Changes in Planktivory and Herbivory Regimes in a Shallow South American Lake (Lake Blanca Chica, Argentina) Over the Last 250 Years
Abstract
:1. Introduction
2. Materials and Methods
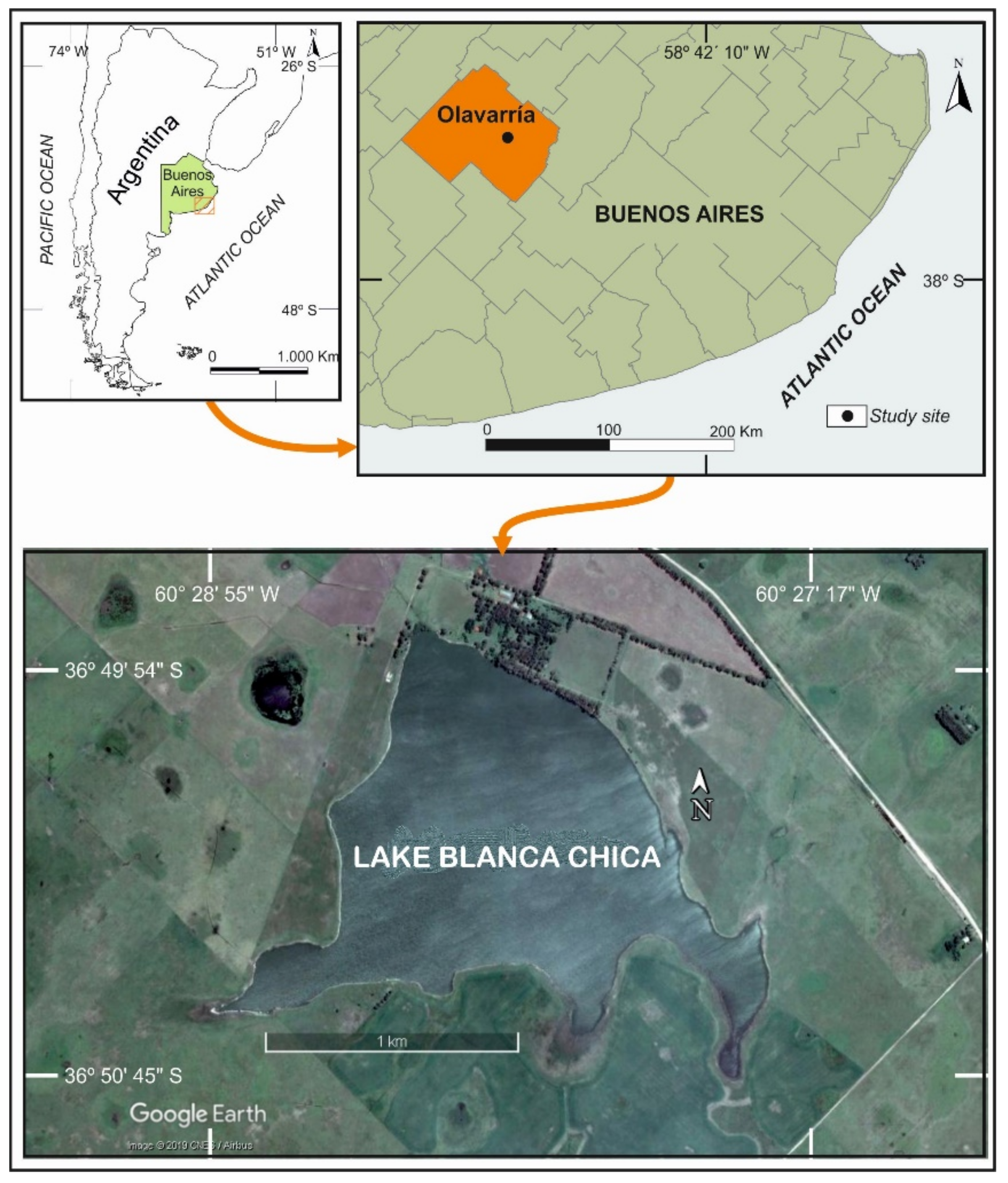
2.1. Study Area
2.2. Core Chronology and Paleolimnological Analyses
2.3. Data Analyses
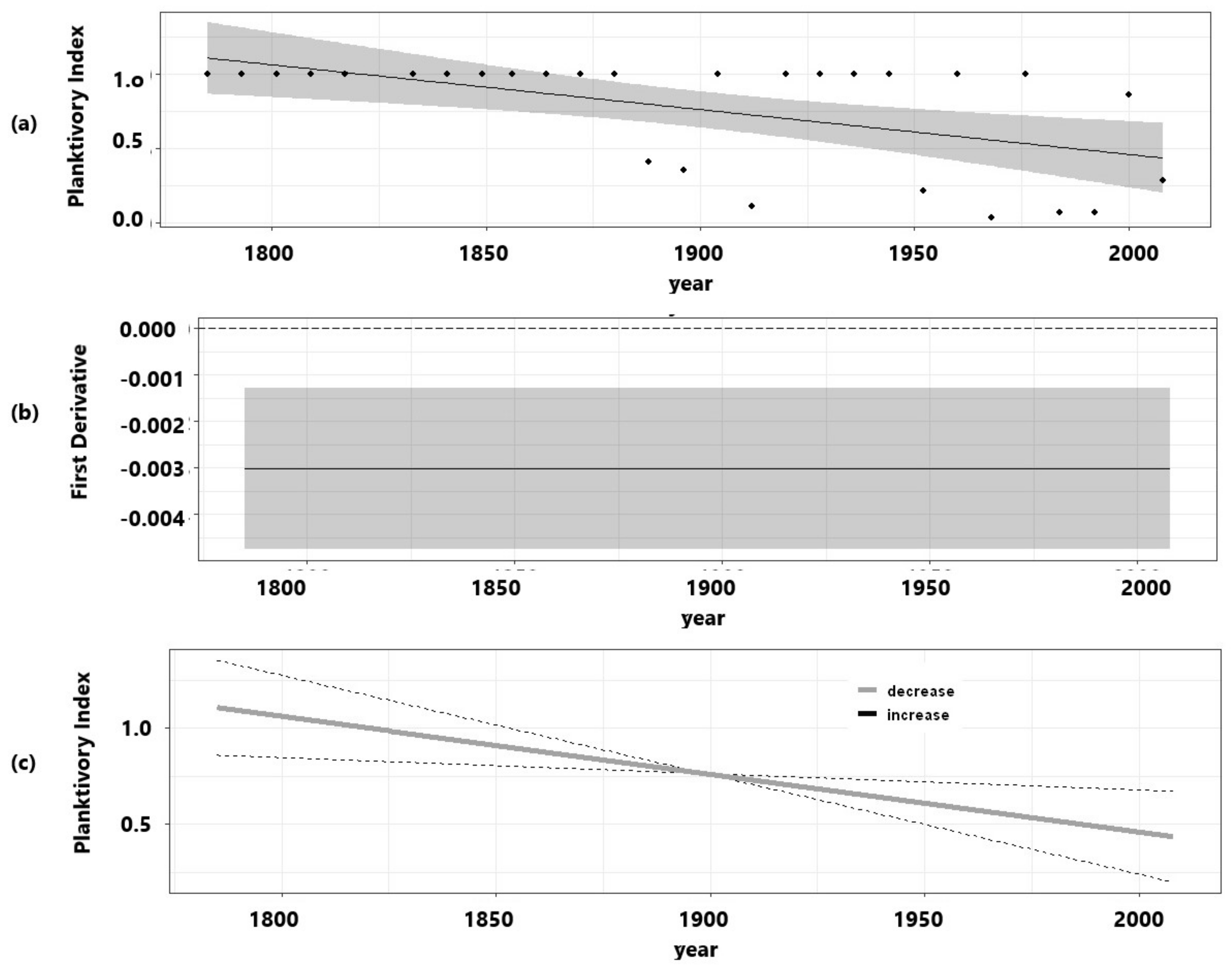
3. Results
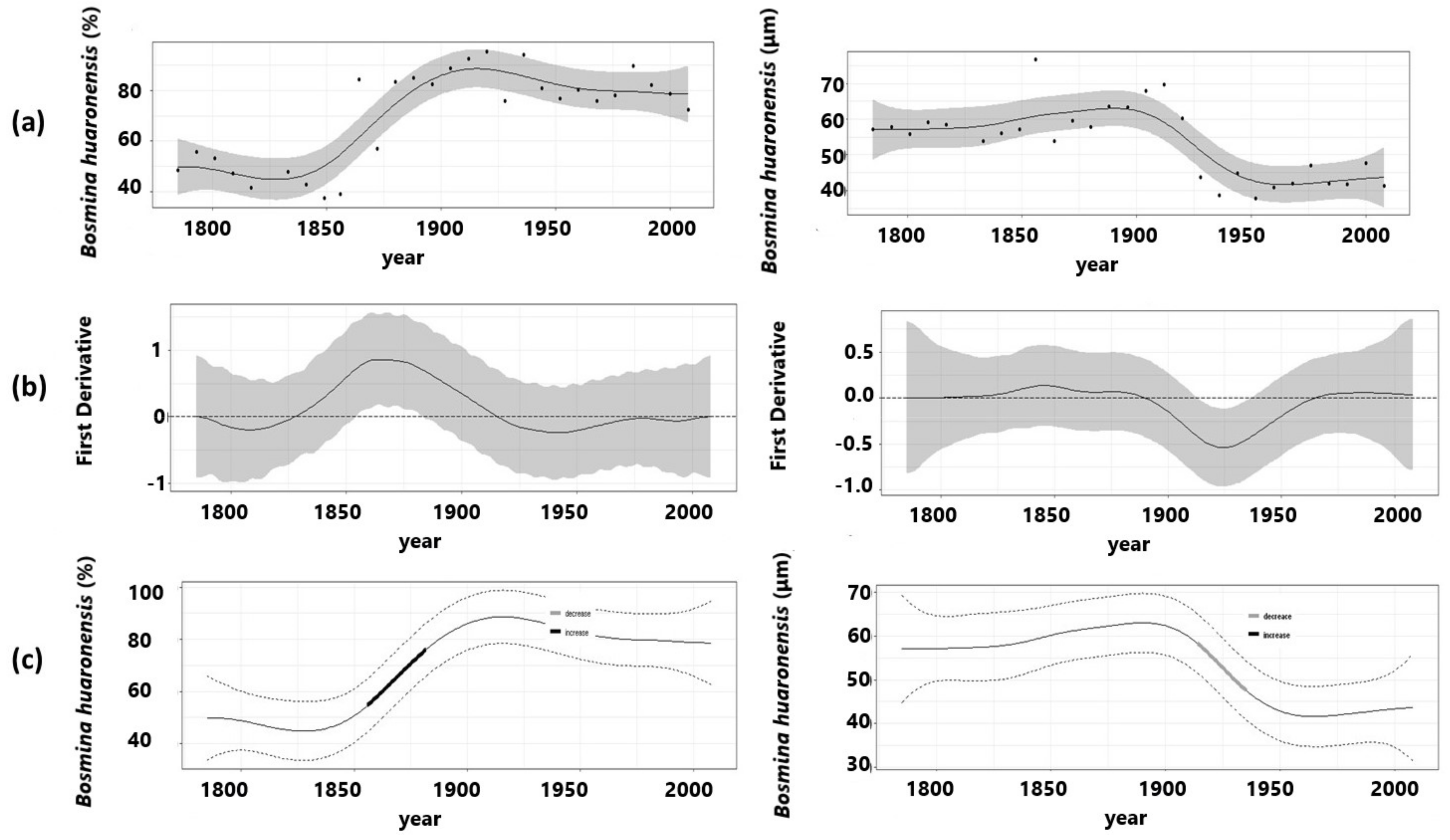
3.1. Herbivory Biomarker
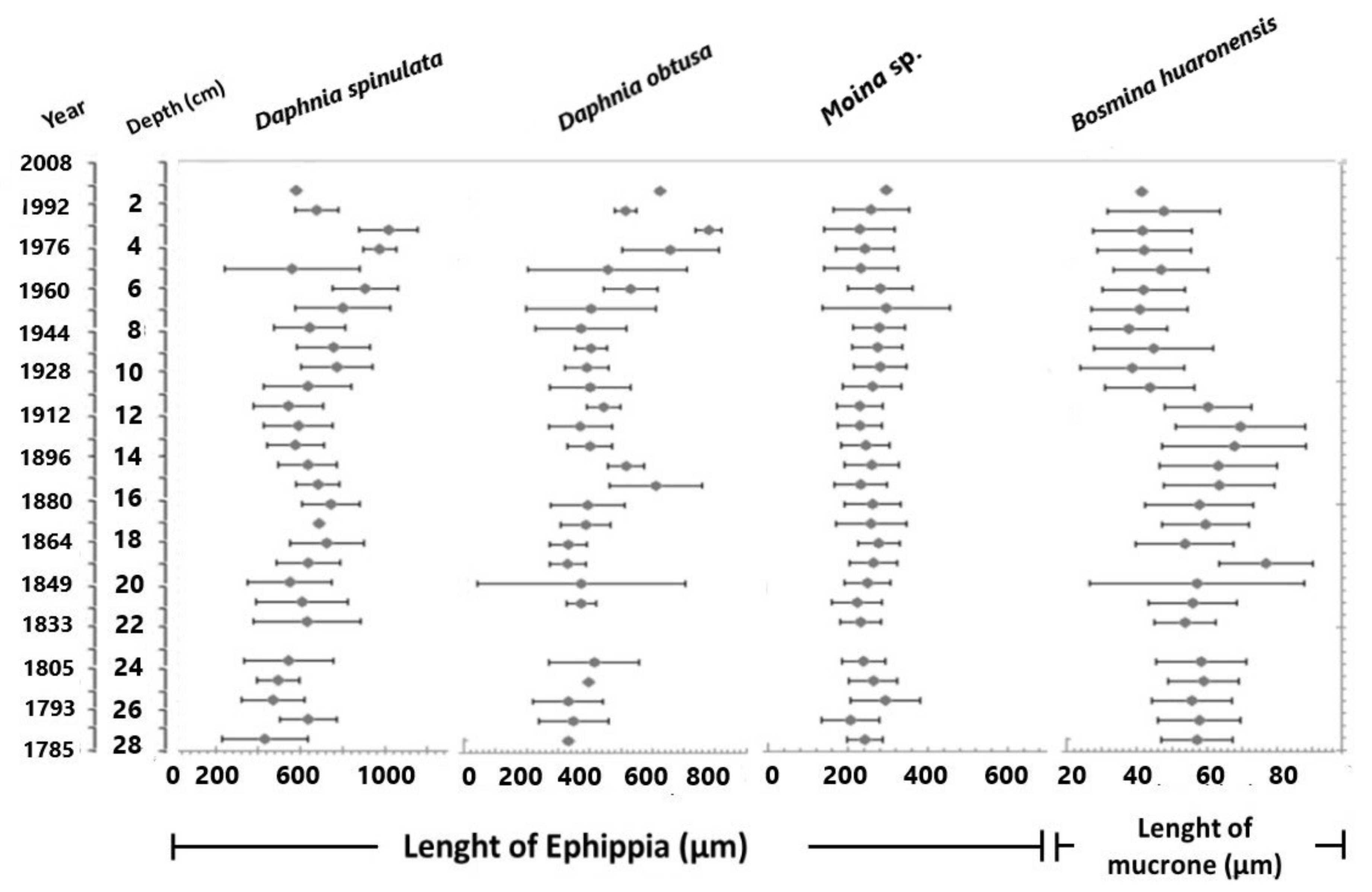
3.2. Ephippial Assemblage
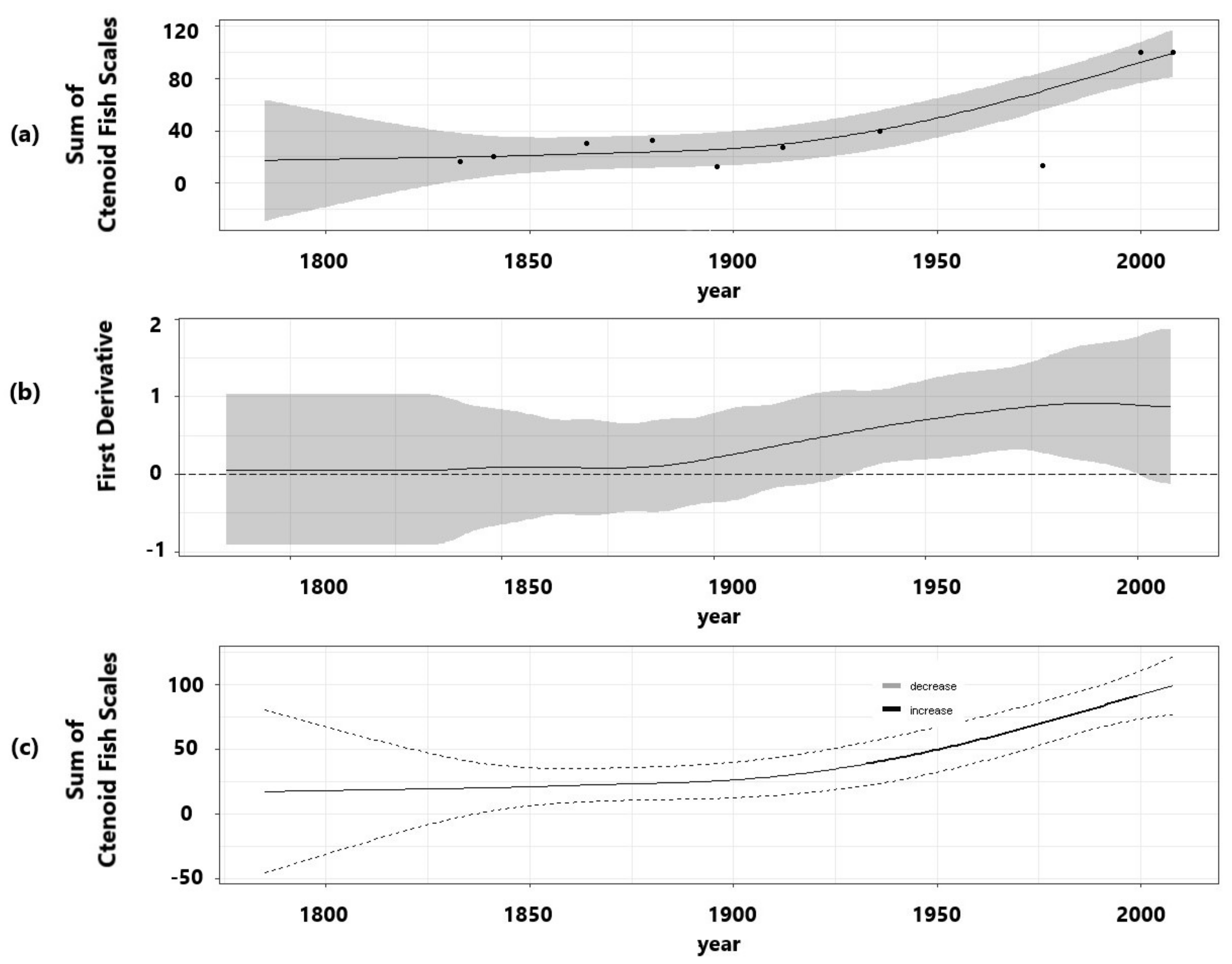
3.3. Fish Predation Indicators
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dumont, H.J.; Van de Velde, I.; Dumont, S. The dry weight estimate of biomass in a selection of Cladocera, Copepoda and Rotifera from the plankton, periphyton and benthos of continental waters. Oecologia 1975, 19, 75–97. [Google Scholar] [CrossRef]
- Sterner, R.W. Role of Zooplankton in Aquatic Ecosystems. In Encyclopedia of Inland Waters; Likens, G.E., Ed.; Academic Press: Oxford, UK, 2009; pp. 678–688. [Google Scholar]
- Nevalainen, L.; Brown, M.; Manca, M. Sedimentary record of cladoceran functionality under eutrophication and re-oligotrophication in Lake Maggiore, Northern Italy. Water 2018, 10, 86. [Google Scholar] [CrossRef] [Green Version]
- Brooks, J.; Dodson, S. Predation, body size, and composition of plankton. Science 1965, 150, 28–35. [Google Scholar] [CrossRef] [PubMed]
- González Sagrario, M.A.; Balseiro, E. The role of macroinvertebrates and fish in regulating the provision by macrophytes of refugia for zooplankton in a warm temperate shallow lake. Freshw. Biol. 2010, 55, 2153–2166. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Søndergaard, M.; Lauridsen, T.; Pedersen, L.J.; Jensen, L. Top-down control in freshwater lakes: The role of nutrient state, submerged macrophytes and water depth. Hydrobiologia 1997, 342, 151–164. [Google Scholar] [CrossRef]
- Korosi, J.B.; Paterson, A.M.; DeSellas, A.M.; Smol, J.P. Linking mean body size of pelagic Cladocera to environmental variables in Precambrian Shield lakes: A paleolimnological approach. J. Limnol. 2008, 67, 22–34. [Google Scholar] [CrossRef] [Green Version]
- Hann, B.J. Methods in Quaternary Ecology #6. Cladocera. Geosci. Can. 1989, 16, 17–26. [Google Scholar]
- DeSellas, A.M.; Paterson, A.M.; Sweetman, J.N.; Smol, J.P. Cladocera assemblages from the surface sediments of south-central Ontario (Canada) lakes and their relationships to measured environmental variables. Hydrobiologia 2008, 600, 105–119. [Google Scholar] [CrossRef]
- Scheffer, M.; Jeppesen, E. Regime Shifts in Shallow Lakes. Ecosystems 2007, 10, 1–3. [Google Scholar] [CrossRef] [Green Version]
- Jeppesen, E.; Leavitt, P.; De Meester, L.; Jensen, J.P. Functional ecology and palaeolimnology: Using cladoceran remains to reconstruct anthropogenic impact. Trends Ecol. Evol. 2001, 16, 191–198. [Google Scholar] [CrossRef]
- Meerhoff, M.; Iglesias, C.; De Mello, F.T.; Clemente, J.M.; Jensen, E.; Lauridsen, T.L.; Jeppesen, E. Effects of habitat complexity on community structure and predator avoidance behaviour of littoral zooplankton in temperate versus subtropical shallow lakes. Freshw. Biol. 2007, 52, 1009–1021. [Google Scholar] [CrossRef]
- Catalan, J.; Pla-Rabés, S.; Wolfe, A.P.; Smol, J.P.; Rühland, K.M.; Anderson, N.J.; Kopáček, J.; Stuchlík, E.; Schmidt, R.; Koinig, K.A.; et al. Global change revealed by palaeolimnological records from remote lakes: A review. J. Paleolimnol. 2013, 49, 513–535. [Google Scholar] [CrossRef]
- Battarbee, R.W. Mountain lakes, pristine or polluted? Limnetica 2005, 24, 1–8. [Google Scholar]
- Carpenter, S.R.; Leavitt, P.R. Temporal variation in a paleolimnological record arising from a trophic cascade. Ecology 1991, 72, 277–285. [Google Scholar] [CrossRef]
- Daley, R.J. Experimental characterization of lacustrine chlorophyll diagenesis. II. Bacterial, viral and herbivore grazing effects. Arch. Hydrobiol. 1973, 72, 409–439. [Google Scholar]
- Leavitt, P.R. A review of factors that regulate carotenoid and chlorophyll deposition and fossil pigment abundance. J. Paleolimnol. 1993, 9, 109–127. [Google Scholar] [CrossRef]
- Davidson, T.A.; Sayer, C.D.; Perrow, M.; Bramm, M.; Jeppesen, E. The simultaneous inference of zooplanktivorous fish and macrophyte density from sub-fossil cladoceran assemblages: A multivariate regression tree approach. Freshw. Biol. 2010, 55, 546–564. [Google Scholar] [CrossRef]
- Davidson, T.A.; Sayer, C.D.; Perrow, M.R.; Bramm, M.; Jeppesen, E. Are the controls of species composition similar for contemporary and sub-fossil cladoceran assemblages? A study of 39 shallow lakes of contrasting trophic status. J. Paleolimnol. 2007, 38, 117–134. [Google Scholar] [CrossRef]
- Hann, B.J.; Leavitt, P.R.; Chang, P.S.S. Cladocera community response to experitmental eutrophication in Lake 227 as recorded in laminated sediments. Can. J. Fish. Aquat. Sci. 1994, 51, 2312–2321. [Google Scholar] [CrossRef]
- Jeppesen, E.; Jensen, J.P.; Amsinck, S.; Landkildehus, F.; Lauridsen, T.; Mitchell, S. Reconstructing the historical change in Daphnia mean size and planktivorous fish abundance in lakes from the size of Daphnia ephippia in the sediment. J. Paleolimnol. 2002, 27, 133–143. [Google Scholar] [CrossRef]
- Verschuren, D.; Marnell, L.F. Fossil zooplankton and the historical status of westslope cutthroat trout in a headwater lake of Glacier National Park, Montana. Trans. Am. Fish. Soc. 1997, 126, 21–34. [Google Scholar] [CrossRef]
- Paggi, J.C. Revisión de las especies argentinas del género Bosmina Bird agrupadas en el subenero Neobosmina Lieder (Crustacea: Cladocera). Acta Zool. Lilloana 1979, 35, 137–162. [Google Scholar]
- Iriondo, M. Quaternary lakes of Argentina. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1989, 70, 81–88. [Google Scholar] [CrossRef]
- Sanzano, V.; Grosman, F.; Colasurdo, P. Estudio limnológico de Laguna Blanca Chica (Olavarría, Provincia de Buenos Aires) durante un período de sequía. Biol. Acuát. 2014, 30, 189–202. [Google Scholar]
- Piovano, E.L.; Ariztegui, D.; Damatto-Moreira, S. Recent environmental changes in Laguna Mar Chiquita (central Argentina): A sedimentary model for a highly variable saline lake. Sedimentology 2002, 49, 1371–1384. [Google Scholar] [CrossRef]
- Guerra, L.; Piovano, E.L.; Córdoba, F.E.; Sylvestre, F.; Damatto, S. The hydrological and environmental evolution of shallow Lake Melincué, central Argentinean Pampas, during the last millennium. J. Hydrol. 2015, 529, 570–583. [Google Scholar] [CrossRef]
- González Sagrario, M.A.; Musazzi, S.; Córdoba, F.E.; Mendiolar, M.; Lami, A. Inferring the occurrence of regime shifts in a shallow lake during the last 250 years based on multiple indicators. Ecol. Indic. 2019. submitted. [Google Scholar]
- Lami, A.; Musazzi, S.; Marchetto, A.; Buchaca, T.; Kerna, M.; Jeppesen, E.; Guilizzoni, P. Sedimentary pigments in 308 alpine lakes and their relation to environmental gradients. Adv. Limnol. 2009, 62, 217–238. [Google Scholar]
- Korhola, A.; Rautio, M. Cladocera and other branchiopod crustaceans. In Tracking Environmental Change Using Lake Sediments; Zoological Indicators; Smol, J.P., Birks, H.J.B., Last, W.M., Eds.; Kluwer Academic Publisher: Dordrecht, The Netherlands, 2001; Volume 4, pp. 5–41. [Google Scholar]
- Kotov, A.A. Separation of Leydigia louisi Jenkin, 1934 from L. leydigi (Schoedler, 1863) (Chydoridae, Anomopoda, Cladocera). Hydrobiologia 2003, 490, 147–168. [Google Scholar] [CrossRef]
- Smirnov, N.N.; Kotov, A.A.; Coronel, J.S. Partial revision of the aduncus-like species of Pleuroxus Baird, 1843 (Chydoridae, Cladocera) from the southern hemisphere with comments on subgeneric differentiation within the genus. J. Nat. Hist. 2006, 40, 1617–1639. [Google Scholar] [CrossRef]
- Szeroczyńska, K.; Sarmaja-Korjonen, K. Atlas of Subfossil Cladocera from Central and Northern Europe; Friends of the lower Vistula Society: Świecie, Poland, 2007; p. 84. [Google Scholar]
- Wood, S.N. Generalized Additive Models: An Introduction with R, 2nd ed.; Chapman and Hall/CRC: Boca Raton FL, USA, 2017. [Google Scholar]
- Simpson, G.L. Modelling palaeoecological time series using generalised additive models. Front. Ecol. Evol. 2018, 6, 149. [Google Scholar] [CrossRef] [Green Version]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R package version 2.2-1. 2015. Available online: http://cran.r-project.org (accessed on 15 November 2018).
- Borcard, D.; Gillet, F.; Legendre, P. Numerical Ecology with R, 1st ed.; Springer: New York, NY, USA, 2011; p. 319. [Google Scholar]
- R Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 15 November 2018).
- Córdoba, F.E.; Piovano, E.L.; Guerra, L.; Mulsow, S.; Sylvestre, F.; Zárate, M. Independent time markers validate 210Pb chronologies for two shallow Argentine lakes in Southern Pampas. Quatern. Int. 2017, 438, 175–186. [Google Scholar] [CrossRef]
- González Sagrario, M.A.; Balseiro, E.; Ituarte, R.; Spivak, E. Macrophytes as refuge or risky area for zooplankton: A balance set by littoral predacious macroinvertebrates. Freshw. Biol. 2009, 54, 1042–1053. [Google Scholar] [CrossRef]
- Perga, M.-E.; Desmet, M.; Enters, D.; Reyss, J.-L. A century of bottom-up- and top-down driven changes on a lake planktonic food web: A paleoecological and paleoisotopic study of Lake Annecy, France. Limnol. Oceanogr. 2010, 55, 803–816. [Google Scholar]
- Amsinck, S.L.; Jeppesen, E.; Landkildehus, F. Inference of past changes in zooplankton community structure and planktivorous fish abundance from sedimentary subfossils—A study of a coastal lake subjected to major fish kill incidents during the past century. Arch. Hydrobiol. 2005, 162, 363–382. [Google Scholar] [CrossRef]
- Korosi, J.B.; Kurek, J.; Smol, J.P. A review on utilizing Bosmina size structure archived in lake sediments to infer historic shifts in predation regimes. J. Plankton Res. 2013, 35, 444–460. [Google Scholar] [CrossRef] [Green Version]
- Brahney, J.; Routledge, R.; Bos, D.G.; Pellatt, M.G. Changes to the productivity and trophic structure of a sockeye salmon Rearing Lake in British Columbia. N. Am. J. Fish. Manag. 2010, 30, 433–444. [Google Scholar] [CrossRef]
- Davidson, T.A.; Sayer, C.D.; Langdon, P.G.; Burgess, A.; Jackson, M. Inferring past zooplanktivorous fish and macrophyte density in a shallow lake: Application of a new regression tree model. Freshw. Biol. 2010, 55, 584–599. [Google Scholar] [CrossRef]
- Manca, M.; Torretta, B.; Comoli, P.; Amsinck, S.L.; Jeppesen, E. Major changes in trophic dynamics in large, deep sub-alpine Lake Maggiore from 1940s to 2002: A high resolution comparative palaeo-neolimnological study. Freshw. Biol. 2007, 52, 2256–2269. [Google Scholar] [CrossRef]
- Acharya, K.; Kyle, M.; Elser, J.J. Effects of stoichiometric dietary mixing on Daphnia growth and reproduction. Oecologia 2003, 138, 333–340. [Google Scholar] [CrossRef]
- Gliwicz, Z.M. Daphnia growth at different concentrations of blue-green filaments. Arch. Hydrobiol. 1990, 120, 51–65. [Google Scholar]
- Davidson, T.A.; Bennion, H.; Jeppesen, E.; Clarke, G.H.; Sayer, C.D.; Morley, D.; Odgaard, B.V.; Rasmussen, P.; Rawcliffe, R.; Salgado, J.; et al. The role of cladocerans in tracking long-term change in shallow lake trophic status. Hydrobiologia 2011, 676, 299–315. [Google Scholar] [CrossRef]



| Indicator | Species | % | r2 | p-Value | Edf | F or Chi * |
|---|---|---|---|---|---|---|
| Ephippia weighted | D. spinulata | 25.2 | 0.205 | 0.01 | 0.870 | 3.362 |
| Abundance (%) | D. obtusa | 25.8 | 0.213 | 0.00918 | 1.532 | 7.294 * |
| Moina sp. | 9.63 | 0.0645 | Ns | 0.924 | 0.207 | |
| Ephippia mean | D. spinulata | 35.4 | 0.329 | 0.000789 | 1 | 14.25 |
| Size | D. obtusa | 37.1 | 0.464 | <1 × 10−6 | 2.714 | 32.01 * |
| Moina sp. | 8.01 | 0.0447 | Ns | 1 | 2.262 | |
| Abundance (%) | B. huaronensis | 81.7 | 0.778 | 9.12 × 10−11 | 4.796 | 3.506 |
| Mucrone mean Length | B. huaronensis | 73.3 | 0.67 | 2.13 × 10−5 | 5.118 | 8.71 |
| Pheophorbide a | 97.5 | 0.955 | <2 × 10−16 | 11.85 | 39.2 | |
| Planktivory index | 29.2 | 0.265 | 0.00106 | 1 | 10.71 * | |
| Fish scales | 64.4 | 0.547 | 2.6 × 10−13 | 2.219 | 52.1 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carrozzo, D.; Musazzi, S.; Lami, A.; Córdoba, F.E.; González Sagrario, M.d.l.Á. Changes in Planktivory and Herbivory Regimes in a Shallow South American Lake (Lake Blanca Chica, Argentina) Over the Last 250 Years. Water 2020, 12, 597. https://0-doi-org.brum.beds.ac.uk/10.3390/w12020597
Carrozzo D, Musazzi S, Lami A, Córdoba FE, González Sagrario MdlÁ. Changes in Planktivory and Herbivory Regimes in a Shallow South American Lake (Lake Blanca Chica, Argentina) Over the Last 250 Years. Water. 2020; 12(2):597. https://0-doi-org.brum.beds.ac.uk/10.3390/w12020597
Chicago/Turabian StyleCarrozzo, David, Simona Musazzi, Andrea Lami, Francisco E. Córdoba, and María de los Ángeles González Sagrario. 2020. "Changes in Planktivory and Herbivory Regimes in a Shallow South American Lake (Lake Blanca Chica, Argentina) Over the Last 250 Years" Water 12, no. 2: 597. https://0-doi-org.brum.beds.ac.uk/10.3390/w12020597






