Apoptosis Induction by OTA and TNF-α in Cultured Primary Rat Hepatocytes and Prevention by Silibinin
Abstract
:1. Introduction
2. Results and Discussion
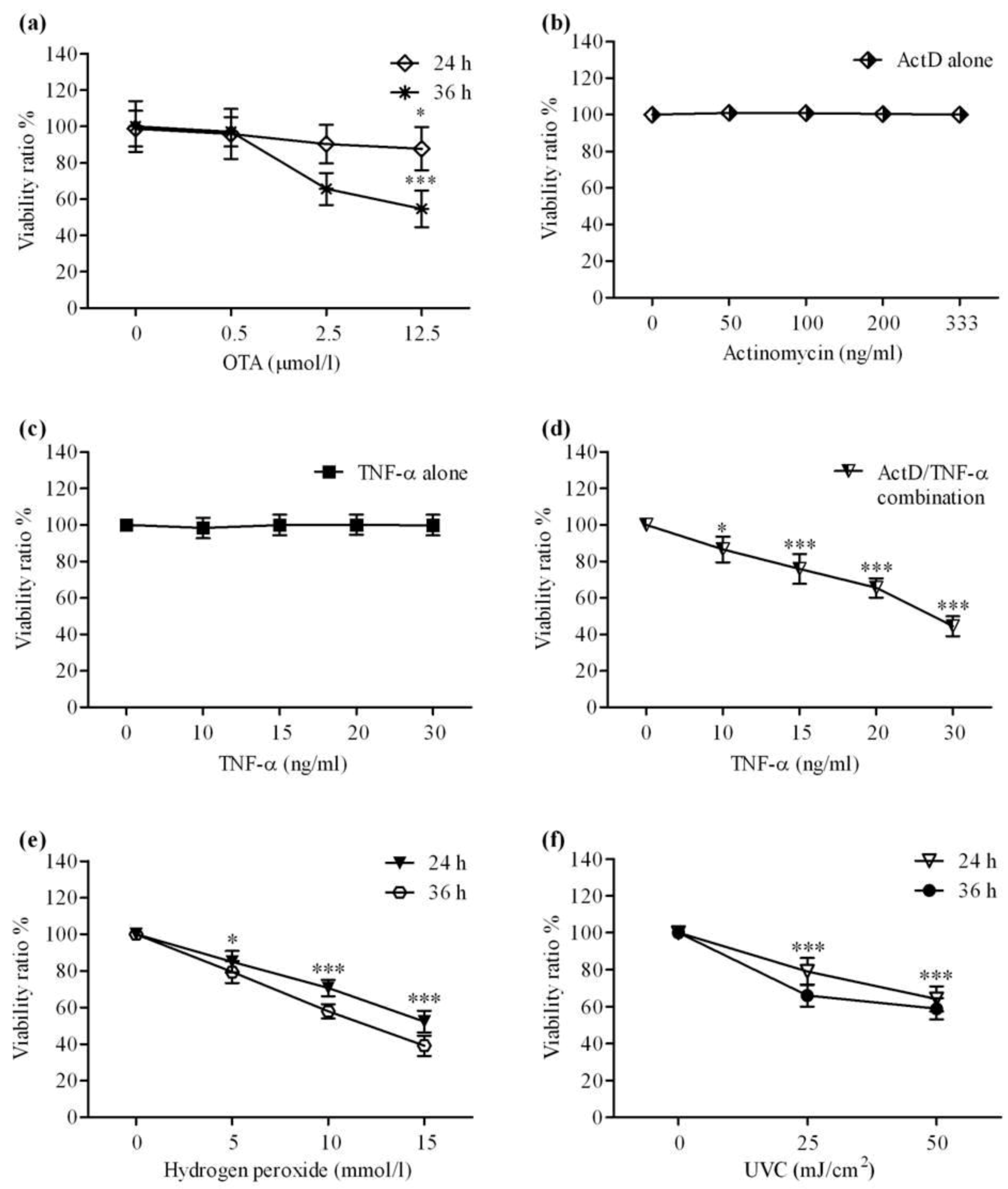
2.1. Cytotoxicity Effects of OTA, ActD, TNF-α, H2O, and UV-C
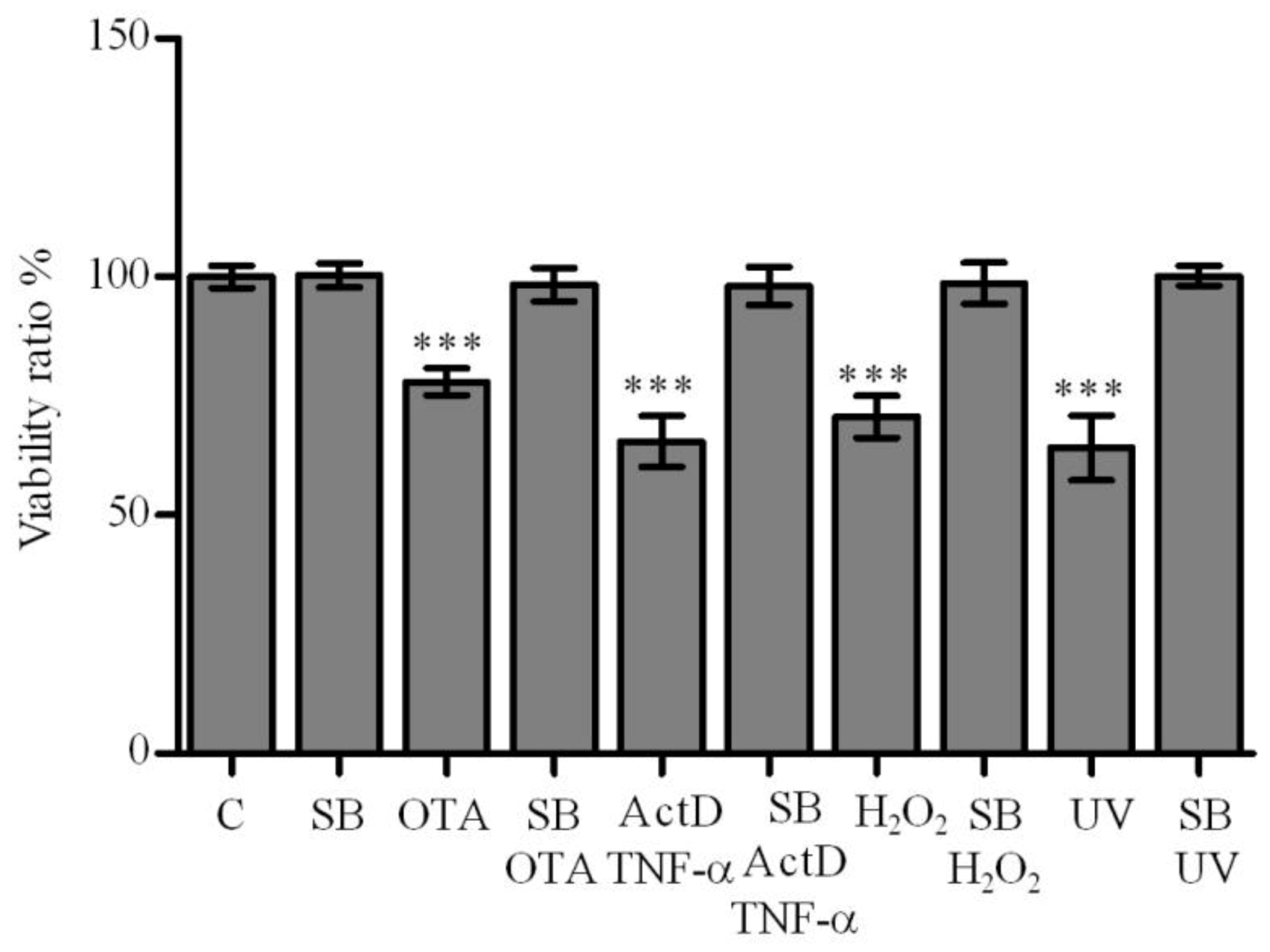
2.2. Effect of Silibinin on OTA, ActD/TNF-α, H2O2 and UV-C Cytotoxicity
2.3. Effect of OTA, ActD/TNF-α, H2O2 and UV-C on Caspase-3 Activity and Anti-apoptotic Effects of Silibinin

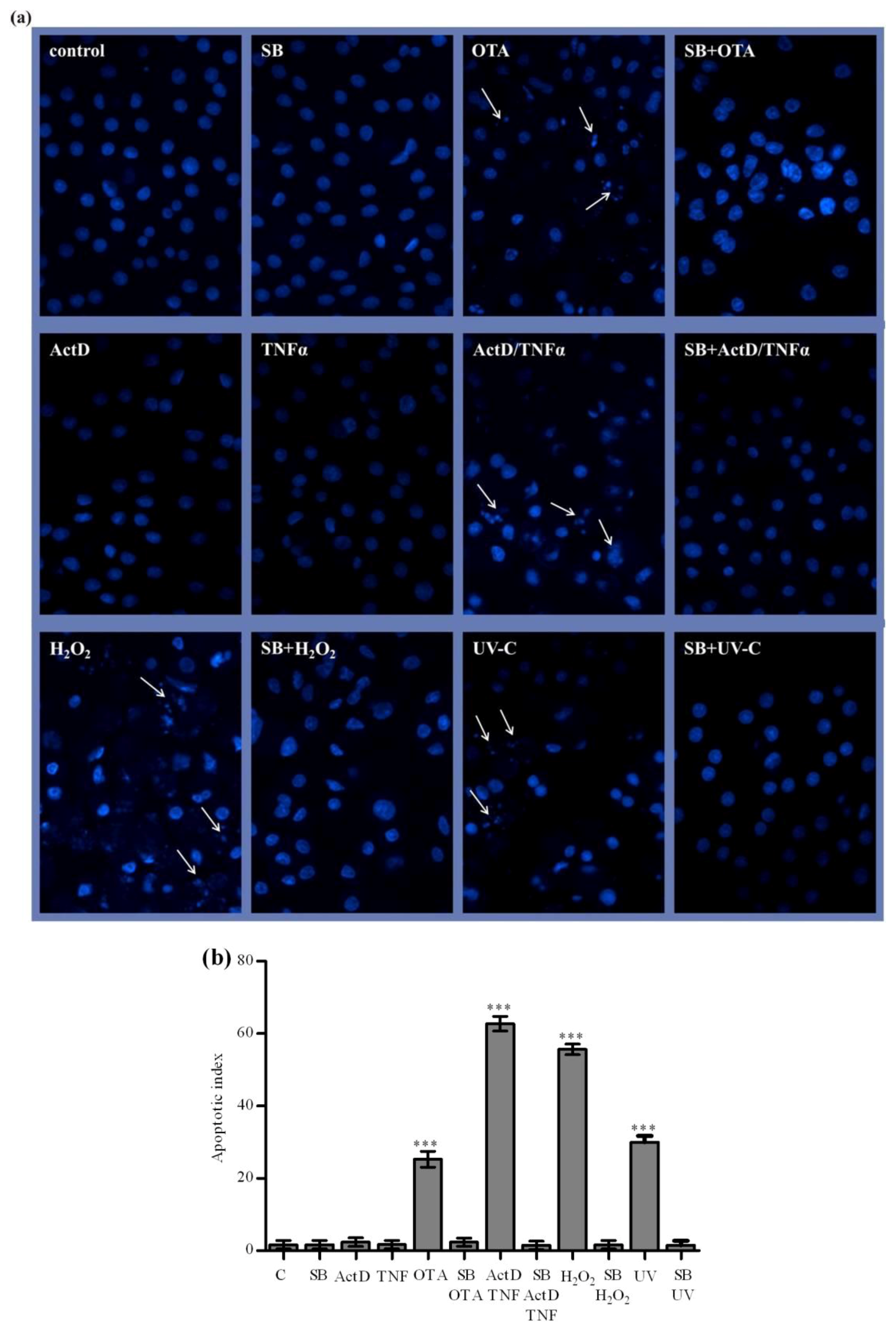
2.4. The Nuclear Damage Caused by OTA, ActD/TNF-α, H2O2 and UV-C and Prevention by Silibinin
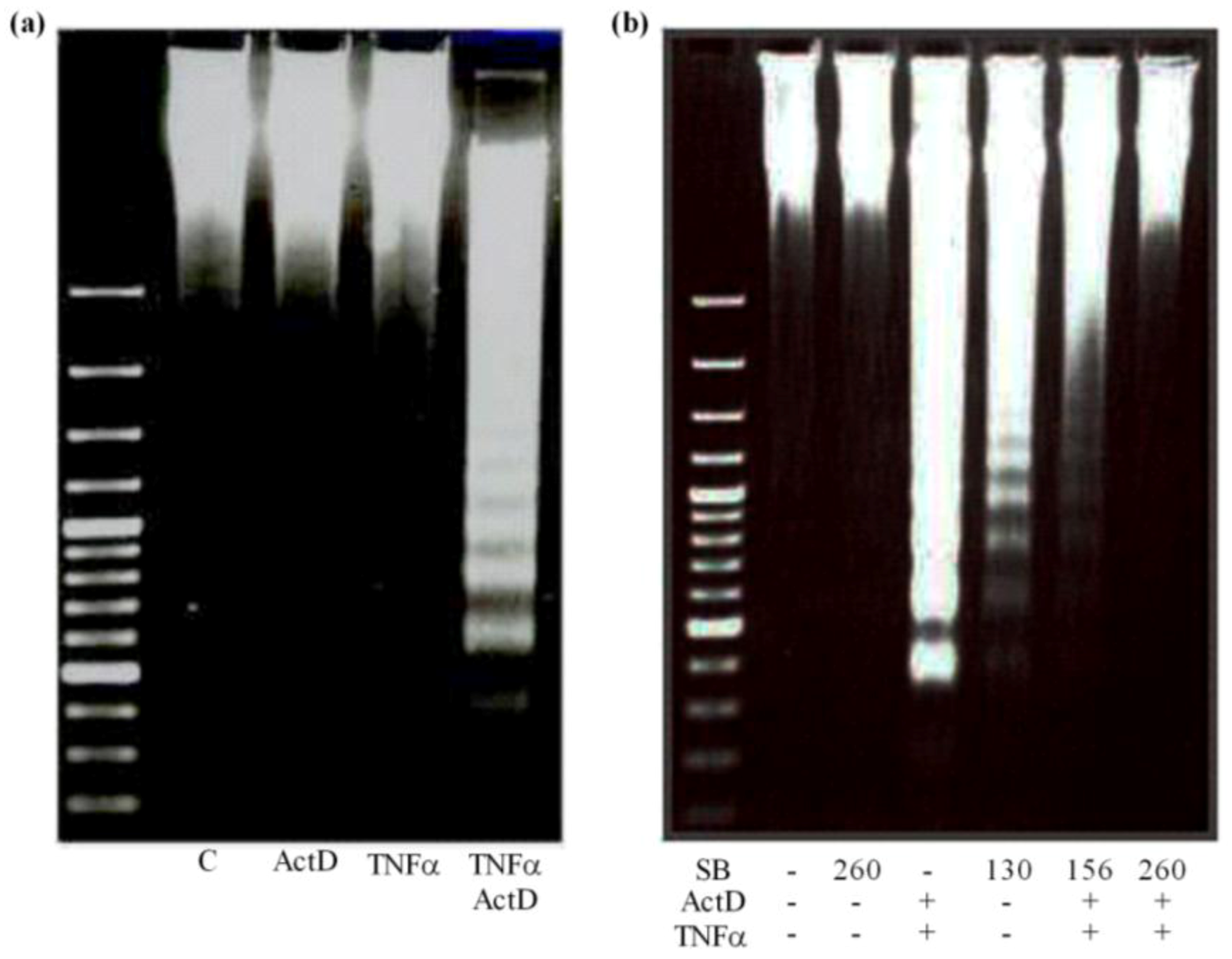
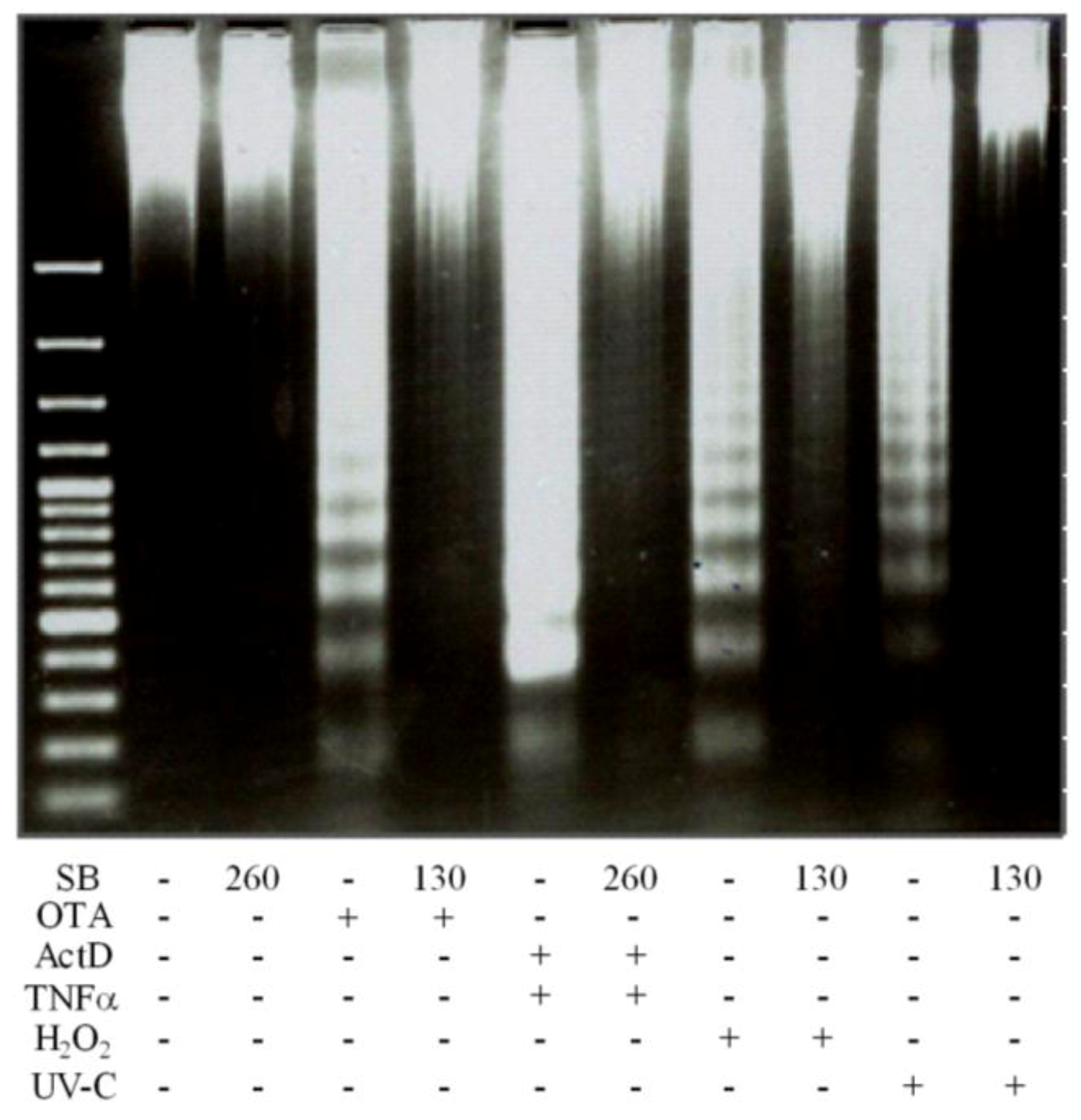
2.5. Development of DNA Ladders by OTA, ActD/TNF-α, H2O2 and UV-C and Prevention by Silibinin
2.6. OTA and Soluble TNF-α Receptor I (sTNFRI)


2.7. OTA Causes Cell Toxicity In Vitro via Oxidative Stress Reactions
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Animals
3.3. Isolation and Culture of Primary Rat Hepatocytes
3.4. Sensitization of Primary Rat Hepatocytes
3.5. Culture of HPCT-1E3 Hepatocytoma Cells
3.6. Cytotoxicity Assay
3.7. Analysis of Hepatocyte Nuclear Morphology
3.8. Caspase-3 Activity
3.9. DNA Ladder Fragmentation Analysis
3.10. Oxidative Stress Determination
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Lawen, A. Apoptosis-An introduction. Bioessays 2003, 25, 888–896. [Google Scholar] [CrossRef]
- Thornberry, N.A.; Lazebnik, Y. Caspases: Enemies within. Science 1998, 281, 1312–1316. [Google Scholar] [CrossRef]
- Riedl, S.J.; Shi, Y. Molecular mechanisms of caspase regulation during apoptosis. Nat. Rev. Mol. Cell Biol. 2004, 5, 897–907. [Google Scholar] [CrossRef]
- Petzinger, E.; Ziegler, K. Ochratoxin A from a toxicological perspective. J. Vet. Pharmacol. Ther. 2000, 23, 91–98. [Google Scholar] [CrossRef]
- Ringot, D.; Chango, A.; Schneider, Y.J.; Larondelle, Y. Toxicokinetics and toxicodynamics of ochratoxin A, an update. Chem. Biol. Interact. 2006, 159, 18–46. [Google Scholar] [CrossRef]
- Aydin, G.; Ozçelik, N.; Ciçek, E.; Soyöz, M. Histopathologic changes in liver and renal tissues induced by Ochratoxin A and melatonin in rats. Hum. Exp. Toxicol. 2003, 22, 383–391. [Google Scholar]
- Atroshi, F.; Biese, I.; Saloniemi, H.; Ali-Vehmas, T.; Saari, S.; Rizzo, A.; Veijalainen, P. Significance of apoptosis and its relationship to antioxidants after ochratoxin A administration in mice. J. Pharm. Pharm. Sci. 2000, 3, 281–291. [Google Scholar]
- Chopra, M.; Link, P.; Michels, C.; Schrenk, D. Characterization of ochratoxin A-induced apoptosis in primary rat hepatocytes. Cell Biol. Toxicol. 2010, 26, 239–254. [Google Scholar] [CrossRef]
- Essid, E.; Petzinger, E. Silibinin pretreatment protects against Ochratoxin A-mediated apoptosis in primary rat hepatocytes. Mycotoxin Res. 2011, 27, 167–176. [Google Scholar] [CrossRef]
- Al-Anati, L.; Essid, E.; Stenius, U.; Beuerlein, K.; Schuh, K.; Petzinger, E. Differential cell sensitivity between OTA and LPS upon Releasing TNF-alpha. Toxins 2010, 2, 1279–1299. [Google Scholar] [CrossRef]
- Leist, M.; Gantner, F.; Jilg, S.; Wendel, A. Activation of the 55 kDa TNF receptor is necessary and sufficient for TNF-induced liver failure, hepatocyte apoptosis, and nitrite releas. J. Immunol. 1995, 154, 1307–1316. [Google Scholar]
- Nagata, S. Apoptosis by death factor. Cell 1997, 88, 355–365. [Google Scholar] [CrossRef]
- Micheau, O.; Tschopp, J. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 2003, 114, 181–190. [Google Scholar] [CrossRef]
- Ding, W.X.; Yin, X.M. Dissection of the multiple mechanisms of TNF-alpha-induced apoptosis in liver injury. J. Cell. Mol. Med. 2004, 8, 445–454. [Google Scholar] [CrossRef]
- Leist, M.; Gantner, F.; Bohlinger, I.; Germann, P.G.; Tiegs, G.; Wendel, A. Murine hepatocyte apoptosis induced in vitro and in vivo by TNF-alpha requires transcriptional arrest. J. Immunol. 1994, 153, 1778–1788. [Google Scholar]
- Hu, C.; Han, J.Q.; Xu, Z.; Xiu, H.M.; Liu, J.D.; Hao, Y. Effects of baicalin on hepatocyte apoptosis induced by TNF-alpha and act D in rats (In Chinese). China J. Chin. Mater. Med. 2001, 26, 124–127. [Google Scholar]
- Bindoli, A.; Cavallini, L.; Siliprandi, N. Inhibitory action of silymarin of lipid peroxide formation in rat liver mitochondria and microsomes. Biochem. Pharmacol. 1977, 26, 2405–2409. [Google Scholar] [CrossRef]
- Kiruthiga, P.V.; Shafreen, R.B.; Pandian, S.K.; Devi, K.P. Silymarin protection against major reactive oxygen species released by environmental toxins: Exogenous H2O2 exposure in erythrocytes. Basic Clin. Pharmacol. Toxicol. 2007, 100, 414–419. [Google Scholar] [CrossRef]
- Asghar, Z.; Masood, Z. Evaluation of antioxidant properties of silymarin and its potential to inhibit peroxyl radicals in vitro. Pak. J. Pharm. Sci. 2008, 21, 249–254. [Google Scholar]
- Al-Anati, L.; Essid, E.; Reinehr, R.; Petzinger, E. Silibinin protects OTA-mediated TNF-alpha release from perfused rat livers and isolated rat Kupffer cells. Mol. Nutr. Food Res. 2009, 53, 460–466. [Google Scholar] [CrossRef]
- Pook, S.H.; Toh, C.K.; Mahendran, R. Combination of thiol antioxidant Silibinin with Brostallicin is associated with increase in the anti-apoptotic protein Bcl-2 and decrease in caspase 3 activity. Cancer Lett. 2006, 238, 146–152. [Google Scholar] [CrossRef]
- Omar, R.F.; Hasinoff, B.B.; Mejilla, F.; Rahimtula, A.D. Mechanism of ochratoxin A stimulated lipid peroxidation. Biochem. Pharmacol. 1990, 40, 1183–1191. [Google Scholar] [CrossRef]
- Gagliano, N.; Donne, I.D.; Torri, C.; Migliori, M.; Grizzi, F.; Milzani, A.; Filippi, C.; Annoni, G.; Colombo, P.; Costa, F.; et al. Early cytotoxic effects of ochratoxin A in rat liver: A morphological, biochemical and molecular study. Toxicology 2006, 225, 214–224. [Google Scholar] [CrossRef]
- Marin-Kuan, M.; Nestler, S.; Verguet, C.; Bezençon, C.; Piguet, D.; Mansourian, R.; Holzwarth, J.; Grigorov, M.; Delatour, T.; Mantle, P.; et al. A toxicogenomics approach to identify new plausible epigenetic mechanisms of ochratoxin a carcinogenicity in rat. Toxicol. Sci. 2006, 89, 120–134. [Google Scholar]
- Cavin, C.; Delatour, T.; Marin-Kuan, M.; Holzhäuser, D.; Higgins, L.; Bezençon, C.; Guignard, G.; Junod, S.; Richoz-Payot, J.; Gremaud, E.; et al. Reduction in antioxidant defenses may contribute to ochratoxin A toxicity and carcinogenicity. Toxicol. Sci. 2007, 96, 30–39. [Google Scholar]
- Adamson, G.M.; Billings, R.E. Tumor necrosis factor induced oxidative stress in isolated mouse hepatocytes. Arch. Biochem. Biophys. 1992, 294, 223–229. [Google Scholar] [CrossRef]
- Kamat, J.P.; Devasagayam, T.P.; Priyadarsini, K.I.; Mohan, H.; Mittal, J.P. Oxidative damage induced by the fullerene C60 on photosensitization in rat liver microsomes. Chem. Biol. Interact. 1998, 114, 145–159. [Google Scholar] [CrossRef]
- Svobodová, A.R.; Galandáková, A.; Sianská, J.; Doležal, D.; Ulrichová, J.; Vostálová, J. Acute exposure to solar simulated ultraviolet radiation affects oxidative stress-related biomarkers in skin, liver and blood of hairless mice. Biol. Pharm. Bull. 2011, 34, 471–479. [Google Scholar] [CrossRef]
- Manna, S.K.; Mukhopadhyay, A.; Van, N.T.; Aggarwal, B.B. Silymarin suppresses TNF-induced activation of NF-kappa B, c-Jun N-terminal kinase, and apoptosis. J. Immunol. 1999, 163, 6800–6809. [Google Scholar]
- Saller, R.; Meier, R.; Brignoli, R. The use of silymarin in the treatment of liver diseases. Drugs 2001, 61, 2035–2063. [Google Scholar] [CrossRef]
- Schümann, J.; Prockl, J.; Kiemer, A.K.; Vollmar, A.M.; Bang, R.; Tiegs, G. Silibinin protects mice from T cell-dependent liver injury. J. Hepatol. 2003, 39, 333–340. [Google Scholar] [CrossRef]
- Comelli, M.C.; Mengs, U.; Schneider, C.; Prosdocimi, M. Toward the definition of the mechanism of action of silymarin: Activities related to cellular protection from toxic damage induced by chemotherapy. Integr. Cancer Ther. 2007, 6, 120–129. [Google Scholar] [CrossRef]
- Luper, S. A review of plants used in the treatment of liver disease: Part 1. Altern. Med. Rev. 1998, 3, 410–421. [Google Scholar]
- Chrungoo, V.J.; Singh, K.; Singh, J. Silymarin mediated differential modulation of toxicity induced by carbon tetrachloride, paracetamol and D-galactosamine in freshly isolated rat hepatocytes. Indian J. Exp. Biol. 1997, 35, 611–617. [Google Scholar]
- Kroemer, G.; Petit, P.; Zamzami, N.; Vayssière, J.L.; Mignotte, B. The biochemistry of programmed cell death. FASEB J. 1995, 9, 1277–1287. [Google Scholar]
- Patel, T.; Gores, G.J.; Kaufmann, S.H. The role of proteases during apoptosis. FASEB J. 1996, 10, 587–597. [Google Scholar]
- Enari, M.; Sakahira, H.; Yokoyama, H.; Okawa, K.; Iwamatsu, A.; Nagata, S. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 1998, 391, 43–50. [Google Scholar] [CrossRef]
- Gekle, M.; Sauvant, C.; Schwerdt, G. Ochratoxin A at nanomolar concentrations: A signal modulator in renal cells. Mol. Nutr. Food Res. 2005, 49, 118–130. [Google Scholar] [CrossRef]
- Assaf, H.; Azouri, H.; Pallardy, M. Ochratoxin A induces apoptosis in human lymphocytes through down regulation of Bcl-xL. Toxicol. Sci. 2004, 79, 335–344. [Google Scholar] [CrossRef]
- Gekle, M.; Schwerdt, G.; Freudinger, R.; Mildenberger, S.; Wilflingseder, D.; Pollack, V.; Dander, M.; Schramek, H. Ochratoxin A induces JNK activation and apoptosis in MDCK-C7 cells at nanomolar concentrations. J. Pharmacol. Exp. Ther. 2000, 293, 837–844. [Google Scholar]
- Scibelli, A.; Tafuri, S.; Ferrante, M.C.; Alimenti, E.; Naso, B.; Lucisano, A.; Staiano, N.; della Morte, R. Ochratoxin A affects COS cell adhesion and signaling. Toxicol. Appl. Pharmacol. 2003, 192, 222–230. [Google Scholar] [CrossRef]
- Sauvant, C.; Holzinger, H.; Gekle, M. The nephrotoxin ochratoxin A induces key parameters of chronic interstitial nephropathy in renal proximal tubular cells. Cell Physiol. Biochem. 2005, 15, 125–134. [Google Scholar] [CrossRef]
- Wang, Y.K.; Hong, Y.J.; Huang, Z.Q. Protective effects of silybin on human umbilical vein endothelial cell injury induced by H2O2 in vitro. Vascul. Pharmacol. 2005, 43, 198–206. [Google Scholar] [CrossRef]
- Li, L.H.; Wu, L.J.; Tashiro, S.; Onodera, S.; Uchiumi, F.; Ikejima, T. Silibinin prevents UV-induced HaCaT cell apoptosis partly through inhibition of caspase-8 pathway. Biol. Pharm. Bull. 2006, 29, 1096–1101. [Google Scholar] [CrossRef]
- Jiang, Y.Y.; Wang, H.J.; Wang, J.; Tashiro, S.; Onodera, S.; Ikejima, T. The protective effect of silibinin against mitomycin C-induced intrinsic apoptosis in human melanoma A375-S2 cells. J. Pharmacol. Sci. 2009, 111, 137–146. [Google Scholar] [CrossRef]
- Zhou, B.; Wu, L.J.; Tashiro, S.; Onodera, S.; Uchiumi, F.; Ikejima, T. Silibinin protects rat cardiac myocyte from isoproterenol-induced DNA damage independent on regulation of cell cycle. Biol. Pharm. Bull. 2006, 29, 1900–1905. [Google Scholar] [CrossRef]
- Tripathi, M.; Singh, B.K.; Kakkar, P. Glycyrrhizic acid modulates t-BHP induced apoptosis in primary rat hepatocytes. Food Chem. Toxicol. 2009, 47, 339–347. [Google Scholar] [CrossRef]
- Feher, J.; Lengyel, G. Silymarin in the treatment of chronic liver diseases: Past and future. Orv. Hetil. 2008, 149, 2413–2418. [Google Scholar] [CrossRef]
- Ashkenazi, A.; Dixit, V.M. Death receptors: Signaling and modulation. Science 1998, 281, 1305–1308. [Google Scholar] [CrossRef]
- Osawa, Y.; Banno, Y.; Nagaki, M.; Brenner, D.A.; Naiki, T.; Nozawa, Y.; Nakashima, S.; Moriwaki, H. TNF-alpha-induced sphingosine 1-phosphate inhibits apoptosis through a phosphatidylinositol 3-kinase/Akt pathway in human hepatocytes. J. Immunol. 2001, 167, 173–180. [Google Scholar]
- Kneuer, C.; Lakoma, C.; Honscha, W. Prediction of acute toxicity in HPCT-1E3 hepatocytoma cells with liver-like transport activities. Altern. Lab. Anim. 2007, 35, 411–420. [Google Scholar]
- Honscha, W.; Petzinger, E. Characterization of the bile acid sensitive methotrexate carrier of rat liver cells. Naunyn Schmiedebergs. Arch. Pharmacol. 1999, 359, 411–419. [Google Scholar] [CrossRef]
- Halwachs, S.; Kneuer, C.; Honscha, W. Endogenous expression of liver-specific drug transporters for organic anions in the rat hepatocytoma fusion cell line HPCT-1E3. Eur. J. Cell Biol. 2005, 84, 677–686. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Worner, W.; Schrenk, D. Influence of liver tumor promoters on apoptosis in rat hepatocytes induced by 2-acetylaminofluorene, ultraviolet light, or transforming growth factor beta 1. Cancer Res. 1996, 56, 1272–1278. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Essid, E.; Dernawi, Y.; Petzinger, E. Apoptosis Induction by OTA and TNF-α in Cultured Primary Rat Hepatocytes and Prevention by Silibinin. Toxins 2012, 4, 1139-1156. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins4111139
Essid E, Dernawi Y, Petzinger E. Apoptosis Induction by OTA and TNF-α in Cultured Primary Rat Hepatocytes and Prevention by Silibinin. Toxins. 2012; 4(11):1139-1156. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins4111139
Chicago/Turabian StyleEssid, Ebtisam, Yousef Dernawi, and Ernst Petzinger. 2012. "Apoptosis Induction by OTA and TNF-α in Cultured Primary Rat Hepatocytes and Prevention by Silibinin" Toxins 4, no. 11: 1139-1156. https://0-doi-org.brum.beds.ac.uk/10.3390/toxins4111139



