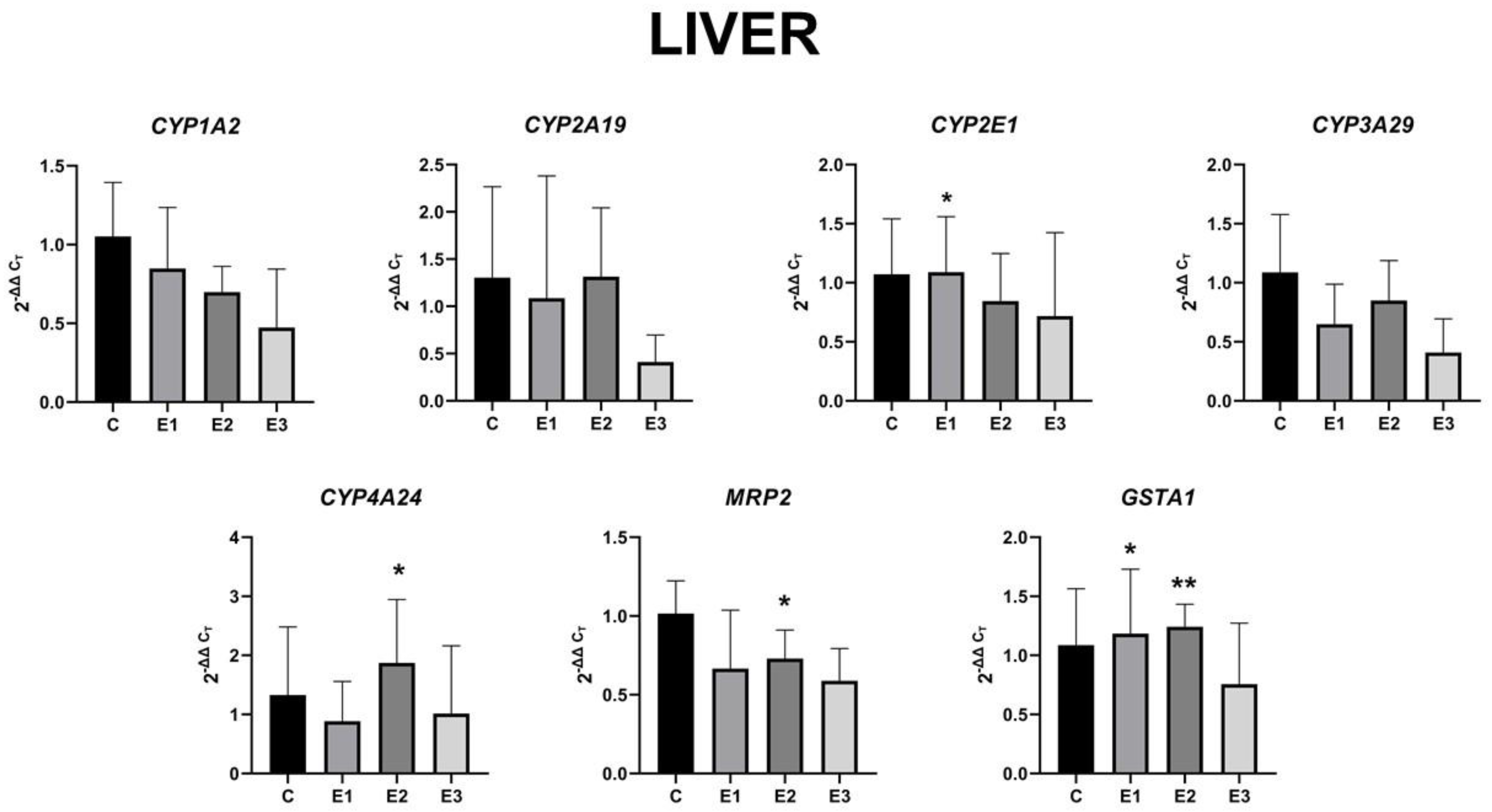
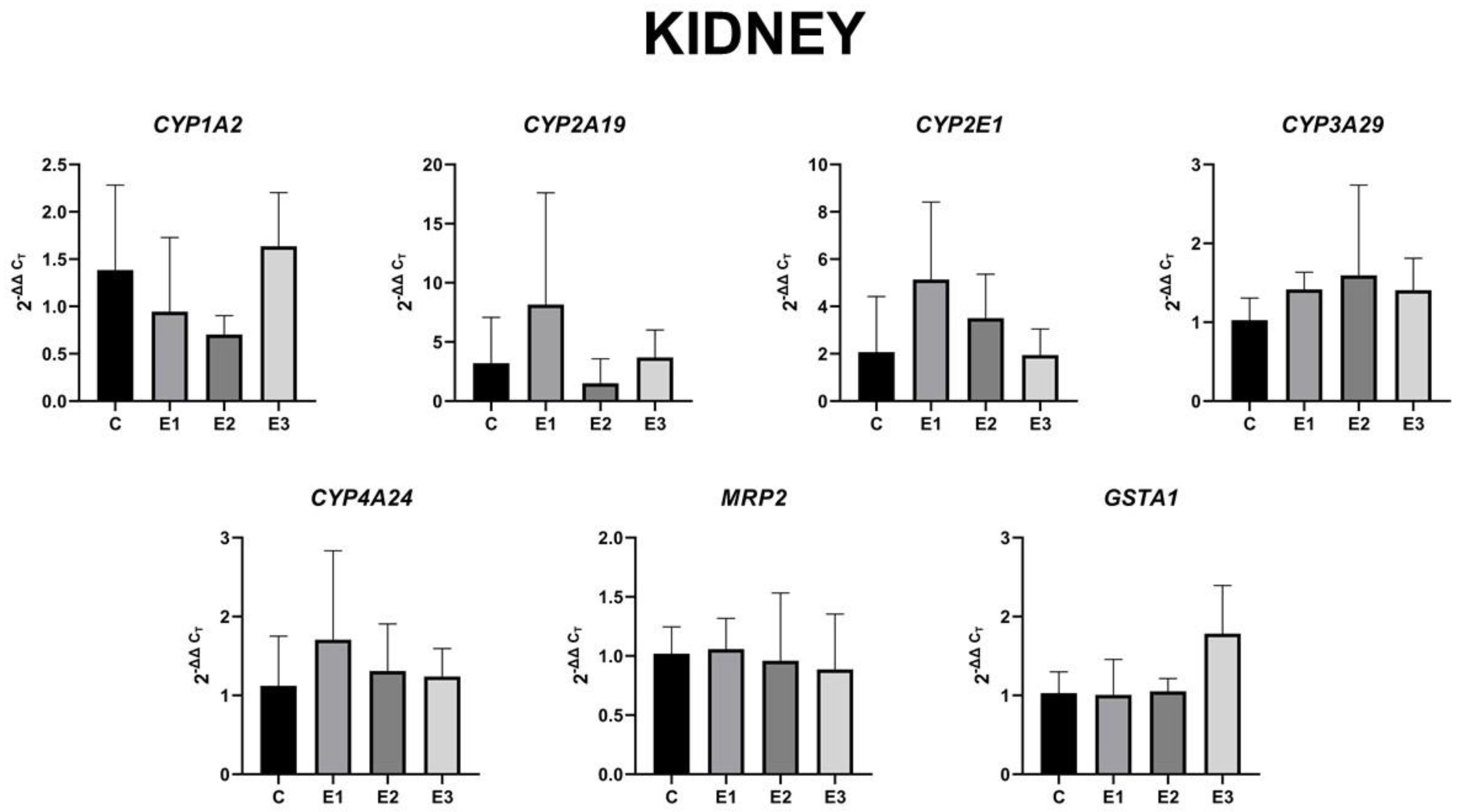
2.4. The Level of Gene Expression
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Experimental Design and Samples Collection
5.2. Feed Characterization
5.3. Plasma Biomarkers Analysis
5.4. Light Microscopy Examination
5.5. RNA Isolation
5.6. RNA Integrity Number (RIN)
5.7. Reverse Transcription
5.8. Primer Design
5.9. Real-Time PCR
5.10. Data Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liew, W.-P.-P.; Mohd-Redzwan, S. Mycotoxin: Its Impact on Gut Health and Microbiota. Front. Cell. Infect. Microbiol. 2018, 8, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desjardins, A.E.; Hohn, T.M. Mycotoxins in Plant Pathogenesis. Mol. Plant.-Microbe Interact. 2007, 10, 147–152. [Google Scholar] [CrossRef] [Green Version]
- Riley, R.T.; Pestka, J. Mycotoxins: Metabolism, mechanisms and biochemical markers. In The Mycotoxin Blue Book, 2nd ed.; Diaz, D., Ed.; Nottingham University Press: Nottingham, UK, 2005; pp. 279–294. [Google Scholar]
- Feddern, V.; Dors, G.C.; Tavernari, F.; Mazzuco, H.; Cunha, J.A.; Krabbe, E.L.; Scheuermann, G.N. Aflatoxins: Importance on animal nutrition. In Aflatoxins Recent Advances and Future Prospects; InTech Open Access: London, UK, 2013; pp. 171–195. [Google Scholar] [CrossRef] [Green Version]
- Medina, A.; Akbar, A.; Baazeem, A.; Rodriguez, A.; Magan, N. Climate change, food security and mycotoxins: Do we know enough? Fungal Biol. Rev. 2017, 31, 143–154. [Google Scholar] [CrossRef] [Green Version]
- Pierron, A.; Alassane-Kpembi, I.; Oswald, I.P. Impact of mycotoxin on immune response and consequences for pig health. Anim. Nutr. 2016, 2, 63–68. [Google Scholar] [CrossRef]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrence in animal feed—Focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Global Mycotoxin Occurrence in Feed: A Ten-Year Survey. Toxins 2020, 11, 375. [Google Scholar] [CrossRef] [Green Version]
- Marin, D.E.; Motiu, M.; Taranu, I. Food Contaminant Zearalenone and Its Metabolites Affect Cytokine Synthesis and Intestinal Epithelial Integrity of Porcine Cells. Toxins 2015, 7, 1979–1988. [Google Scholar] [CrossRef] [PubMed]
- Alassane-Kpembi, I.; Kolf-clauw, M.; Gauthier, T.; Abrami, R.; Abiola, F.A.; Oswald, I.P.; Puel, O. New insights into mycotoxin mixtures: The toxicity of low doses of Type B trichothecenes on intestinal epithelial cells is synergistic. Toxicol. Appl. Pharmacol. 2013, 272, 191–198. [Google Scholar] [CrossRef]
- Broom, L.J.; Wood, M.; Park, E.; Kingdom, U. Organic acids for improving intestinal health of poultry. Worlds Poult. Sci. J. 2015, 71, 630–642. [Google Scholar] [CrossRef]
- Filazi, A.; Sireli, U.T. Occurrence of aflatoxins in food. In Aflatoxins: Recent Advances and Future Prospects; Mehdi, R.-A., Ed.; InTech Open Access: London, UK, 2012; pp. 143–170. [Google Scholar]
- Seetha, A.; Munthali, W.; Msere, H.W.; Swai, E.; Muzanila, Y.; Sichone, E.; Tsusaka, T.W.; Rathore, A.; Okori, P. Occurrence of aflatoxins and its management in diverse cropping systems of central Tanzania. Mycotoxin Res. 2017, 33, 323–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ismail, A.; Gonçalves, B.L.; de Neeff, D.V.; Ponzilacqua, B.; Coppa, C.F.S.C.; Hintzsche, H.; Sajid, M.; Cruz, A.G.; Corassin, C.H.; Oliveira, C.A.F. Aflatoxin in foodstuffs: Occurrence and recent advances in decontamination. Food Res. Int. 2018, 113, 74–85. [Google Scholar] [CrossRef]
- Negash, D. Citation: Negash D (2018) A Review of Aflatoxin: Occurrence, Prevention, and Gaps in Both Food and Feed Safety. J. Appl. Microbiol. Res. 2018, 1, 1–35. [Google Scholar] [CrossRef] [Green Version]
- Arroyo-Manzanares, N.; Rodríguez-Estévez, V.; Arenas-Fernández, P.; García-Campaña, A.M.; Gámiz-Gracia, L. Occurrence of Mycotoxins in Swine Feeding from Spain. Toxins 2019, 11, 342. [Google Scholar] [CrossRef] [Green Version]
- Nazhand, A.; Durazzo, A.; Lucarini, M.; Souto, E.B.; Santini, A. Characteristics, Occurrence, Detection and Detoxification of Aflatoxins in Foods and Feeds. Foods 2020, 9, 644. [Google Scholar] [CrossRef] [PubMed]
- Dhama, K.; Singh, K.P. Aflatoxins- Hazard to Livestock and Poultry Production: A Review. J. Immunol. Immunopathol. 2014, 9, 1–15. [Google Scholar] [CrossRef]
- Devreese, M.; De Backer, P.; Croubels, S. Overview of the most important mycotoxins for the pig and poultry husbandry Overzicht van de meest belangrijke mycotoxines voor de varkens-en pluimveehouderij. Vlaams Diergeneeskd. Tijdschr. 2013, 82, 171–180. [Google Scholar] [CrossRef]
- Jatfa, J.W.; Wachida, N.M.; Ijabo, H.M.; Adamu, S.S. Aflatoxicosis Associated with Swine Stillbirth in the Piggery Farm University of Agriculture Makurdi. Curr. Trends Biomedical. Eng. Biosci. 2018, 13, 555873. [Google Scholar]
- Lee, H.S.; Lindahl, J.; Nguyen-Viet, H.; Khong, N.V.; Nghia, V.B.; Xuan, H.N.; Grace, D. An investigation into aflatoxin M1in slaughtered fattening pigs and awareness of aflatoxins in Vietnam. BMC Vet. Res. 2017, 13, 1–7. [Google Scholar] [CrossRef]
- Wilfred, E.G.; Dungworth, D.L.; Moulton, J.E. Pathologic Effects of Aflatoxin in Pigs. Vet. Pathol. 1968, 5, 370–384. [Google Scholar] [CrossRef]
- Dilkin, P.; Zorzete, P.; Mallmann, C.A.; Gomes, J.D.F.; Utiyama, C.E.; Oetting, L.L.; Corrêa, B. Toxicological effects of chronic low doses of aflatoxin B1 and fumonisin B1-containing Fusarium moniliforme culture material in weaned piglets. Food Chem. Toxicol. 2003, 41, 1345–1353. [Google Scholar] [CrossRef]
- Obuseh, F.A.; Jolly, P.E.; Jiang, Y.; Shuaib, F.M.B.; Waterbor, J.; Ellis, W.O.; Piyathilake, C.J.; Desmond, R.A.; Afriyie-Gyawu, E.; Phillips, T.D. Aflatoxin B1 albumin adducts in plasma and aflatoxin M1 in urine are associated with plasma concentrations of vitamins A and E. Int. J. Vitam. Nutr. Res. 2010, 80, 355–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfohl-leszkowicz, A.; Manderville, R.A. An Update on Direct Genotoxicity as a Molecular Mechanism of Ochratoxin A Carcinogenicity. Chem. Res. Toxicol. 2012, 25, 252–262. [Google Scholar] [CrossRef]
- Arbillaga, L.; Azqueta, A.; Van Delft, J.H.M.; López, A.; Cerain, D. In vitro gene expression data supporting a DNA non-reactive genotoxic mechanism for ochratoxin A. Toxicol. Appl. Pharmacol. 2007, 220, 216–224. [Google Scholar] [CrossRef]
- Asrani, R.K.; Patial, V.; Thakur, M. Ochratoxin A: Possible mecanisms of toxicity. In Ochratoxins-Biosynthesis, Detection and Toxicity; Porter, D., Ed.; Nova Science Publishers Inc.: New York, NY, USA, 2016; pp. 57–89. [Google Scholar]
- Marin, D.E.; Braicu, C.; Gras, M.A.; Pistol, G.C.; Petric, R.C.; Neagoe, I.B.; Palade, M.; Taranu, I.S.C. Low level of ochratoxin A affects genome-wide expression in kidney of pig. Toxicon 2017, 136, 67–77. [Google Scholar] [CrossRef]
- Ringot, D.; Chango, A.; Schneider, Y.J.; Larondelle, Y. Toxicokinetics and toxicodynamics of ochratoxin A, an update. Chem. Biol. Interact. 2006, 159, 18–46. [Google Scholar] [CrossRef] [PubMed]
- Kőszegi, T.; Poór, M. Ochratoxin a: Molecular interactions, mechanisms of toxicity and prevention at the molecular level. Toxins 2016, 8, 111. [Google Scholar] [CrossRef]
- Bayman, P.; Baker, J.L. Ochratoxins: A global perspective. Mycopathologia 2006, 162, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Peeradon, T.; Tichakorn, S.; Anupon, T.; Supatra, P. Modulation of Edible Plants on Hepatocellular Carcinoma Induced by Aflatoxin B. In Phytochemicals in Human Health; InTech Open Access: London, UK, 2020; pp. 1–23. [Google Scholar] [CrossRef]
- Guyonnet, D.; Belloir, C.; Suschetet, M.; Bon, A. Le Mechanisms of protection against aflatoxin B 1 genotoxicity in rats treated by organosulfur compounds from garlic. Carcinogenesis 2002, 23, 1335–1341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moudgil, V.; Redhu, D.; Dhanda, S.; Singh, J. A review of molecular mechanisms in the development of hepatocellular carcinoma by aflatoxin and hepatitis B and C viruses. J. Environ. Pathol. Toxicol. Oncol. 2013, 32, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xing, L.; Zhang, M.; Wang, J.; Zheng, N. The Toxic Effects of Aflatoxin B1 and Aflatoxin M1 on Kidney through Regulating L-Proline and Downstream Apoptosis. BioMed. Res. Intern. 2018, 2018, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanora, A.; Maes, D. The role of mycotoxins in pig reproduction: A review. Vet. Med. 2009, 54, 565–576. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Zhao, L.; Fan, Y.; Jia, Y.; Sun, L.; Ma, S.; Ji, C.; Ma, Q.; Zhang, J. Occurrence of mycotoxins in feed ingredients and complete feeds obtained from the Beijing region of China. J. Anim. Sci. Biotechnol. 2014, 5, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Svoboda, M.; Blahová, J.; Honzlová, A.; Kalinová, J.; Macharáčková, P.; Rosmus, J.; Mejzlík, V.; Kúkol, P.; Vlasáková, V.; Mikulková, K. Multiannual occurrence of mycotoxins in feed ingredients and complete feeds for pigs in the Czech Republic. Acta. Vet. Brno. 2019, 88, 291–301. [Google Scholar] [CrossRef] [Green Version]
- Khoshal, A.K.; Novak, B.; Martin, P.G.P.; Jenkins, T.; Neves, M.; Schatzmayr, G.; Oswald, I.P.; Pinton, P. Worldwide Finished Pig Feed and Their Combined Toxicity in Intestinal Cells. Toxins 2019, 11, 727. [Google Scholar] [CrossRef] [Green Version]
- Ma, R.; Zhang, L.; Liu, M.; Su, Y.; Xie, W.; Zhang, N. Individual and Combined Occurrence of Mycotoxins in Feed Ingredients and Complete Feeds in China. Toxins 2018, 10, 113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freitas, B.V.; Mota, M.M.; Del Santo, T.A.; Afonso, E.R.; Silva, C.C.; Utimi, N.B.P.; Barbosa, L.C.G.S.; Vilela, F.G.; Araújo, L.F. Mycotoxicosis in Swine: A Review. J. Anim. Prod. Adv. 2012, 2, 174–181. [Google Scholar]
- Adunphatcharaphon, S.; Petchkongkaew, A.; Greco, D.; D’Ascanio, V.; Visessanguan, W.; Avantaggiato, G. The Effectiveness of Durian Peel as a Multi-Mycotoxin Adsorbent. Toxins 2020, 8, 108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in Occurrence, Importance, and Mycotoxin Control Strategies: Prevention and Detoxification in Foods. Foods 2020, 28, 137. [Google Scholar] [CrossRef] [PubMed]
- Solís-Cruz, B.; Hernández-Patlán, D.; Beyssac, E.; Latorre, J.D.; Hernandez-Velasco, X.; Merino-Guzman, R.; Tellez, G.; López-Arellano, R. Evaluation of Chitosan and Cellulosic Polymers as Binding Adsorbent Materials to Prevent Aflatoxin B1, Fumonisin B1, Ochratoxin, Trichothecene, Deoxynivalenol, and Zearalenone Mycotoxicoses Through an In Vitro Gastrointestinal Model for Poultry. Polymers 2017, 19, 529. [Google Scholar] [CrossRef] [PubMed]
- Čolović, R.; Puvača, N.; Cheli, F.; Avantaggiato, G.; Greco, D.; Đuragić, O.; Kos, J.; Pinotti, L. Decontamination of Mycotoxin-Contaminated Feedstuffs and Compound Feed. Toxins 2019, 11, 617. [Google Scholar] [CrossRef] [Green Version]
- Vila-Donat, P.; Marín, S.; Sanchis, V.; Ramos, A.J. A review of the mycotoxin adsorbing agents, with an emphasis on their multi-binding capacity, for animal feed decontamination. Food Chem. Toxicol. 2018, 114, 246–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badr, A.N.; Abdel-Razek, A.G.; Youssef, M.; Shehata, M.; Hassanein, M.M.; Amra, H. Natural Antioxidants: Preservation Roles and Mycotoxicological Safety of Food. Egypt. J. Chem. 2021, 64, 285–298. [Google Scholar] [CrossRef]
- Gugliandolo, E.; Peritore, A.F.; D’Amico, R.; Licata, P.; Crupi, R. Evaluation of Neuroprotective Effects of Quercetin against Aflatoxin B1-Intoxicated Mice. Animals 2020, 21, 898. [Google Scholar] [CrossRef] [PubMed]
- Antonissen, G.; Devreese, M.; De Baere, S.; Martel, A.; Van Immerseel, F.; Croubels, S. Impact of Fusarium mycotoxins on hepatic and intestinal mRNA expression of cytochrome P450 enzymes and drug transporters, and on the pharmacokinetics of oral enrofloxacin in broiler chickens. Food Chem. Toxicol. 2017, 101, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Zaragozá, C.; Villaescusa, L.; Monserrat, J.; Zaragozá, F.; Álvarez-Mon, M. Potential Therapeutic Anti-Inflammatory and Immunomodulatory Effects of Dihydroflavones, Flavones, and Flavonols. Molecules 2020, 25, 1017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Mycotoxins. Children’s Health and the Environment. Available online: https://www.who.int/ceh/capacity/mycotoxins.pdf (accessed on 3 January 2021).
- Perrone, G.; Ferrara, M.; Medina, A.; Pascale, M.; Magan, N. Toxigenic Fungi and Mycotoxins in a Climate Change Scenario: Ecology, Genomics, Distribution, Prediction and Prevention of the Risk. Microorganisms 2020, 8, 1496. [Google Scholar] [CrossRef]
- Joubrane, K.; Mnayer, D.; El Khoury, A.; El Khoury, A.; Awad, E. Co-Occurrence of Aflatoxin B1 and Ochratoxin A in Lebanese Stored Wheat. J. Food Prot. 2020, 83, 1547–1552. [Google Scholar] [CrossRef]
- Ibañez-Vea, M.; González-Peñas, E.; Lizarraga, E.; López de Cerain, A. Co-occurrence of aflatoxins, ochratoxin A and zearalenone in barley from a northern region of Spain. Food Chem. 2012, 1, 35–42. [Google Scholar] [CrossRef]
- Gamze, N.K.; Fatih, O.; Bulent, K. Co-occurrence of aflatoxins and ochratoxin A in cereal flours commercialised in Turkey. Food Control. 2015, 54, 275–281. [Google Scholar] [CrossRef]
- Ozbey, F.; Kabak, B. Natural co-occurrence of aflatoxins and ochratoxin A in spices. Food Control. 2012, 28, 354–361. [Google Scholar] [CrossRef]
- Santos Pereira, C.; Cunha, S.; Fernandes, J.O. Prevalent Mycotoxins in Animal Feed: Occurrence and Analytical Methods. Toxins 2019, 11, 290. [Google Scholar] [CrossRef] [Green Version]
- Taranu, I.; Marin, D.E.; Palade, M.; Pistol, G.C.; Chedea, V.S.; Gras, M.A.; Rotar, C. Assessment of the e ffi cacy of a grape seed waste in counteracting the changes induced by a fl atoxin B1 contaminated diet on performance, plasma, liver and intestinal tissues of pigs after weaning. Toxicon 2019, 162, 24–31. [Google Scholar] [CrossRef]
- Nilova, L.; Malyutenkova, S. The possibility of using powdered sea-buckthorn in the development of bakery products with antioxidant properties. Agro. Res. 2018, 16, 1444–1456. [Google Scholar] [CrossRef]
- Balogh, K.; Hausenblasz, J.; Weber, M.; Erdélyi, M.; Fodor, J.; Mézes, M. Effects of ochratoxin A on some production traits, lipid peroxide and glutathione redox status of weaned piglets. Acta. Vet. Hung. 2007, 55, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Marin, D.E.; Taranu, I. Ochratoxin A and its effects on immunity. Toxin Rev. 2015, 34, 11–20. [Google Scholar] [CrossRef]
- Ayed-Boussema, I.; Pascussi, J.M.; Zaied, C.; Maurel, P.; Bacha, H.; Hassen, W. CYP1A2 gene expression in primary cultured human hepatocytes: A possible activation of nuclear receptors. Drug Chem. Toxicol. 2012, 35, 71–80. [Google Scholar] [CrossRef]
- Jiang, Z.; Gu, L.; Liang, X.; Cao, B.; Zhang, J.; Guo, X. The Effect of Selenium on CYP450 Isoform Activity and Expression in Pigs. Biol. Trace. Elem. Res. 2020, 196, 454–462. [Google Scholar] [CrossRef]
- Altay, A.; Bozoğlu, F. Salvia fruticosa Modulates mRNA Expressions and Activity Levels of Xenobiotic Metabolizing CYP1A2, CYP2E1, NQO1, GPx, and GST Enzymes in Human. Nutr. Cancer 2017, 69, 892–903. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, M.K.; Zamaratskaia, G.; Ekstrand, B. Gender-related Differences in Cytochrome P450 in Porcine Liver-Implication for Activity, Expression and Inhibition by Testicular Steroids. Reprod. Domest. Anim. 2011, 46, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Nwafor, I.C.; Shale, K.; Achilonu, M.C. Chemical Composition and Nutritive Benefits of Chicory (Cichorium intybus) as an Ideal Complementary and/or Alternative Livestock Feed Supplement. Sci. World J. 2017, 2017, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dietrich, C. Antioxidant Functions of the Aryl Hydrocarbon Receptor. Stem. Cells. Int. 2016, 2016, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kapelyukh, Y.; Henderson, C.J.; Scheer, N.; Rode, A.; Wolf, C.R. Defining the Contribution of CYP1A1 and CYP1A2 to Drug Metabolism Using Humanized CYP1A1/1A2 and Cyp1a1/Cyp1a2 Knockout Mice. Drug Metab. Dispos. 2019, 47, 907–918. [Google Scholar] [CrossRef]
- Sansen, S.; Yano, J.K.; Rosamund, L.; Schoch, G.A.; Keith, J.; Stout, C.D.; Johnson, E.F.; Sansen, S.; Yano, J.K.; Reynald, R.L.; et al. Adaptations for the Oxidation of Polycyclic Aromatic. J. Biol. Chem. 2007, 282, 14348–14355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schelstraete, W.; De Clerck, L.; Govaert, E.; Mil, J.; Devreese, M.; Deforce, D.; D. Bocxlaer, J.; Croubels, S. Characterization of Porcine Hepatic and Intestinal Drug Metabolizing CYP450: Comparison with Human Orthologues from A Quantitative, Activity and Selectivity Perspective. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, J.; Mu, P.; Deng, Y. Mycotoxins: Cytotoxicity and biotransformation in animal cells. Toxicol. Res. (Camb.) 2016, 5, 377–387. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.; Desai, L.P. Reciprocal regulation of TGF- β and reactive oxygen species: A perverse cycle for fi brosis. Redox Biol. 2015, 6, 565–577. [Google Scholar] [CrossRef] [Green Version]
- Muller, G.F. Effect of transforming growth factor- b 1 on cytochrome P450 expression: Inhibition of CYP1 mRNA and protein expression in primary rat hepatocytes. Arch. Toxicol 2000, 74, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Penner, N.; Woodward, C.; Prakash, C. Appendix: Drug Metabolizing Enzymes and Biotransformation Reactions. ADME 2012, 2012, 545–565. [Google Scholar] [CrossRef]
- Pyo, M.C.; Shin, H.S.; Jeon, G.Y.; Lee, K.-W. Synergistic Interaction of Ochratoxin A and Acrylamide Toxins in Human Kidney and Liver Cells. Biol Pharm Bull. 2020, 43, 1346–1355. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, L.; Yang, T.; Feng, Y.L.; Vaziri, N.D.; Liu, B.L.; Liu, Q.Q.; Guo, Y. Aryl hydrocarbon receptor activation mediates kidney disease and renal cell carcinoma. J. Transl. Med. 2019, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, M.K.; Zamaratskaia, G. Regulation of Porcine Hepatic Cytochrome P450—Implication for Boar Taint. CSBJ 2014, 11, 106–112. [Google Scholar] [CrossRef] [Green Version]
- Burkina, V.; Rasmussen, M.K.; Olünychenko, Y.; Zamaratskaia, G. Porcine cytochrome 2A19 and 2E1. Basic Clin. Pharmacol. Toxicol. 2019, 124, 32–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pitarque, M.; Rodriguez-Antona, C.; Oscarson, M.; Ingelman-Sundberg, M. Transcriptional regulation of the human CYP2A6 gene. J. Pharmacol. Exp. Ther. 2005, 313, 814–822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunius, C.; Andersson, K.; Zamaratskaia, G. Expression and activities of hepatic cytochrome P450 (CYP1A, CYP2A and CYP2E1) in entire and castrated male pigs. Animal 2012, 6, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Yokota, S.; Higashi, E.; Fukami, T.; Yokoi, T.; Nakajima, M. Human CYP2A6 is regulated by nuclear factor-erythroid 2 related factor 2. Biochem. Pharmacol. 2011, 81, 289–294. [Google Scholar] [CrossRef] [Green Version]
- Tanner, J.; Tyndale, R.F. Variation in CYP2A6 Activity and Personalized Medicine. J. Pers Med. 2017, 6, 18. [Google Scholar] [CrossRef] [Green Version]
- Diaz, G.J.; Murcia, H.W.; Cepeda, S.M.; Boermans, H.J. The role of selected cytochrome P450 enzymes on the bioactivation of aflatoxin B1 by duck liver microsomes. Avian Pathol. 2010, 39, 279–286. [Google Scholar] [CrossRef]
- Kalbolandi, S.M.; Gorji, A.V.; Babaahmadi-Rezaei, H.; Mansouri, E. Luteolin confers renoprotection against ischemia–reperfusion injury via involving Nrf2 pathway and regulating miR320. Mol. Biol. Rep. 2019, 46, 4039–4047. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Hussein, O.E.; Abd El-Twab, S.M.; Hozayen, W.G. Ferulic acid protects against methotrexate nefrotoxicity via activation of Nrf2/ARE/HO-1 signaling and PPARγ and suppression of NF-kB/NLRP3 inflammasome axis. Food Funct. 2019, 10, 4593–4607. [Google Scholar] [CrossRef]
- Tambyrajah, W.S.; Doran, E.; Wood, J.D.; Mcgivan, J.D. The pig CYP2E1 promoter is activated by COUP-TF1 and HNF-1 and is inhibited by androstenone. Arch. Biochem. Biophys. 2004, 431, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Park, E.C.; Kim, S.I.; Hong, Y.; Hwang, J.W.; Cho, G.; Cha, H.; Han, J.; Yun, C.; Park, S.; Jang, I.; et al. Inhibition of CYP4A Reduces Hepatic Endoplasmic Reticulum Stress and Features of Diabetes in Mice. Gastroenterology 2014, 147, 860–869. [Google Scholar] [CrossRef]
- Raucy, J.L.; Lasker, J.; Ozaki, K.; Zoleta, V. Regulation of CYP2E1 by Ethanol and Palmitic Acid and CYP4A11 by Clofibrate in Primary Cultures of Human Hepatocytes. Toxicol. Sci. 2004, 241, 233–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sung, M.; Kim, I.; Park, M.; Whang, Y.; Lee, M. Differential effects of dietary fatty acids on the regulation of CYP2E1 and protein kinase C in human hepatoma HepG2 cells. J. Med. Food 2004, 7, 197–203. [Google Scholar] [CrossRef]
- Wang, W.; Zhai, S.; Xia, Y.; Wang, H.; Ruan, D.; Zhou, T.; Zhu, Y.; Zhang, H.; Zhang, M.; Ye, H.; et al. Ochratoxin A induces liver inflammation: Involvement of intestinal microbiota. Microbiome 2019, 7, 1–14. [Google Scholar] [CrossRef]
- Lin, Q.; Kang, X.; Li, X.; Wang, T.; Liu, F.; Jia, J.; Jin, Z.; Id, Y.X. NF-κB-mediated regulation of rat CYP2E1 by two independent signaling pathways. PLoS ONE 2019, 14, e0225531. [Google Scholar] [CrossRef]
- Meyer, C.; Nadkarni, V.; Nadkarni, K.; Stumvoll, M.; Gerich, J. Human kidney free fatty acid and glucose uptake: Evidence for a renal glucose-fatty acid cycle. Am. J. Phisiol. 1997, 273, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Pu, Q.; Wang, E.; Yu, C. Celery extract inhibits mouse CYP2A5 and human CYP2A6 activities via different mechanisms. Oncol. Lett 2016, 12, 5309–5314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ling, D.; Salvaterra, P.M. Robust RT-qPCR Data Normalization: Validation and Selection of Internal Reference Genes during Post-Experimental Data Analysis. PLoS ONE 2011, 6, e17762. [Google Scholar] [CrossRef] [Green Version]
- Messina, A.; Nannelli, A.; Fiorio, R.; Longo, V.; Gervasi, P.G. Expression and inducibility of and CYP2B22, 3A22, 3A29, 3A46 by rifampicin in the respiratory and olfactory mucosa of pig. Toxicology 2009, 260, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Arias, C.A.; Crespo-Sempre, S.; Sanchis, V.; Ramos, A.J. Modulation of the xenobiotic transformation system and inflammatory response by ochratoxin A exposure using a co-culture system of Caco-2 and HepG2 cells. Food Chem. Toxicol. 2015, 86, 245–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rasmussen, M.K. Porcine cytochrome P450 3A: Current status on expression and regulation. Arch. Toxicol. 2020, 94, 1899–1914. [Google Scholar] [CrossRef]
- Zepnik, H.; Pa, A.; Schauer, U.; Dekant, W. Ochratoxin A-Induced Tumor Formation: Is There a Role of Reactive Ochratoxin A Metabolites? Toxicol. Sci. 2001, 59, 59–67. [Google Scholar] [CrossRef]
- Robertson, G.; Leclercq, I.; Farrell, G.C.; Steatosis, C.F.N.; Ii, S. Nonalcoholic Steatosis and Steatohepatitis II. Cytochrome. Am. J. Physiol. Gastrointest Liver Pysiol. 2020, 281, 1135–1139. [Google Scholar] [CrossRef]
- Leclercq, I.A.; Gonzalez, F.J.; Graham, R.; Leclercq, I.A.; Farrell, G.C.; Field, J.; Bell, D.R.; Gonzalez, F.J.; Robertson, G.R. CYP2E1 and CYP4A as microsomal catalysts of lipid peroxides in murine nonalcoholic steatohepatitis. J. Clin. Investig. 2000, 105, 1067–1075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Xu, W.; Zhai, T.; You, J.; Chen, Y. Silibinin ameliorates hepatic lipid accumulation and oxidative stress in mice with non-alcoholic steatohepatitis by regulating CFLAR-JNK pathway. Acta Pharm. Sin. B 2019, 9, 745–757. [Google Scholar] [CrossRef]
- Stading, R.; Couroucli, X.; Lingappan, K.; Moorthy, B. The role of cytochrome P450 (CYP) enzymes in hyperoxic lung injury. Expert Opin. Drug Metab. Toxicol. 2020, 13, 1–8. [Google Scholar] [CrossRef]
- Ruan, D.; Zhu, Y.W.; Fouad, A.M.; Yan, S.J.; Chen, W.; Zhang, Y.N.; Xia, W.G.; Wang, S.; Jiang, S.Q.; Yang, L.; et al. Dietary curcumin enhances intestinal antioxidant capacity in ducklings via altering gene expression of antioxidant and key detoxification enzymes. Poult. Sci. 2018, 98, 3705–3714. [Google Scholar] [CrossRef] [PubMed]
- Jedlitschky, G.; Hoffmann, U.; Kroemer, H.K. Structure and function of the MRP2 (ABCC2) protein and its role. Expert Opin. Drug Metab. Toxicol. 2006, 2, 351–366. [Google Scholar] [CrossRef]
- Gekle, M.; Mildenberger, S.; Freudinger, R.; Silbernagl, S. pH of endosomes labelled by receptor-mediated and fluid-phase endocytosis and its possible role for the regulation of endocy-totic uptake. In Studies in Honour of Karl Julius Ultrich. An Australian Symposium; Poronnik, P., Cook, D.I., Young, J.A., Eds.; Wild & Woolley: Glebe, Australia, 1994; pp. 45–49. [Google Scholar]
- Zhang, J.; Pan, Z.; Moloney, S.; Sheppard, A. RNA-Seq Analysis Implicates Detoxification Pathways in Ovine Mycotoxin Resistance. PLoS ONE 2014, 9, e99975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghunath, A.; Sundarraj, K.; Nagarajan, R.; Arfuso, F.; Bian, J. Redox Biology Antioxidant response elements: Discovery, classes, regulation and potential applications. Redox Biol. 2018, 17, 297–314. [Google Scholar] [CrossRef]
- Ghadiri, S.; Spalenza, V.; Dellafiora, L.; Badino, P.; Barbarossa, A.; Dall, C.; Nebbia, C.; Girolami, F. Toxicology in Vitro Modulation of aflatoxin B1 cytotoxicity and aflatoxin M1 synthesis by natural antioxidants in a bovine mammary epithelial cell line. Toxicol. Vitr. 2019, 57, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Corcuera, L.; Vettorazzi, A.; Arbillaga, L.; Pérez, N.; Gloria, A.; Azqueta, A.; González-peñas, E.; García-jalón, J.A.; López, A.; Cerain, D. Genotoxicity of Aflatoxin B1 and Ochratoxin A after simultaneous application of the in vivo micronucleus and comet assay. Food Chem. Toxicol. 2015, 76, 116–124. [Google Scholar] [CrossRef]
- Shin, H.S.; Lee, H.J.; Pyo, M.C.; Ryu, D.; Lee, K.-W. Ochratoxin A-Induced Hepatotoxicity through Phase I and Phase II Reactions Regulated by AhR in Liver Cells. Toxins (Basel) 2019, 11, 377. [Google Scholar] [CrossRef] [Green Version]
- Boudra, H.S.; Saivin, S.; Buffiere, C.; Morgavi, D.P. Short communication: Toxicokinetics of ochratoxin A in dairy ewes and carryover to milk following a single or long-term ingestion of contaminated feed. J. Dairy Sci. 2013, 96, 6690–6696. [Google Scholar] [CrossRef] [Green Version]
- Festing, S.; Wilkinson, R. The ethics of animal research. Talking Point on the use of animals in scientific research. EMBO Rep. 2007, 8, 526–530. [Google Scholar] [CrossRef] [Green Version]
- Taranu, I.; Braicu, C.; Marin, D.E.; Pistol, G.C.; Motiu, M.; Balacescu, L.; Beridan Neagoe, I.; Burlacu, R. Exposure to zearalenone mycotoxin alters in vitro porcine intestinal epithelial cells by differential gene expression. Toxicol. Lett. 2015, 232, 310–325. [Google Scholar] [CrossRef]
- Taranu, I.; Habeanu, M.; Gras, M.A.; Pistol, G.C.; Lefter, N.; Palade, M.; Ropota, M.; Chedea, V.S.; Marin, D.E. Assessment of the effect of grape seed cake inclusion in the diet of healthy fattening- finishing pigs. J. Anim. Physiol. Anim. Nutr. (Berl) 2017, 102, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hermenean, A.; Damache, G.; Albu, P.; Ardelean, A.; Ardelean, G.; Puiu Ardelean, D.; Horge, M.; Nagy, T.; Braun, M.; Zsuga, M.; et al. Histopatological alterations and oxidative stress in liver and kidney of Leuciscus cephalus following exposure to heavy metals in the Tur River, North Western Romania. Ecotoxicol. Environ. Saf. 2015, 119, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, A.; Nijveen, H.; Rao, X.; Bisseling, T. Primer3Plus, an enhanced web interface to Primer3. Nucleic Acids Res. 2007, 35, 71–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altschup, S.F.; Gish, W.; Pennsylvania, T.; Park, U. Basic Local Alignment Search Tool 2Department of Computer Science. J. Mol. Biol. 1990, 403–410. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real- Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Sandercock, D.A.; Coe, J.E.; Di, P.; Edwards, S.A. Research in Veterinary Science Determination of stable reference genes for RT-qPCR expression data in mechanistic pain studies on pig dorsal root ganglia and spinal cord. Res. Vet. Sci. 2017, 114, 493–501. [Google Scholar] [CrossRef] [PubMed]
| Byproducts | DM (103 °C) % | CP (%) | EE (%) | Ash (%) | Carbohydrates (mg/g) | |||
|---|---|---|---|---|---|---|---|---|
| Fructose | Glucose | Sucrose | Maltose | |||||
| Sea buckthorn meal | 84.48 | 15.67 | 10.28 | 2.75 | 9.78 | 7.68 | 8.03 | 0.43 |
| Grapeseed meal | 90.85 | 11.32 | 6.17 | 3.34 | 8.34 | 5.60 | 3.49 | 0.54 |
| Saturated Fatty Acids | Sea Buckthorn Meal | Grapeseed Meal | Unsaturated Fatty Acids | Sea Buckthorn Meal | Grapeseed Meal |
|---|---|---|---|---|---|
| Butiric (4:0) | 0.07 | 0.12 | Miristoleic (14:1) | 0.09 | 0.05 |
| Caproic (6:0) | 0.07 | 0.16 | Pentadecenoic (C15:1) | 0.00 | 0.08 |
| Caprilic (10:0) | 0.20 | 0.18 | Palmitoleic (C16:1n-7) | 14.28 | 0.33 |
| Capric (10:0) | 0.24 | 0.17 | Heptadecenoic (17:1) | 0.05 | 0.00 |
| Lauric (12:0) | 0.03 | 0.03 | Oleic cis (C18:1n-9) | 31.07 | 14.66 |
| Miristic (C14:0) | 0.93 | 0.59 | Linoleic cis (C18:2n-6) | 18.59 | 67.35 |
| Pentadecanoic (15:0) | 0.17 | 0.07 | Linolenic (C18:3n-6) | 0.00 | 0.04 |
| Palmitic (C16:0) | 24.32 | 9.69 | α -Linolenic (C18:3n-3) | 6.09 | 0.94 |
| Heptadecanoic (17:0) | 0.12 | 0.09 | Octadecatetraenoic (C18:4n-3) | 0.28 | 0.23 |
| Stearic (C18:0) | 2.00 | 3.56 | Eicosadienoic (C20:2n-6) | 0.44 | 0.21 |
| Arachidonic (C20:4n-6) | 0.00 | 0.20 | |||
| Eicosapentaenoic (C20:5n-3) | 0.19 | 0.26 | |||
| Lignoceric (C24:0) | 0.25 | 0.31 | |||
| Nervonic (C24:1n-9) | 0.00 | 0.13 | |||
| Other fatty acids | 0.51 | 0.55 | |||
| Σ SFA | 28.40 | 14.98 | |||
| Σ UFA | 71.09 | 84.47 | |||
| Σ MUFA | 45.49 | 15.25 | |||
| Σ PUFA | 25.60 | 69.23 | |||
| SFA/UFA | 0.399 | 0.177 | |||
| PUFA/MUFA | 0.563 | 4.541 | |||
| Linoleic/α-Linolenic | 3.05 | 71.64 |
| Flavonoids (mg/g) | Sea Buckthorn Meal | Grapeseed Meal | Phenolic Acids (mg/g) | Sea Buckthorn Meal | Grapeseed Meal |
|---|---|---|---|---|---|
| Catechin | 0.119 | 0.378 | Vanillic acid | 0.008 | 0.062 |
| Epicatechin | 0.397 | 0.271 | Caffeic acid | 0.003 | 0.001 |
| Rutin | 0.021 | 0.009 | P-Coumaric acid | 0.041 | 0.005 |
| Quercetin | 0.019 | 0.005 | Ferulic acid | 0.500 | 0.063 |
| Luteolin | 0.077 | 0.008 |
| Macroelements (%) | Sea Buckthorn Meal | Grapeseed Meal | Microelements (ppm) | Sea Buckthorn Meal | Grapeseed Meal |
|---|---|---|---|---|---|
| Calcium (Ca) | 0.04 | 0.79 | Copper (Cu) | 7.26 | 15.46 |
| Phosphor (P) | 0.34 | 0.35 | Iron (Fe) | 625.77 | 89.65 |
| Natrium (Na) | 0.117 | 0127 | Manganese (Mn) | 22.34 | 18.27 |
| Kalium (K) | 1.69 | 0.89 | Zinc (Zn) | 21.90 | 18.66 |
| Magnesium (Mg) | 0.127 | 0.005 |
| Control | E1 | E2 | E3 | |||||
|---|---|---|---|---|---|---|---|---|
| Mean | SEM | Mean | SEM | Mean | SEM | Mean | SEM | |
| Total protein (g/dL) | 5.34 | 0.10 | 5.08 | 0.82 | 5.05 | 0.18 | 5.41 | 0.85 |
| Bilirubin (mg/dL) | 0.35 | 0.04 | 0.43 | 0.09 | 0.31 | 0.02 | 0.30 | 0.06 |
| ALAT (U/L) | 49.44 | 2.36 | 48.33 | 1.49 | 47.22 | 1.95 | 50.24 | 3.56 |
| ASAT (U/L) | 38.50 | 2.92 | 41.24 | 3.98 | 39.96 | 3.05 | 41.04 | 3.28 |
| ALP (U/L) | 247.58 a | 11.1 | 279.88 ac | 28.3 | 311.44 bc | 25.4 | 273.22 ac | 15.9 |
| GGT (U/L) | 26.3 a | 2.55 | 26.67 ac | 2.27 | 34.02 bc | 3.34 | 29.92 ac | 1.88 |
| Albumin (g/L) | 3.00 | 0.00 | 3.00 | 0.00 | 3.02 | 0.01 | 3.18 | 0.02 |
| Creatinine (mg/dL) | 0.92 | 0.33 | 0.96 | 0.03 | 0.94 | 0.03 | 0.87 | 0.05 |
| MAV | Control Group | E1 Group | E2 Group | E3 Group |
|---|---|---|---|---|
| Liver | 1 | 1 | 3.5 ± 0.55 *** | 2.5 ± 0.55 ***/^ |
| Kidney | 1 | 1 | 3.7 ± 0.52 *** | 2.3 ± 0.52 ***/^^ |
| GenBank Accession Number | Gene | PCR Product Length (bp) | Primer Name | Primer Sequence |
|---|---|---|---|---|
| XM021085497 | TATA-box binding protein | 124 | tbp-F | 5′-GATGGACGTTCGGTTTAGG-3′ |
| tbp-R | 5′-AGCAGCACAGTACGAGCAA-3′ | |||
| XM005659862 | ribosomal protein L4 | 122 | rpl4-F | 5′-CAAGAGTAACTACAACCTTC-3′ |
| rpl4-R | 5′-GAACTCTACGATGAATCTTC-3′ | |||
| NM213978 | beta-2-microglobulin | 172 | b2m-F | 5′-CCGCCCCAGATTGAAATTGA-3′ |
| b2m-R | 5′-GCTTATCGAGAGTCACGTGC-3′ | |||
| NM001159614 | cytochrome P450, family 1, subfamily A, polypeptide 2 | 173 | cyp1a2-F | 5′-CTCTTCCGACACACCTCCTT-3′ |
| cyp1a2-R | 5′-AATCTCTCTGGCCGGAACTC-3′ | |||
| NM214417 | cytochrome P450 2A19 | 174 | cyp2a19-F | 5′-CTCATGAAGATCAGCCAGCG-3′ |
| cyp2a19-R | 5′-GCCATAGCCTTTGAAGAGCC-3′ | |||
| XM005657509 | cytochrome P450, family 2, subfamily E, polypeptide 1 | 150 | cyp2e1-F | 5′-ACCTCATTCCCTCCAACCTG-3′ |
| cyp2e1-R | 5′-CTGGCTTAAACTTCTCCGGC-3′ | |||
| NM214423 | cytochrome P450 3A29 | 205 | cyp3a29-F | 5′-ATTGCTGTCTCCGACCTTCA-3′ |
| cyp3a29-R | 5′-TGGGTTGTTGAGGGAATCGA-3′ | |||
| XM021096706 | cytochrome P450 4A24 | 157 | cyp4a24-F | 5′-CTCTATCCGCCAGTACCAGG-3′ |
| cyp4a24-R | 5′-ATGGGTCAAACTCCTCTGGG-3′ | |||
| XM021073710 | ATP binding cassette subfamily C member 2 | 172 | mrp2-F | 5′-AGCAGTACACCGTTGGAGAA-3′ |
| mrp2-R | 5′-ATCACCCCAACACCTGCTAA-3′ | |||
| NM214389 | glutathione S-transferase alpha 1 | 186 | gsta1-F | 5′-GCCCATGGTTGAGATTGACG-3′ |
| gsta1-R | 5′-TTTTCATTGGGTGGGCACAG-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popescu, R.G.; Bulgaru, C.; Untea, A.; Vlassa, M.; Filip, M.; Hermenean, A.; Marin, D.; Țăranu, I.; Georgescu, S.E.; Dinischiotu, A. The Effectiveness of Dietary Byproduct Antioxidants on Induced CYP Genes Expression and Histological Alteration in Piglets Liver and Kidney Fed with Aflatoxin B1 and Ochratoxin A. Toxins 2021, 13, 148. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020148
Popescu RG, Bulgaru C, Untea A, Vlassa M, Filip M, Hermenean A, Marin D, Țăranu I, Georgescu SE, Dinischiotu A. The Effectiveness of Dietary Byproduct Antioxidants on Induced CYP Genes Expression and Histological Alteration in Piglets Liver and Kidney Fed with Aflatoxin B1 and Ochratoxin A. Toxins. 2021; 13(2):148. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020148
Chicago/Turabian StylePopescu, Roua Gabriela, Cristina Bulgaru, Arabela Untea, Mihaela Vlassa, Miuta Filip, Anca Hermenean, Daniela Marin, Ionelia Țăranu, Sergiu Emil Georgescu, and Anca Dinischiotu. 2021. "The Effectiveness of Dietary Byproduct Antioxidants on Induced CYP Genes Expression and Histological Alteration in Piglets Liver and Kidney Fed with Aflatoxin B1 and Ochratoxin A" Toxins 13, no. 2: 148. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020148







