Isolation of Textile Waste Cellulose Nanofibrillated Fibre Reinforced in Polylactic Acid-Chitin Biodegradable Composite for Green Packaging Application
Abstract
:1. Introduction
2. Materials and Methodology
2.1. Materials
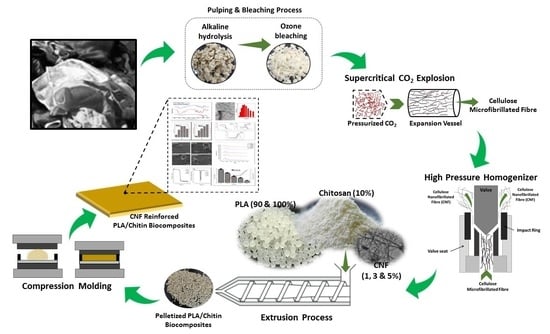
2.2. Textile Waste Cellulose Nanofibrillated Fibre Isolation and Characterisation
2.3. Preparation of Textile Waste Cellulose Nanofibrillated Fibre Reinforced in PLA/Chitin Biocomposite
2.4. Characterisation of Textile Waste Cellulose Nanofibrillated Fibre Reinforced in PLA/Chitin Biocomposite
3. Results and Discussion
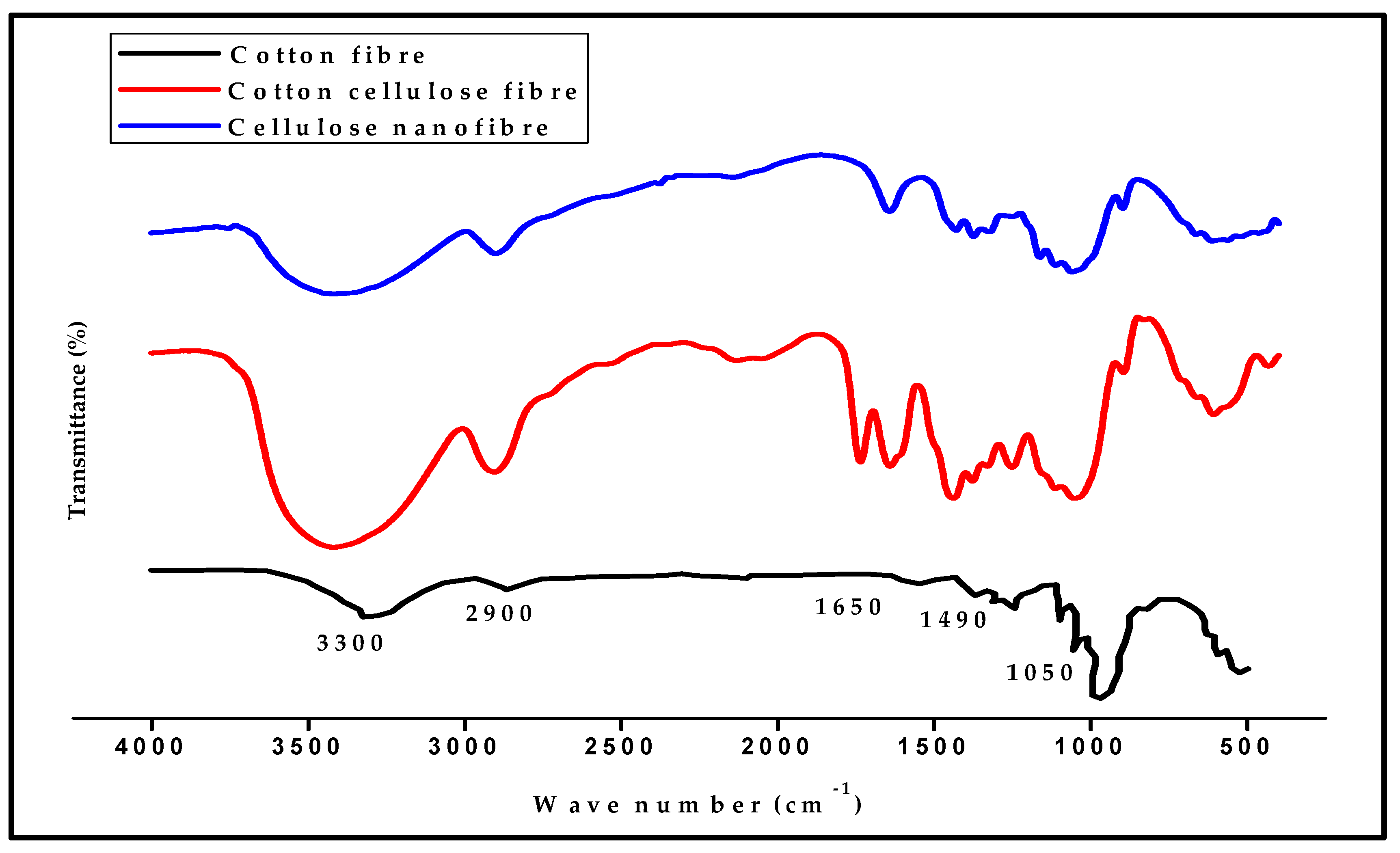
3.1. Properties of Textile Waste Cellulose Nanofibrillated Fibre
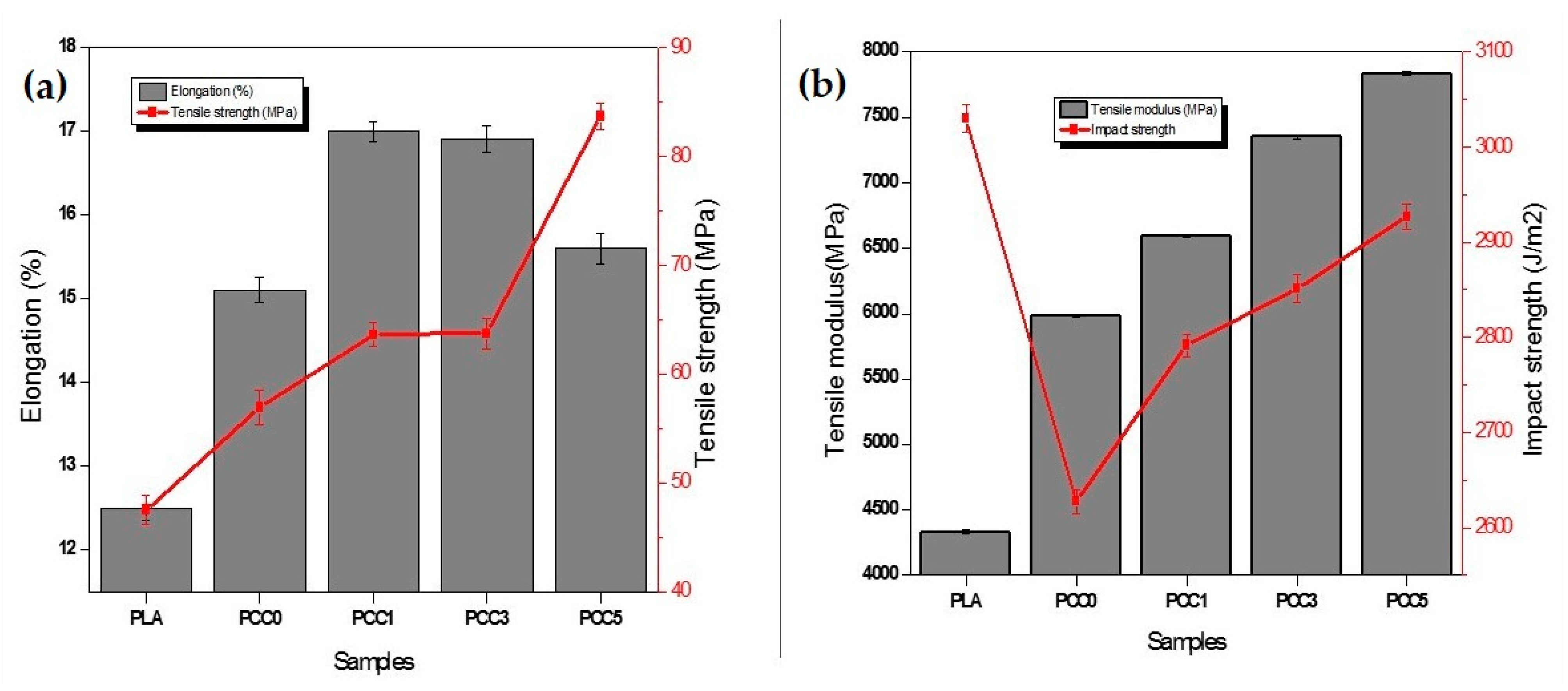
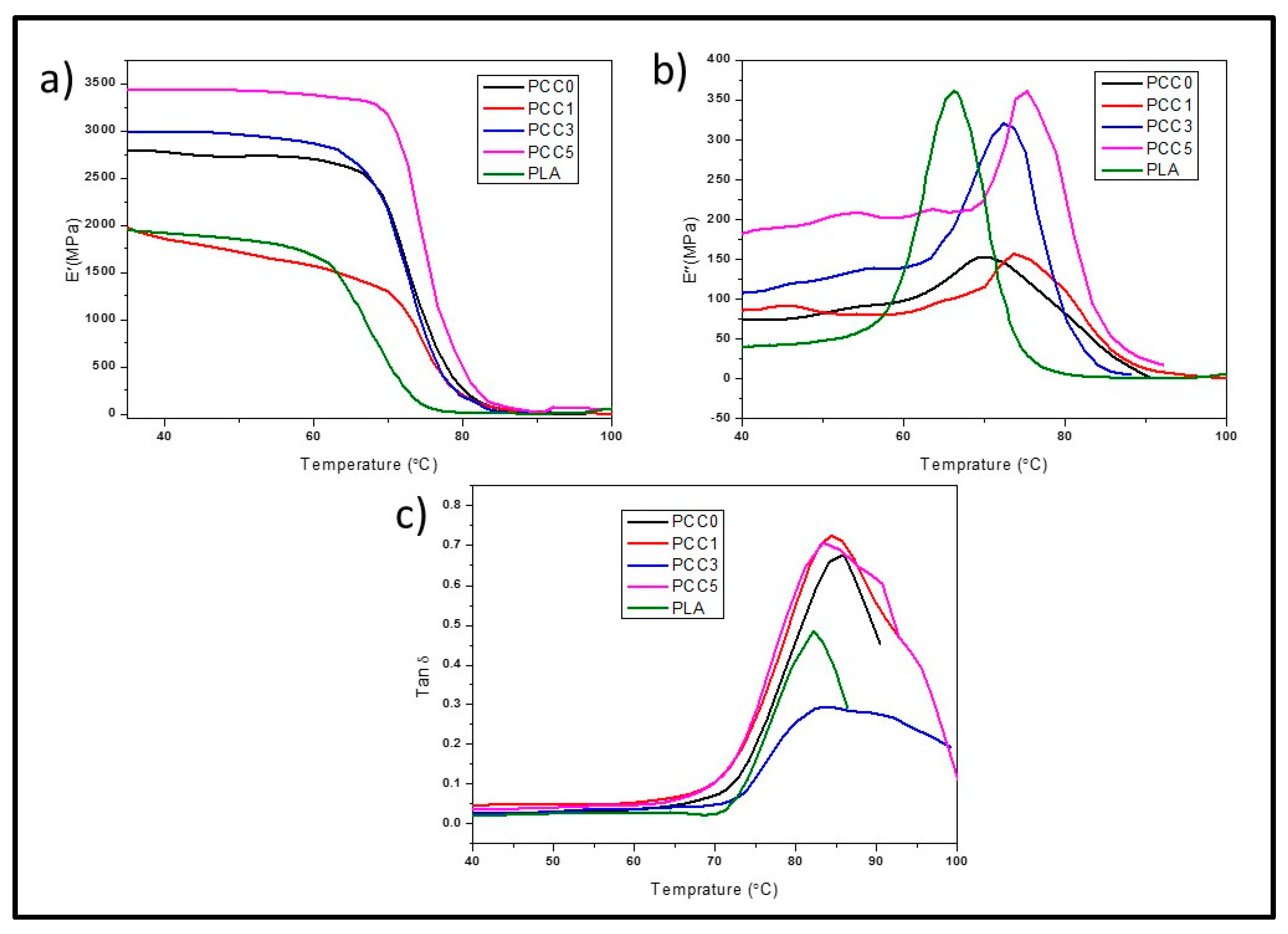
3.2. Characterisation of Textile Waste Cellulose nanofibre Reinforced in Polylactic Acid-Chitin Biocomposite
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Wei, R.; Zimmermann, W. Microbial enzymes for the recycling of recalcitrant petroleum-based plastics: How far are we? Microb. Biotechnol. 2017, 10, 1308–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmed, J.; Varshney, S.K. Polylactides—Chemistry, properties and green packaging technology: A review. Int. J. Food Prop. 2011, 14, 37–58. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Adnan, A.; Yahya, E.B.; Olaiya, N.; Safrida, S.; Hossain, M.; Balakrishnan, V.; Gopakumar, D.A.; Abdullah, C.; Oyekanmi, A. A Review on plant cellulose nanofibre-based aerogels for biomedical applications. Polymers 2020, 12, 1759. [Google Scholar] [CrossRef] [PubMed]
- Alshabanat, M. Morphological, thermal, and biodegradation properties of LLDPE/treated date palm waste composite buried in a soil environment. J. Saudi Chem. Soc. 2019, 23, 355–364. [Google Scholar] [CrossRef]
- Siracusa, V. Microbial degradation of synthetic biopolymers waste. Polymers 2019, 11, 1066. [Google Scholar] [CrossRef] [Green Version]
- Sadasivuni, K.K.; Saha, P.; Adhikari, J.; Deshmukh, K.; Ahamed, M.B.; Cabibihan, J.J. Recent advances in mechanical properties of biopolymer composites: A review. Polym. Compos. 2020, 41, 32–59. [Google Scholar] [CrossRef]
- Hermawan, D.; Lai, T.K.; Jafarzadeh, S.; Gopakumar, D.A.; Hasan, M.; Owolabi, F.T.; Aprilia, N.S.; Rizal, S.; Abdul Khalil, H.P.S. Development of seaweed-based bamboo microcrystalline cellulose films intended for sustainable food packaging applications. BioResources 2019, 14, 3389–3410. [Google Scholar]
- Olaiya, N.; Surya, I.; Oke, P.; Rizal, S.; Sadiku, E.; Ray, S.S.; Farayibi, P.; Hossain, M.S.; Abdul Khalil, H.P.S. Properties and characterization of a PLA–chitin–starch biodegradable polymer composite. Polymers 2019, 11, 1656. [Google Scholar] [CrossRef] [Green Version]
- Rizal, S.; Abdullah, C.; Olaiya, N.; Sri Aprilia, N.; Zein, I.; Surya, I.; Abdul Khalil, H.P.S. Preparation of Palm Oil Ash Nanoparticles: Taguchi Optimization Method by Particle Size Distribution and Morphological Studies. Appl. Sci. 2020, 10, 985. [Google Scholar] [CrossRef] [Green Version]
- Olaiya, N.G.; Nuryawan, A.; Oke, P.K.; Abdul Khalil, H.P.S.; Rizal, S.; Mogaji, P.; Sadiku, E.; Suprakas, S.; Farayibi, P.K.; Ojijo, V. The role of two-step blending in the properties of starch/chitin/polylactic acid biodegradable composites for biomedical applications. Polymers 2020, 12, 592. [Google Scholar] [CrossRef] [Green Version]
- Surya, I.; Olaiya, N.; Rizal, S.; Zein, I.; Sri Aprilia, N.; Hasan, M.; Yahya, E.B.; Sadasivuni, K.; Abdul Khalil, H.P.S. Plasticizer Enhancement on the Miscibility and Thermomechanical Properties of Polylactic Acid-Chitin-Starch Composites. Polymers 2020, 12, 115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siakeng, R.; Jawaid, M.; Ariffin, H.; Sapuan, S.; Asim, M.; Saba, N. Natural fiber reinforced polylactic acid composites: A review. Polym. Compos. 2019, 40, 446–463. [Google Scholar] [CrossRef]
- Sharma, A.; Thakur, M.; Bhattacharya, M.; Mandal, T.; Goswami, S. Commercial application of cellulose nano-composites—A review. Biotechnol. Rep. 2019, 21, e00316. [Google Scholar] [CrossRef] [PubMed]
- Chuayjuljit, S.; Su-uthai, S.; Charuchinda, S. Poly (vinyl chloride) film filled with microcrystalline cellulose prepared from cotton fabric waste: Properties and biodegradability study. Waste Manag. Res. 2010, 28, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yao, Z.; Zhou, J.; Zhang, Y. Reuse of waste cotton cloth for the extraction of cellulose nanocrystals. Carbohydr. Polym. 2017, 157, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Rosson, L.; Wang, X.; Byrne, N. Upcycling of waste textiles into regenerated cellulose fibres: Impact of pretreatments. J. Text. Inst. 2020, 111, 630–638. [Google Scholar] [CrossRef]
- Clarkson, C.M.; El Awad Azrak, S.M.; Chowdhury, R.; Shuvo, S.N.; Snyder, J.; Schueneman, G.; Ortalan, V.; Youngblood, J.P. Melt spinning of cellulose nanofibril/polylactic acid (CNF/PLA) composite fibers for high stiffness. ACS Appl. Polym. Mater. 2018, 1, 160–168. [Google Scholar] [CrossRef]
- Yang, Z.; Li, X.; Si, J.; Cui, Z.; Peng, K. Morphological, mechanical and thermal properties of poly (lactic acid)(PLA)/cellulose nanofibrils (CNF) composites nanofiber for tissue engineering. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2019, 34, 207–215. [Google Scholar] [CrossRef]
- Li, J.; Li, J.; Feng, D.; Zhao, J.; Sun, J.; Li, D. Comparative Study on Properties of Polylactic Acid Nanocomposites with Cellulose and Chitin Nanofibers Extracted from Different Raw Materials. J. Nanomater. 2017, 2017, 7193263. [Google Scholar] [CrossRef]
- Hassan, M.M.; Koyama, K. Thermomechanical and viscoelastic properties of green composites of PLA using chitin micro-particles as fillers. J. Polym. Res. 2020, 27, 27. [Google Scholar] [CrossRef]
- Nasrin, R.; Biswas, S.; Rashid, T.U.; Afrin, S.; Jahan, R.A.; Haque, P.; Rahman, M.M. Preparation of Chitin-PLA laminated composite for implantable application. Bioact. Mater. 2017, 2, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Thambiraj, S.; Shankaran, D.R. Preparation and physicochemical characterization of cellulose nanocrystals from industrial waste cotton. Appl. Surf. Sci. 2017, 412, 405–416. [Google Scholar] [CrossRef]
- Wei, D.W.; Wei, H.; Gauthier, A.C.; Song, J.; Jin, Y.; Xiao, H. Superhydrophobic modification of cellulose and cotton textiles: Methodologies and applications. J. Bioresour. Bioprod. 2020, 5, 1–15. [Google Scholar] [CrossRef]
- Atiqah, M.; Gopakumar, D.A.; FAT, O.; Pottathara, Y.B.; Rizal, S.; Aprilia, N.; Hermawan, D.; Paridah, M.; Thomas, S.; Abdul Khalil, H.P.S. Extraction of Cellulose Nanofibers via Eco-friendly Supercritical Carbon Dioxide Treatment Followed by Mild Acid Hydrolysis and the Fabrication of Cellulose Nanopapers. Polymers 2019, 11, 1813. [Google Scholar] [CrossRef] [Green Version]
- Prabaharan, M.; Rao, J.V. Study on ozone bleaching of cotton fabric–process optimisation, dyeing and finishing properties. Coloration Technol. 2001, 117, 98–103. [Google Scholar] [CrossRef]
- Ghasemi, S.; Behrooz, R.; Ghasemi, I.; Yassar, R.S.; Long, F. Development of nanocellulose-reinforced PLA nanocomposite by using maleated PLA (PLA-g-MA). J. Thermoplast. Compos. Mater. 2018, 31, 1090–1101. [Google Scholar] [CrossRef]
- Safdari, F.; Bagheriasl, D.; Carreau, P.J.; Heuzey, M.C.; Kamal, M.R. Rheological, mechanical, and thermal properties of polylactide/cellulose nanofiber biocomposites. Polym. Compos. 2018, 39, 1752–1762. [Google Scholar] [CrossRef]
- Luo, X.; Wang, X. Preparation and characterization of nanocellulose fibers from NaOH/Urea pretreatment of oil palm fibers. BioResources 2017, 12, 5826–5837. [Google Scholar] [CrossRef]
- Du, H.; Liu, C.; Zhang, Y.; Yu, G.; Si, C.; Li, B. Sustainable preparation and characterization of thermally stable and functional cellulose nanocrystals and nanofibrils via formic acid hydrolysis. J. Bioresour. Bioprod. 2017, 2, 10–15. [Google Scholar]
- Wang, Q.; Yuan, T.; Liu, S.; Yang, G.; Li, W.; Yang, R. Enzymatic activation of dissolving pulp with cationic polyacrylamide to enhance cellulase adsorption. J. Bioresour. Bioprod. 2017, 2, 16–19. [Google Scholar]
- García-Campo, M.J.; Boronat, T.; Quiles-Carrillo, L.; Balart, R.; Montanes, N. Manufacturing and characterization of toughened poly (lactic acid)(PLA) formulations by ternary blends with biopolyesters. Polymers 2018, 10, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- George, K.; Komalan, C.; Kumar, P.; Varughese, K.; Thomas, S. Dynamic Mechanical Analysis of Binary and Ternary Polymer Blends Based on Nylon Copolymer/EPDM Rubber and EPM Grafted Maleic Anhydride Compatibilize. eXPRESS Polym. Lett. 2007, 1, 641–653. [Google Scholar]
- Goertzen, W.K.; Kessler, M. Dynamic mechanical analysis of carbon/epoxy composites for structural pipeline repair. Compos. Part B Eng. 2007, 38, 1–9. [Google Scholar] [CrossRef]
- Wijesena, R.N.; Tissera, N.D.; Abeyratne, C.; Bangamuwa, O.M.; Ludowyke, N.; Dahanayake, D.; Gunasekara, S.; de Silva, N.; de Silva, R.M.; de Silva, K.N. In-situ formation of supramolecular aggregates between chitin nanofibers and silver nanoparticles. Carbohydr. Polym. 2017, 173, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Stark, N.M.; Rowlands, R.E. Effects of wood fiber characteristics on mechanical properties of wood/polypropylene composites. Wood Fiber Sci. 2003, 35, 167–174. [Google Scholar]
- Rizal, S.; Gopakumar, D.A.; Thalib, S.; Huzni, S.; Abdul Khalil, H.P.S. Interfacial compatibility evaluation on the fiber treatment in the Typha fiber reinforced epoxy composites and their effect on the chemical and mechanical properties. Polymers 2018, 10, 1316. [Google Scholar] [CrossRef] [Green Version]
- Jin, E.; Guo, J.; Yang, F.; Zhu, Y.; Song, J.; Jin, Y.; Rojas, O.J. On the polymorphic and morphological changes of cellulose nanocrystals (CNC-I) upon mercerization and conversion to CNC-II. Carbohydr. Polym. 2016, 143, 327–335. [Google Scholar] [CrossRef]
- Martelli-Tosi, M.; Torricillas, M.d.S.; Martins, M.A.; Assis, O.B.G.d.; Tapia-Blácido, D.R. Using commercial enzymes to produce cellulose nanofibers from soybean straw. J. Nanomater. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Liao, Y.; Liu, C.; Coppola, B.; Barra, G.; Di Maio, L.; Incarnato, L.; Lafdi, K. Effect of porosity and crystallinity on 3D printed PLA properties. Polymers 2019, 11, 1487. [Google Scholar] [CrossRef] [Green Version]
- Mihai, M.; Huneault, M.A.; Favis, B.D.; Li, H. Extrusion foaming of semi-crystalline PLA and PLA/thermoplastic starch blends. Macromol. Biosci. 2007, 7, 907–920. [Google Scholar] [CrossRef] [Green Version]
- Kulachenko, A.; Denoyelle, T.; Galland, S.; Lindström, S.B. Elastic properties of cellulose nanopaper. Cellulose 2012, 19, 793–807. [Google Scholar] [CrossRef]
- Tham, C.; Hamid, Z.A.A.; Ahmad, Z.; Ismail, H. Surface engineered Poly (Lactic Acid)(PLA) Microspheres by Chemical Treatment for Drug Delivery System; Trans Tech Publ.: Zurich, Switzerland, 2014; Volume 594. [Google Scholar]
- Mokhena, T.; Sefadi, J.; Sadiku, E.; John, M.; Mochane, M.; Mtibe, A. Thermoplastic processing of PLA/cellulose nanomaterials composites. Polymers 2018, 10, 1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gazzotti, S.; Rampazzo, R.; Hakkarainen, M.; Bussini, D.; Ortenzi, M.A.; Farina, H.; Lesma, G.; Silvani, A. Cellulose nanofibrils as reinforcing agents for PLA-based nanocomposites: An in situ approach. Compos. Sci. Technol. 2019, 171, 94–102. [Google Scholar] [CrossRef]
- Trifol, J.; Plackett, D.; Sillard, C.; Hassager, O.; Daugaard, A.E.; Bras, J.; Szabo, P. A comparison of partially acetylated nanocellulose, nanocrystalline cellulose, and nanoclay as fillers for high-performance polylactide nanocomposites. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Pang, X.; Zhuang, X.; Tang, Z.; Chen, X. Polylactic acid (PLA): Research, development and industrialization. Biotechnol. J. 2010, 5, 1125–1136. [Google Scholar] [CrossRef] [PubMed]



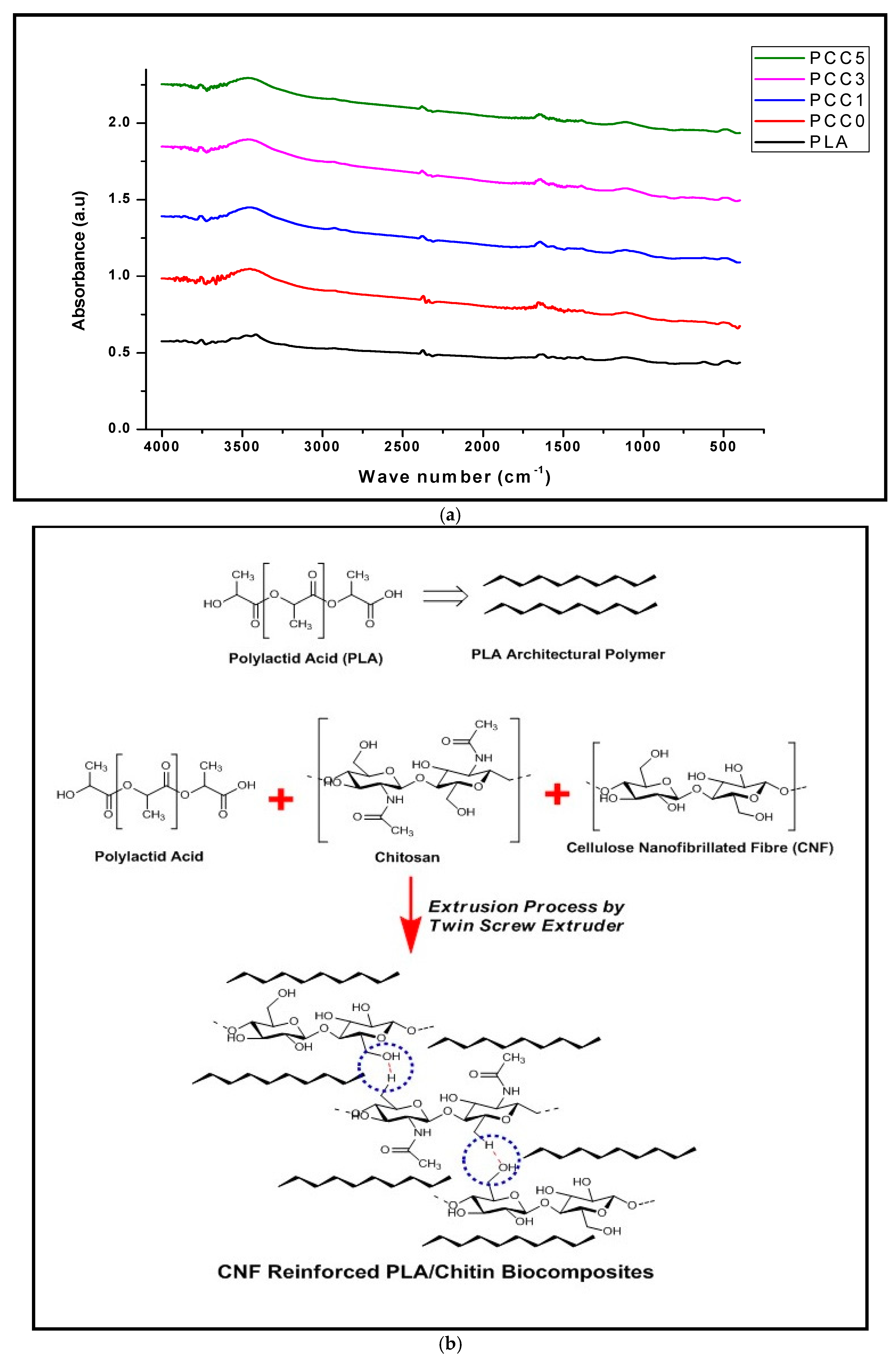

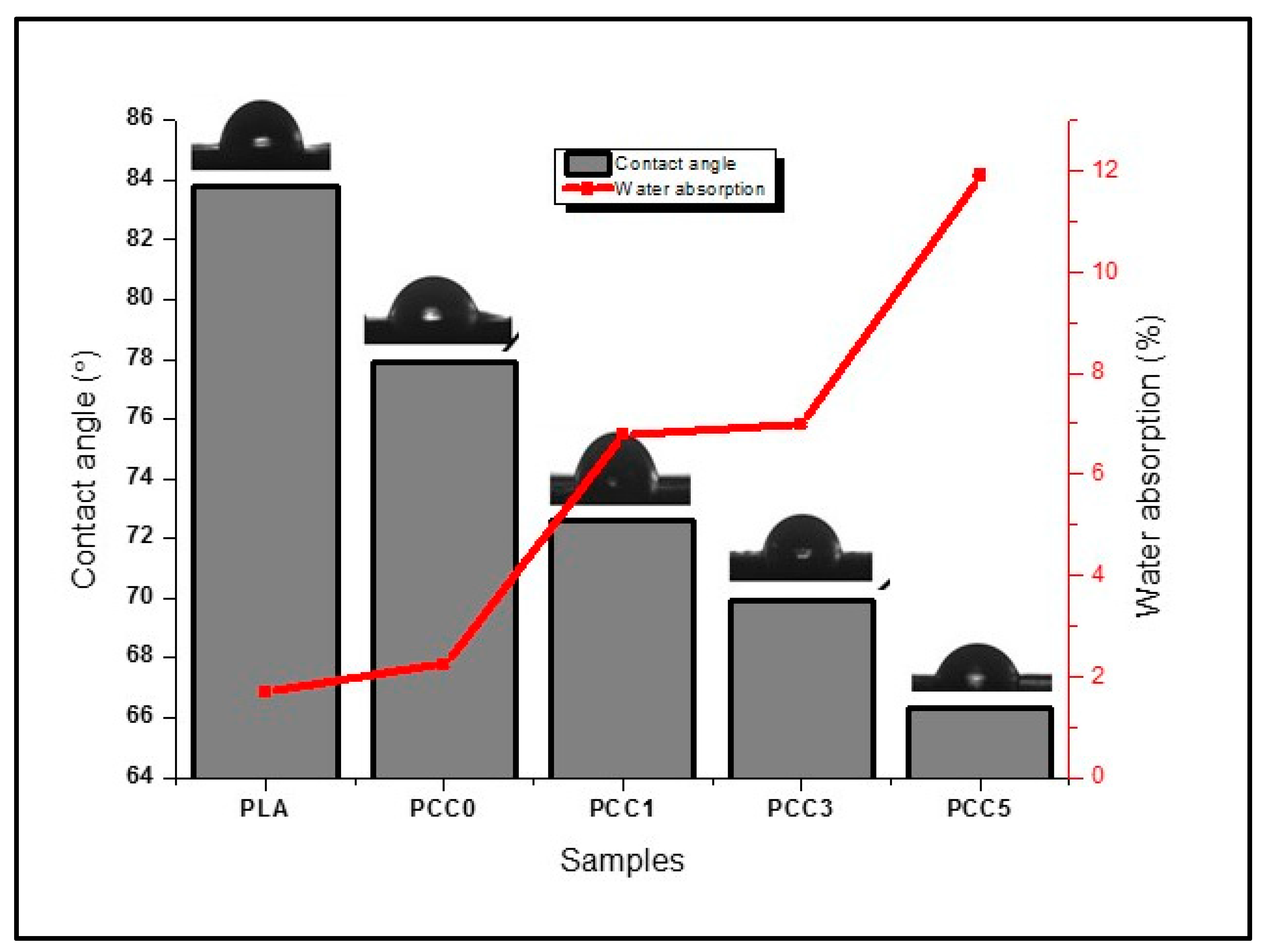
| Sample Name | Polylactic Acid (Wt%) | Chitosan (Wt%) | CNF (Wt%) |
|---|---|---|---|
| PLA | 100 | 0 | 0 |
| PCC0 | 90 | 10 | 0 |
| PCC1 | 90 | 10 | 1 |
| PCC3 | 90 | 10 | 3 |
| PCC5 | 90 | 10 | 5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rizal, S.; Olaiya, F.G.; Saharudin, N.I.; Abdullah, C.K.; N. G., O.; Mohamad Haafiz, M.K.; Yahya, E.B.; Sabaruddin, F.A.; Ikramullah; Khalil H. P. S., A. Isolation of Textile Waste Cellulose Nanofibrillated Fibre Reinforced in Polylactic Acid-Chitin Biodegradable Composite for Green Packaging Application. Polymers 2021, 13, 325. https://0-doi-org.brum.beds.ac.uk/10.3390/polym13030325
Rizal S, Olaiya FG, Saharudin NI, Abdullah CK, N. G. O, Mohamad Haafiz MK, Yahya EB, Sabaruddin FA, Ikramullah, Khalil H. P. S. A. Isolation of Textile Waste Cellulose Nanofibrillated Fibre Reinforced in Polylactic Acid-Chitin Biodegradable Composite for Green Packaging Application. Polymers. 2021; 13(3):325. https://0-doi-org.brum.beds.ac.uk/10.3390/polym13030325
Chicago/Turabian StyleRizal, Samsul, Funmilayo G. Olaiya, N. I. Saharudin, C. K. Abdullah, Olaiya N. G., M. K. Mohamad Haafiz, Esam Bashir Yahya, F. A. Sabaruddin, Ikramullah, and Abdul Khalil H. P. S. 2021. "Isolation of Textile Waste Cellulose Nanofibrillated Fibre Reinforced in Polylactic Acid-Chitin Biodegradable Composite for Green Packaging Application" Polymers 13, no. 3: 325. https://0-doi-org.brum.beds.ac.uk/10.3390/polym13030325