Ochratoxin A in Portugal: A Review to Assess Human Exposure
Abstract
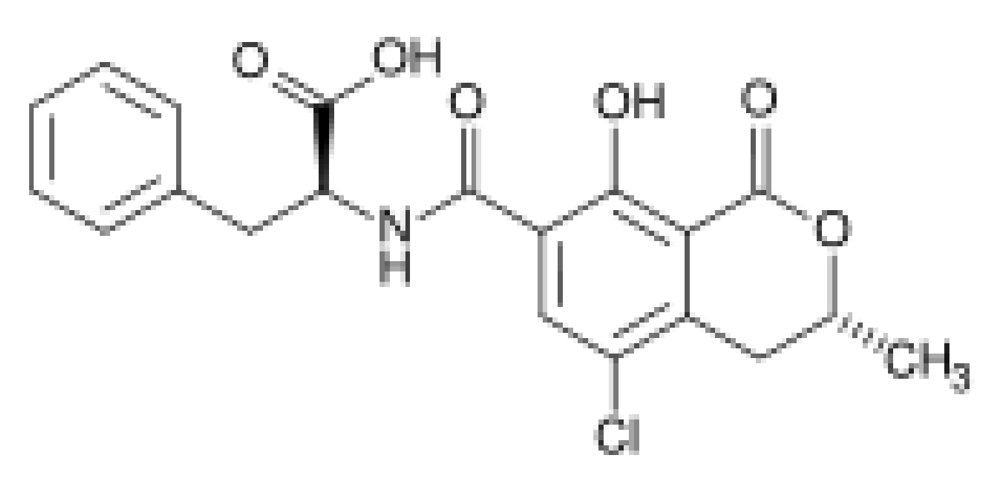
:1. Introduction
2. Portugal Portrayal
3. Exposure Assessment
3.1. Biological fluids contamination
3.2. Food contamination
4. Exposure Estimate
| Portuguese population exposure | ||||||
|---|---|---|---|---|---|---|
| Commodity | Consumption | Reference | Average level contamination | Reference | EDI | Contribution (%) |
| Cereals | 389.9 | [103] | 0.64 | [54] | 3.839 | 96.47 |
| Swine meat | 116.44 | 0.01 | [100] | 0.018 | 0.45 | |
| Chicken meat | 60 | 0 | 0.000 | 0 | ||
| Turkey meat | 10.7 | 0.02 | 0.003 | 0.08 | ||
| Wine | 123.01 | 0.063 | [30] | 0.119 | 3 | |
| Sum of dietary intake | 3.979 | |||||
| Portuguese population exposure | ||||
|---|---|---|---|---|
| Commodity | Consumption | Average level contamination | EDI | Contribution (%) |
| Cereal (wheat) flour | 235.1 | 0.19 | 0.69 | 84.7 |
| Coffee | 10.1 | 0.6 | 0.09 | 11.5 |
| Sweet wine | 1.1 | 0.01 | 0.00 | 0 |
| Rosé wine | 149.3 | 0.01 | 0.02 | 2.8 |
| Beer | 177.3 | 0.003 | 0.01 | 1 |
| Sum of dietary intake | 0.81 | |||
| Portuguese population exposure | Porto population exposure | ||||||
|---|---|---|---|---|---|---|---|
| Bread type | Consumption | Average level contamination | References | EDI | Contribution (%) | EDI | Contribution (%) |
| Wheat bread | 117.2 | 0.238 | [45,67,71] | 0.430 | 74.17 | 0.430 | 65.88 |
| Maize bread | 29.3 | 0.332 | [45,67] | 0.150 | 25.83 | - | - |
| Avintes bread* | 29.3 | 0.494 | [67] | - | - | 0.223 | 34.12 |
| 0.580 | Sum of dietary intake | 0.653 | Sum of dietary intake | ||||
5. Conclusions
Acknowledgements
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar]
- Van der Merwe, K.J.; Steyn, P.S.; Fourie, L.; Scott, De B.; Theron, J.J. Ochratoxin A, a toxic metabolite produced by Aspergillus ochraceus With. Nature 1965, 205, 1112–1113. [Google Scholar] [PubMed]
- Shotwell, O.L.; Hesseltine, C.W.; Goulden, M.L. Ochratoxin A: Occurrence as Natural Contaminant of a Corn Sample. Appl. Microbiol. 1969, 17, 765–766. [Google Scholar] [PubMed]
- Van Walbeek, W.; Scott, P.M.; Harwig, J.; Lawrence, J.W. Penicillium viridicatum Westling: A new source of ochratoxin A. Can. J. Microbiol. 1969, 15, 1281–1285. [Google Scholar]
- Duarte, S.C.; Pena, A.; Lino, C.M. A review on ochratoxin A occurrence and effects of processing of cereal and cereal derived food products. Food Microbiol. 2010, 27, 187–198. [Google Scholar]
- Zinedine, A.; Blesa, J.; Mahnine, N.; El Abidi, A.; Montesano, D.; Mañes, J. Pressurized liquid extraction coupled to liquid chromatography for the analysis of ochratoxin A in breakfast and infants cereals from Morocco. Food Control 2010, 21, 132–135. [Google Scholar]
- Clark, H.A.; Snedeker, S.M. Ochratoxin A: Its cancer risk and potential for exposure. J. Toxicol. Environ. Health B 2006, 9, 265–296. [Google Scholar] [CrossRef]
- Legarda, T.M.; Burdaspal, P.A. Occurrence of ochratoxin A in samples of bread marketed in Spain and twelve other countries. Alimentaria 2001, 321, 89–96. [Google Scholar]
- Zimmerli, B.; Dick, R. Determination of ochratoxin A at the ppt level in human blood, serum, milk and some foodstuffs by high-performance liquid chromatography with enhanced fluorescence detection and immunoaffinity column cleanup" methodology and Swiss data. J. Chromatog. B 1995, 666, 85–99. [Google Scholar]
- Visconti, A.; Pascale, M.; Centonze, G. Determination of ochratoxin A in domestic and imported beers in Italy by immunoaffinity clean-up and liquid chromatography. J. Chromatog. A 2000, 888, 321–326. [Google Scholar]
- Levi, C.P.; Trenk, H.L.; Mohr, H.K. Study of the occurrence of ochratoxin A in green coffee beans. J. Assoc. Off. Anal. Chem. 1974, 57, 866–870. [Google Scholar]
- Jørgensen, K. Survey of pork, poultry, coffee, beer and pulses for ochratoxin A. Food Addit. Contam. 1998, 15, 550–554. [Google Scholar]
- Curtui, V.G; Gareis, M.; Usleber, E.; Märtlbauer, E. Survey of Romanian slaughtered pigs for the occurrence of mycotoxins ochratoxins A and B, and zearalenone. Food Addit. Contam. 2001, 18, 730–738. [Google Scholar] [PubMed]
- Skaug, M.A. Analysis of Norwegian milk and infant formulas for ochratoxin A. Food Addit. Contam. 1999, 16, 75–78. [Google Scholar] [Green Version]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef] [PubMed]
- Palli, D.; Miraglia, M.; Saieva, C.; Masala, G.; Cava, E.; Colatosti, M. Corsi, A.M.; Corsi, A.M.; Russo, A.; Brera, C. Serum Levels of Ochratoxin A in Healthy Adults in Tuscany: Correlation with Individual Characteristics and between Repeat Measurements. Cancer Epidemiol. Biom. Prev. 1999, 8, 265–269. [Google Scholar]
- Gilbert, J.; Brereton, P.; MacDonald, S. Assessment of dietary exposure to ochratoxin A in the UK using a duplicate diet approach and analysis of urine and plasma samples. Food Addit. Contam. 2001, 18, 1088–1093. [Google Scholar]
- Skaug, M.A.; Helland, I.; Solvoll, K.; Saugstad, O.D. Presence of ochratoxin A in human milk in relation to dietary intake. Food Addit. Contam. 2001, 18, 321–327. [Google Scholar] [Green Version]
- Ratola, N.; Abade, E.; Simões, T.; Venâncio, A.; Alves, A. Evolution of ochratoxin A content from must to wine in Port Wine microvinification. Anal. Bioanal. Chem. 2005, 382, 405–411. [Google Scholar] [Green Version]
- Duarte, S.C.; Pena, A.; Lino, C.M. Ochratoxin A non-conventional exposure sources—A review. Microchem. J. 2009, 93, 115–120. [Google Scholar]
- Pfohl-Leszkowicz, A. Ochratoxin A and aristolochic acid involvement in nephropathies and associated urothelial tract tumours. Arh. Hig. Rada Toksikol. 2009, 60, 465–483. [Google Scholar]
- IARC, Some Naturally Occurring Substances, Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins, Monograph No. 56; International Agency for Research on Cancer: Lyon, France, 1993.
- Jørgensen, K.; Petersen, A. Content of ochratoxin A in paired kidney and meat samples from healthy Danish slaughter pigs. Food Addit. Contam. 2002, 19, 562–567. [Google Scholar]
- Pfohl-Leszkowicz, A.; Tozlovanu, M.; Manderville, R.; Peraica, M.; Castegnaro, M.; Stefanovic, V. New molecular and field evidences for the implication of mycotoxins but not aristolochic acid in human nephropathy and urinary tract tumor. Mol. Nutr. Food Res. 2007, 51, 1131–1146. [Google Scholar]
- Grosso, F.; Saïd, S.; Mabrouk, I.; Fremy, J.M.; Castegnaro, M.; Jemmali, M.; Dragacci, S. New data on the occurrence of ochratoxin A in human sera from patients affected or not by renal diseases in Tunisia. Food Chem. Toxicol. 2003, 41, 1133–1140. [Google Scholar] [CrossRef] [PubMed]
- Instituto Geográfico Português. Atlas de Portugal—Um país de área repartida. Available online: http://www.igeo.pt/atlas/Cap1/Cap1.html (accessed on 5 May 2010).
- Minerva. Ambiente e Diversidade na Europa: uma viagem pelos principais Biomas. Available online: http://www.minerva.uevora.pt/eschola2002/medit.htm (accessed on 19 January 2010).
- Paterson, R.R.M.; Lima, N. How will climate change affect mycotoxins in food? Food Res. Int. 2009. [Google Scholar]
- Alea. Actualidades do INE. Available online: http://alea-estp.ine.pt/ (accessed on 19 January 2010).
- Festas, I.; Herbert, P.; Santos, L.; Cabral, M.; Barros, P.; Alves, A. Ochratoxin A in some Portuguese Wines: Method validation and screening in Port wine and Vinho Verde. Am. J. Enol. Vitic. 2000, 51, 150–154. [Google Scholar]
- Shephard, G.S.; Van der Westhuizen, L.; Sewram, V. Biomarkers of exposure to fumonisin mycotoxins: A review. Food Addit. Contam. 2007, 24, 1196–1201. [Google Scholar]
- Castegnaro, M.; Canadas, D.; Vrabcheva, T.; Petkova-Bocharova, T.; Chernozemsky, I.N.; Pfohl-Leszkowicz, A. Balkan endemic nephropathy: Role of ochratoxins A through biomarkers. Mol. Nutr. Food Res. 2006, 50, 519–529. [Google Scholar]
- Lino, C.M.; Baeta, M.L.; Henri, M.; Dinis, A.M.P.; Pena, A.S.; Silveira, M.I.N. Levels of ochratoxin A in serum from urban and rural Portuguese populations and estimation of exposure degree. Food Chem. Toxicol. 2008, 46, 879–885. [Google Scholar]
- Erkekoğlu, P.; Sabuncuoğlu, S.; Aydın, S.; Şahin, G.; Giray, B. Determination of seasonal variations in serum ochratoxin A levels in healthy population living in some regions of Turkey by enzyme-linked immunosorbent assay. Toxicon 2010, 55, 507–513. [Google Scholar] [PubMed]
- Dinis, A.M.P.; Lino, C.M.; Pena, A.S. Ochratoxin A in nephropathic patients from two cities of central zone in Portugal. J. Pharm. Biomed. Anal. 2007, 44, 553–557. [Google Scholar]
- Sangare-Tigori, B.; Moukha, S.; Kouadio, J.H.; Dano, D.S.; Betbeder, A.-M.; Achour, A.; Creppy, E.E. Ochratoxin A in human blood in Abidjan, Côte d’Ivoire. Toxicon 2006, 47, 894–900. [Google Scholar]
- Thuvander, A.; Paulsen, J.E.; Axberg, K.; Johansson, N.; Vidnes, A.; Enghardt-Barbieri, H.; Trygg, K.; Lund-Larsen, K.; Jahrl, S.; Widenfalk, A.; Bosnes, V.; Alexander, J.; Hult, K.; Olsen, M. Levels of ochratoxin A in blood from Norwegian and Swedish blood donors and their possible correlation with food consumption. Food Chem. Toxicol. 2001, 39, 1145–1151. [Google Scholar]
- Domijan, A.M.; Peraica, M.; Fuchs, R.; Lucić, A.; Radić, B.; Balija, M.; Bosanac, I.; Grgičević, D. Ochratoxin A in blood of healthy population in Zagreb. Arh hig Rada Toksikol. 1999, 50, 263–271. [Google Scholar]
- Assaf, H.; Betbeder, A.-M.; Creppy, E.E.; Pallardy, M.; Azouri, H. Ochratoxin A levels in human plasma and foods in Lebanon. Hum. Exp. Toxicol. 2004, 23, 495–501. [Google Scholar]
- Ostry, V.; Malir, F.; Roubal, T.; Skarkova, J.; Ruprich, J.; Cerna, M.; Creppy, E.E. Monitoring of mycotoxin biomarkers in the Czech Republic. Mycot. Res. 2005, 21, 49–52. [Google Scholar]
- JECFA. Safety Evaluation of Certain Mycotoxins in Food. WHO Food Additives Series 59. Available online: http://whqlibdoc.who.int/publications/2008/9789241660594_eng.pdf (accessed on 18 November 2009).
- Duarte, S.; Bento, J.; Pena, A.; Lino, C.M.; Delerue-Matos, C.; Oliva-Teles, T.; Morais, S.; Correia, M.; Oliveira, M.B.P.P.; Alves, M.R.; Pereira, J.A. Monitoring of ochratoxin A exposure of the Portuguese population through a nationwide urine survey—Winter 2007. Sci. Total Environ. 2010, 408, 1195–1198. [Google Scholar]
- Pena, A.; Seifrtová, M.; Lino, C.M.; Silveira, M.I.N.; Solich, P. Estimation of ochratoxin A in Portuguese population: New data on the occurrence in human urine by high performance liquid chromatography with fluorescence detection. Food Chem. Toxicol. 2006, 4, 1449–1454. [Google Scholar]
- Manique, R.; Pena, A.; Lino, C.M.; Moltó, J.C.; Mañez, J. Ochratoxin A in themorning and afternoon portions of urine from Coimbra and Valencia populations. Toxicon 2008, 51, 1281–1287. [Google Scholar]
- Duarte, S.C; Bento, J.M.V.; Pena, A.; Lino, C.M. Ochratoxin A exposure assessment of the inhabitants of Lisbon during winter 2007/2008 through bread and urine analysis. Food Addit. Contam. 2009, 10, 1411–1420. [Google Scholar]
- Petkova-Bocharova, T.; Castegnaro, M.; Pfohl-Leszkowicz, A.; Garren, L.; Grosso, F.; Nikolov, I.; Vrabcheva, T.; Dragacci, S.; Chernozemsky, I.N. Analysis of ochratoxin A in serum and urine of inhabitants from an area with Balkan endemic nephropathy: A one month follow up study. Facta Univ. Ser. Med. Biol. 2003, 10, 62–68. [Google Scholar]
- Breitholtz-Emanuelsson, A.; Minervini, F.; Hult, K.; Visconti, A. Ochratoxin A in human serum samples collected in southern Italy from healthy individuals and individuals suffering from different kidney disorders. Nat. Toxins 1994, 2, 366–370. [Google Scholar]
- Fazecas, B.; Tar, A.; Kovacs, M. Ochratoxin a content of urine samples of healthy humans in Hungary. Acta Vet. Hung. 2005, 53, 35–44. [Google Scholar]
- EFSA. Opinion of the Scientific Panel on Contaminants in the Food Chain on a Request from the Commission Related to Ochratoxin A in Food. Question No. EFSA-Q-2005-154. EFSA J. 2006, 365, 1–56.
- Ueno, Y.; Maki, S.; Lin, J.; Furuya, M.; Sugiura, Y.; Kawamura, O. A 4-Year Study of Plasma Ochratoxin A in a Selected Population in Tokyo by Immunoassay and Immunoaffinity Column-linked HPLC. Food Chem. Toxicol. 1998, 36, 445–449. [Google Scholar]
- Miraglia, M.; Brera, C. Assessment of Dietary Intake of Ochratoxin A by the Population of EU Member States. SCOOP/2002,Task 3.2.7; European Union: Brussels, Belgium, 2002. [Google Scholar]
- Verger, P.; Volatier, J.L.; Dufour, A. Estimation des niveaux théOriques d’ingestion d’aflatoxines et d’ochratoxine. In Mycotoxines dans l’alimentation: évaluation et gestion du risque; Pfohl-Leszkowicz, A., Ed.; Tec & Doc Lavoisier: Paris, France, 1999; pp. 371–384. Chapter XIV. [Google Scholar]
- Czerwiecki, L.; Czajkowska, D.; Witkowska-Gwiazdowska, A. On ochratoxin A and fungal flora in Polish cereals from conventional and ecological farms. Part 2: Occurrence of ochratoxin A and fungi in cereals in 1998. Food Addit. Contam. 2002, 19, 1051–1057. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; Moltó, J.C.; Lino, C.M.; Mañes, J. Determination of ochratoxin A in organic and non-organic cereals and cereal products from Spain and Portugal. Food Chem. 2008, 107, 525–530. [Google Scholar]
- EC. Commission Regulation (EC) No. 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Food Stuffs. OJEUL 2006, 364, 5–24.
- Pena, A.; Cerejo, F.; Lino, C.; Silveira, I. Determination of ochratoxin A in Portuguese rice samples by high performance liquid chromatography with fluorescence detection. Anal. Bioanal. Chem. 2005, 382, 1288–1293. [Google Scholar]
- Gonzalez, L.; Juan, C.; Soriano, J.M.; Moltó, J.C.; Manes, J. Occurrence and daily intake of ochratoxin A of organic and non-organic rice and rice products. Int. J. Food Microbiol. 2006, 107, 223–227. [Google Scholar]
- Nguyen, M.T.; Tozlovanu, M.; Tran, T.L.; Pfohl-Leszkowicz, A. Occurrence of aflatoxin B1, citrinin and ochratoxin A in rice in five provinces of central region in Vietnam. Food Chem. 2007, 105, 42–47. [Google Scholar]
- Zinedine, A.; Soriano, J.M.; Juan, C.; Mojemmi, B.; Moltó, J.C.; Bouclouze, A.; Cherrah, Y.; Idrissi, L.; El Aouad, R.; Mañes, J. Incidence of ochratoxin A in rice and dried fruits from Rabat and Salé area, Morocco. Food Addit. Contam. 2007, 24, 285–291. [Google Scholar]
- Ghali, R.; Hmaissia-khlifa, K.; Ghorbel, H.; Maaroufi, K.; Hedili, A. Incidence of aflatoxins, ochratoxin A and zearalenone in tunisian foods. Food Control 2008, 19, 921–924. [Google Scholar]
- Zaied, C.; Abid, S.; Zorgui, L.; Bouaziz, C.; Chouchane, S.; Jomaa, M.; Bacha, H. Natural occurrence of ochratoxin A in Tunisian cereals. Food Control 2009, 20, 218–222. [Google Scholar]
- Vega, M.; Muñoz, K.; Sepúlveda, C.; Aranda, M.; Campos, V.; Villegas, R.; Villarroel, O. Solid-phase extraction and HPLC determination of ochratoxin A in cereals products on Chilean market. Food Control 2009, 20, 631–634. [Google Scholar]
- INE. Anuário estatístico de Portugal 2006. Available online: http://www.ine.pt/ine/acess/pub_detalhe.jsp? boui_aux =11796801 (accessed on 12 June 2008).
- INE. Estatísticas da produção industrial 2006. Available online: http://www.ine.pt/ine/acess/pub_detalhe.jsp?boui_aux=16671129 (accessed on 12 June 2008).
- Juan, C.; Lino, C.M.; Pena, A.; Moltó, J.C.; Mañes, J.; Silveira, I. Determination of ochratoxin A in maize bread samples by LC with fluorescence detection. Talanta 2007, 73, 246–250. [Google Scholar]
- Juan, C.; Pena, A.; Lino, C.; Moltó, J.C.; Mañes, J. Levels of ochratoxin A in wheat and maize bread from the central zone of Portugal. Int. J. Food Microbiol. 2008, 127, 284–289. [Google Scholar]
- Duarte, S.C.; Tanello, A.; Pena, A.; Lino, C.M.; Matos, C.D.; Oliveira, M.B.P.P.; Alves, M.R. Evaluation of ochratoxin A exposure degree in two Portuguese cities through wheat and maize bread consumption during the winter 2007. Food Control 2010, 21, 702–707. [Google Scholar] [CrossRef]
- Bullerman, L.B.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 140–146. [Google Scholar]
- Valle-Algarra, F.M.; Mateo, E.M.; Medina, Á.; Mateo, F.; Gimeno-Adelantado, J.V.; Jiménez, M. Changes in ochratoxin A and type B trichothecenes contained in wheat flour during dough fermentation and breadbaking. Food Addit. Contam. Part A 2009, 26, 896–906. [Google Scholar] [CrossRef]
- Arroyo, M.; Aldred, D.; Magan, N. Environmental factors and weak organic acid interactions have differential effects on control of growth and ochratoxin A production by Penicillium verrucosum isolates in bread. Int. J. Food Microbiol. 2005, 98, 223–231. [Google Scholar]
- Bento, J.M.V.; Pena, A.; Lino, C.M.; Pereira, J.A. Determination of ochratoxin A content in wheat bread samples collected from the Algarve and Bragança regions, Portugal: Winter 2007. Microchem. J. 2009, 91, 165–169. [Google Scholar]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007, 11, 1633–1644. [Google Scholar]
- Cengiz, M.; Oruç, H.H.; Uzunoğlu, I.; Sonal, S. Ochratoxin A levels in different types of bread and flour. Uludag Univ. J. Fac. Vet. Med. 2007, 26, 7–10. [Google Scholar]
- González-Osnaya, L.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Dietary intake of ochratoxin A from conventional and organic bread. Int. J. Food Microbiol. 2007, 118, 87–91. [Google Scholar]
- Zummo, N.; Scott, G.E. Interaction of Fusarium moniliforme and Aspergillus flavus on kernel infection and aflatoxin contamination in maize ears. Plant Dis. 1992, 76, 771–773. [Google Scholar]
- Puntarić, D.; Bosnir, J.; Smit, Z.; Skes, I.; Baklaic, Z. Ochratoxin A in corn and wheat: Geographical association with endemic nephropathy. Croat. Med. J. 2001, 42, 175–180. [Google Scholar] [PubMed]
- Ratola, N.; Martins, N.; Alves, A. Ochratoxin A in wines-assessing global uncertainty associated with the results. Anal. Chim. Acta 2004, 513, 319–324. [Google Scholar]
- Serra, R.; Mendonça, C.; Venâncio, A. Fungi and ochratoxin A detected in healthy grapes for wine Production. Lett. Appl. Microbiol. 2006, 42, 42–47. [Google Scholar]
- OIV, Reduction de L’Ochratoxine A dans les Vins. Resolution CST 1/2002; Off. Int. de la Vigne et du Vin: Paris, France, 2002.
- Rosa, C.A.R.; Magnoli, C.E.; Fraga, M.E.; Dalcero, A.M.; Santana, D.M.N. Occurrence of ochratoxin A in wine and grape juice marketed in Rio de Janeiro, Brazil. Food Addit. Contam. 2004, 21, 358–364. [Google Scholar] [PubMed]
- Stander, M.A.; Steyn, P.S. Survey of ochratoxin A in South African wines. S. Afr. J. Enol. Vitic. 2002, 23, 9–13. [Google Scholar]
- Shephard, G.S.; Fabiani, A.; Stockenström, S.; Mshicileli, N.; Sewram, V. Quantitation of Ochratoxin A in South African Wines. J. Agric. Food Chem. 2003, 51, 1102–1106. [Google Scholar] [CrossRef] [PubMed]
- Berente, B.; Móricz, A.; H.-Otta, K; Záraya, G.; Lékó, L.; Rácz, L. Determination of ochratoxin A in Hungarian wines. Microchem. J. 2005, 79, 103–107. [Google Scholar] [CrossRef]
- Var, I.; Kabak, B. Occurrence of ochratoxin A in Turkish wines. Microchem. J. 2007, 86, 241–247. [Google Scholar]
- Bellí, N.; Marin, S.; Duaigues, A.; Ramos, A.J.; Sanchis, V. Ochratoxin A in wines, musts and grape juices from Spain. J. Sci. Food Agric. 2004, 84, 591–594. [Google Scholar]
- Burdaspal, P.; Legarda, T. Occurrence of ochratoxin A in sweet wines produced in Spain and other Countries. Food Addit. Contam. 2007, 24, 976–986. [Google Scholar]
- EC. Comiission regulation. No 123/2005 of 26 January 2005 amending Regulation (EC) No. 466/2001 as regards ochratoxin A. Off. J. EU 2006, L-25, 3–5.
- Alvito, P.C.; Sizoo, E.A; Almeida, C.M.M.; van Egmond, H.P. Occurrence of aflatoxins and ochratoxin A in baby foods in portugal. Food Anal. Method 2010, 3, 22–30. [Google Scholar] [CrossRef]
- Kuiper-Goodman, T.; Hilts, C.; Billiard, S.M.; Kiparissis, Y.; Richard, I.D.K.; Hayward, S. Health risk assessment of ochratoxin A for all age-sex strata in a market Economy. Food Addit. Contam. 2010, 27, 212–240. [Google Scholar]
- Araguás, C.; González-Peñas, E.; López de Cerain, A. Study on ochratoxin A in cereal-derived products from Spain. Food Chem. 2005, 92, 459–464. [Google Scholar]
- Lombaert, G.A.; Pellaers, P.; Roscoe, V.; Mankotia, M.; Neil, R.; Scott, P.M. Mycotoxins in infant cereal foods from the Canadian retail market. Food. Addit. Contam. 2003, 20, 494–504. [Google Scholar]
- Kabak, B. Ochratoxin A in cereal-derived products in Turkey: Occurrence and exposure assessment. Food Chem. Toxicol. 2009, 47, 348–352. [Google Scholar]
- Schiavone, A.; Cavallero, C.; Girotto, L.; Pozzo, L.; Antoniazzi, S.; Cavallarin, L. A survey on the occurrence of ochratoxin A in feeds and sera collected in conventional and organic poultry farms in Northern Italy. Ital. J. Anim. Sci. 2008, 7, 495–503. [Google Scholar]
- Petzinger, E.; Weidenbach, A. Mycotoxins in the food chain: The role of ochratoxins. Livest. Prod. Sci. 2002, 76, 245–250. [Google Scholar]
- Hagelberg, S.; Hult, K.; Fuchs, R. Toxicokinetics of ochratoxin A in several species and its plasma-binding properties. J. Appl. Toxicol. 1989, 9, 91–96. [Google Scholar]
- Monaci, L.; Palmisano, F. Determination of ochratoxin A in foods: State-of-the-art and analytical challenges. Anal. Bioanal. Chem. 2004, 378, 96–103. [Google Scholar]
- Jiménez, A.M.; De Cerain, A.L.; Gonzalez-Penas, E.; Bello, J. Determination of ochratoxin A in pig liver-derived pâtés by high-performance liquid chromatography. Food Addit. Contam. 2001, 18, 559–563. [Google Scholar]
- Curtui, V.G.; Gareis, M. A simple HPLC method for the determination of the mycotoxins ochratoxin A and B in blood serum of swine. Food Addit. Contam. 2001, 18, 635–643. [Google Scholar]
- Chiavaro, E.; Lepiani, A.; Colla, F.; Bettoni, P.; Pari, E.; Spotti, E. Ochratoxin A determination in ham by immunoaffinity clean-up and a quick fluorometric method. Food Addit. Contam. 2002, 19, 575–581. [Google Scholar]
- Guillamont, E.M.; Lino, C.M.; Baeta, M.L.; Pena, A.S.; Silveira, M.I.N.; Vinuesa, J.M. A comparative study of extraction apparatus in HPLC analysis of ochratoxin A in muscle. Anal. Bioanal. Chem. 2005, 383, 570–575. [Google Scholar]
- Monaci, L.; Tantillo, G.; Palmisano, F. Determination of ochratoxin A in pig tissues by liquid-liquid extraction and clean-up and high-performance liquid chromatography. Anal. Bioanal. Chem. 2004, 378, 1777–1782. [Google Scholar]
- Matrella, R.; Monaci, L.; Milillo, M.A.; Palmisano, F.; Tantillo, M.G. Ochratoxin A determination in paired kidneys and muscle samples from swines slaughtered in southern Italy. Food Control 2006, 17, 114–117. [Google Scholar]
- INE. Base de dados—Consumo anual humano per capita. Available online: http://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_pesquisa&frm_accao=PESQUISAR&frm_show_page_num=1&frm_modo_pesquisa=PESQUISA_SIMPLES&frm_modo_texto=MODO_TEXTO_ALL&frm_texto=consumo+anual&frm_imgPesquisar=++ (accessed on 18 January 2010).
- GEMS/Food, Regional diets: Regional per Capita Consumption of Raw and Semi-processed Agricultural Commodities. Global Environment Monitoring System/Food Contamination Monitoring and Assessment Programme, Food Safety Department, WHO: Geneva, Switzerland, 2003.
- SCF, Opinion on of the European Comission’s Scientific Committee on food on ochratoxin A. SCF, European Union: Brussels, Belgium, 1998; Expressed 17.09.98.
- Leblanc, J.-C.; Tard, A.; Volatier, J.-L.; Verger, P. Estimated dietary exposure to principal food mycotoxins from The First French Total Diet Study. Food Addit. Contam. 2005, 22, 652–672. [Google Scholar]
- JECFA. Safety Evaluation of Certain Mycotoxins in Food. WHO Food Additives Series 47. Available online: http://www.inchem.org/documents/jecfa/jecmono/v47je01.htm (accessed on 10 August 2008).
- EFSA. Opinion of the Scientific Panel on Contaminants in the Food Chain on a Request from the Commission Related to Ochratoxin A in Food. Question no. EFSA-Q-2005-154. EFSA J. 2006, 365, 1–56.
- Kuiper-Goodman, T.; Scott, P.M. Risk assessment of the mycotoxin ochratoxin A. Biomed. Environ. Sci. 1989, 2, 179–248. [Google Scholar]
- Mantle, P.G. Experimental mycotoxic nephropathies and Balkan endemic nephropathy. Facta Univ. Ser. Med. Biol. 2002, 9, 64–65. [Google Scholar]
- Pfohl-Leszkowicz, A.; Molinie, A.; Tozlovanu, M.; Manderville, R.A. Combined toxic effects of ochratoxin A and citrinin, in vitro and in vivo. In Food Contaminants; Mycotoxins and Food Allergen; Siantar, D.P., Trucksess, M.W., Scott, P.M., Herman, E.M., Eds.; ACS Symposium Series, American Chemical Society: Washington, DC, USA, 2008; Volume 1001, pp. 56–80. [Google Scholar]
- Miraglia, M.; Brera, C.; Colatosti, M. Application of biomarkers to assessment of risk to human health from exposure to mycotoxins. Microchem. J. 1996, 54, 472–477. [Google Scholar]
- Coronel, M.B.; Sanchis, V.; Ramos, A.J.; Marin, S. Assessment of the exposure to ochratoxin A in the province of Lleida, Spain. Food Chem. Toxicol. 2009, 47, 2847–2852. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Duarte, S.C.; Pena, A.; Lino, C.M. Ochratoxin A in Portugal: A Review to Assess Human Exposure. Toxins 2010, 2, 1225-1249. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins2061225
Duarte SC, Pena A, Lino CM. Ochratoxin A in Portugal: A Review to Assess Human Exposure. Toxins. 2010; 2(6):1225-1249. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins2061225
Chicago/Turabian StyleDuarte, Sofia C., Angelina Pena, and Celeste M. Lino. 2010. "Ochratoxin A in Portugal: A Review to Assess Human Exposure" Toxins 2, no. 6: 1225-1249. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins2061225