Genome-Wide Association Study on Adiponectin-Mediated Suppression of HDL-C Levels in Taiwanese Individuals Identifies Functional Haplotypes in CDH13
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. GWAS Population
2.3. Genomic DNA Extraction and Genotyping
2.4. Laboratory Examinations and Assays
2.5. Mediation Analysis
2.6. Construct
2.7. Transfection and Functional Reporter Assay
2.8. Statistical Analysis
3. Results
3.1. Subsection
3.1.1. Characteristics of the TWB Participants
3.1.2. Genome Wide Association Study for HDL-C in the TWB Participants
3.1.3. Mediation Analysis of the Association between HDL-C and rs4783244 in the TWB Population
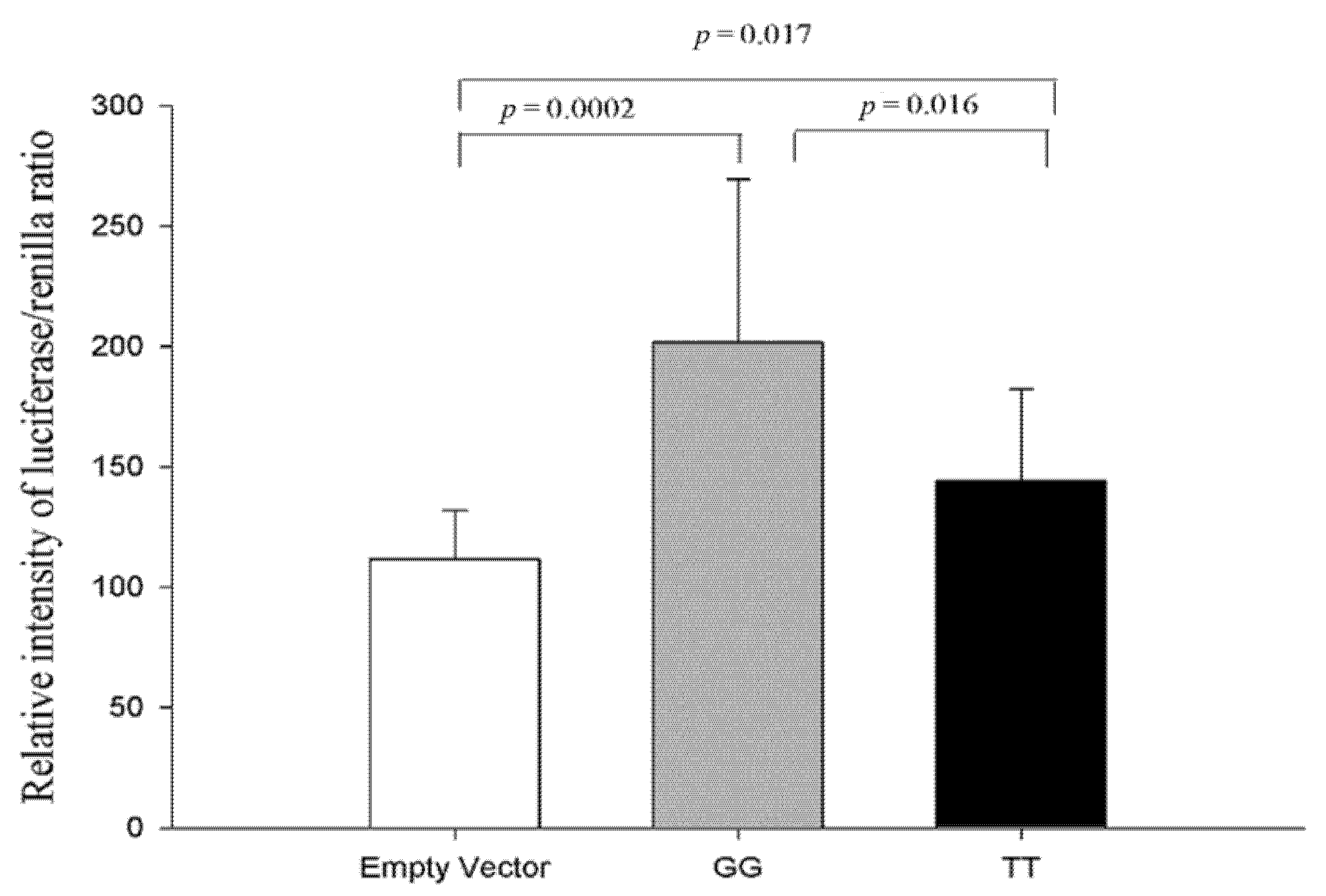
3.1.4. Functional Analysis of Two Haplotypes in the CDH13 Proximal Intron 1 Promoter Region
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Yamauchi, T.; Kamon, J.; Ito, Y.; Tsuchida, A.; Yokomizo, T.; Kita, S.; Sugiyama, T.; Miyagishi, M.; Hara, K.; Tsunoda, M.; et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 2003, 423, 762–769. [Google Scholar] [CrossRef]
- Matsuzawa, Y.; Funahashi, T.; Kihara, S.; Shimomura, I. Adiponectin and Metabolic Syndrome. Arter. Thromb. Vasc. Biol. 2004, 24, 29–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, P.H.; Jiang, Y.-D.; Chen, W.J.; Chang, C.-C.; Lee, T.-C.; Sun, S.; Chuang, L.-M. Genetic and environmental influences on adiponectin, leptin, and BMI among adolescents in Taiwan: A multivariate twin/sibling analysis. win Res. Hum. Genet. 2008, 11, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Menzaghi, C.; Trischitta, V.; Doria, A. Genetic influences of adiponectin on insulin resistance, type 2 diabetes, and cardiovascular disease. Diabetes 2007, 56, 1198–1209. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.S.; Chuang, L.M. Human genetics of adiponectin in the metabolic syndrome. J. Mol. Med. 2005, 84, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Narkiewicz, K.; De Toni, R.; Aldighieri, E.; Williams, C.; Rossi, G.P. Heritability of plasma adiponectin levels and body mass index in twins. J. Clin. Endocrinol. Metab. 2007, 92, 3082–3088. [Google Scholar] [CrossRef] [Green Version]
- Henneman, P.; Aulchenko, Y.; Frants, R.; Zorkoltseva, I.; Zillikens, M.; Frölich, M.; Oostra, B.; van Dijk, K.W.; Van Duijn, C.M. Genetic architecture of plasma adiponectin overlaps with the genetics of metabolic syndrome-related traits. Diabetes Care 2010, 33, 908–913. [Google Scholar] [CrossRef] [Green Version]
- Jee, S.H.; Sull, J.W.; Lee, J.-E.; Shin, C.; Park, J.; Kimm, H.; Cho, E.-Y.; Shin, E.-S.; Yun, J.E.; Park, J.W.; et al. Adiponectin concentrations: A genome-wide association study. Am. J. Hum. Genet. 2010, 87, 545–552. [Google Scholar] [CrossRef] [Green Version]
- Chung, C.M.; Lin, T.-H.; Chen, J.-W.; Leu, H.-B.; Yang, H.-C.; Ho, H.-Y.; Ting, C.-T.; Sheu, S.-H.; Tsai, W.-C.; Lin, S.-J.; et al. A genome-wide association study reveals a quantitative trait locus of adiponectin on CDH13 that predicts cardiometabolic outcomes. Diabetes 2011, 60, 2417–2423. [Google Scholar] [CrossRef] [Green Version]
- Gao, H.; Kim, Y.-M.; Chen, P.; Igase, M.; Kawamoto, R.; Kim, M.K.; Kohara, K.; Lee, J.; Miki, T.; Ong, R.T.-H.; et al. Genetic variation in CDH13 is associated with lower plasma adiponectin levels but greater adiponectin sensitivity in East Asian populations. Diabetes 2013, 62, 4277–4283. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Gao, H.; Li, H.; Tabara, Y.; Nakatochi, M.; Chiu, Y.-F.; Park, E.J.; Wen, W.; Adair, L.S.; Borja, J.B.; et al. A meta-analysis of genome-wide association studies for adiponectin levels in East Asians identifies a novel locus near WDR11-FGFR2. Hum. Mol. Genet. 2014, 23, 1108–1119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morisaki, H.; Yamanaka, I.; Iwai, N.; Miyamoto, Y.; Kokubo, Y.; Okamura, T.; Okayama, A.; Morisaki, T. CDH13 gene coding T-cadherin influences variations in plasma adiponectin levels in the Japanese population. Hum. Mutat. 2012, 33, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Spracklen, C.N.; Iyengar, A.K.; Vadlamudi, S.; Raulerson, C.K.; Jackson, A.U.; Brotman, S.M.; Wu, Y.; Cannon, M.E.; Davis, J.P.; Crain, A.T.; et al. Adiponectin GWAS loci harboring extensive allelic heterogeneity exhibit distinct molecular consequences. PLoS Genet. 2020, 16, e1009019. [Google Scholar] [CrossRef]
- Weissglas-Volkov, D.; Pajukanta, P. Genetic causes of high and low serum HDL-cholesterol. J. Lipid Res. 2010, 51, 2032–2057. [Google Scholar] [CrossRef] [Green Version]
- Khetarpal, S.A.; Rader, D.J. Genetics of lipid traits: Genome-wide approaches yield new biology and clues to causality in coronary artery disease. Biochim. Biophys. Acta 2014, 1842, 2010–2020. [Google Scholar] [CrossRef] [Green Version]
- Willer, C.J.; Schmidt, E.M.; Sengupta, S.; Peloso, G.M.; Gustafsson, S.; Kanoni, S.; Ganna, A.; Chen, J.; Buchkovich, M.L.; Mora, S.; et al. Discovery and refinement of loci associated with lipid levels. Nat. Genet. 2013, 45, 1274–1283. [Google Scholar] [PubMed] [Green Version]
- Brunham, L.R.; Hayden, M.R. Human genetics of HDL: Insight into particle metabolism and function. Prog. Lipid Res. 2015, 58, 14–25. [Google Scholar] [CrossRef] [Green Version]
- Dron, J.; Hegele, R.A. Genetics of Lipid and Lipoprotein Disorders and Traits. Curr. Genet. Med. Rep. 2016, 4, 130–141. [Google Scholar] [CrossRef] [Green Version]
- Alwin, D.F.; Hauser, R.M. The Decomposition of Effects in Path Analysis. Am. Sociol. Rev. 1975, 40, 37. [Google Scholar] [CrossRef] [Green Version]
- Baron, R.M.; Kenny, D.A. The moderator-mediator variable distinction in social psychological research: Conceptual, strategic, and statistical considerations. J. Pers. Soc. Psychol. 1986, 51, 1173–1182. [Google Scholar] [CrossRef] [PubMed]
- James, L.R.; Brett, J.M. Mediators, moderators, and tests for mediation. J. Appl. Psychol. 1984, 69, 307. [Google Scholar] [CrossRef]
- Sobel, M.E. Effect analysis and causation in linear structural equation models. Psychometrika 1990, 55, 495–515. [Google Scholar] [CrossRef]
- Tzelgov, J.; Henik, A. Suppression situations in psychological research: Definitions, implications, and applications. Psychol. Bull. 1991, 109, 524. [Google Scholar] [CrossRef]
- Teng, M.-S.; Hsu, L.A.; Wu, S.; Chou, H.H.; Chang, C.J.; Sun, Y.Z.; Juan, S.H.; Ko, Y.L. Mediation analysis reveals a sex-dependent association between ABO gene variants and TG/HDL-C ratio that is suppressed by sE-selectin level. Atherosclerosis 2013, 228, 406–412. [Google Scholar] [CrossRef]
- Teng, M.-S.; Hsu, L.-A.; Wu, S.; Sun, Y.-C.; Juan, S.-H.; Ko, Y.-L. Association of CDH13 genotypes/haplotypes with circulating adiponectin levels, metabolic syndrome, and related metabolic phenotypes: The role of the suppression effect. PLoS ONE 2015, 10, e0122664. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.-H.; Yang, J.-H.; Chiang, C.; Hsiung, C.-N.; Wu, P.-E.; Chang, L.-C.; Chu, H.-W.; Chang, J.; Yuan-Tsong, C.; Yang, S.-L.; et al. Population structure of Han Chinese in the modern Taiwanese population based on 10,000 participants in the Taiwan Biobank project. Hum. Mol. Genet. 2016, 25, 5321–5331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacKinnon, D.P. Analysis of mediating variables in prevention and intervention research. NIDA Res. Monogr. 1994, 139, 127–153. [Google Scholar] [PubMed]
- Sobel, M.E. Asymptotic confidence intervals for indirect effects in structural equation models. Sociol. Methodol. 1982, 13, 290–312. [Google Scholar] [CrossRef]
- Sobel, M.E. Direct and indirect effects in linear structural equation models. Sociol. Methods Res. 1987, 16, 155–176. [Google Scholar] [CrossRef]
- MacKinnon, D.P.; Krull, J.L.; Lockwood, C.M. Equivalence of the mediation, confounding and suppression effect. Prev. Sci. 2000, 1, 173–181. [Google Scholar] [CrossRef]
- Liao, Y.-H.; Er, L.-K.; Wu, S.; Ko, Y.-L.; Teng, M.-S. Functional haplotype of LIPC induces triglyceride-mediated suppression of HDL-C levels according to genome-wide association studies. Genes 2021, 12, 148. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Waterworth, D.M.; Stirnadel, H.A.; Pollin, T.I.; Barter, P.J.; Kesäniemi, Y.A.; Mahley, R.W.; McPherson, R.; Waeber, G.; Bersot, T.P.; et al. Genome-wide linkage and association analyses to identify genes influencing adiponectin levels: The GEMS Stud. Obesity 2009, 17, 737–744. [Google Scholar] [CrossRef] [Green Version]
- Heid, I.M.; Henneman, P.; Hicks, A.; Coassin, S.; Winkler, T.; Aulchenko, Y.S.; Fuchsberger, C.; Song, K.; Hivert, M.-F.; Waterworth, D.M.; et al. Clear detection of ADIPOQ locus as the major gene for plasma adiponectin: Results of genome-wide association analyses including 4659 European individuals. Atherosclerosis 2010, 208, 412–420. [Google Scholar] [CrossRef] [Green Version]
- Putku, M.; Kals, M.; Inno, R.; Kasela, S.; Org, E.; Kožich, V.; Milani, L.; Laan, M. CDH13 promoter SNPs with pleiotropic effect on cardiometabolic parameters represent methylation QTLs. Hum. Genet. 2015, 134, 291–303. [Google Scholar] [CrossRef] [Green Version]
- Kadowaki, T.; Sekikawa, A.; Okamura, T.; Takamiya, T.; Kashiwagi, A.; Zaky, W.R.; Maegawa, H.; El-Saed, A.; Nakamura, Y.; Evans, R.W.; et al. Higher levels of adiponectin in American than in Japanese men despite obesity. Metabolism 2006, 55, 1561–1563. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, T.; Nio, Y.; Maki, T.; Kobayashi, M.; Takazawa, T.; Iwabu, M.; Okada-Iwabu, M.; Kawamoto, S.; Kubota, N.; Kubota, T.; et al. Targeted disruption of AdipoR1 and AdipoR2 causes abrogation of adiponectin binding and metabolic actions. Nat. Med. 2007, 13, 332–339. [Google Scholar] [CrossRef]
- Ouchi, N.; Kihara, S.; Arita, Y.; Okamoto, Y.; Maeda, K.; Kuriyama, H.; Hotta, K.; Nishida, M.; Takahashi, M.; Muraguchi, M.; et al. Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-κB signaling through a cAMP-dependent pathway. Circulation 2000, 102, 1296–1301. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-H.; Klein, R.L.; El-Shewy, H.M.; Luttrell, D.K.; Luttrell, L.M. The adiponectin receptors AdipoR1 and AdipoR2 activate ERK1/2 through a Src/Ras-dependent pathway and stimulate cell growth. Biochemistry 2008, 47, 11682–11692. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Total | Men | Women | p value | |
|---|---|---|---|---|
| Number | 2199 | 986 | 1213 | |
| Age (years) | 48.1 ± 10.8 | 48.6 ± 11.1 | 48.2 ± 10.7 | 0.444 |
| BMI (kg/m2) | 24.2 ± 3.5 | 25.1 ± 3.4 | 23.4 ± 3.5 | <0.001 |
| Waist circumference (cm) | 84.8 ± 9.4 | 87.4 ± 9.2 | 80.4 ± 9.2 | <0.001 |
| Waist-to-hip ratio | 0.9 ± 0.2 | 0.90 ± 0.2 | 0.84 ± 0.1 | <0.001 |
| Current smokers (%) | 396 (18.0%) | 329 (33.4%) | 67 (5.5%) | <0.001 |
| Total cholesterol (mg/dL) | 195.1 ± 35.9 | 194.6 ± 35.4 | 195.6 ± 36.5 | 0.500 |
| LDL-C (mg/dL) | 122.5 ± 32.9 | 124.8 ± 32. | 120.5 ± 32.7 | 0.002 |
| HDL-C (mg/dL) | 55.1 ± 13.4 | 49.7 ± 11.4 | 59.4 ± 13.3 | <0.001 |
| TG (mg/dL) | 117.9 ± 91.9 | 138.9 ± 105.8 | 100.8 ± 74.7 | <0.001 |
| Adiponectin (mg/L) | 3.5 ± 1.9 | 2.88 ± 1.45 | 4.01 ± 1.97 | <0.001 |
| CHR | Gene | SNP | Chr. Location | Minor Allele | p | p * |
|---|---|---|---|---|---|---|
| 16 | CDH13 | rs4783244 | 82,662,268 | T | 0.8439 | 1.05 × 10−7 |
| 16 | CDH13 | rs28597883 | 82,647,566 | C | 0.4147 | 7.66 × 10−6 |
| 16 | CDH13 | rs3865188 | 82,650,717 | T | 0.3587 | 7.84 × 10−6 |
| 16 | CDH13 | rs12444222 | 82,672,920 | G | 0.1005 | 1.40 × 10−5 |
| 16 | CDH13 | rs4783248 | 82,674,055 | G | 0.11 | 2.29 × 10−5 |
| 16 | CDH13 | rs12446731 | 82,676,142 | A | 0.1179 | 3.28 × 10−5 |
| 16 | CDH13 | rs12925602 | 82,655,901 | A | 0.1391 | 5.01 × 10−5 |
| 16 | CDH13 | rs9652670 | 82,671,477 | G | 0.1597 | 7.10 × 10−5 |
| 16 | CDH13 | rs3865186 | 82,646,972 | A | 0.3461 | 7.90 × 10−5 |
| 16 | CDH13 | rs9940180 | 82,653,444 | T | 0.9354 | 8.00 × 10−5 |
| rs4783244 | ||
|---|---|---|
| Criterion 1 | α | |
| regression coefficient | −0.079 | |
| Standard error | 0.006 | |
| p value | 3.68 × 10−37 | |
| Criterion 2 | β | |
| regression coefficient | 0.21 | |
| Standard error | 0.01 | |
| #p value | 1.12 × 10−93 | |
| γ′ | ||
| regression coefficient | 0.015 | |
| Standard error | 0.003 | |
| * p value | 7.17 × 10−8 | |
| Criterion 3 | αβ + γ′ | |
| regression coefficient | −0.001 | |
| Standard error | 0.003 | |
| * p value | 0.731 | |
| Criterion 4 | αβ | |
| regression coefficient | −0.017 | |
| Standard error | 0.001 | |
| p value (Sobel test) | <10−8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Er, L.-K.; Wu, S.; Cheng, T.; Ko, Y.-L.; Teng, M.-S. Genome-Wide Association Study on Adiponectin-Mediated Suppression of HDL-C Levels in Taiwanese Individuals Identifies Functional Haplotypes in CDH13. Genes 2021, 12, 1582. https://0-doi-org.brum.beds.ac.uk/10.3390/genes12101582
Er L-K, Wu S, Cheng T, Ko Y-L, Teng M-S. Genome-Wide Association Study on Adiponectin-Mediated Suppression of HDL-C Levels in Taiwanese Individuals Identifies Functional Haplotypes in CDH13. Genes. 2021; 12(10):1582. https://0-doi-org.brum.beds.ac.uk/10.3390/genes12101582
Chicago/Turabian StyleEr, Leay-Kiaw, Semon Wu, Tzuyu Cheng, Yu-Lin Ko, and Ming-Sheng Teng. 2021. "Genome-Wide Association Study on Adiponectin-Mediated Suppression of HDL-C Levels in Taiwanese Individuals Identifies Functional Haplotypes in CDH13" Genes 12, no. 10: 1582. https://0-doi-org.brum.beds.ac.uk/10.3390/genes12101582






