Zearalenone Induces Endothelial Cell Apoptosis through Activation of a Cytosolic Ca2+/ERK1/2/p53/Caspase 3 Signaling Pathway
Abstract
:1. Introduction
2. Results
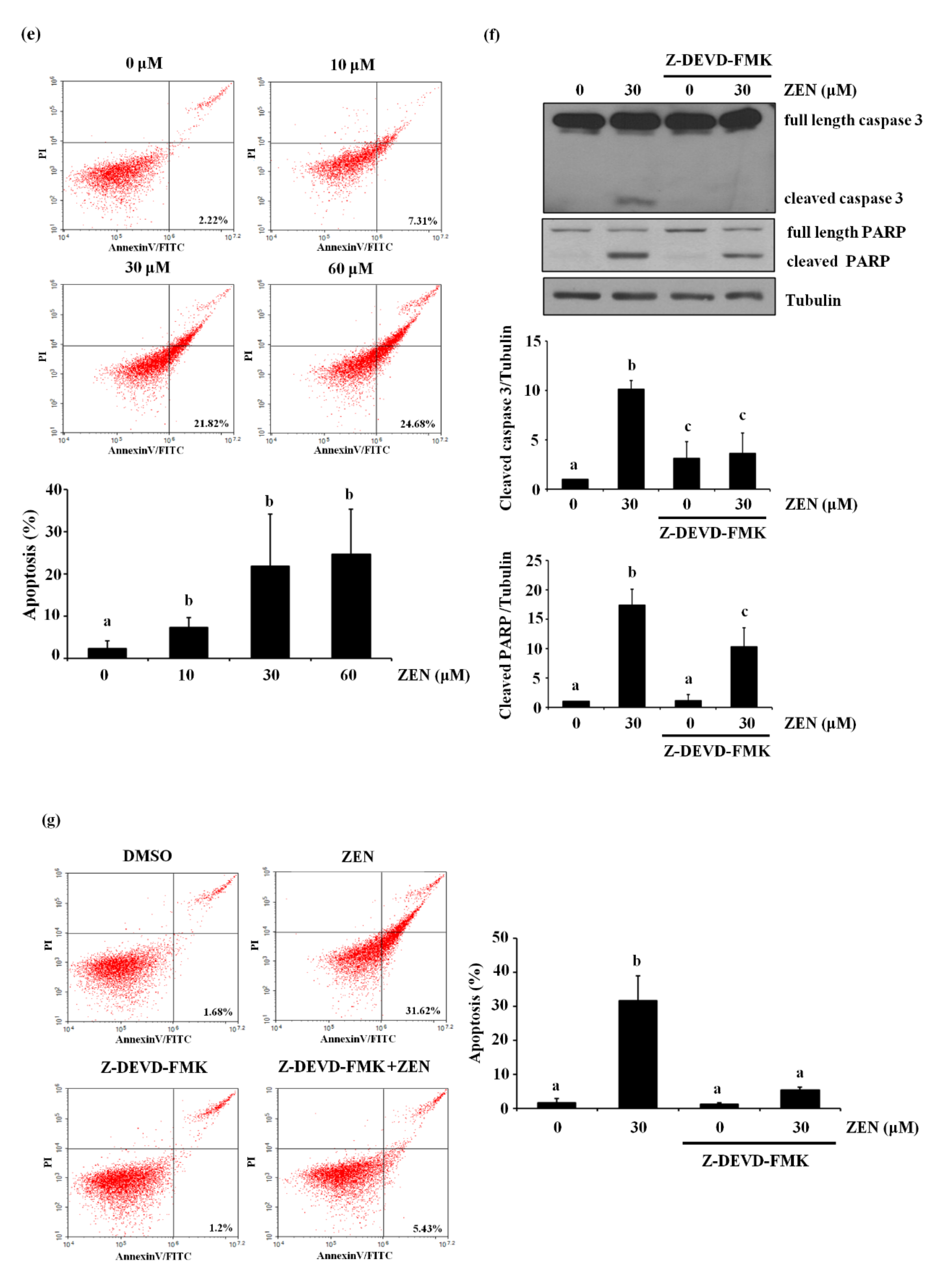
2.1. Zearalenone Increases Apoptotic Death of BAECs
2.2. ZEN-Induced Apoptosis Is Mediated through Phosphorylation of ERK1/2
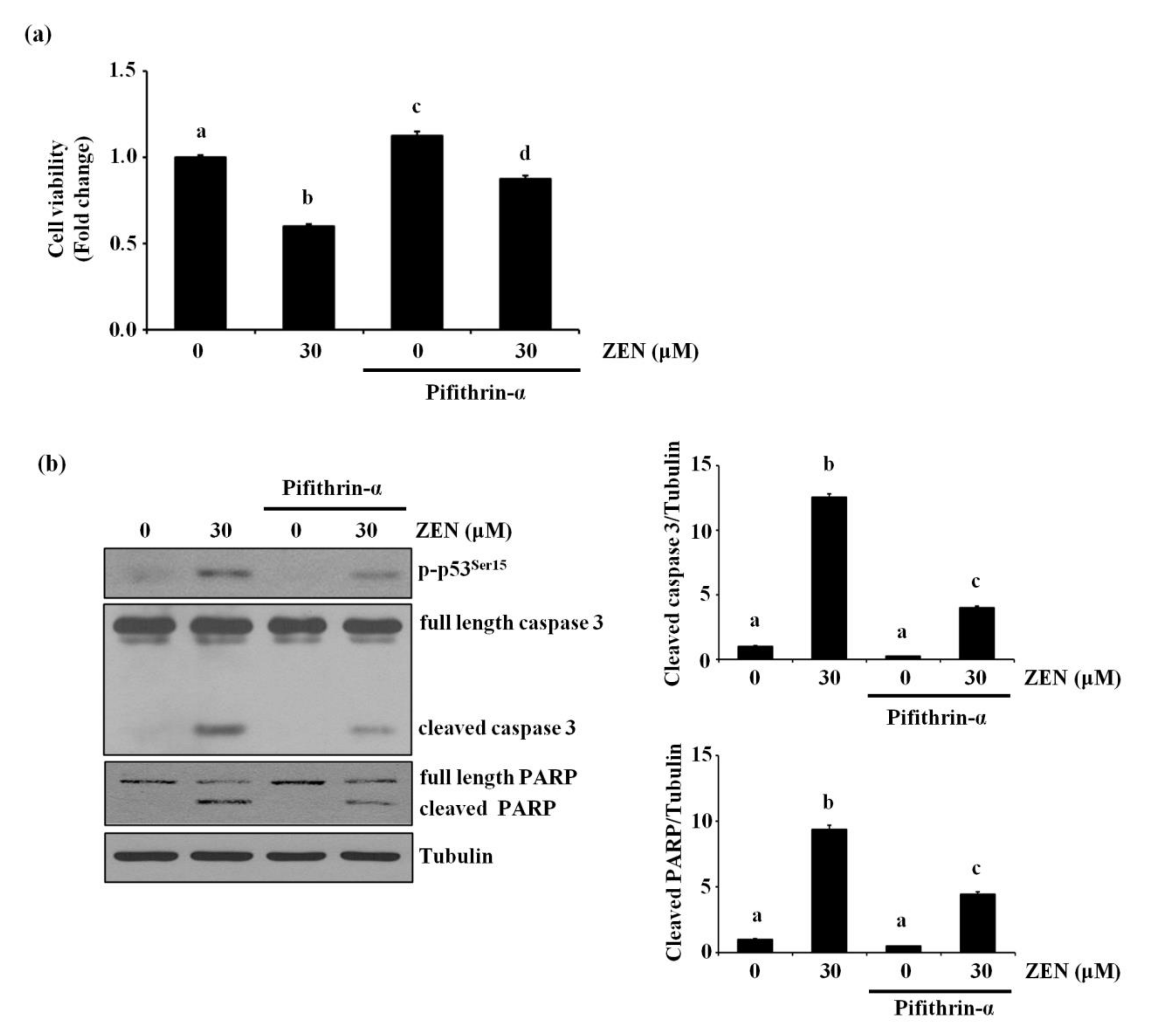
2.3. p53 Is Involved in ZEN-Induced Apoptosis of BAECs
2.4. ERK1/2 Is an Upstream Mediator of p53-Mediating ZEN-Induced EC Apoptosis
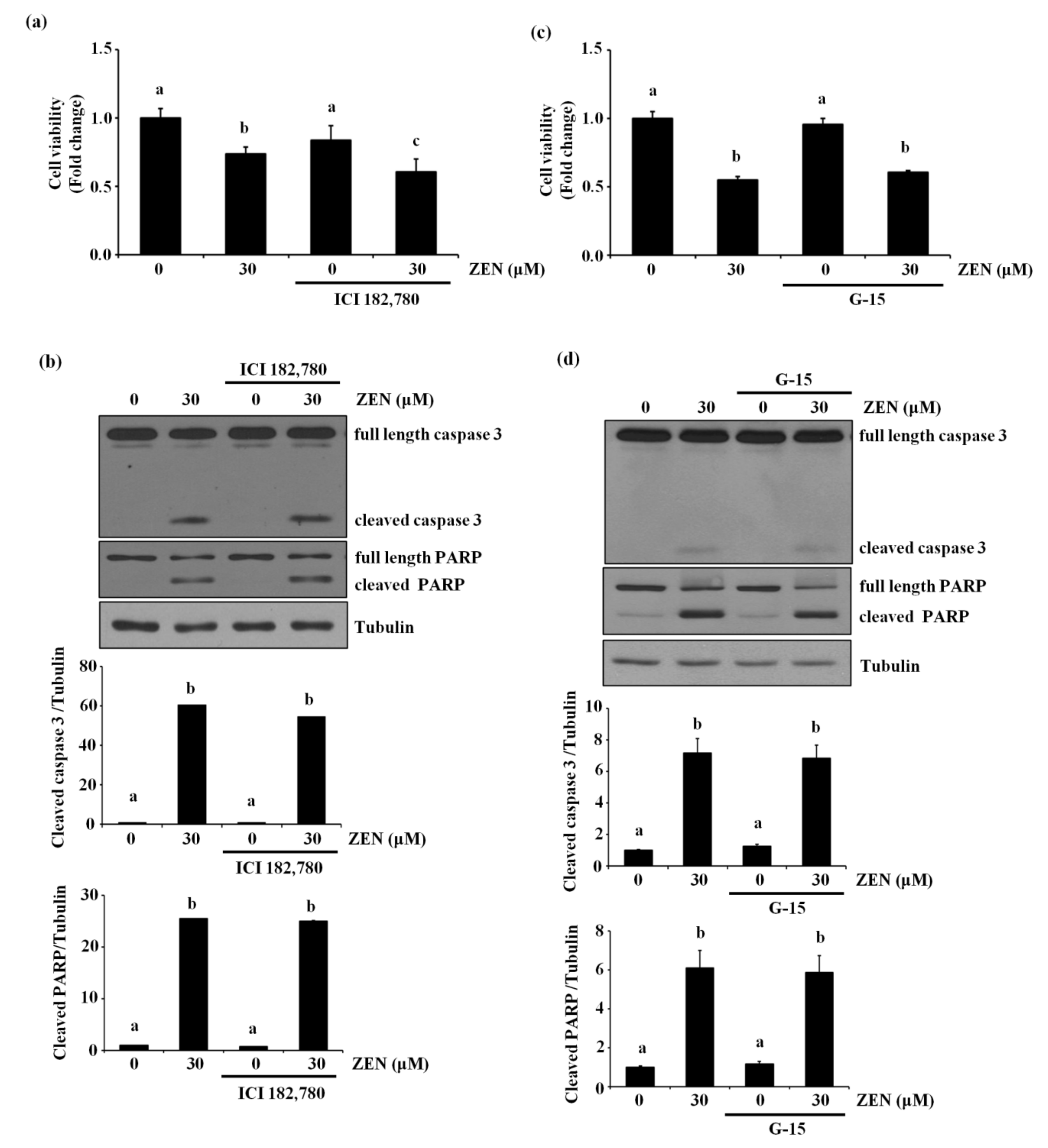
2.5. The Estrogen Receptor Is Not Involved in ZEN-Stimulated Apoptosis
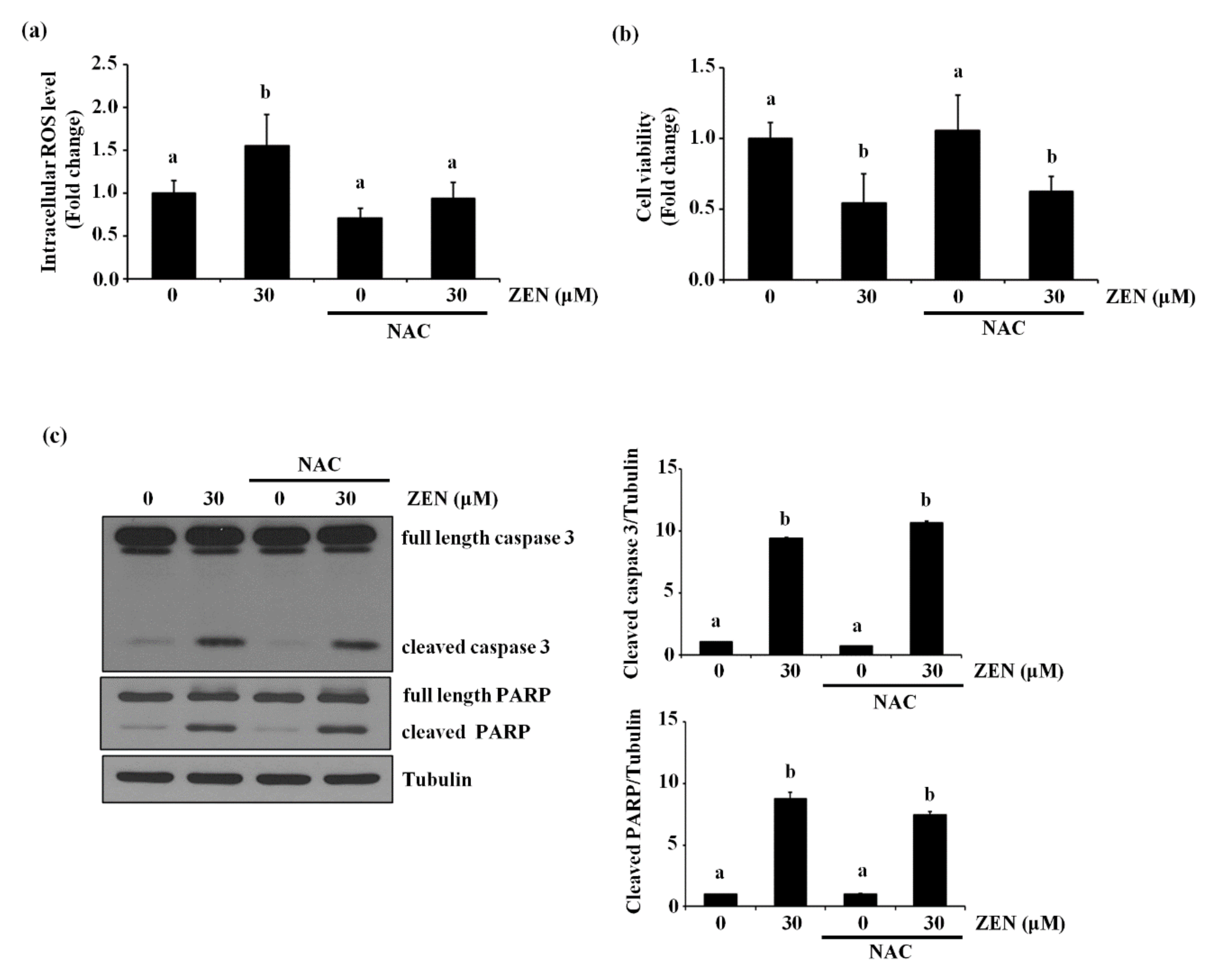
2.6. ZEN Induces Reactive Oxygen Species (ROS), But ROS Are Not Involved in ZEN-Induced EC Apoptosis
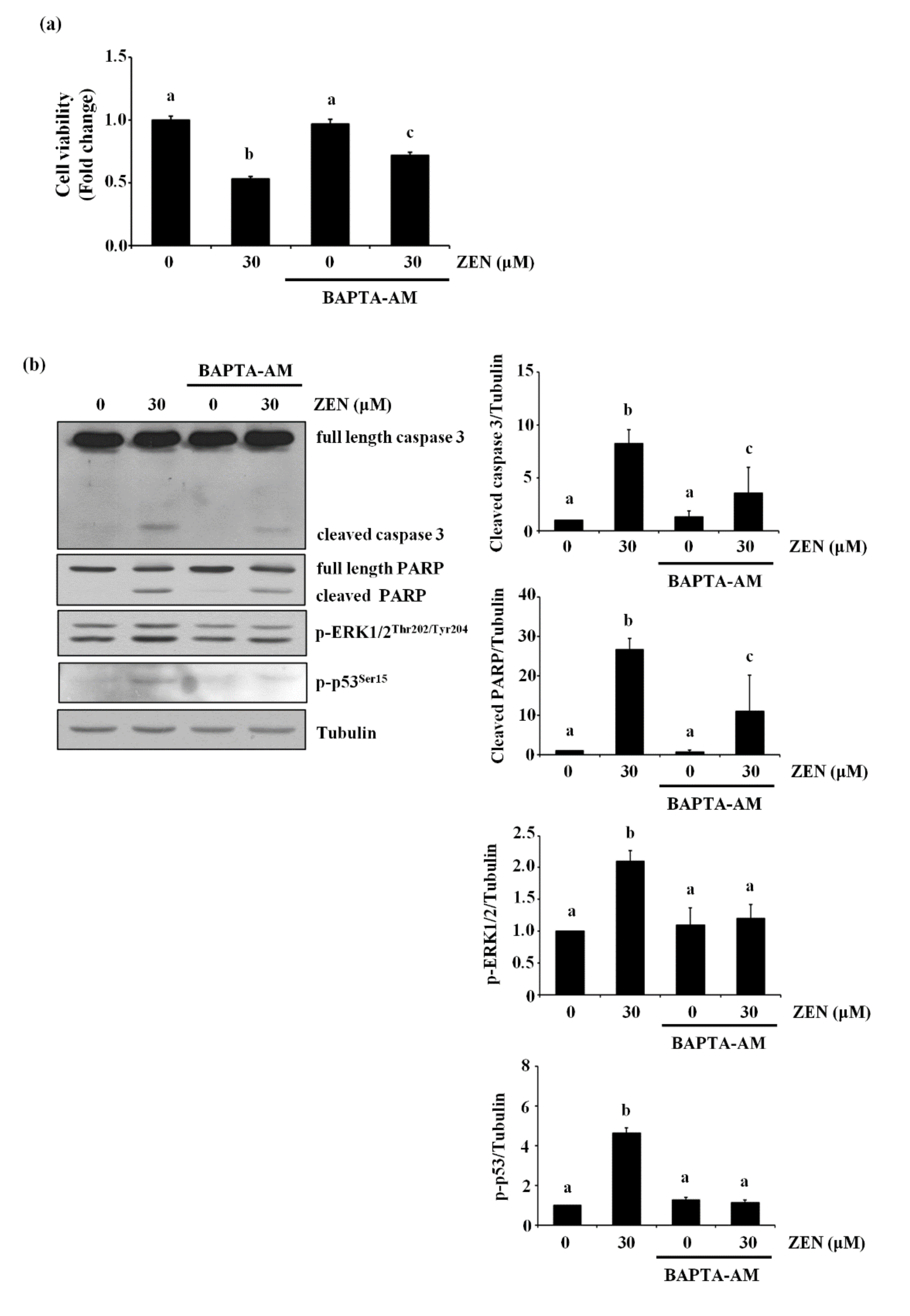
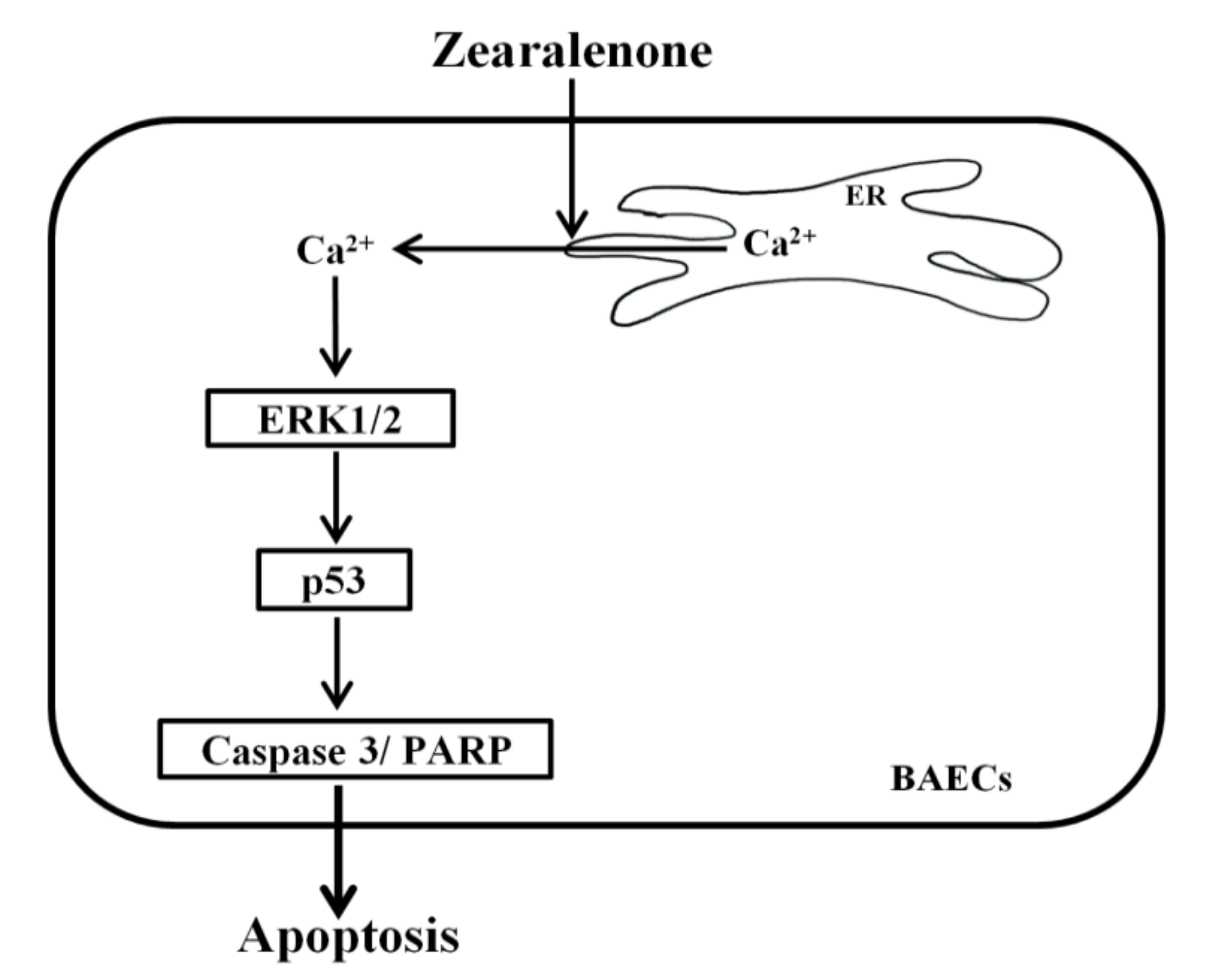
2.7. ZEN Mediates Apoptosis through a Cytosolic Ca2+-Dependent Pathway
2.8. The Endoplasmic Reticulum (ER) Is the Organelle Responsible for Cytosolic Ca2+ Release by ZEN
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Materials
5.2. Cell Culture and Drug Treatments
5.3. Cell Viability Assay
5.4. Annexin V-Fluorescein Isothiocynate (FITC)/Propidium Iodide (PI) Double Staining
5.5. Detection of Intracellular ROS Levels
5.6. Small Interfering RNA (siRNA) Transfection
5.7. Western Blot Analyses
5.8. Reverse Transcription-Polymerase Chain Reaction (PCR)
5.9. Measurement of Cytosolic Ca2+ Levels
5.10. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hossain, Z.; Mari, N.; Goto, T. The relationship between ergosterol and mycotoxin contamination in maize from various countries. Mycotoxin Res. 2015, 31, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Belhassen, H.; Jimenez-Diaz, I.; Ghali, R.; Ghorbel, H.; Molina-Molina, J.M.; Olea, N.; Hedili, A. Validation of a UHPLC-MS/MS method for quantification of zearalenone, alpha-zearalenol, beta-zearalenol, alpha-zearalanol, be-ta-zearalanol and zearalanone in human urine. J. Chromatogr. B 2014, 962, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Rogowska, A.; Pomastowski, P.; Sagandykova, G.; Buszewski, B. Zearalenone and its metabolites: Effect on human health, metabolism and neutralisation methods. Toxicon 2019, 162, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Feng, N.; Wang, Y.; Noll, L.; Xu, S.; Liu, X.; Lu, N.; Zou, H.; Gu, J.; Yuan, Y.; et al. Effects of zearalenone and its derivatives on the synthesis and secretion of mammalian sex steroid hormones: A review. Food Chem. Toxicol. 2019, 126, 262–276. [Google Scholar] [CrossRef]
- Adibnia, E.; Razi, M.; Malekinejad, H. Zearalenone and 17 beta-estradiol induced damages in male rats reproduction potential; evidence for ERalpha and ERbeta receptors expression and steroidogenesis. Toxicon 2016, 120, 133–146. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Wei, C.; Li, Y.; Liu, Y.; Wang, Y.; Pan, J.; Liu, J.; Wu, Y.; Cui, S. Zearalenone and alpha-zearalenol inhibit the synthesis and secretion of pig follicle stimulating hormone via the non-classical estrogen membrane receptor GPR30. Mol. Cell. Endocrinol. 2018, 461, 43–54. [Google Scholar] [CrossRef]
- Zatecka, E.; Ded, L.; Elzeinova, F.; Kubátová, A.; Dorosh, A.; Margaryan, H.; Dostalova, P.; Korenkova, V.; Hošková, K.; Pěknicová, J. Effect of zearalenone on reproductive parameters and expression of selected testicular genes in mice. Reprod. Toxicol. 2014, 45, 20–30. [Google Scholar] [CrossRef]
- Gao, F.; Jiang, L.-P.; Chen, M.; Geng, C.-Y.; Yang, G.; Ji, F.; Zhong, L.-F.; Liu, X.-F. Genotoxic effects induced by zearalenone in a human embryonic kidney cell line. Mutat. Res. Toxicol. Environ. Mutagen. 2013, 755, 6–10. [Google Scholar] [CrossRef]
- Lu, J.; Yu, J.Y.; Lim, S.S.; Son, Y.O.; Kim, D.H.; Lee, S.A.; Shi, X.; Lee, J.C. Cellular mechanisms of the cytotoxic effects of the zearalenone metabolites alpha-zearalenol and beta-zearalenol on RAW264 7 macrophages. Toxicol. In Vitro 2013, 27, 1007–1017. [Google Scholar] [CrossRef]
- Zheng, W.-L.; Wang, B.-J.; Wang, L.; Shan, Y.-P.; Zou, H.; Song, R.-L.; Wang, T.; Gu, J.-H.; Yuan, Y.; Liu, X.-Z.; et al. ROS-Mediated Cell Cycle Arrest and Apoptosis Induced by Zearalenone in Mouse Sertoli Cells via ER Stress and the ATP/AMPK Pathway. Toxins 2018, 10, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Venkataramana, M.; Nayaka, S.C.; Anand, T.; Rajesh, R.; Aiyaz, M.; Divakara, S.; Murali, H.; Prakash, H.; Rao, P.L. Zearalenone induced toxicity in SHSY-5Y cells: The role of oxidative stress evidenced by N-acetyl cysteine. Food Chem. Toxicol. 2014, 65, 335–342. [Google Scholar] [CrossRef]
- Lin, P.; Chen, F.; Sun, J.; Zhou, J.; Wang, X.; Wang, N.; Li, X.; Zhang, Z.; Wang, A.; Jin, Y. Mycotoxin zearalenone induces apoptosis in mouse Leydig cells via an endoplasmic reticulum stress-dependent signalling pathway. Reprod. Toxicol. 2015, 52, 71–77. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, W.; Bian, X.; Yuan, Y.; Gu, J.; Liu, X.; Liu, Z.; Bian, J. Zearalenone induces apoptosis and cytoprotective autophagy in primary Leydig cells. Toxicol. Lett. 2014, 226, 182–191. [Google Scholar] [CrossRef]
- Yu, J.-Y.; Zheng, Z.-H.; Son, Y.-O.; Shi, X.; Jang, Y.-O.; Lee, J.-C. Mycotoxin zearalenone induces AIF- and ROS-mediated cell death through p53- and MAPK-dependent signaling pathways in RAW264.7 macrophages. Toxicol. Vitr. 2011, 25, 1654–1663. [Google Scholar] [CrossRef]
- Anderson, C.; Majeste, A.; Hanus, J.; Wang, S. E-Cigarette Aerosol Exposure Induces Reactive Oxygen Species, DNA Damage, and Cell Death in Vascular Endothelial Cells. Toxicol. Sci. 2016, 154, 332–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ouyang, J.; Li, R.; Shi, H.; Zhong, J. Curcumin Protects Human Umbilical Vein Endothelial Cells against H2O2-Induced Cell Injury. Pain Res. Manag. 2019, 2019, 3173149. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, S.; Alam Khan, S.; Kindelin, A.; Mohseni, T.; Bhatia, K.; Hoda, N.; Ducruet, A.F. Acetyl-11-keto-β-boswellic acid (AKBA) Attenuates Oxidative Stress, Inflammation, Complement Activation and Cell Death in Brain Endothelial Cells Following OGD/Reperfusion. Neuro Mol. Med. 2019, 21, 505–516. [Google Scholar] [CrossRef]
- Gajecki, M. Zearalenone—Undesirable substances in feed. Pol. J. Veter-Sci. 2002, 5, 117–122. [Google Scholar]
- Zheng, W.; Wang, B.; Li, X.; Wang, T.; Zou, H.; Gu, J.; Yuan, Y.; Liu, X.; Bai, J.; Bian, J.; et al. Zearalenone Promotes Cell Proliferation or Causes Cell Death? Toxins 2018, 10, 184. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Zhao, X.; Ni, C.; Dai, Y.; Guo, Y. Zearalenone regulates endometrial stromal cell apoptosis and migration via the promotion of mitochondrial fission by activation of the JNK/Drp1 pathway. Mol. Med. Rep. 2018, 17, 7797–7806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pistol, G.C.; Braicu, C.; Motiu, M.; Gras, M.A.; Marin, D.E.; Stancu, M.; Calin, L.; Israel-Roming, F.; Berindan-Neagoe, I.; Taranu, I. Zearalenone mycotoxin affects immune mediators, MAPK signalling molecules, nuclear receptors and ge-nome-wide gene expression in pig spleen. PLoS ONE 2015, 10, e0127503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayed-Boussema, I.; Bouaziz, C.; Rjiba, K.; Valenti, K.; Laporte, F.; Bacha, H.; Hassen, W. The mycotoxin Zearalenone induces apoptosis in human hepatocytes (HepG2) via p53-dependent mitochondrial signaling pathway. Toxicol. Vitr. 2008, 22, 1671–1680. [Google Scholar] [CrossRef]
- Jilani, K.; Lang, F. Ca2+-dependent suicidal erythrocyte death following zearalenone exposure. Arch. Toxicol. 2013, 87, 1821–1828. [Google Scholar] [CrossRef]
- Wang, Y.C.; Deng, J.L.; Xu, S.W.; Peng, X.; Zuo, Z.C.; Cui, H.M.; Ren, Z.H.; Wang, Y. Effects of zearalenone on calcium homeostasis of splenic lymphocytes of chickens in vitro. Poult. Sci. 2012, 91, 1956–1963. [Google Scholar] [CrossRef]
- Wang, M.; Kern, A.M.; Hülskötter, M.; Greninger, P.; Singh, A.; Pan, Y.; Chowdhury, D.; Krause, M.; Baumann, M.; Benes, C.H.; et al. EGFR-Mediated Chromatin Condensation Protects KRAS-Mutant Cancer Cells against Ionizing Radiation. Cancer Res. 2014, 74, 2825–2834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cagnol, S.; Chambard, J.C. ERK and cell death: Mechanisms of ERK-induced cell death—Apoptosis, autophagy and se-nescence. FEBS J. 2010, 277, 2–21. [Google Scholar] [CrossRef]
- Chen, F.; Li, Q.; Zhang, Z.; Lin, P.; Lei, L.; Wang, A.; Jin, Y. Endoplasmic Reticulum Stress Cooperates in Zeara-lenone-Induced Cell Death of RAW 264.7 Macrophages. Int. J. Mol. Sci. 2015, 16, 19780–19795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Celli, A.; Crumrine, D.; Meyer, J.M.; Mauro, T.M. Endoplasmic Reticulum Calcium Regulates Epidermal Barrier Response and Desmosomal Structure. J. Investig. Dermatol. 2016, 136, 1840–1847. [Google Scholar] [CrossRef] [Green Version]
- Sano, R.; Reed, J.C. ER stress-induced cell death mechanisms. Biochim. Biophys. Acta (BBA) Bioenerg. 2013, 1833, 3460–3470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romine, I.C.; Wiseman, R.L. PERK Signaling Regulates Extracellular Proteostasis of an Amyloidogenic Protein During Endoplasmic Reticulum Stress. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Zhu, L.; Yuan, H.; Guo, C.; Lu, Y.; Deng, S.; Yang, Y.; Wei, Q.; Wen, L.; He, Z. Zearalenone induces apoptosis and necrosis in porcine granulosa cells via a caspase-3- and caspase-9-dependent mitochondrial signaling pathway. J. Cell. Physiol. 2012, 227, 1814–1820. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, H.; Lu, Z.; Yu, X.; Lv, C.; Tian, Y.; Sui, D. Pseudo-Ginsenoside Rh2 induces A549 cells apoptosis via the Ras/Raf/ERK/p53 pathway. Exp. Ther. Med. 2018, 15, 4916–4924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, S.-H.; Wang, S.-M.; Syu, J.-P.; Chen, Y.; Wang, S.-D.; Peng, Y.-S.; Kuo, M.-F.; Kung, H.-N. Andrographolide Induces Apoptosis of C6 Glioma Cells via the ERK-p53-Caspase 7-PARP Pathway. BioMed Res. Int. 2014, 2014, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.Y.; Shin, S.J.; Kim, H.S. ERK1/2 activation mediated by the nutlin3induced mitochondrial translocation of p53. Int. J. Oncol. 2013, 42, 1027–1035. [Google Scholar] [CrossRef]
- Lee, S.Y.; Choi, H.C.; Choe, Y.J.; Shin, S.J.; Lee, S.H.; Kim, H.S. Nutlin-3 induces BCL2A1 expression by activating ELK1 through the mitochondrial p53-ROS-ERK1/2 pathway. Int. J. Oncol. 2014, 45, 675–682. [Google Scholar] [CrossRef]
- Holstein, D.M.; Berg, K.A.; Leeb-Lundberg, L.M.; Olson, M.S.; Saunders, C. Calcium-sensing receptor-mediated ERK1/2 activation requires Galphai2 coupling and dynamin-independent receptor internalization. J. Biol. Chem. 2004, 279, 10060–10069. [Google Scholar] [CrossRef] [Green Version]
- Veeranna; Kaji, T.; Boland, B.; Odrljin, T.; Mohan, P.; Basavarajappa, B.S.; Peterhoff, C.; Cataldo, A.; Rudnicki, A.; Amin, N.; et al. Calpain mediates calcium-induced activation of the erk1,2 MAPK pathway and cytoskeletal phosphorylation in neurons: Relevance to Alzheimer’s disease. Am. J. Pathol. 2004, 165, 795–805. [Google Scholar]
- Schmitt, J.M.; Wayman, G.A.; Nozaki, N.; Soderling, T.R. Calcium Activation of ERK Mediated by Calmodulin Kinase I. J. Biol. Chem. 2004, 279, 24064–24072. [Google Scholar] [CrossRef] [Green Version]
- Li, D.W.-C.; Liu, J.-P.; Mao, Y.-W.; Xiang, H.; Wang, J.; Ma, W.-Y.; Dong, Z.; Pike, H.M.; Brown, R.E.; Reed, J.C. Calcium-activated RAF/MEK/ERK Signaling Pathway Mediates p53-dependent Apoptosis and Is Abrogated by αB-Crystallin through Inhibition of RAS Activation. Mol. Biol. Cell 2005, 16, 4437–4453. [Google Scholar] [CrossRef] [Green Version]
- Shin, D.-H.; Leem, D.-G.; Shin, J.-S.; Kim, J.-I.; Kim, K.-T.; Choi, S.Y.; Lee, M.-H.; Choi, J.-H.; Lee, K.-T. Compound K induced apoptosis via endoplasmic reticulum Ca2+ release through ryanodine receptor in human lung cancer cells. J. Ginseng Res. 2018, 42, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Wang, B.; Cai, P.; Zheng, W.; Zou, H.; Gu, J.; Yuan, Y.; Liu, X.; Liu, Z.; Bian, J. ZEA-induced autophagy in TM4 cells was mediated by the release of Ca(2+) activates CaMKKbeta-AMPK signaling pathway in the endoplasmic reticulum. Toxicol. Lett. 2020, 323, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gajęcka, M. The effect of low-dose experimental zearalenone intoxication on the immunoexpression of estrogen receptors in the ovaries of pre-pubertal bitches. Pol. J. Veter-Sci. 2012, 15, 685–691. [Google Scholar] [CrossRef]
- Qin, G.; Wu, L.; Liu, H.; Pang, Y.; Zhao, C.; Wu, S.; Wang, X.; Chen, T. Artesunate induces apoptosis via a ROS-independent and Bax-mediated intrinsic pathway in HepG2 cells. Exp. Cell Res. 2015, 336, 308–317. [Google Scholar] [CrossRef]
- Lu, J.J.; Meng, L.H.; Cai, Y.J.; Chen, Q.; Tong, L.J.; Lin, L.P.; Ding, J. Dihydroartemisinin induces apoptosis in HL-60 leu-kemia cells dependent of iron and p38 mitogen-activated protein kinase activation but independent of reactive oxygen species. Cancer Biol. Ther. 2008, 7, 1017–1023. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.P.; Lee, J.Y.; Jeong, J.K.; Bae, S.W.; Lee, H.K.; Jo, I. Nongenomic Stimulation of Nitric Oxide Release by Estrogen Is Mediated by Estrogen Receptor α Localized in Caveolae. Biochem. Biophys. Res. Commun. 1999, 263, 257–262. [Google Scholar] [CrossRef]
- Kim, J.-Y.; Choi, J.-Y.; Lee, H.-J.; Byun, C.J.; Park, J.-H.; Park, J.H.; Cho, H.-S.; Cho, S.-J.; Jo, S.A.; Jo, I. The Green Tea Component (-)-Epigallocatechin-3-Gallate Sensitizes Primary Endothelial Cells to Arsenite-Induced Apoptosis by Decreasing c-Jun N-Terminal Kinase-Mediated Catalase Activity. PLoS ONE 2015, 10, e0138590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, M.H.; Liu, J.F.; Chiang, Y.C.; Hu, S.C.; Hsu, L.F.; Lin, Y.C.; Lin, Z.C.; Lee, H.C.; Chen, M.C.; Huang, C.L.; et al. Arto-carpin, an isoprenyl flavonoid, induces p53-dependent or independent apoptosis via ROS-mediated MAPKs and Akt ac-tivation in non-small cell lung cancer cells. Oncotarget 2017, 8, 28342–28358. [Google Scholar] [CrossRef] [Green Version]
- Cho, D.H.; Choi, Y.J.; Jo, S.A.; Jo, I. Nitric oxide production and regulation of endothelial nitric-oxide synthase phos-phorylation by prolonged treatment with troglitazone: Evidence for involvement of peroxisome proliferator-activated receptor (PPAR) gamma-dependent and PPARgamma-independent signaling pathways. J. Biol. Chem. 2004, 279, 2499–2506. [Google Scholar] [PubMed] [Green Version]
- Kim, H.Y.; Yu, Y.; Oh, S.-Y.; Wang, K.-K.; Kim, Y.-R.; Jung, S.-C.; Kim, H.S.; Jo, I. Far-Infrared Irradiation Inhibits Adipogenic Differentiation and Stimulates Osteogenic Differentiation of Human Tonsil-Derived Mesenchymal Stem Cells: Role of Protein Phosphatase 2B. Cell. Physiol. Biochem. 2019, 52, 240–253. [Google Scholar] [CrossRef] [PubMed]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.-J.; Oh, S.-Y.; Jo, I. Zearalenone Induces Endothelial Cell Apoptosis through Activation of a Cytosolic Ca2+/ERK1/2/p53/Caspase 3 Signaling Pathway. Toxins 2021, 13, 187. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13030187
Lee H-J, Oh S-Y, Jo I. Zearalenone Induces Endothelial Cell Apoptosis through Activation of a Cytosolic Ca2+/ERK1/2/p53/Caspase 3 Signaling Pathway. Toxins. 2021; 13(3):187. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13030187
Chicago/Turabian StyleLee, Hyeon-Ju, Se-Young Oh, and Inho Jo. 2021. "Zearalenone Induces Endothelial Cell Apoptosis through Activation of a Cytosolic Ca2+/ERK1/2/p53/Caspase 3 Signaling Pathway" Toxins 13, no. 3: 187. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins13030187




