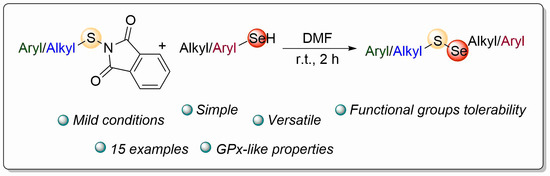
Towards New Catalytic Antioxidants: A Simple and Mild Synthesis of Selenenylsulfides
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Experimental Section
3.2. General Procedure for the Synthesis of Selenenylsulfides 3
3.2.1. Synthesis of (2,4-Dimethoxyphenyl)(phenylselanyl)sulfane 3a
3.2.2. Synthesis of 1-((Phenylselanyl)thio)naphthalen-2-ol 3b
3.2.3. Synthesis of 4-((Phenylselanyl)thio)-2,6-dipropylbenzene-1,3-diol 3c
3.2.4. Synthesis of (2-Chlorocyclohexyl)(phenylselanyl)sulfane 3h
3.2.5. Synthesis of (2,4-Dimethoxyphenyl)(p-tolylselanyl)sulfane 3i
3.2.6. Synthesis of (2-Chlorocyclohexyl)(p-tolylselanyl)sulfane 3j
3.2.7. Synthesis of (2,4-Dimethoxyphenyl)((4-methoxyphenyl)selanyl)sulfane 3k
3.2.8. Synthesis of (2-Chlorocyclohexyl)((4-methoxyphenyl)selanyl)sulfane 3l
3.2.9. Synthesis of (2-Chlorocyclohexyl)((2-methoxyphenyl)selanyl)sulfane 3m
3.3. GPx-Like Catalytic Activity Measurments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Notes and References
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef] [Green Version]
- Lü, J.-M.; Lin, P.H.; Yao, Q.; Chen, C. Chemical and molecular mechanisms of antioxidants: Experimental approaches and model systems. J. Cell. Mol. Med. 2010, 14, 840–860. [Google Scholar] [CrossRef]
- Sarma, B.K.; Mugesh, G. Thiol cofactors for selenoenzymes and their synthetic mimics. Org. Biomol. Chem. 2008, 6, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Mugesh, G.; du Mont, W.W.; Sies, H. Chemistry of biologically important synthetic organoselenium compounds. Chem. Rev. 2001, 101, 2125–2179. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, B.; Koketsu, M. Biologically significant selenium-containing heterocycles. Coord. Chem. Rev. 2011, 255, 2968–2990. [Google Scholar]
- Mónica Álvarez-Pérez, M.; Wesam, A.; Małgorzata, A.M.; Handzlik, J.; Domínguez-Álvarez, E. Selenides and diselenides: A review of their anticancer and chemopreventive activity. Molecules 2018, 23, 628. [Google Scholar] [CrossRef]
- Wessjohann, L.A.; Schneider, A. Synthesis of selenocysteine and its derivatives with an emphasis on selenenylsulfide (-Se-S-) formation. Chem. Biodivers. 2008, 5, 375–388. [Google Scholar] [CrossRef]
- Angeli, A.; Tanini, D.; Capperucci, A.; Malevolti, G.; Turco, F.; Ferraroni, M.; Supuran, C.T. Synthesis of different thio-scaffolds bearing sulfonamide with subnanomolar carbonic anhydrase II and IX inhibitory properties and X-ray investigations for their inhibitory mechanism. Bioorg. Chem. 2018, 81, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Angeli, A.; Tanini, D.; Capperucci, A.; Supuran, C.T. First evaluation of organotellurium derivatives as carbonic anhydrase I, II, IV, VII and IX inhibitors. Bioorg. Chem. 2018, 76, 268–272. [Google Scholar] [CrossRef]
- Reich, H.J.; Hondal, R.J. Why Nature Chose Selenium. ACS Chem. Biol. 2016, 11, 821–841. [Google Scholar] [CrossRef]
- Bhuyan, B.J.; Mugesh, G. Biological and biochemical aspects of selenium compounds. In Organoselenium Chemistry: Synthesis and Reactions; Wirth, T., Ed.; Wiley-VCH Verlag & Co.: Weinheim, Germany, 2012. [Google Scholar]
- Nogueira, C.W.; Zeni, G.; Rocha, J.B.T. Organoselenium and Organotellurium Compounds: Toxicology and Pharmacology. Chem. Rev. 2004, 104, 6255–6286. [Google Scholar] [CrossRef] [PubMed]
- Orian, L.; Toppo, S. Organochalcogen peroxidase mimetics as potential drugs: A long story of a promise still unfulfilled. Free Radic. Biol. Med. 2014, 66, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Mugesh, G.; Singh, H.B. Synthetic organoselenium compounds as antioxidants: Glutathione peroxidase activity. Chem. Soc. Rev. 2000, 29, 347–357. [Google Scholar] [CrossRef]
- Zimmerman, M.T.; Bayse, C.A.; Ramoutar, R.R.; Brumaghim, J.L. Sulfur and selenium antioxidants: Challenging radical scavenging mechanisms and developing structure-activity relationships based on metal binding. J. Inorg. Biochem. 2015, 145, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Haratake, M.; Tachibana, Y.; Emaya, Y.; Yoshida, S.; Fuchigami, T.; Nakayama, M. Synthesis of nanovesicular Glutathione Peroxidase mimics with a selenenylsulfide-bearing lipid. ACS Omega 2016, 1, 58–65. [Google Scholar] [CrossRef]
- Boutureira, O.; Bernardes, G.J.L.; Fernàndez-Gonzàlez, M.; Anthony, D.C.; Davis, B.G. Selenenylsulfide-linked homogeneous glycopeptides and glycoproteins: Synthesis of human “Hepatic Se Metabolite A”. Angew. Chem. Int. Ed. 2012, 57, 1432–1436. [Google Scholar] [CrossRef]
- Cheng, Q.; Sandalova, T.; Lindqvist, Y.; Arnér, E.S.J. Crystal structure and catalysis of the selenoprotein Thioredoxin Reductase 1. J. Biol. Chem. 2009, 284, 3998–4008. [Google Scholar] [CrossRef] [PubMed]
- Engman, L.; Andersson, C.; Morgenstern, R.; Cotgreave, I.A.; Andersson, C.-M.; Hallberg, A. Evidence for a common selenolate intermediate in the glutathione peroxidase-like catalysis of α-(phenylselenenyl) ketones and diphenyl diselenide. Tetrahedron 1994, 50, 2929–2938. [Google Scholar] [CrossRef]
- Venkateswarlu, C.; Gautam, V.; Chandrasekaran, S. Synthesis of mixed glycosyl disulfides/selenosulfides using benzyltriethylammonium tetrathiomolybdate as a sulfur transfer reagent. Carbohydr. Res. 2015, 402, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Ahrika, A.; Auger, J.; Paris, J. Stabilisation of 2-nitrophenyl-selenosulfide, -diselenide and -thioselenide ions in N,N-dimethylacetamide. N. J. Chem. 1999, 23, 679–681. [Google Scholar] [CrossRef]
- Potapov, V.A.; Amosova, S.V.; Petrov, P.A.; Romanenko, L.S.; Keiko, V.V. Exchange reactions of dialkyl dichalcogenides. Sulfur Lett. 1992, 15, 121–126. [Google Scholar]
- Kumar, K.; Kandasamy, K.; Singh, H.B.; Wolmershäuser, G.; Butcher, R.J. Chelate ring size effect on the reactivity of [2-(2-phenyl-5,6-dihydro-4h-1,3-oxazinyl)]lithium and Se···N interactions in low-valent organoselenium compounds: facile isolation of diorganotriselenide. Organometallics 2004, 23, 4199–4208. [Google Scholar] [CrossRef]
- Tanini, D.; Grechi, A.; Dei, S.; Teodori, E.; Capperucci, A. An easy one-step procedure for the synthesis of novel β-functionalised tellurides. Tetrahedron 2017, 73, 5646–5653. [Google Scholar] [CrossRef]
- Capperucci, A.; Salles, C.; Scarpelli, S.; Tanini, D. Selective access to sulfurated and selenated heterocycles by intramolecular cyclization of β-substituted sulfides and selenides. Phosphorus Sulfur Silicon Relat. Elem. 2017, 192, 172–174. [Google Scholar] [CrossRef]
- Tanini, D.; Capperucci, A.; Degl’Innocenti, A. Bis-(trimethylsilyl)selenide in the Selective Synthesis of β-Hydroxy, β-Mercapto, and β-Amino Diorganyl Diselenides and Selenides Through Ring Opening of Strained Heterocycles. Eur. J. Org. Chem. 2015, 357–369. [Google Scholar] [CrossRef]
- Capperucci, A.; Tanini, D. Silicon-assisted synthesis and functionalization of sulfurated and selenated compounds. Phosphorus Sulfur Silicon Relat. Elem. 2015, 190, 1320–1338. [Google Scholar] [CrossRef]
- Tanini, D.; Barchielli, G.; Benelli, F.; Degl’Innocenti, A.; Capperucci, A. Aziridines ring opening by silyl chalcogenides: A stereoselective access to polyfunctionalized molecules as precursor of sulfurated and selenated heterocycles. Phosphorus Sulfur Silicon Relat. Elem. 2015, 190, 1265–1270. [Google Scholar] [CrossRef]
- Capperucci, A.; Tanini, D.; Borgogni, C.; Degl’Innocenti, A. Thiosilane- and organoselenosilane-mediated novel access to 3,7- disubstituted-1,2,5- trithiepanes and −1,2,5-dithiaselenepanes. Heteroat. Chem. 2014, 25, 678–683. [Google Scholar] [CrossRef]
- Viglianisi, C.; Bonardi, C.; Ermini, E.; Capperucci, A.; Menichetti, S.; Tanini, D. Selenosilane-Promoted Selective Mild Transformation of N-Thiophthalimides into Symmetric Disulfides. Synthesis 2019, 51, 1819–1824. [Google Scholar] [CrossRef]
- Tanini, D.; Tiberi, C.; Gellini, C.; Salvi, P.R.; Capperucci, A. A Straightforward access to stable β-functionalized alkyl selenols. Adv. Synth. Catal. 2018, 360, 3367–3375. [Google Scholar] [CrossRef]
- Angeli, A.; Tanini, D.; Nocentini, A.; Capperucci, A.; Ferraroni, M.; Gratteri, P.; Supuran, C.T. Selenols: A new class of carbonic anhydrase inhibitors. Chem. Commun. 2019, 55, 648–651. [Google Scholar] [CrossRef]
- Tanini, D.; Grechi, A.; Ricci, L.; Dei, S.; Teodori, E.; Capperucci, A. Novel functionalized organotellurides with enhanced thiol peroxidase catalytic activity. New J. Chem. 2018, 42, 6077–6083. [Google Scholar] [CrossRef] [Green Version]
- Tanini, D.; D’Esopo, V.; Chen, D.; Barchielli, G.; Capperucci, A. Novel sulfur and selenium-containing antioxidants: Synthesis and evaluation of their GPx-like activity. Phosphorus Sulfur Silicon Relat. Elem. 2017, 192, 166–168. [Google Scholar] [CrossRef]
- Kumakura, F.; Mishra, B.; Priyadarsini, K.I.; Iwaoka, M. A watersoluble cyclic selenide with enhanced glutathione peroxidase-like catalytic activities. Eur. J. Org. Chem. 2010, 3, 440–445. [Google Scholar] [CrossRef]
- T50 is the time required, in seconds, to halve the initial thiol concentration after the addition of H2O2.
- Bhabak, K.P.; Mugesh, G. A simple and efficient strategy to enhance the antioxidant activities of amino-substituted glutathione peroxidase mimics. Chem. Eur. J. 2008, 14, 8640–8651. [Google Scholar] [CrossRef]
- Bailly, F.; Azaroual, N.; Bernier, J.L. Design, synthesis and glutathione peroxidase-like properties of ovothiol-derived diselenides. Bioorg. Med. Chem. 2003, 11, 4623–4630. [Google Scholar] [CrossRef]
- In our hands, S-Se bond exhibited a certain degree of instability. However, selenenylsulfides 3h and 3l proved to be stable for longer times (few days) with respect to those required to perform the GPx-like activity measurements (1 or 2 h). A possible mechanism for the catalytic action of selenylsulfides involves: (i) nucleophilic attack of one of the thiol moieties of DTT onto the Se atom of 3, with the formation of a selenenylsulfide bearing the DTT skeleton; (ii) intramolecular nucleophilic attack of the residual SH moiety onto the S atom of the selenenylsulfide with formation of DTTox and an arylselenol; (iii) H2O2 oxidation of the selenol to the corresponding selenenic acid (ArSeOH); (iv) eventually, ArSeOH undergoes redox reactions with DTT and H2O2, as reported for the catalytic activity of diselenides (see reference [42]). Notably, in this first step (i), likely the rate-determining one, the attack at selenium is both kinetically and thermodynamically favoured over attack at sulfur (see references [40,41,42,43]). On the other hand, the attack at sulfur is favoured in the second step (ii), due to the formation of a six membered cyclic disulfide (DTTox). Further investigations in order to better elucidate the reaction mechanism are currently ongoing.
- Bortoli, M.; Wolters, L.P.; Orian, L.; Bickelhaupt, F.M. Addition-Elimination or Nucleophilic Substitution? Understanding the Energy Profiles for the Reaction of Chalcogenolates with Dichalcogenides. J. Chem. Theory Comput. 2016, 12, 2752–2761. [Google Scholar] [CrossRef]
- Bhabak, K.P.; Mugesh, G. Functional Mimics of Glutathione Peroxidase: Bioinspired Synthetic Antioxidants. Acc. Chem. Res. 2010, 43, 1408–1419. [Google Scholar] [CrossRef]
- Bachrach, S.M.; Demoin, D.W.; Luk, M.; Miller, J.V., Jr. Nucleophilic Attack at Selenium in Diselenides and Selenosulfides. A Computational Study. J. Phys. Chem. A 2004, 108, 4040–4046. [Google Scholar] [CrossRef]
- Capozzi, G.; Gori, L.; Menichetti, S.; Nativi, C. Phthalimidesulfenyl Chloride. Part 4. Addition to Acetylenes and Synthetic Utilization of their Adducts. J. Chem. Soc. Perkin Trans. 1 1992, 1923–1928. [Google Scholar] [CrossRef]
- Capozzi, G.; Nativi, C.; Menichetti, S.; Rosi, A.; Franck, R.W.G. Phthalimidesulfenyl Chloride Part 6. The First Example of an alpha-Oxothione Acting as Heterodiene: Synthesis of 2,3-Dihydro-1,4-Oxathiines. Tetrahedron Lett. 1993, 34, 4253–4257. [Google Scholar] [CrossRef]
- Capozzi, G.; Menichetti, S.; Neri, S.; Skowronska, A. Fluoride Ion Promoted Synthesis of Thiiranes. Synlett 1994, 267–268. [Google Scholar] [CrossRef]
- Menichetti, S.; Aversa, M.C.; Cimino, F.; Contini, A.; Tomaino, A.; Viglianisi, C. Synthesis and ‘double-faced’ antioxidant activity of polyhydroxylated 4-thiaflavans. Org. Biomol. Chem. 2005, 3, 3066–3072. [Google Scholar] [CrossRef]
- Lodovici, M.; Menichetti, S.; Viglianisi, C.; Caldini, S.; Giuliani, E. Polyhydroxylated 4-Thiaflavans as Multipotent Antioxidants: Protective Effect on Oxidative DNA Damage in vitro. Bioorg. Med. Chem. Lett. 2006, 16, 1957–1960. [Google Scholar] [CrossRef] [PubMed]
- Viglianisi, C.; Marcantoni, E.; Carapacchi, V.; Menichetti, S.; Marsili, L. A base-mediated mild sulfenylation of indoles and pyrrole with α-acylthiones. Eur. J. Org. Chem. 2014, 6405–6410. [Google Scholar] [CrossRef]
- Singh, D.; Deobald, A.M.; Camargo, L.R.S.; Tabarelli, G.; Rodrigues, O.E.D.; Braga, A.L. An Efficient One-Pot Synthesis of Symmetrical Diselenides or Ditellurides from Halides with CuO Nanopowder/Se0 or Te0/Base. Org. Lett. 2010, 12, 3288–3291. [Google Scholar] [CrossRef]



| Entry | Solvent | Base | Temperature | Time | 3a:4a:5a a | Conversion (%) a |
|---|---|---|---|---|---|---|
| 1 | CHCl3 | Et3N | 0 °C to r.t. | 2 h b | 36:32:32 | 71 c |
| 2 | CHCl3 | Et3N | −78 °C | 4 h | 34:33:33 | 88 c |
| 3 | CH3CN | Et3N | 0 °C to r.t. | 4 h | 18:41:41 | 52 c |
| 4 | PhMe | Et3N | 0 °C to r.t. | 24 h | 42:29:29 | 67 c |
| 5 | DMF | Et3N | 0 °C to r.t. | 2 h | 32:34:34 | >95 |
| 6 | DMF | Cs2CO3/TBAl | 0 °C to r.t. | 2 h | 44:28:28 | >95 |
| 7 | DMF | - | r.t. | 4 h | 72:14:14 | >95 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tanini, D.; Bonardi, C.; Viglianisi, C.; Capperucci, A.; Menichetti, S. Towards New Catalytic Antioxidants: A Simple and Mild Synthesis of Selenenylsulfides. Catalysts 2019, 9, 333. https://0-doi-org.brum.beds.ac.uk/10.3390/catal9040333
Tanini D, Bonardi C, Viglianisi C, Capperucci A, Menichetti S. Towards New Catalytic Antioxidants: A Simple and Mild Synthesis of Selenenylsulfides. Catalysts. 2019; 9(4):333. https://0-doi-org.brum.beds.ac.uk/10.3390/catal9040333
Chicago/Turabian StyleTanini, Damiano, Chiara Bonardi, Caterina Viglianisi, Antonella Capperucci, and Stefano Menichetti. 2019. "Towards New Catalytic Antioxidants: A Simple and Mild Synthesis of Selenenylsulfides" Catalysts 9, no. 4: 333. https://0-doi-org.brum.beds.ac.uk/10.3390/catal9040333