Novel Structural Variation and Evolutionary Characteristics of Chloroplast tRNA in Gossypium Plants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Identification of tRNAs
2.2. Structural Analysis of tRNAs
2.3. Sequence Alignment
2.4. Phylogenetic Tree Construction
2.5. Analysis of Disparity Index
2.6. Transition/Transversion Analysis
2.7. Evolutionary Analysis of Single Nucleotide Polymorphisms
2.8. Calculation of Mutation Rate
2.9. Duplication/Loss Analysis of tRNA Genes
3. Results
3.1. Basic Characteristics of Cotton Chloroplast tRNAs
3.2. Diversification of tRNA Structure
3.3. Chloroplast tRNA Contained Introns
3.4. Chloroplast tRNAs with Non-Typical Features
3.5. Cotton Chloroplast tRNAs Were Derived from Several Evolutionary Ancestors
3.6. Transition/Transversion of tRNAs
3.7. Evolutionary Characteristics of Single Nucleotide Polymorphisms
3.8. Mutation Rate of Chloroplast Genome
3.9. tRNA Duplication/Loss Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Arnon, D.I. The chloroplast as a complete photosynthetic unit. Science 1955, 122, 9–16. [Google Scholar] [CrossRef]
- Roughan, P.G.; Holland, R.; Slack, C.R. On the control of long-chain-fatty acid synthesis in isolated intact spinach (Spinacia oleracea) chloroplasts. Biochem. J. 1979, 184, 193–202. [Google Scholar] [CrossRef] [Green Version]
- Neuhaus, H.E.; Emes, M.J. Nonphotosyntheticmetabolism inplastids. Annu. Rev. Plant Biol. 2000, 51, 111–140. [Google Scholar] [CrossRef] [PubMed]
- Spetea, C.; Hundal, T.; Lundin, B.; Heddad, M.; Adamska, I.; Andersson, B. Multiple evidence for nucleotide metabolism in the chloroplast thylakoid lumen. Proc. Natl. Acad. Sci. USA 2004, 101, 1409–1414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holsinger, K.E.; Soltis, P.S.; Soltis, D.E.; Doyle, J.J. Molecular systematics of plants. Syst. Bot. 1993, 18, 539. [Google Scholar] [CrossRef]
- Gross, J.; Meurer, J.; Bhattacharya, D. Evidence of a chimeric genome in the cyanobacterial ancestor of plastids. BMC Evol. Biol. 2008, 8, 117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, C.-P.; Wu, C.-S.; Huang, Y.-Y.; Chaw, S.-M. The complete chloroplast genome of ginkgo biloba reveals the mechanism of inverted repeat contraction. Genome Biol. Evol. 2012, 4, 374–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, P.A.T.; Kim, J.S.; Kim, J.-H. The complete chloroplast genome of colchicine plants (Colchicum autumnale L. and Gloriosa superba L.) and its application for identifying the genus. Planta 2015, 242, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Ruf, S.; Karcher, D.; Bock, R. Determining the transgene containment level provided by chloroplast transformation. Proc. Natl. Acad. Sci. USA 2007, 104, 6998–7002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, L.-Y.; Tang, T.-Y.; Li, F.-W.; Su, H.-J.; Chiou, W.-L.; Huang, Y.-M.; Wang, C.-N. Organelle genome inheritance in Deparia Ferns (Athyriaceae, Aspleniineae, Polypodiales). Front. Plant Sci. 2018, 9, 486. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.-J.; Cheng, C.-L.; Chang, C.-C.; Wu, C.-L.; Su, T.-M.; Chaw, S.-M. Dynamics and evolution of the inverted repeat-large single copy junctions in the chloroplast genomes of monocots. BMC Evol. Biol. 2008, 8, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fish, L.E.; Jagendorf, A.T. High rates of potein synthesis by isolated chloroplasts. Plant Physiol. 1982, 70, 1107–1114. [Google Scholar] [CrossRef] [Green Version]
- Shinozaki, K.; Hayashida, N.; Sugiura, M. Nicotiana chloroplast genes for components of the photosynthetic apparatus. Photosynth. Res. 1988, 18, 7–31. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, S.; Ignatova, Z. Emerging roles of tRNA in adaptive translation, signalling dynamics and disease. Nat. Rev. Genet. 2014, 16, 98–112. [Google Scholar] [CrossRef]
- Brumfield, R.T.; Beerli, P.; Nickerson, D.A.; Edwards, S.V. Single nucleotide polymorphisms (SNPs) as markers in phylogeography. Trends Ecol. Evol. 2003, 18, 249–256. [Google Scholar] [CrossRef]
- Rafalski, A. Applications of single nucleotide polymorphisms in crop genetics. Curr. Opin. Plant Biol. 2002, 5, 94–100. [Google Scholar] [CrossRef]
- Holley, R.W.; Apgar, J.; Everett, G.A.; Madison, J.T.; Marquisee, M.; Merrill, S.H.; Penswick, J.R.; Zamir, A. Structure of a ribonucleic acid. Science 1965, 147, 1462–1465. [Google Scholar] [CrossRef] [PubMed]
- Nicoghosian, K.; Bigras, M.; Sankoff, D.; Cedergren, R. Archetypical features in tRNA families. J. Mol. Evol. 1987, 26, 341–346. [Google Scholar] [CrossRef]
- Giegé, R.; Puglisi, J.D.; Florentz, C. tRNA structure and aminoacylation efficiency. Prog. Nucleic Acid Res. Mol. Biol. 1993, 45, 129–206. [Google Scholar] [CrossRef] [PubMed]
- De Pouplana, L.R.; Dedon, P.C. More than an adaptor molecule: The emerging role of tRNA in cell signaling and disease. FEBS Lett. 2014, 588, 4267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blee, E.; Joyard, J. Envelope membranes from spinach chloroplasts are a site of metabolism of fatty acid hydroperoxides. Plant Physiol. 1996, 110, 445–454. [Google Scholar] [CrossRef] [Green Version]
- Kanai, A. Disrupted tRNA genes and tRNA fragments: A perspective on tRNA gene evolution. Life 2015, 5, 321–331. [Google Scholar] [CrossRef]
- Anderson, P.; Ivanov, P. tRNA fragments in human health and disease. FEBS Lett. 2014, 588, 4297–4304. [Google Scholar] [CrossRef] [Green Version]
- Kashdan, M.A.; Dudock, B.S. The gene for a spinach chloroplast isoleucine tRNA has a methionine anticodon. J. Biol. Chem. 1982, 257, 11191–11194. [Google Scholar] [CrossRef]
- Yasukawa, T.; Kirino, Y.; Ishii, N.; Holt, I.; Jacobs, H.T.; Makifuchi, T.; Fukuhara, N.; Ohta, S.; Suzuki, T.; Watanabe, K. Wobble modification deficiency in mutant tRNAs in patients with mitochondrial diseases. FEBS Lett. 2005, 579, 2948–2952. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morscher, R.J.; Ducker, G.S.; Li, S.H.-J.; Mayer, J.A.; Gitai, Z.; Sperl, W.; Rabinowitz, J.D. Mitochondrial translation requires folate-dependent tRNA methylation. Nat. Cell Biol. 2018, 554, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Mohanta, T.K.; Syed, A.S.; Ameen, F.; Bae, H. Novel genomic and evolutionary perspective of cyanobacterial tRNAs. Front. Genet. 2017, 8, 200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohanta, T.K.; Bae, H. Analyses of genomic tRNA reveal presence of novel tRNAs in Oryza sativa. Front. Genet. 2017, 8, 90. [Google Scholar] [CrossRef] [Green Version]
- Mohanta, T.K.; Khan, A.L.; Hashem, A.; Allah, E.F.A.; Yadav, D.; Al-Harrasi, A. Genomic and evolutionary aspects of chloroplast tRNA in monocot plants. BMC Plant Biol. 2019, 19, 39. [Google Scholar] [CrossRef]
- Fryxell, P.A. A classification of Gossypium L. (Malvaceae). Taxon 1969, 18, 585–591. [Google Scholar] [CrossRef]
- Beasley, J.O. The origin of American Tetraploid Gossypium species. Am. Nat. 1940, 74, 285–286. [Google Scholar] [CrossRef]
- Li, F.; Fan, G.; Lu, C.; Xiao, G.; Zou, C.; Kohel, R.J.; Ma, Z.; Shang, H.; Ma, X.; Wu, J.; et al. Genome sequence of cultivated Upland cotton (Gossypium hirsutum TM-1) provides insights into genome evolution. Nat. Biotechnol. 2015, 33, 524–530. [Google Scholar] [CrossRef] [Green Version]
- Zhang, T.; Hu, Y.; Jiang, W.; Fang, L.; Guan, X.; Chen, J.; Zhang, J.; A Saski, C.; E Scheffler, B.; Stelly, D.; et al. Sequencing of allotetraploid cotton (Gossypium hirsutum L. acc. TM-1) provides a resource for fiber improvement. Nat. Biotechnol. 2015, 33, 531–537. [Google Scholar] [CrossRef] [Green Version]
- Sun, R.; Wang, K.; Guo, T.; Jones, D.C.; Cobb, J.; Zhang, B.; Wang, Q. Genome-wide identification of auxin response factor (ARF) genes and its tissue-specific prominent expression in Gossypium raimondii. Funct. Integr. Genom. 2015, 15, 481–493. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.; Tang, Z.; Wang, M.; Gao, W.; Tu, L.; Jin, X.; Chen, L.; He, Y.; Zhang, L.; Zhu, L.; et al. The genome sequence of Sea-Island cotton (Gossypium barbadense) provides insights into the allopolyploidization and development of superior spinnable fibres. Sci. Rep. 2016, 5, 17662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Q.; Jones, D.C.; Li, W.; Xie, F.; Ma, J.; Sun, R.; Wang, Q.; Zhu, S.; Zhang, B. Genome-wide identification of R2R3-MYB genes and expression analyses during abiotic stress in Gossypium raimondii. Sci. Rep. 2016, 6, 22980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, L.-F.; Li, Y.; Chen, Y.; Li, X.-B. Improved drought and salt tolerance of Arabidopsis thaliana by ectopic expression of a cotton (Gossypium hirsutum) CBF gene. Plant Cell Tissue Organ Cult. 2016, 124, 583–598. [Google Scholar] [CrossRef]
- Salih, H.; Gong, W.; He, S.; Sun, G.; Sun, J.; Du, X. Genome-wide characterization and expression analysis of MYB transcription factors in Gossypium hirsutum. BMC Genet. 2016, 17, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.; Wang, G.; Zhang, X.; Zhang, X.; Qiao, P.; Long, L.; Yuan, Y.; Cai, Y. Genome-wide identification of the TIFY gene family in three cultivated Gossypium species and the expression of JAZ genes. Sci. Rep. 2017, 7, 42418. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, F.; Yang, D.-G.; Li, W.; Zhou, X.-J.; Pei, X.-Y.; Liu, Y.-G.; He, K.-L.; Zhang, W.-S.; Ren, Z.-Y.; et al. Comparative chloroplast genomics of Gossypium species: Insights into repeat sequence variations and phylogeny. Front. Plant Sci. 2018, 9, 376. [Google Scholar] [CrossRef] [Green Version]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Lowe, T.M.; Eddy, S.R. tRNAscan-SE: A program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res. 1997, 25, 0955–0964. [Google Scholar] [CrossRef] [PubMed]
- Lowe, T.M.; Chan, P.P. tRNAscan-SE On-line: Integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res. 2016, 44, W54–W57. [Google Scholar] [CrossRef]
- Mitchell, C. MultAlin–multiple sequence alignment. Bioinformatics 1993, 9, 614. [Google Scholar] [CrossRef]
- Mohanta, T.K.; Arora, P.K.; Mohanta, N.; Parida, P.; Bae, H. Identification of new members of the MAPK gene family in plants shows diverse conserved domains and novel activation loop variants. BMC Genom. 2015, 16, 1–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohanta, T.K.; Mohanta, N.; Parida, P.; Panda, S.K.; Ponpandian, L.N.; Bae, H. Genome-wide identification of mitogen-activated protein kinase gene family across fungal lineage shows presence of novel and diverse activation loop motifs. PLoS ONE 2016, 11, e0149861. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Nei, M.; Dudley, J.; Tamura, K. MEGA: A biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief. Bioinform. 2008, 9, 299–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- Tamura, K. Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G+C-content biases. Mol. Biol. Evol. 1992, 9, 678–687. [Google Scholar] [CrossRef] [Green Version]
- Librado, P.; Rozas, J. DnaSP v5: A software for comprehensive analysis of DNA polymorphism data. Bioinformatics 2009, 25, 1451–1452. [Google Scholar] [CrossRef] [Green Version]
- Saitou, N.; Ueda, S. Evolutionary rates of insertion and deletion in noncoding nucleotide sequences of primates. Mol. Biol. Evol. 1994, 11, 504–512. [Google Scholar] [CrossRef]
- Chen, Z.; Feng, K.; Grover, C.E.; Li, P.; Liu, F.; Wang, Y.; Xu, Q.; Shang, M.; Zhou, Z.; Cai, X.; et al. Chloroplast DNA structural variation, phylogeny, and age of divergence among Diploid Cotton species. PLoS ONE 2016, 11, e0157183. [Google Scholar] [CrossRef] [PubMed]
- Grover, C.E.; Gallagher, J.P.; Jareczek, J.J.; Page, J.T.; Udall, J.A.; Gore, M.; Wendel, J.F. Re-evaluating the phylogeny of allopolyploid Gossypium L. Mol. Phylogenet. Evol. 2015, 92, 45–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, K.; Durand, D.; Farach-Colton, M. NOTUNG: A program for dating gene duplications and optimizing gene family trees. J. Comput. Biol. 2000, 7, 429–447. [Google Scholar] [CrossRef]
- Vernot, B.; Stolzer, M.; Goldman, A.; Durand, D. Reconciliation with non-binary species trees. J. Comput. Biol. 2008, 15, 981–1006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumazawa, Y.; Nishida, M. Sequence evolution of mitochondrial tRNA genes and deep-branch animal phylogenetics. J. Mol. Evol. 1993, 37, 380–398. [Google Scholar] [CrossRef]
- Shepotinovskaya, I.; Uhlenbeck, O.C. tRNA residues evolved to promote translational accuracy. RNA 2013, 19, 510–516. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Q.; Fu, X.; Zhang, T.; Zhou, T.; Yue, M.; Liu, J.; Li, Z. Phylogeny and evolution of chloroplast tRNAs in Adoxaceae. Ecol. Evol. 2021, 11, 1294–1309. [Google Scholar] [CrossRef]
- Tong, K.-L.; Wong, J.-F. Anticodon and wobble evolution. Gene 2004, 333, 169–177. [Google Scholar] [CrossRef]
- Iqbal, M.J.; Aziz, N.; Saeed, N.A.; Zafar, Y.; Malik, K.A. Genetic diversity evaluation of some elite cotton varieties by RAPD analysis. Theor. Appl. Genet. 1997, 94, 139–144. [Google Scholar] [CrossRef]
- Kumar, P.; Anaya, J.; Mudunuri, S.B.; Dutta, A. Meta-analysis of tRNA derived RNA fragments reveals that they are evolutionarily conserved and associate with AGO proteins to recognize specific RNA targets. BMC Biol. 2014, 12, 1–14. [Google Scholar] [CrossRef]
- Charette, M.W.G.M. Pseudouridine in RNA: What, where, how, and why. IUBMB Life 2000, 49, 341–351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.-T.; Hou, Y.-K.; Yang, T.; Zhang, S.-Y.; Yue, M.; Liu, J.; Li, Z. Evolutionary analysis of chloroplast tRNA of Gymnosperm revealed the novel structural variation and evolutionary aspect. PeerJ 2020, 8, e10312. [Google Scholar] [CrossRef]
- Nardi, F.; Carapelli, A.; Fanciulli, P.P.; Dallai, R.; Frati, F. The complete mitochondrial DNA sequence of the basal Hexapod Tetrodontophora bielanensis: Evidence for Heteroplasmy and tRNA translocations. Mol. Biol. Evol. 2001, 18, 1293–1304. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jühling, T.; Duchardt-Ferner, E.; Bonin, S.; Wöhnert, J.; Pütz, J.; Florentz, C.; Betat, H.; Sauter, C.; Mörl, M. Small but large enough: Structural properties of armless mitochondrial tRNAs from the nematode Romanomermis culicivorax. Nucleic Acids Res. 2018, 46, 9170–9180. [Google Scholar] [CrossRef]
- Wang, W.; Chen, X.; Wolin, S.L.; Xiong, Y. Structural basis for tRNA mimicry by a bacterial Y RNA. Structure 2018, 26, 1635–1644. [Google Scholar] [CrossRef] [Green Version]
- Yoshihisa, T. Handling tRNA introns, archaeal way and eukaryotic way. Front. Genet. 2014, 5, 213. [Google Scholar] [CrossRef] [Green Version]
- Kawach, O.; Voß, C.; Wolff, J.; Hadfi, K.; Maier, U.-G.; Zauner, S. Unique tRNA introns of an enslaved algal cell. Mol. Biol. Evol. 2005, 22, 1694–1701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoser, S.M.; Hoffmann, A.; Meindl, A.; Gamper, M.; Fallmann, J.; Bernhart, S.H.; Müller, L.; Ploner, M.; Misslinger, M.; Kremser, L.; et al. Intronic tRNAs of mitochondrial origin regulate constitutive and alternative splicing. Genome Biol. 2020, 21, 1–35. [Google Scholar] [CrossRef]
- Adams, P.L.; Stahley, M.R.; Kosek, A.B.; Wang, J.; Strobel, S.A. Crystal structure of a self-splicing group I intron with both exons. Nat. Cell Biol. 2004, 430, 45–50. [Google Scholar] [CrossRef]
- Wilusz, J.E. Controlling translation via modulation of tRNA levels. Wiley Interdiscip. Rev. RNA 2015, 6, 453–470. [Google Scholar] [CrossRef] [Green Version]
- Köhrer, C.; Mandal, D.; Gaston, K.W.; Grosjean, H.; Limbach, P.A.; Rajbhandary, U.L. Life without tRNAIle-lysidine synthetase: Translation of the isoleucine codon AUA in Bacillus subtilis lacking the canonical tRNA2Ile. Nucleic Acids Res. 2013, 42, 1904–1915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonawane, K.D.; Tewari, R. Conformational Preferences of Hypermodified Nucleoside Lysidine (k2C) Occurring at “Wobble” position in Anticodon Loop of tRNAIle. Nucleosides Nucleotides Nucleic Acids 2008, 27, 1158–1174. [Google Scholar] [CrossRef]
- Weber, F.; Dietrich, A.; Weil, J.-H.; Maréchal-Drouard, L. A potato mitochondrial isoleucine tRNA is coded for by a mitochondrial gene possessing a methionine anticodon. Nucleic Acids Res. 1990, 18, 5027–5030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soma, A.; Ikeuchi, Y.; Kanemasa, S.; Kobayashi, K.; Ogasawara, N.; Ote, T.; Kato, J.-I.; Watanabe, K.; Sekine, Y.; Suzuki, T. An RNA-modifying enzyme that governs both the codon and amino acid specificities of isoleucine tRNA. Mol. Cell 2003, 12, 689–698. [Google Scholar] [CrossRef]
- Shrestha, B.; Weng, M.-L.; Theriot, E.C.; Gilbert, L.E.; Ruhlman, T.A.; Krosnick, S.E.; Jansen, R.K. Highly accelerated rates of genomic rearrangements and nucleotide substitutions in plastid genomes of Passiflora subgenus Decaloba. Mol. Phylogenet. Evol. 2019, 138, 53–64. [Google Scholar] [CrossRef]
- Zhao, Z.; Fu, Y.-X.; Hewett-Emmett, D.; Boerwinkle, E. Investigating single nucleotide polymorphism (SNP) density in the human genome and its implications for molecular evolution. Gene 2003, 312, 207–213. [Google Scholar] [CrossRef]
- Yarham, J.W.; McFarland, R.; Taylor, R.W.; Elson, J.L. A proposed consensus panel of organisms for determining evolutionary conservation of mt-tRNA point mutations. Mitochondrion 2012, 12, 533–538. [Google Scholar] [CrossRef]
- Jelesko, J.G.; Harper, R.; Furuya, M.; Gruissem, W. Rare germinal unequal crossing-over leading to recombinant gene formation and gene duplication in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 1999, 96, 10302–10307. [Google Scholar] [CrossRef] [Green Version]
- Xiao, H.; Jiang, N.; Schaffner, E.; Stockinger, E.J.; Van Der Knaap, E. A retrotransposon-mediated gene duplication underlies morphological variation of tomato fruit. Science 2008, 319, 1527–1530. [Google Scholar] [CrossRef]
- Rensing, S.A. Gene duplication as a driver of plant morphogenetic evolution. Curr. Opin. Plant Biol. 2014, 17, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Panchy, N.; Lehti-Shiu, M.; Shiu, S.-H. Evolution of gene duplication in plants. Plant Physiol. 2016, 171, 2294–2316. [Google Scholar] [CrossRef] [PubMed] [Green Version]

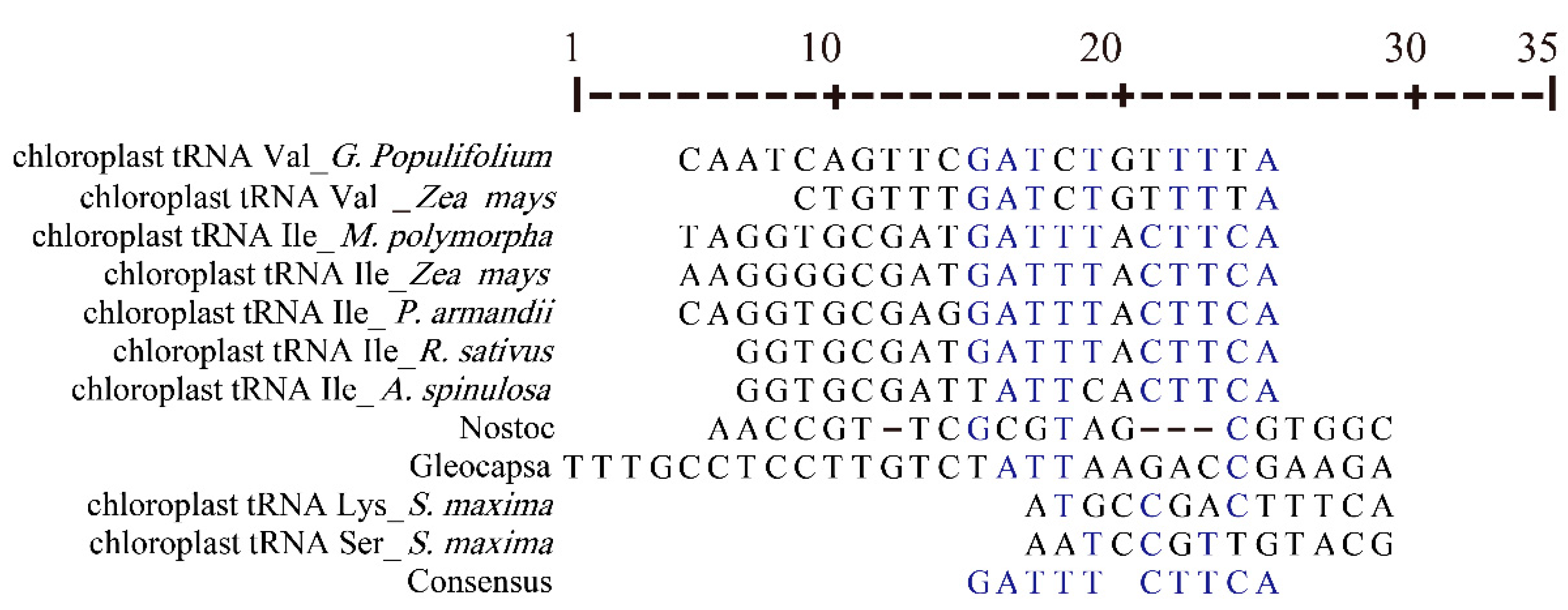
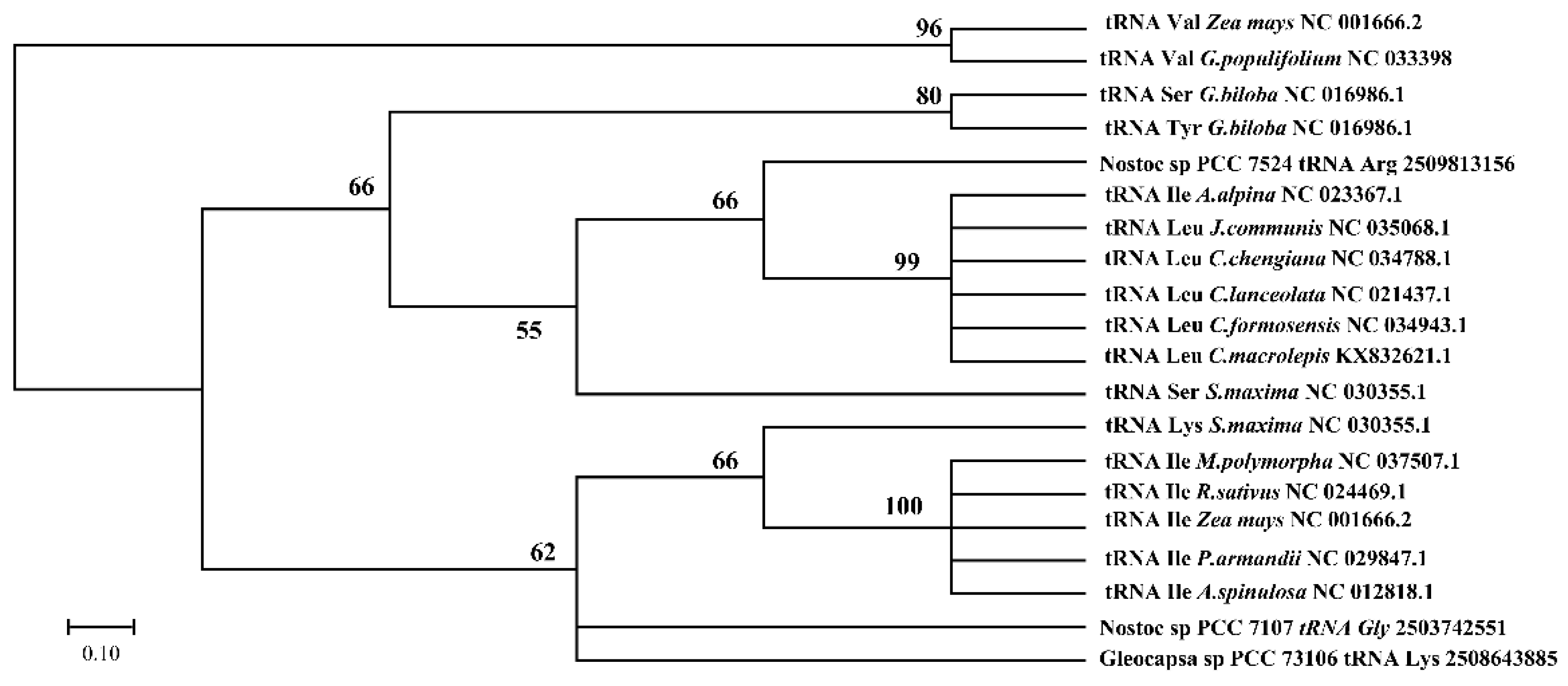

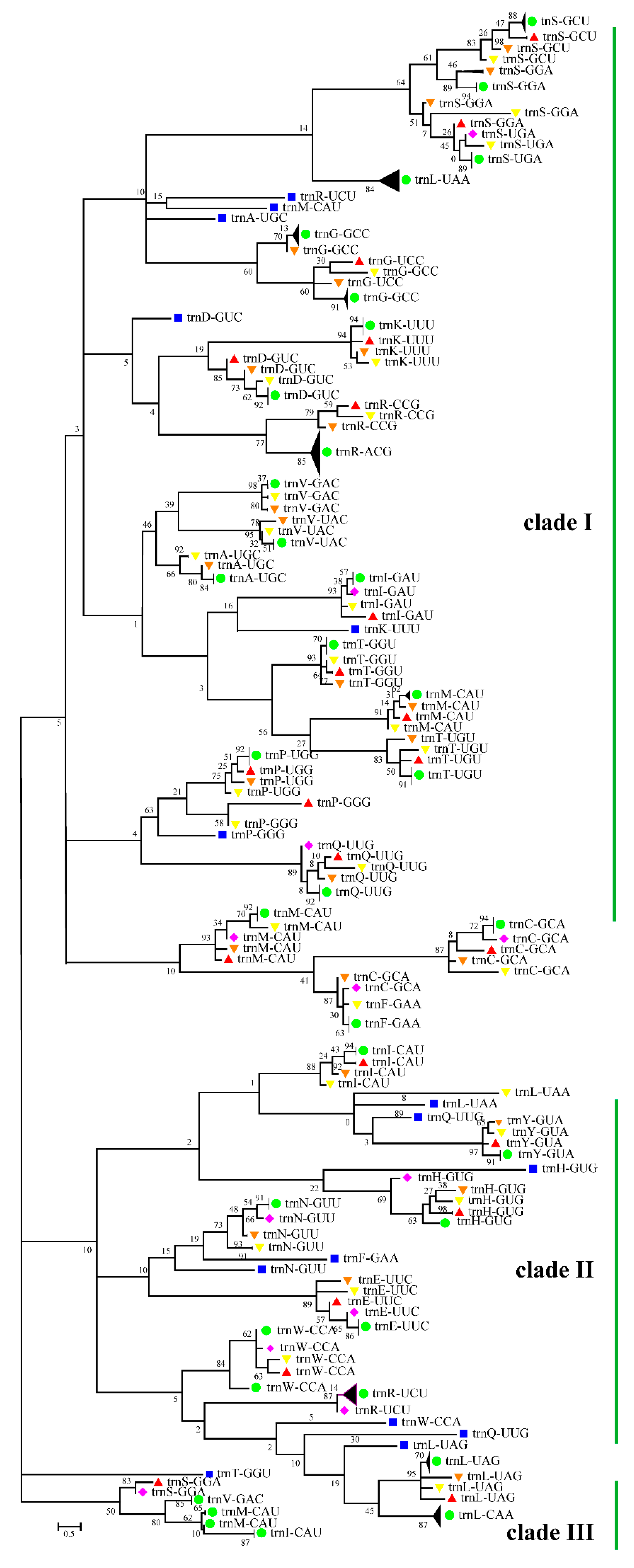


| Karyotype | Species | Accession Number | Wild/Cultivars |
|---|---|---|---|
| A2 | Gossypium arboreum | NC_016712 | cultivars |
| B1 | G. anomalum | NC_023213 | wild |
| C2 | G. robinsonii | NC_018113 | wild |
| D3-k | G. klotzschianum | NC_033394 | wild |
| E2 | G. somalense | NC_018110 | wild |
| F1 | G. longicalyx | NC_023216 | wild |
| G1 | G. bickii | NC_023214 | wild |
| K2 | G. populifolium | NC_033398 | wild |
| AD1 | G. hirsutum | HQ901196 | cultivars |
| AD2 | G. barbadense | HQ901199 | cultivars |
| tRNA Isotype | Number of tRNAs | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| A2 1 | B1 2 | C2 3 | D3-K 4 | E2 5 | F1 6 | AD1 7 | AD2 8 | G1 9 | K2 10 | |
| Alanine | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Glycine | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Proline | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Threonine | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Valine | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 |
| Serine | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 |
| Arginine | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 |
| Leucine | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| Phenylalanine | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Asparagine | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Lysine | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Aspartate | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Glutamate | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Histidine | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Glutamine | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Isoleucine | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| Methionine | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 1 | 2 |
| Tyrosine | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Cysteine | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Tryptophan | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Selenocysteine | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Suppressor | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total | 37 | 37 | 37 | 37 | 37 | 37 | 37 | 37 | 36 | 37 |
| tRNA Isotype | AC-Arm | D-Arm | D-Loop | ANC-Arm | ANC-Loop | Variable Region | Ψ-Arm | Ψ-Loop |
|---|---|---|---|---|---|---|---|---|
| Alanine | GGGGAUA | GCUC | AGUUGGUA | CCGCU | CUUGCAU | AUGUC | AGCGG | UUCGAGU |
| Arginine | GXGXCX2 | Gx3 | AX2GGAUA | ***** | CUXCXAA | GU | GG | UUCGAAU |
| Asparagine | GUCGGGA | GCUC | AGUUGGUA | GUCGG | CUGUUAA | UGGUC | GUAGG | UUCGAAU |
| Aspartate | GGGAUUG | GUUC | AAUUGGUCA | CCGCC | CUGUCAA | AAGCU | GCGGG | UUCGAGC |
| Cysteine | GGCGACA | GCC | GAGCGGUAA | GGGGA | CUGCAAA | UAUUC | CCCAG | UUCAAAU |
| Glutamate | GX2CX3 | GX3 | AGXGGUX1–3 | CX2CX | CUUUCAX | X2GX1–2 | X3GX | UUCXAXU |
| Glutamine | UGGGGCG | GCC | AAGUGGUAA | CGGG | UUUUGGU | CUAUGC | GGAGG | UUCGAAU |
| Glycine | GCGGAUA | GU | CGAAUGGUAAA | UCUCU | UUGCCAA | AGAC | GCGGG | UUCGAUU |
| Histidine | GCGGAUG | GCC | AAGUGGAUCAA | GUGGA | UUGUGAA | CAUGC | GCGGG | UUCAAUU |
| Isoleucine | GCAUCCA | GCU | GAAUGGUUAA | CCCAA | CUCAUAA | AAUUC | GUAGG | UUCAAUU |
| Leucine | GX6 | GXG | AAAUXGX3–4A | X3GX | CUX4A | XGX9–12 | X3GG | UUCXAGU |
| Lysine | GGGUUGC | ACUC | AACGGUA | UCGG | CUUUUAA | CUAGUU | CCGGG | UUCGAGU |
| Methionine | XCX5–6 | X3G | AGUX5–6 | ***** | XUCAUAX | X2GUC | AUXGG | UUCAAAU |
| Phenylalanine | GUCGGGA | GCUC | AGUUGGUA | GAGGA | CUGAAAA | GUGUC | ACCAG | UUCAAAU |
| Proline | AGGGAUG | GCGC | AGCUUGGUA | UUUGU | UUUGGGU | AUGUC | ACGGG | UUCAAAU |
| Serine | GGAGAGA | GCX1–2 | X4GX3–4A | X2GX1–2 | XUXGXAX | X4GX15–19 | GAGGG | UUCGAAU |
| Threonine | XGCCX0–4 | XCUC | AGXGGUA | XCGCX | X3GUAA | X2GUC | AUCGG | UUCX3U |
| Tryptophan | GCGCUCU | GUUC | AGUUCGGUA | UGGGU | CUCCAAA | AUGUC | GUAGG | UUCAAAU |
| Tyrosine | GGGUCGA | CCCG | AGCGGUUAA | ACGGA | CUGUAAA | GGCA | GCUGG | UUCAAAU |
| Valine | AGGGAUA | ACUC | AGCGGUA | UCACC | UUGACGU | AAGUC | AUCAG | UUCGAGC |
| Integrated Clades | Types of tRNAs |
|---|---|
| clade I | tRNASer, tRNALeu, tRNAArg, tRNAMet, tRNAAla, tRNAGly, tRNAAsp, tRNALys, tRNAVal, tRNAIle, tRNAThr, tRNAPro, tRNAGln, tRNACys, tRNAPhe |
| clade II | tRNAIle, tRNALeu, tRNAGln, tRNATyr, tRNAHis, tRNAAsn, tRNAPhe, tRNAGlu, tRNATrp, tRNAArg |
| clade III | tRNAThr, tRNASer, tRNAVal, tRNAMet, tRNAIle |
| Genome Region | Length (bp) | Value | % |
|---|---|---|---|
| Total substitutions | 162,231 | 2709 | 1.67 |
| Coding regions | 79,244 | 906 | 1.14 |
| Non-synonymous | / | 681.58 | 0.86 |
| Synonymous | / | 224.42 | 0.28 |
| dN/dS | / | 3.04 | / |
| Intron | 21,443 | 299 | 1.39 |
| Intergenic spacer | 51,920 | 1504 | 2.90 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, T.-T.; Yang, Y.; Song, X.-Y.; Gao, X.-Y.; Zhang, X.-L.; Zhao, J.-J.; Zhou, K.-H.; Zhao, C.-B.; Li, W.; Yang, D.-G.; et al. Novel Structural Variation and Evolutionary Characteristics of Chloroplast tRNA in Gossypium Plants. Genes 2021, 12, 822. https://0-doi-org.brum.beds.ac.uk/10.3390/genes12060822
Zhang T-T, Yang Y, Song X-Y, Gao X-Y, Zhang X-L, Zhao J-J, Zhou K-H, Zhao C-B, Li W, Yang D-G, et al. Novel Structural Variation and Evolutionary Characteristics of Chloroplast tRNA in Gossypium Plants. Genes. 2021; 12(6):822. https://0-doi-org.brum.beds.ac.uk/10.3390/genes12060822
Chicago/Turabian StyleZhang, Ting-Ting, Yang Yang, Xiao-Yu Song, Xin-Yu Gao, Xian-Liang Zhang, Jun-Jie Zhao, Ke-Hai Zhou, Chang-Bao Zhao, Wei Li, Dai-Gang Yang, and et al. 2021. "Novel Structural Variation and Evolutionary Characteristics of Chloroplast tRNA in Gossypium Plants" Genes 12, no. 6: 822. https://0-doi-org.brum.beds.ac.uk/10.3390/genes12060822





