Attempt to Develop Rat Disseminated Intravascular Coagulation Model Using Yamakagashi (Rhabdophis tigrinus) Venom Injection
Abstract
:1. Introduction
2. Results
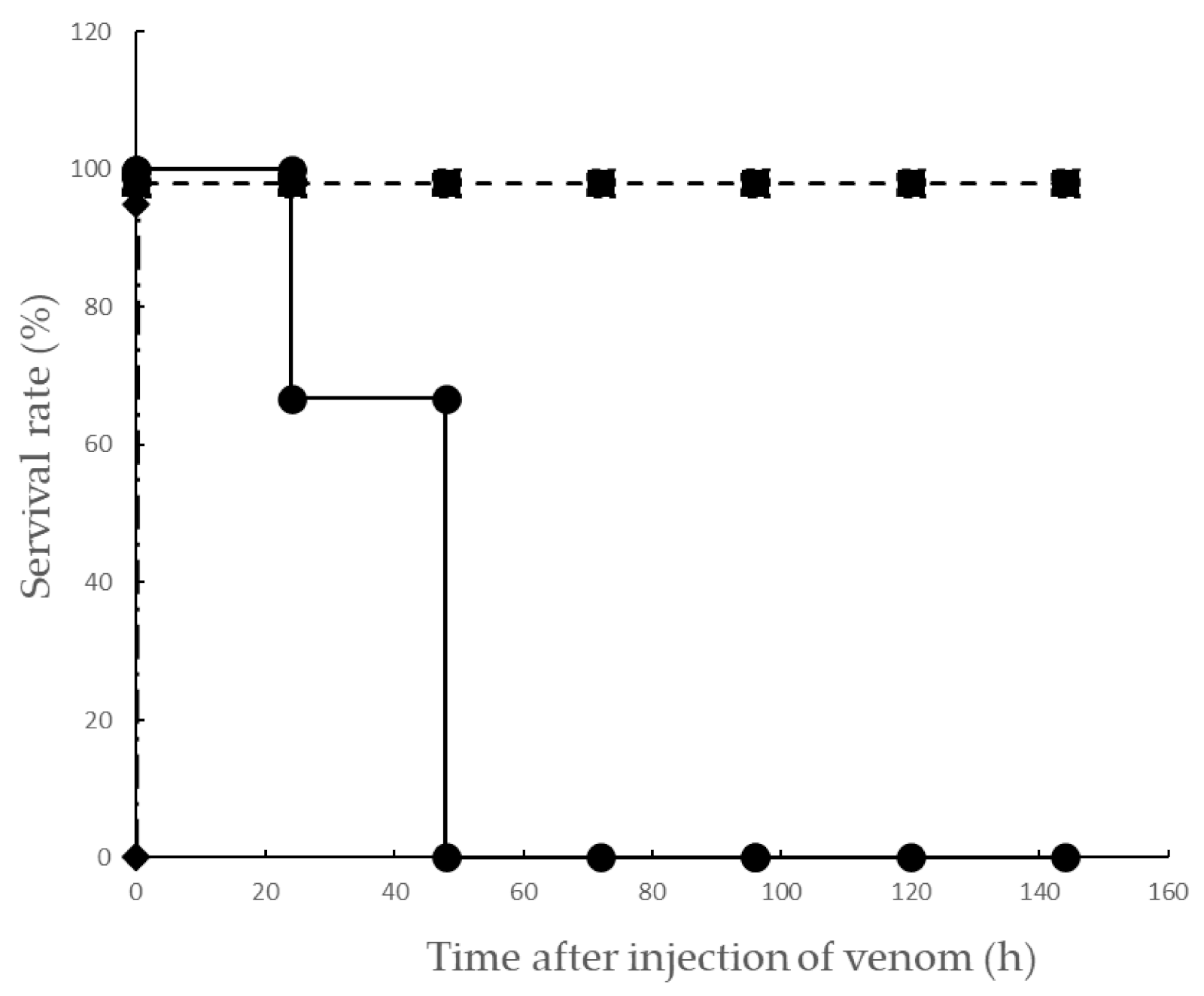
2.1. Effect on Lethality Induced by Yamakagashi Venom in Rats
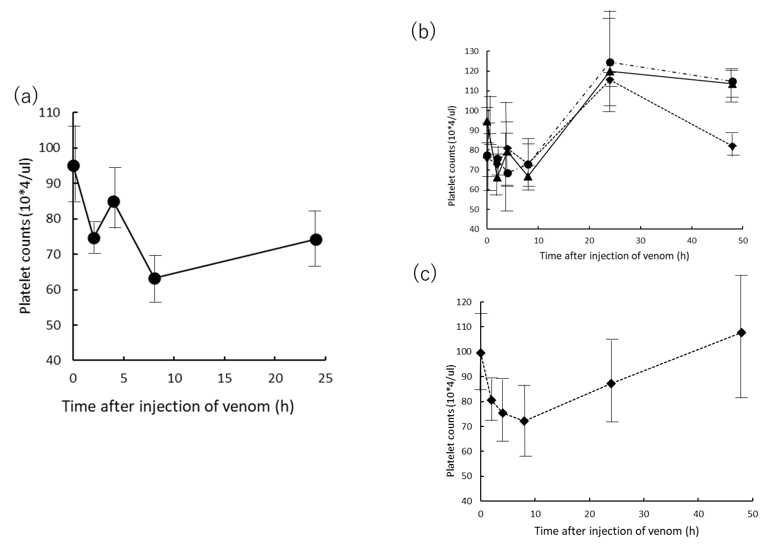
2.2. Effects of Platelet Counts of Rat Plasma Injected with Yamakagashi Venom
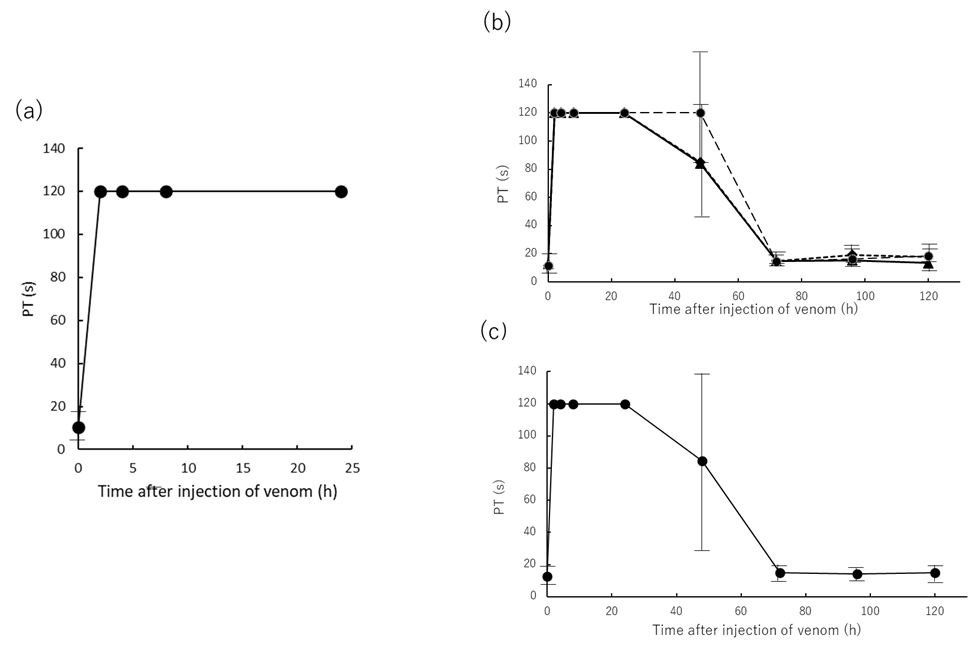
2.3. Effects of Prothrombin Time of Rat Plasma Injected with Yamakagashi Venom
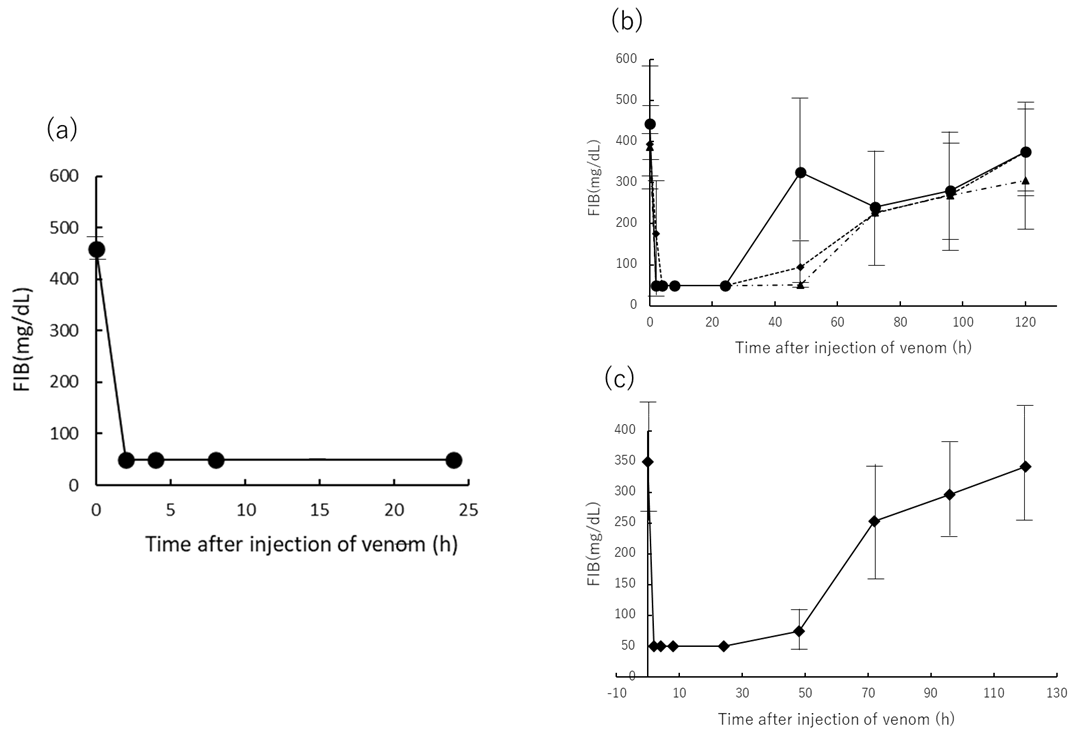
2.4. Effects of FIB Concertation of Rat Plasma Injected Yamakagashi Venom
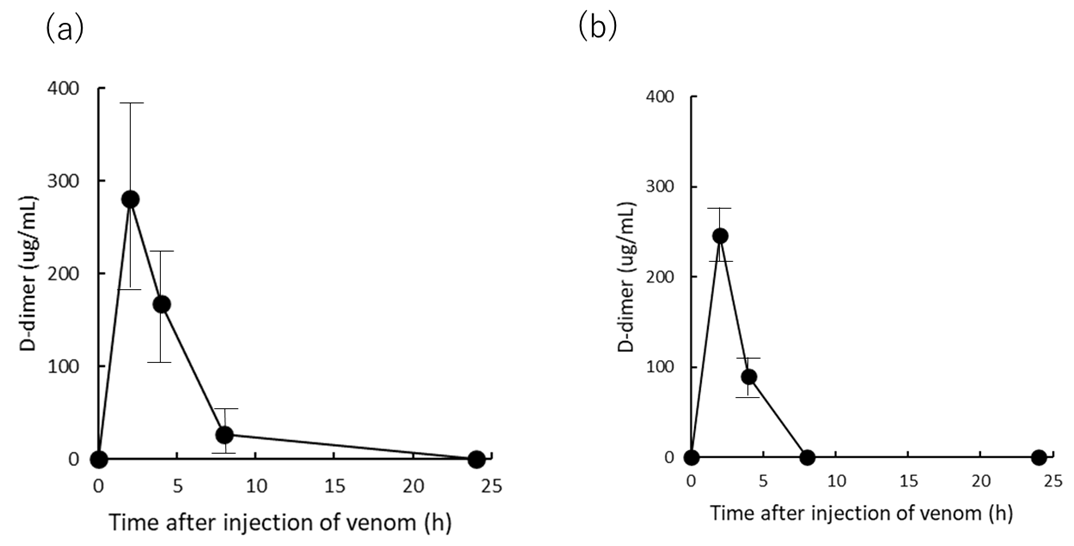
2.5. Effects of D-Dimer Concertation of Rat Plasma Injected with Yamakagashi Venom
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Animal Preparation
5.2. Snake Venoms
5.3. Anti-Snake Venom Equine Antibody
5.4. Experimental Protocols
5.5. Principle of Test in the Measurement of Platelet Counts
5.6. Principle of Test in the Measurement of PT
5.7. Principle of Test in the Measurement of FIB
5.8. Principle of Test in the Measurement of D-Dimer
5.9. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levi, M. Disseminated Intravascular Coagulation in Cancer: An Update. Semin. Thromb. Hemost. 2019, 45, 342–347. [Google Scholar] [CrossRef]
- Refaai, M.A.; Riley, P.; Mardovina, T.; Bell, P.D. The Clinical Significance of Fibrin Monomers. Thromb. Haemost. 2018, 118, 1856–1866. [Google Scholar] [CrossRef] [Green Version]
- Levi, M. Pathogenesis and diagnosis of disseminated intravascular coagulation. Int. J. Lab. Hematol. 2018, 40, 15–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jourdain, M.; Tournoys, A.; Leroy, X.; Mangalaboyi, J.; Fourrier, F.; Goudemand, J.; Gosselin, B.; Vallet, B.; Chopin, C. Effect of N omega-nitro-L-arginine methyl ester on the endovenom-induced disseminated intravascular coagulation in porcine sep-tic shock. Crit. Care. Med. 1997, 25, 452–459. [Google Scholar] [CrossRef]
- Iba, T.; Levi, M.; Levy, J.H. Sepsis-Induced Coagulopathy and Disseminated Intravascular Coagulation. Semin. Thromb. Hemost. 2020, 46, 089–095. [Google Scholar] [CrossRef]
- Isbister, G.K. Snakebite Doesn’t Cause Disseminated Intravascular Coagulation: Coagulopathy and Thrombotic Microangiopathy in Snake Envenoming. Semin. Thromb. Hemost. 2010, 36, 444–451. [Google Scholar] [CrossRef]
- Tibbaas, J.; Monagle, P. Tiger snake envenomation and disseminated intravascular coagulation (DIC). Pathology 2002, 34, 481–482. [Google Scholar]
- Glatstein, M.; Lerman, L.; Friedman, S.; Carbell, G.; Munchak, I.; Valla, U.; Scolnik, D.; Hoyte, C. Severe disseminated intra-vascular coagulation in a child envenomated by Echis coloratus and successful treatment with monovalent equine immuno-globulin G antivenom. Toxicon 2019, 167, 82–86. [Google Scholar] [CrossRef]
- Yau, C.S.; Randy, C.F.C.; Lixin, X.; Jack, H.W.; Tzi, B.N.; Wai, Y.C. Snake venom venoms: Toxicity and medical applications. Appl. Microbiol. Biotechnol. 2016, 100, 6165–6181. [Google Scholar]
- Wedasingha, S.; Isbister, G.; Silva, A. Bedside Coagulation Tests in Diagnosing Venom-Induced Consumption Coagulopathy in Snakebite. Toxins 2020, 12, 583. [Google Scholar] [CrossRef]
- Berling, I.; Isbister, G.K. Hematologic Effects and Complications of Snake Envenoming. Transfus. Med. Rev. 2015, 29, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Mori, A. Home Range and Movements of Rhabdophis tigrinus in a Mountain Habitat of Kyoto, Japan. Cur. Herpetol. 2014, 33, 8–20. [Google Scholar] [CrossRef]
- Ichiki, T.; Kohda, F.; Hiramatsu, T.; Saiki, R.; Sakai, A.; Furune, M. Early pathology in venom-induced consumption coag-ulopathy by Rhabdophis tigrinus (Yamakagashi) envenomation. Clin. Toxicol. 2019, 57, 668–671. [Google Scholar] [CrossRef]
- Komori, K.; Konishi, M.; Maruta, Y.; Toriba, M.; Sakai, A.; Matsuda, A.; Hori, T.; Nakatani, M.; Minamino, N.; Akizawa, A. Characterization of a novel metalloprotease in duvernoy’s gland of Rhabdophis tigrinus. J. Toxicol. Sci. 2006, 31, 157–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakai, A.; Honma, M.; Sawai, Y. Study on the pathogenesis of envenomation by the Japanese colubrid snake, Yamakagashi, Rhabdophis Trgrinus Tigrinus 1. Study on the toxicity of the venom. Snake 1983, 15, 7–13. [Google Scholar]
- Mittleman, M.B.; Goris, R.C. Envenomation from the bite of the Japanese colubrid snake Rhabdophis tigrinus (Boie). Herpetologica 1974, 30, 113–119. [Google Scholar]
- Mori, K.; Hisa, S.; Suzuki, S.; Sugai, K.; Sakai, H.; Kikuchi, T.; Hiwatashi, N.; Shishido, H.; Goto, Y.; Takahashi, T. A case of severe defibrination syndrome due to snake (Rhabdophis tigrinus) bite. Rinsho Ketsueki 1983, 24, 256–262. [Google Scholar]
- Sakai, A.; Hatsuse, M.; Sawai, Y. Study on the pathogenesis of envenomation by the Japanese colubrid snake, Yamakagashi, Rhabdophis tigrinus tigrinus. Snake 1990, 22, 11–19. [Google Scholar]
- Akimoto, R.; Watanabe, Y.; Sakai, A.; Kawamura, Y.; Sawai, Y. A case of defibrinnation syndrome due to Japanese colubrid snake, Yamakagashi (Rhabdophis t. tigrinus), bite treated with antivenom. Snake 1991, 23, 36–39. [Google Scholar]
- Boral, B.M.; Williams, D.J.; Boral, L.I. Disseminated intravascular coagulation. Am. J. Clin. Pathol. 2016, 146, 670–680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komori, Y.; Hifumi, T.; Yamamoto, A.; Sakai, A.; Ato, M.; Sawabe, K.; Nikai, T. Comparative Study of Biological Activities of Venom from Colubrid Snakes Rhabdophis tigrinus (Yamakagashi) and Rhabdophis lateralis. Toxins 2017, 9, 373. [Google Scholar] [CrossRef] [Green Version]
- Chapin, J.C.; Hajjar, K.A. Fibrinolysis and the control of blood coagulation. Blood Rev. 2015, 29, 17–24. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Qu, J.; Xiong, M.; Qiao, Y.; Wang, D.; Liu, F.; Li, D.; Hu, M.; Zhang, J.; Wang, F.; et al. Role of vascular endotheli-al cells in disseminated intravascular coagulation induced by seawater immersion in a rat trauma model. Biomed. Res. Int. 2017, 2017, 5147532. [Google Scholar] [CrossRef] [Green Version]
- Schaeffer, R.C.; Briston, C.; Chilton, S.M.; Carlson, R.W. Disseminated intravascular coagulation following Echis carinatus venom in dogs: Effects of a synthetic thrombin inhibitor. J. Lab. Clin. Med. 1986, 107, 488–497. [Google Scholar]
- Berthelsen, L.O.; Krstensen, A.T.; Tranholm, M. Animal models of DIC and their relevance to human DIC: A systemic review. Thromb. Res. 2011, 128, 103–116. [Google Scholar] [CrossRef]
- Warrell, A.D. Snake bite. Lancet 2010, 375, 77–88. [Google Scholar] [CrossRef]
- Hifumi, T.; Sakai, A.; Kondo, Y.; Yamamoto, A.; Morine, N.; Ato, M.; Shibayama, K.; Umezawa, K.; Kiriu, N.; Kato, H.; et al. Venomous snake bites: Clinical diagnosis and treatment. J. Intensiv. Care 2015, 3, 16. [Google Scholar] [CrossRef] [Green Version]
- WHO. Guidelines for the Management of Snakebites, 2nd ed.; WHO: Geneva, Switzerland, 2016; Available online: https://www.who.int/publications/i/item/9789290225300 (accessed on 17 February 2021).
- Snake Antivenom Immunoglobulins. Available online: https://www.who.int/bloodproducts/animal_sera/en/ (accessed on 17 February 2021).
- Silva, H.A.; Ryan, N.M.; Silva, H.J. Adverse reactions to snake antivenom, and their prevention and treatment. Br. J. Clin. Pharmacol. 2015, 81, 446–452. [Google Scholar] [CrossRef] [Green Version]
- Wada, H.; Matsumoto, T.; Yamashita, Y. Diagnosis and treatment of disseminated intravascular coagulation (DIC) accord-ing to four DIC guidelines. J. Intensive Care 2014, 2, 15. [Google Scholar] [CrossRef] [Green Version]
- Bulfone, T.C.; Samuel, S.P.; Bickler, P.E.; Lewin, M.R. Developing Small Molecule Therapeutics for the Initial and Adjunctive Treatment of Snakebite. J. Trop. Med. 2018, 2018, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Lewin, M.; Samuel, S.; Merkel, J.; Bickler, P. Varespladib (LY315920) Appears to Be a Potent, Broad-Spectrum, Inhibitor of Snake Venom Phospholipase A2 and a Possible Pre-Referral Treatment for Envenomation. Toxins 2016, 8, 248. [Google Scholar] [CrossRef] [Green Version]
- Preciado, L.M.; Comer, J.; Núñez, V.; Rey-Súarez, P.; Pereañez, J.A. Inhibition of a Snake Venom Metalloproteinase by the Flavonoid Myricetin. Molecules 2018, 23, 2662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albulescu, L.O.; Xie, C.; Ainsworth, S.; Alsolaiss, J.; Crittenden, E.; Dawson, C.A.; Softley, R.; Bartlett, K.E.; Harrison, R.A.; Kool, J.; et al. A therapeutic combination of two small molecule toxin inhibitors provides broad preclinical efficacy against viper snakebite. Nat. Com. 2020, 11, 6094. [Google Scholar]
- Morokuma, K.; Kobori, N.; Fukuda, T.; Uchida, T.; Sakai, A.; Toriba, I.; Ohkuma, K.; Nakai, K.; Kurata, T.; Tamahashi, M. Experimental manufacture of equine antivenom against Yamakagashi (Rhabdophis tigrinus). Jpn. J. Infect. Dis. 2011, 64, 397–402. [Google Scholar]
- Kario, K.; Matsuo, T.; Kabayashi, H.; Matsuo, M.; Yamamoto, K.; Sakurai, G.; Baba, M. Rapid quantitative evaluation of plasma D-dimer levels in thrombotic states using an automated latex photometric immunoassay. Thromb. Res. 1992, 66, 179–189. [Google Scholar] [CrossRef]
| Group and Time (h) | Hematuria | Plasma Hemolysis |
|---|---|---|
| 300 μg IM 1 | ||
| 0 | −, −, − | −, −, − |
| 2 | ++, +, + | +, +, + |
| 4 | ++, +, + | +, +, + |
| 8 | ++, +, + | +, +, + |
| 24 | +, + | +, + |
| 250 μg IM 2 | ||
| 0 | −, −, − | −, −, − |
| 2 | +, +, + | +, −, − |
| 4 | +, +, ++ | +, −, + |
| 8 | +, −, + | −, −, − |
| 24 | −, −, − | −, −, − |
| 48 | −, −, − | −, −, − |
| 300 μg IM+Antitoxin 3 | ||
| 0 | ±, −, − | −, −, − |
| 2 | +, ++, + | +, −, − |
| 4 | +, +, + | +, −, + |
| 8 | −, −, + | −, +, − |
| 24 | −, −, − | −, −, − |
| 48 | −, −, − | −, −, − |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamamoto, A.; Ito, T.; Hifumi, T. Attempt to Develop Rat Disseminated Intravascular Coagulation Model Using Yamakagashi (Rhabdophis tigrinus) Venom Injection. Toxins 2021, 13, 160. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020160
Yamamoto A, Ito T, Hifumi T. Attempt to Develop Rat Disseminated Intravascular Coagulation Model Using Yamakagashi (Rhabdophis tigrinus) Venom Injection. Toxins. 2021; 13(2):160. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020160
Chicago/Turabian StyleYamamoto, Akihiko, Takashi Ito, and Toru Hifumi. 2021. "Attempt to Develop Rat Disseminated Intravascular Coagulation Model Using Yamakagashi (Rhabdophis tigrinus) Venom Injection" Toxins 13, no. 2: 160. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13020160





