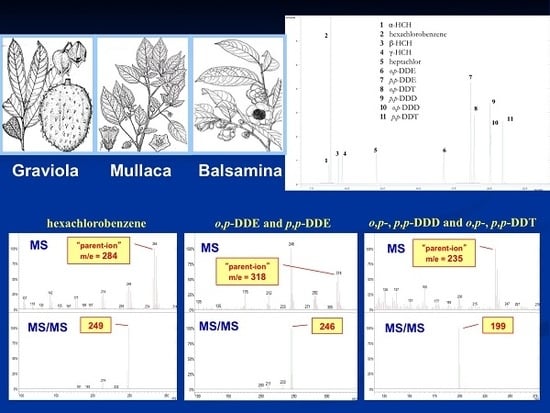
Contamination of Plants from Amazonia by Environmental Pollution
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plants Description
2.2. Chemicals and Reagents
2.3. Plants Preparation
2.4. Extraction
2.5. Clean Up
2.6. Gas Chromatographic-Tandem Mass Spectrometry (GC-MS/MS) and Multi Reaction Monitoring (MRM) Analysis
2.7. Detection Criteria, Analyte Recovery and Repeatability
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schwarzenbach, R.P.; Gschwend, P.M.; Imboden, D.M. Environmental Organic Chemistry, 3rd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2017; ISBN 978-1-118-76723-8. [Google Scholar]
- LRTAP Convention. Protocol to the 1979 Convention on Long-Range Transboundary Air Pollution on Persistent Organic Pollutants (POPs). 1998. Available online: http://ec.europa.eu/world/agreements/prepareCreateTreatiesWorkspace/treatiesGeneralData.do?step=0&redirect=true&treatyId=6201 (accessed on 26 October 2017).
- Tanabe, S. POPs—Need for target research on high risk stage. Mar. Pollut. Bull. 2004, 48, 609–610. [Google Scholar] [CrossRef] [PubMed]
- Wania, F.; Mackay, D. Tracking the distribution of Persistent Organic Pollutants. Environ. Sci. Technol. 1996, 30, 390A–396A. [Google Scholar] [CrossRef] [PubMed]
- Fernández, P.; Grimalt, J.O. On the Global Distribution of Persistent Organic Pollutants. Chimia 2003, 57, 514–521. [Google Scholar] [CrossRef]
- Daly, G.L.; Wania, F. Organic Contaminants in Mountains. Environ. Sci. Technol. 2005, 39, 385–398. [Google Scholar] [CrossRef] [PubMed]
- AMAP. AMAP Assessment 2002: Persistent Organic Pollutants in the Arctic; Arctic Monitoring and Assessment Programme (AMAP): Oslo, Norway, 2004; ISBN 82-7971-019-1. Available online: https://www.amap.no/documents/doc/amap-assessment-2002-persistent-organic-pollutants-in-the-arctic/96 (accessed on 26 October 2017).
- Vilanova, R.; Fernandez, P.; Martinez, C.; Grimalt, J.O. Organochlorine Pollutants in Remote Mountain Lake Waters. J. Environ. Qual. 2001, 30, 1286–1295. [Google Scholar] [CrossRef] [PubMed]
- Prospero, J.M.; Glaccum, R.A.; Nees, R.T. Atmospheric transport of soil dust from Africa to South America. Nature 1981, 289, 570–572. [Google Scholar] [CrossRef]
- Garrison, V.H.; Majewski, M.S.; Foreman, W.T.; Genualdi, S.A.; Mohammed, A.; Massey Simonich, S.L. Persistent organic contaminants in Saharan dust air masses in West Africa, Cape Verde and the eastern Caribbean. Sci. Total Environ. 2014, 468–469, 530–543. [Google Scholar] [CrossRef] [PubMed]
- McKim, J.M. Physiological and biochemical mechanisms that regulate the accumulation and toxicity of environmental chemicals in fish. In Bioavailability—Physical, Chemical and Biological Interactions; Hamelink, J.L., Landrum, P.F., Bergman, H.L., Benson, W.H., Eds.; CRC Press, Lewis Publishers: Boca Raton, FL, USA, 1994; pp. 179–202. ISBN 9781566700863. [Google Scholar]
- Mackay, D.; Fraser, A. Bioaccumulation of Persistent Organic Chemicals: Mechanisms and Models. Environ. Pollut. 2000, 110, 375–391. [Google Scholar] [CrossRef]
- World Health Organization. Some Non-Heterocyclic Polycyclic Aromatic Hydrocarbons and Some Related Exposures. In Iarc Monographs on the Evaluation of Carcinogenic Risks to Humans; World Health Organization: Geneva, Switzerland, 2010; Volume 92, Available online: http://monographs.iarc.fr/ENG/Monographs/vol92/index.php (accessed on 27 October 2017).
- Lang, V. Polichlorinated biphenyls in the environment. J. Chromatogr. A 1992, 595, 1–43. [Google Scholar] [CrossRef]
- Tanabe, S. PCB problems in the future: Foresight from current knowledge. Environ. Pollut. 1988, 50, 5–28. [Google Scholar] [CrossRef]
- United Nations Environment Programme. UNEP 2009 Annual Report. Available online: http://hdl.handle.net/20.500.11822/7824 (accessed on 24 February 2018).
- World Health Organization. WHO Position on DDT Use in Disease Vector Control under the Stockholm Convention on Persistent Organic Pollutants; World Health Organization: Geneva, Switzerland, 2004; Available online: http://apps.who.int/iris/bitstream/10665/68631/1/WHO_HTM_RBM_2004.53.pdf (accessed on 24 February 2018).
- Bidleman, T.F.; Jantunen, M.L.; Karakus, P.K.; Wong, F. Chiral persistent organic pollutants as tracers of atmospheric sources and fate: Review and prospects for investigating climate change influences. Atmos. Pollut. Res. 2012, 3, 371–382. [Google Scholar] [CrossRef]
- McLachlan, M.S.; Welsch-Pausch, K.; Tolls, J. Field Validation of a Model of the Uptake of Gaseous SOC in Lolium multiflorum (Welsh Ray Grass). J. Environ. Sci. Technol. 1995, 29, 1998–2004. [Google Scholar] [CrossRef] [PubMed]
- Welsch-Pausch, K.; McLachlan, M.S.; Umlauf, G. Determination of the Principal Pathways of Polychlorinated Dibenzo-p-dioxins and Dibenzofurans to Lolium multiflorum (Welsh Ray Grass). Environ. Sci. Technol. 1995, 29, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Desalme, D.; Binet, P.; Bernard, N.; Gilbert, D.; Toussaint, M.-L.; Chiapusio, G. Atmospheric phenanthrene transfer and effects on two grassland species and their root symbionts: A microcosm study. Environ. Exp. Bot. 2011, 71, 146–151. [Google Scholar] [CrossRef]
- Huang, D.; Peng, P.; Xu, Y.; Deng, Y.; Deng, H. Distribution and deposition of polycyclic aromatic hydrocarbons in precipitation in Guangzhou, South China. J. Environ. Sci. 2009, 21, 654–660. [Google Scholar] [CrossRef]
- Callén, M.S.; Cruz, M.T.; López, J.M.; Mastral, A.M. PAH in airborne particulate matter. Fuel Process. Technol. 2011, 92, 176–182. [Google Scholar] [CrossRef]
- Dias, A.P.L.; Rinaldi, M.C.S.; Domingos, M. Foliar accumulation of polycyclic aromatic hydrocarbons in native tree species from the Atlantic Forest (SE—Brazil). Sci. Total Environ. 2016, 544, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Najmuddin, S.U.F.S.; Romli, M.F.; Hamid, M.; Alitheen, N.B.; Rahman, N.M.A.N.A. Anti-cancer effect of Annona muricata Linn Leaves Crude Extract (AMCE) on breast cancer cell line. BMC Complement. Altern. Med. 2016, 16, 311. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, A.; Figadere, B.; Zafra-Polo, M.-C.; Barrachina, I.; Estornell, E.; Diego Cortes, D. Acetogenins from Annonaceae: Recent progress in isolation, synthesis and mechanisms of action. Nat. Prod. Rep. 2005, 22, 269–303. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.C.; Haynes, L.J.; Magnus, K.E.; Plimmer, J.R.; Sherrat, H.S.A. Pharmacological screening of some West Indian medicinal plants. J. Pharm. Pharmacol. 1962, 14, 556–612. [Google Scholar] [CrossRef] [PubMed]
- Zeng, P.C.; Feng, E.W.; Oberlies, N.H.; McLaughlin, J.L. Five new Monotetrahydrofuran Ring Acetogenins from the Leaves of Annona muricata. J. Nat. Prod. 1996, 59, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.E.; Zeng, L.; Gu, Z.-M.; Zhao, G.-X.; Zhang, Y.; Schwedler, J.T.; McLaughlin, J.L.; Sastrodihardjo, S. Muricatocins A and B, Two New Bioactive Monotetrahydrofuran Annonaceous Acetogenins from the Leaves of Annona muricata. J. Nat. Prod. 1995, 58, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.B.P.; Brustolim, D.; Santos, L.A.; Bellintani, M.C.; Paiva, F.P.; Ribeiro, Y.M.; Tomassini, T.C.B.; Ribeiro dos Santos, R. Physalins B, F and G, seco-steroids purified from Physalis angulata L., inhibit lymphocyte function and allogeneic transplant rejection. Int. Immunopharmacol. 2006, 6, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.; Jaw, S.M.; Chen, P.M. Inhibitory effects of physalin B and physalin F on various human leukemia cells in vitro. Anticancer Res. 1992, 12, 1155–1162. [Google Scholar] [PubMed]
- Bastos, G.N.T.; Silveira, A.J.A.; Salgado, C.G.; Picanço-Diniz, D.L.W.; do Nascimento, J.L.M. Physalis angulata extract exerts anti-inflammatory effects in rats by inhibiting different pathways. J. Ethnopharmacol. 2008, 118, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Thevissen, K.; François, I.E.; Sijtsma, L.; van Amerongen, A.; Schaaper, W.M.; Meloen, R.; Posthuma-Trumpie, T.; Broekaert, W.F.; Cammue, B.P. Antifungalactivity of synthetic peptides derived from Impatients balsamina antimicrobial peptides Ib-AMP1 and Ib-AMP4. Peptides 2005, 26, 1113–1119. [Google Scholar] [CrossRef] [PubMed]
- Hamelin, G.; Brochu, C.; Moore, S. Application of tandem mass spectrometry with an ion trap detector to the analysis of polychlorinated dibenzo-p-dioxins/polychlorinated dibenzofurans and polychlorinated biphenyls. Organohalogen Compd. 1995, 23, 125–130. [Google Scholar]
- Gruppo di Lavoro per i Residui di Antiparassitari della Commissione Permanente di Coordinamento Interregionale per i Problemi Relativi al Controllo Ufficiale dei Prodotti Alimentary. Metodi multiresiduo per l’analisi di residui antiparassitari in prodotti vegetali. In Rapporti ISTISAN 97/23; Istituto Superiore di Sanità: Roma, Italy, 1997; Available online: http://www.iss.it/binary/publ/cont/1123-3117_1997_23_1.1146226165.pdf (accessed on 24 February 2018).
- De Felip, E.; Miniero, R. Procedimenti analitici adottati per il rilevamento di microcontaminanti in sedimenti lagunari. In Rapporti ISTISAN 99/28; IstitutoSuperiore di Sanità: Roma, Italy, 1999. [Google Scholar]
- Anastassiades, M.; Lehotay, S.J.; Stajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [PubMed]
- EPA 3620B Method by “United States Environmental Protection Agency”. Available online: http://www.cromlab.es/Articulos/Metodos/EPA/3000/3620B.PDF (accessed on 26 October 2017).
- Falcinelli, S.; Giorgini, M.; Sebastiani, B. Phytosterols and Fatty Acids analytical determination on Lodoicea maldivica fruit. In Applied Engineering Sciences; Deng, W., Ed.; Taylor & Francis Group Publishers: London, UK, 2015; Chapter 19; pp. 99–104. ISBN 978-1-138-02649-0. [Google Scholar]
- Falcinelli, S.; Malfatti, D.; Giorgini, M.; Sebastiani, B. Determination of Volatile Aroma Composition Profiles of Coco de Mèr (Lodoicea maldivica) Fruit: Analytical Study by HS-SPME and GC/MS Techniques. In ICCSA 2017, Part III, Lecture Notes in Computer Science LNCS 10406; Gervasi, O., Murgante, B., Misra, S., Borruso, G., Torre, C.M., Rocha, A.M.A.C., Taniar, D., Apduhan, B.O., Stankova, E., Cuzzocrea, A., Eds.; Springer: Cham, Switzerland, 2017; pp. 44–59. [Google Scholar] [CrossRef]
- Falcinelli, S.; Bettoni, M.; Giorgini, F.; Giorgini, M.; Sebastiani, B. Chemical Characterization of “Coco de Mer” (Lodoicea maldivica) Fruit: Phytosterols and Fatty Acids Composition. In ICCSA 2015, Part II, Lecture Notes in Computer Science LNCS 9156; Gervasi, O., Murgante, B., Misra, S., Gavrilova, M.L., Rocha, A.M.A.C., Torre, C.M., Taniar, D., Apduhan, B.O., Eds.; Springer: Cham, Switzerland, 2015; pp. 308–323. [Google Scholar] [CrossRef]
- Sebastiani, B.; Giorgini, M.; Falcinelli, S. Chemical characterization of Lodoicea maldivica fruit. Chem. Biodivers. 2017, 14, e1700109. [Google Scholar] [CrossRef] [PubMed]
- “EU Pesticides Database” by the European Commission. Available online: http://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/public/ (accessed on 26 October 2017).
- Calamari, D.; Bacci, E.; Focardi, S.; Gaggi, C.; Morosini, M.; Vighi, M. Role of Plant Biomass in the Global Environmental Partitioning of Chlorinated Hydrocarbons. Environ. Sci. Technol. 1991, 25, 1489–1495. [Google Scholar] [CrossRef]
- Traina, M.E.; Urbani, E.; Rescia, M.; Mantovani, A. L’insetticida lindano: Identificazione dei rischi possibili per la riproduzione umana. In Rapporti ISTISAN 01/03; Istituto Superiore di Sanità: Roma, Italy, 2001. [Google Scholar]
- Simonich, S.L.; Hites, R.A. Global distribution of persistent organochlorine compounds. Science 1995, 269, 1851–1854. [Google Scholar] [CrossRef]
- Dachs, J.; Bayona, J.M.; Ittekkot, V.; Albaiges, J. Monsoon-driven vertical fluxes of organic pollutants in the Western Arabian Sea. Environ. Sci. Technol. 1999, 33, 3949–3956. [Google Scholar] [CrossRef]
- Pacyna, J.M.; Oehme, M. Long-range transport of some organic compounds to the Norwegian Arctic. Atmos. Environ. 1988, 22, 243–257. [Google Scholar] [CrossRef]
- Brubaker, W.W., Jr.; Hites, R.A. OH Reaction Kinetics of Gas-Phase α- and γ-Hexachlorocyclohexane and Hexachlorobenzene. Environ. Sci. Technol. 1998, 32, 766–769. [Google Scholar] [CrossRef]
- Cortes, D.R.; Basu, I.; Sweet, C.W.; Brice, K.A.; Hoff, R.M.; Hites, R.A. Temporal trends in gas-phase concentrations of chlorinated pesticides measured at the shores of the Great Lakes. Environ. Sci. Technol. 1998, 32, 1920–1927. [Google Scholar] [CrossRef]
- Miglioranza, K.S.B.; Aizpún de Moreno, J.E.; Moreno, V.J.; Osterrieth, M.L.; Escalante, A.H. Fate of organochlorine pesticides in soils and terrestrial biota of ‘‘Los Padres’’ pond watershed, Argentina. Environ. Poll. 1999, 105, 91–99. [Google Scholar] [CrossRef]
- Tsutsui, K.; Ubuka, T.; Kanda, S.; Oka, Y.; Osugi, T.; Ukena, K.; Tachibana, T.; Sakamoto, T.; Amano, M.; Ando, H.; et al. Handbook of Hormones Comparative Endocrinology for Basic and Clinical Research, 1st ed.; Takei, Y., Ando, H., Tsutsui, K., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 978-0-12-801028-0. [Google Scholar]
- UNEP/IOC/IAEA. Sampling of Selected Marine Organisms and Sample Preparation for the Analysis of Chlorinated Hydrocarbons; United Nations Environment Programme: Nairobi, Kenya, 1991. [Google Scholar]
- Hellou, J.; Lebeuf, M.; Rudi, M. Review on DDT and metabolites in birds and mammals of aquatic ecosystems. Environ. Rev. 2013, 21, 53–69. [Google Scholar] [CrossRef]
- Bosch, C.; Grimalt, J.O.; Fernández, P. Enantiomeric fraction and isomeric composition to assess sources of DDT residues in soils. Chemosphere 2015, 138, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Griffing, S.M.; Gamboa, D.; Udhayakumar, V. The history of 20th century malaria control in Peru. Malar. J. 2013, 12, 303. [Google Scholar] [CrossRef] [PubMed]
- Brennan, T. Globalization and Its Terrors: Daily Life in the West, 1st ed.; ROUTLEDGE Psychology Press, Taylor & Francis Group: Abingdon, UK, 2003; ISBN 978-0415285223. [Google Scholar]
- Cliath, M.M.; Spencer, W.F. Dissipation of pesticides from soil by volatilization of degradation products. I. Lindane and DDT. Environ. Sci. Technol. 1972, 6, 910–914. [Google Scholar] [CrossRef]
- Da, C.; Liu, G.; Tang, Q.; Li, J. Distribution, sources, and ecological risks of organochlorine pesticides in surface sediments from the Yellow River Estuary, China. Environ. Sci. Process. Impacts 2013, 15, 2288–2296. [Google Scholar] [CrossRef] [PubMed]
- Doong, R.; Sun, Y.; Liao, P.; Peng, C.; Wu, S. Distribution and fate of organochlorine pesticide residues in sediments from the selected rivers in Taiwan. Chemosphere 2002, 48, 237–246. [Google Scholar] [CrossRef]
- Bekvar, N.; Lotufo, G.R. DDT and Other Organohalogen Pesticides in Aquatic Organisms. University of Nebraska; Lincoln DigitalCommons@University of Nebraska: Lincoln, NE, USA, 2011; Available online: http://digitalcommons.unl.edu/cgi/viewcontent.cgi?article=1247&context=usepapapers (accessed on 27 October 2017).
- Mishra, M.; Kumar, B.; Singh, S.K.; Goel, G.; Gaur, R.; Prakash, D.; Kumar, S.; Sharma, C.S. Hexachlorohexane (HCH) & Dichlorodimethyltrichloroethane (DDT) in soils from Northern Uttar Pradesh, India. Eur. J. Exp. Biol. 2011, 1, 162–168. [Google Scholar]
- Manaca, M.N.; Grimalt, J.O.; Garí, M.; Sacarlal, J.; Sunyer, J.; Gonzalez, R.; Dobaño, C.; Menendez, C.; Alonso, P.L. Assessment of exposure to DDT and metabolites after indoor residual spraying through the analysis of thatch material from rural African dwellings. Environ. Sci. Pollut. Res. Int. 2012, 19, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Zhu, T.; Yao, B.; Hu, S. Contribution of Dicofol to the current DDT pollution in China. Environ. Sci. Technol. 2005, 39, 4385–4390. [Google Scholar] [CrossRef] [PubMed]
- Breivik, K.; Sweetman, A.; Pacyna, J.M.; Jones, K.C. Towards a global historical emission inventory for selected PCB congeners—A mass balance approach: 1. Global production and consumption. Sci. Total Environ. 2002, 290, 181–198. [Google Scholar] [CrossRef]
- Schuster, J.K.; Gioia, R.; Sweetman, A.J.; Jones, K.C. Temporal trends and controlling factors for polychlorinated biphenyls in the UK atmosphere (1991–2008). Environ. Sci. Technol. 2010, 44, 8068–8074. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Gong, S.L.; Zhao, T.L.; Neary, L.; Barrie, L.A. GEM/POPs: A global 3-D dynamic model for semi-volatile persistent organic pollutants; Part 2: Global transports and budgets of PCBs. Atmos. Chem. Phys. 2007, 7, 4015–4025. [Google Scholar] [CrossRef]

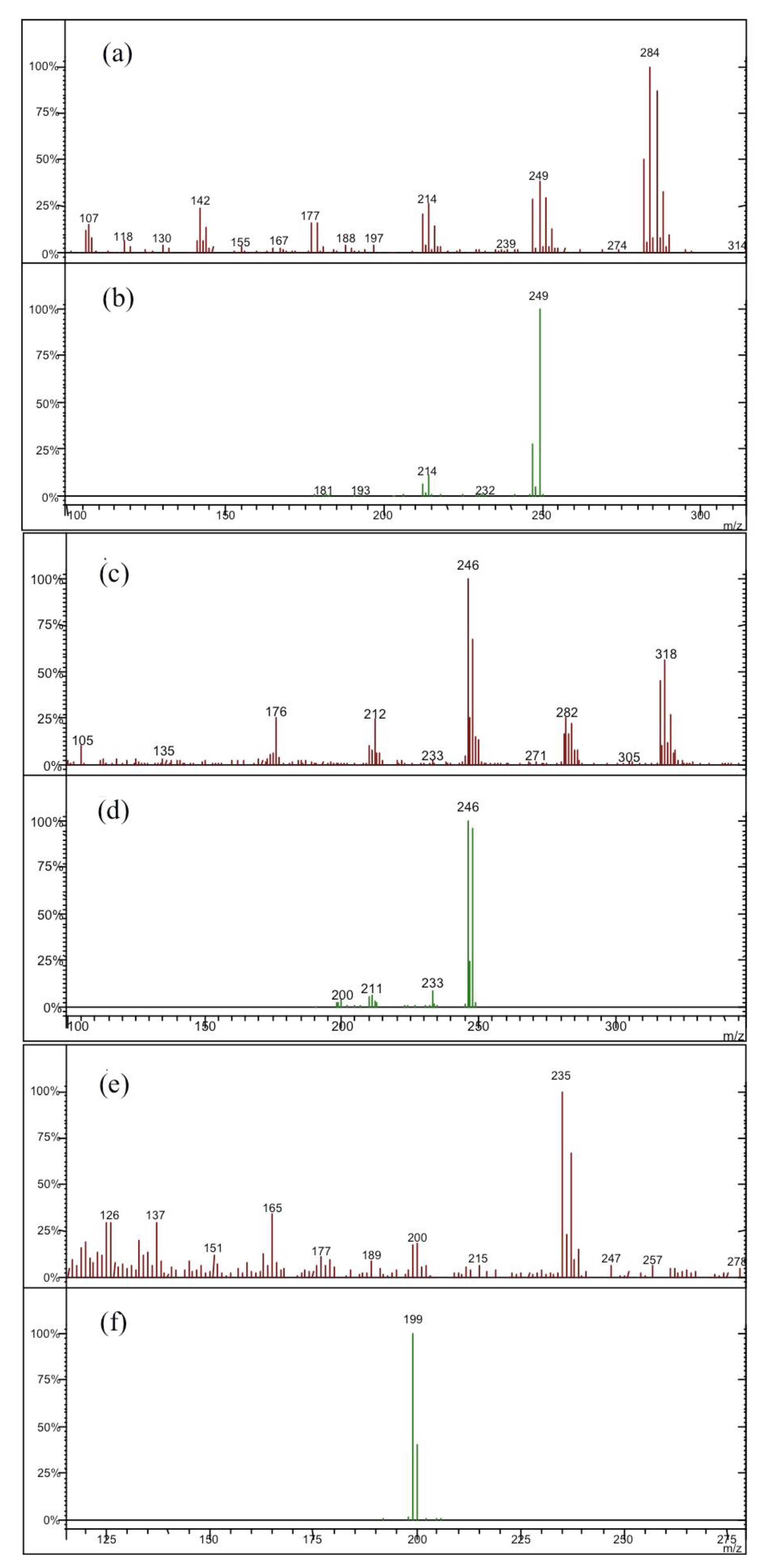
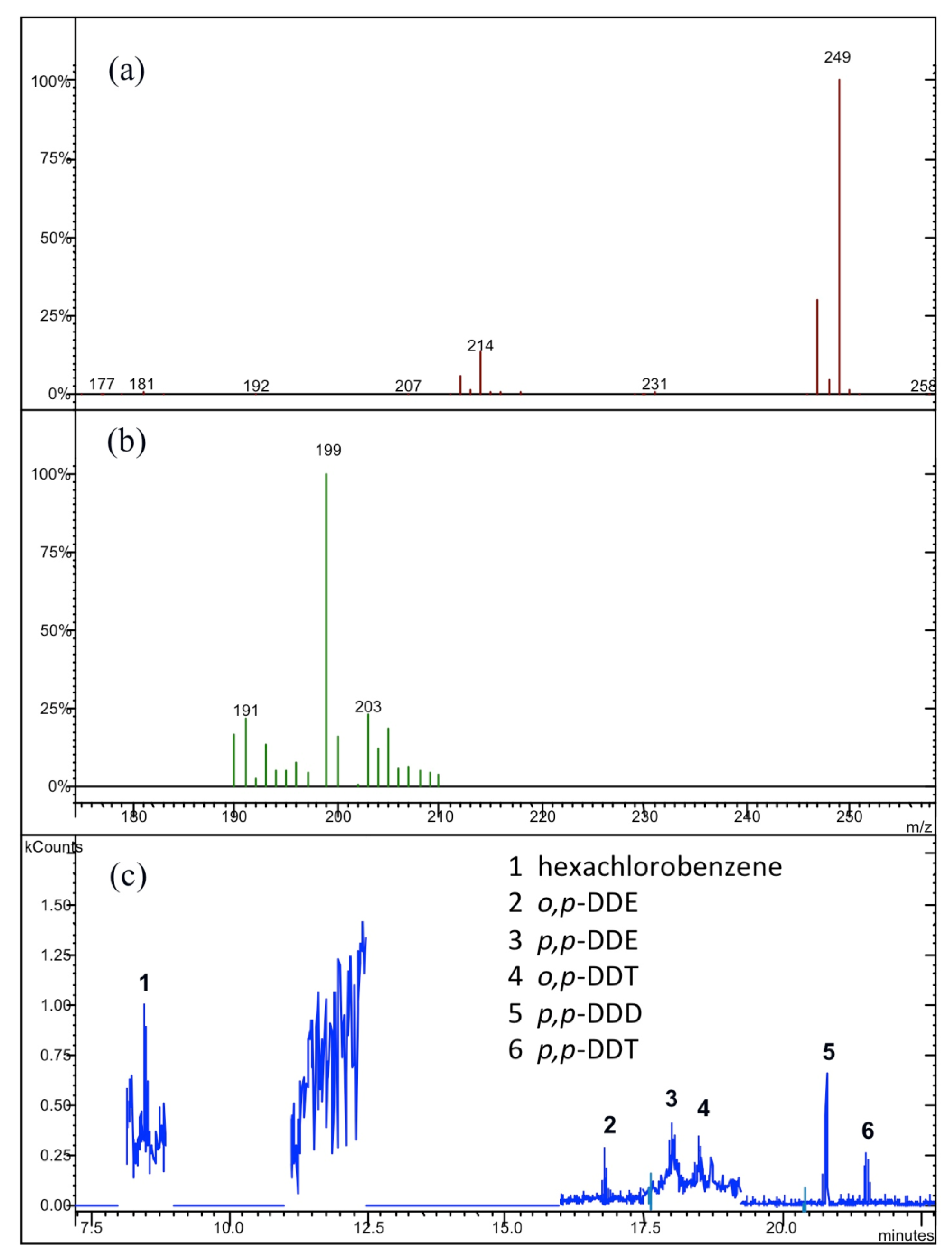



| Compound | Parent Ion Mass | Window (u.m.a.) | Waveform | CID 1 (V) |
|---|---|---|---|---|
| α-HCH 2 | 219 [M-Cl2] | 1 | Resonant | 0.75 |
| β-HCH | ||||
| γ-HCH (lindane) | ||||
| hexachlorobenzene | 284 [M+2] | 1 | Resonant | 2.00 |
| heptachlor | 272 [M-C5H5Cl] | 1 | Resonant | 1.00 |
| o,p-DDE 3 | 318 [M+2] | 1 | Resonant | 2.00 |
| p,p-DDE | ||||
| o,p-DDD 4 | 235 [M-CH2Cl2] | 1 | Resonant | 1.50 |
| p,p-DDD | ||||
| o,p-DDT 5 | 235 [M-CCl3] | 1 | Resonant | 1.50 |
| p,p-DDT |
| Compound | Graviola (ng/g d.w. 1) | Mullaca (ng/g d.w. 1) | Balsamina (ng/g d.w. 1) |
|---|---|---|---|
| α-HCH 2 | <0.030 | <0.030 | <0.030 |
| β-HCH 2 | <0.030 | <0.030 | <0.030 |
| γ-HCH 2 (lindane) | <0.030 | <0.030 | <0.030 |
| hexachlorobenzene | 0.041 | <0.007 | <0.007 |
| heptachlor | <0.020 | <0.020 | <0.020 |
| o,p-DDE | 0.040 | 0.062 | <0.030 |
| p,p-DDE | 0.129 | 0.093 | 0.615 |
| o,p-DDD | <0.020 | <0.020 | 0.164 |
| p,p-DDD | 0.248 | 0.109 | 0.426 |
| o,p-DDT | 0.102 | <0.020 | <0.020 |
| p,p-DDT | 0.054 | 0.085 | 0.124 |
| Total | 0.614 | 0.349 | 1.329 |
| Sample | Graviola (ng/g d.w. 1) | Mullaca (ng/g d.w. 1) | Balsamina (ng/g d.w. 1) |
|---|---|---|---|
| 1 | 0.449 | 0.651 | 1.373 |
| 2 | 0.920 | 0.220 | 0.970 |
| 3 | 0.584 | 0.291 | 1.525 |
| 4 | 0.532 | 0.502 | 1.458 |
| 5 | 0.446 | 0.108 | 0.787 |
| 6 | 0.753 | 0.322 | 1.861 |
| Averaged value 2 | 0.614 ± 0.171 | 0.349 ± 0.180 | 1.329 ± 0.357 |
| Compound Ratio | Graviola | Mullaca | Balsamina |
|---|---|---|---|
| DDT/DDE | 0.9 | 0.5 | 0.2 |
| DDD/DDE | 1.9 | 1.2 | 0.7 |
| (DDE + DDD)/DDTs | 0.7 | 0.6 | 0.8 |
| o,p-DDT/p,p-DDT | 1.9 | 0 | 0 |
| Compound | Graviola (ng/g d.w.) | Mullaca (ng/g d.w.) | Balsamina (ng/g d.w.) |
|---|---|---|---|
| TCB 28 | <0.004 | <0.004 | <0.004 |
| TeCB 52 | <0.003 | <0.003 | <0.003 |
| PeCB 101-118 | <0.006 | <0.006 | <0.006 |
| HxCB 138-153 | <0.013 | <0.013 | <0.013 |
| HpCB 180 | <0.011 | <0.011 | <0.011 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sebastiani, B.; Falcinelli, S. Contamination of Plants from Amazonia by Environmental Pollution. Environments 2018, 5, 33. https://0-doi-org.brum.beds.ac.uk/10.3390/environments5030033
Sebastiani B, Falcinelli S. Contamination of Plants from Amazonia by Environmental Pollution. Environments. 2018; 5(3):33. https://0-doi-org.brum.beds.ac.uk/10.3390/environments5030033
Chicago/Turabian StyleSebastiani, Bartolomeo, and Stefano Falcinelli. 2018. "Contamination of Plants from Amazonia by Environmental Pollution" Environments 5, no. 3: 33. https://0-doi-org.brum.beds.ac.uk/10.3390/environments5030033