3.1. Biomass and Biochar Properties
The composition of biomass is presented in
Table 3. The moisture content of the biomass was 1.73%. Low moisture content is commonly reported in the literature for wood waste biomass [
13,
14]. The ash content (31.5%) is similar to results reported by Chen and Chen [
41] for hardwood (38.55%). Biochar yield for the three different pyrolysis temperatures 350, 450 and 550 °C are shown in
Table 3. Biochar yield had an inverse relation with pyrolysis temperature. This behaviour was expected as wood biomass has a high content of cellulose, lignin and hemicellulose [
14,
42]. In addition, a decrease in yield as the temperature increased can be attributed to great decomposition of primary biomass or to secondary decomposition of biochar according to Ola and Jekayinfa [
43]. The P content in the raw biochar ranged 0.0482–0.345 mg·g
−1, as shown in
Table 3. Experiments were performed to determine the amount of P released from raw biochar to evaluate its availability. The release of P from biochar was very low, thus it is unlikely to be readily available.
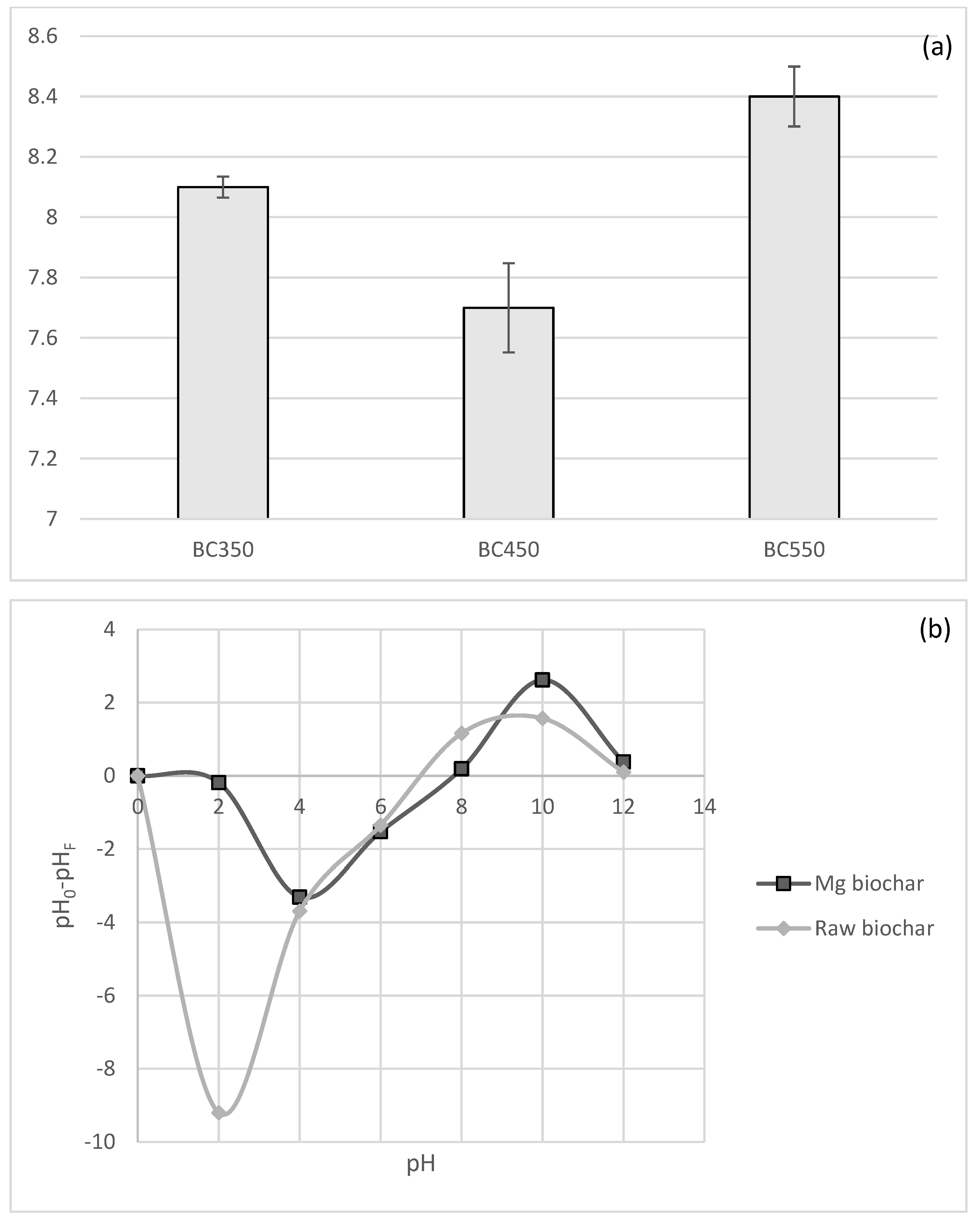
The pH of biochar prepared at 350, 450 and 550 °C was alkaline in nature with values 8.1, 8.2 and 8.4, respectively. The increase in pH values with increase in pyrolysis temperature can be caused by the separation of alkali salts from organic materials, as well as the loss of acidic functional groups and the appearance of basic functional groups [
42,
44]. Oginni and Yakaboylu [
45] reported an increase in pH after pyrolysis of wood biomass from longleaf pine, red oak and hard maple at 500 °C; their pH values were 8.6, 9.0 and 9.8, respectively. The modified biochar, Mg-biochar, had alkaline nature (pH = 8.5). Generally, very little change in the pH was observed after the modification process.
Figure 2 shows the SEM and EDS spectrum of the biochar (before modification) with MgCl2 and after modification (Mg-Biochar). The SEM scans reveal that the pore size of the Mg-Biochar is larger and more developed. Furthermore, the EDS spectra confirms that the Mg presence is more prominent in the Mg-Biochar than in the unmodified biochar.
The specific surface area of the biochar prepared at 500 °C was 1.49 m
2·g
−1 and the pore volume is 0.000427 m
3·g
−1. Similar results were reported for brown marine macroalgae and woody derived biochar by Oginni and Yakaboylu [
45] and Jung and Ahn [
7] with SSAs of 2.39 and 0.26 m
2 g
−1, respectively. For the Mg-biochar (500 °C) produced here, the SSA was 0.97 m
2·g
−1 and the pore volume was 0.0003 m
3·g
−1. The development of larger pores with wider diameters in the modified can explain the reduction in the BET SSA. As the pore diameter grew, it consumed the smaller pores surrounding it, resulting in loss of nano-surface area. This is not necessarily bad because, for many of the small pores, despite providing higher surface area, the surface area and the active sites within them may not be accessible. Other studies have reflected the influence of residence time and pore clogging to be the some of the causes that affect SSA. Lu and Zong [
46] reported an increase of SSA in maize straw and rice biochar pyrolysed at 550 °C when residence time was increased from 2 to 5 h. Li and Dong [
47] noted how high pyrolysis temperatures can cause the pores to collapse reducing the SSA of biochar. Furthermore, it is important to note that the MB method measures the mesoporous surface area of the biochar, which means MB may not reflect the total surface area available for adsorption which includes the micropores. It was also found that the pH
pzc of the biochar increased from 7 (raw) to 8 for the Mg-biochar (550 °C), as shown in
Figure 3.
FTIR analysis was performed to analyse the functional groups in both raw and modified biochar. The resulting FTIR spectra are shown in
Figure 4. Biochar produced at 350 °C exhibited a broad band between 3000 and 3500 cm
−1 (peak maximum at 3327 cm
−1) which represents vibrations of hydrogen-bonded O-H groups (alcoholic and phenolic) as well as the double-bonded groups C=O broad band between 1500 and 2000 cm
−1 (peak maximum at 1689 cm
−1) [
14]. For all raw biochar, adsorption in the region between 2843 and 2981 cm
−1 C-H stretching is allocated to aliphatic functional groups and another strong band at 1055 cm
−1 represents the C-O stretching oxygenated groups. The strong band at 1224 cm
−1 represents phenolic -OH groups [
42]. As temperature increases from 350 to 550 °C, OH functional groups, as well as C=O groups, decrease.
The strong aromatic band of C=C at 1602 cm
−1 is observed in the biochar synthesised at 350 °C, and this vibration is strongly present until the temperature reached 550 °C, at which the band is reduced. This behaviour in biochar produced at high temperatures can be related to their low H/C ratio according to Zhang and Chen [
48]. The intensities of C-H (2843 and 2981 cm
−1) aliphatic stretching, -OH phenolic (1224 cm
−1) and C-O bands (1055 cm
−1) exhibited a significant decrease because of the dehydration and degradation of ligneous and cellulosic compounds [
43]. Functional groups present after modification of biochar by MgCl
2 and adsorption of phosphorus are shown in
Figure 4. There is a significant peak for all biochar in the band stretching from 1500 to 2000 cm
−1 (1578 cm
−1), indicating functional groups C=C and C=O aromatic bonds for modified biochar and modified biochar loaded with phosphorus as well as for raw biochar loaded with phosphorus. For modified biochar in the bands of 3299 and 699 cm
−1, groups assigned Mg-OH and Mg-O were detected confirming the incorporation of Mg in the biochar [
25,
49]. Another significant change for biochar after modification was a sharp increase in the band 1577 cm
−1 for all biochar samples except raw biochar, which can be attributed to the aromatic functional groups C=C and C=O. Compared with raw biochar, Mg-biochar contained groups in the intensity bands from 2500 to 3000 cm
−1 (2906 cm
−1) and 1000 to 1500 cm
−1 and these were assigned to CH
2 groups and C-O groups [
50]. After the HW-biochar and Mg-biochar were loaded with phosphorus, a significant increase in the stretching bands of 1408, 1023, 874 and 559 cm
−1 were observed for HW-biochar with functional groups C-H alkene, C-O and C-H aromatic out of plane. Regarding modified biochar, with the loading of P, the peaks located at 3299 and 699 cm
−1 disappeared, indicating this group can be linked to the adsorption of phosphorus into the Mg-biochar [
25].
3.2. Adsorption of Phosphorus by HW-Biochar and Mg-Biochar
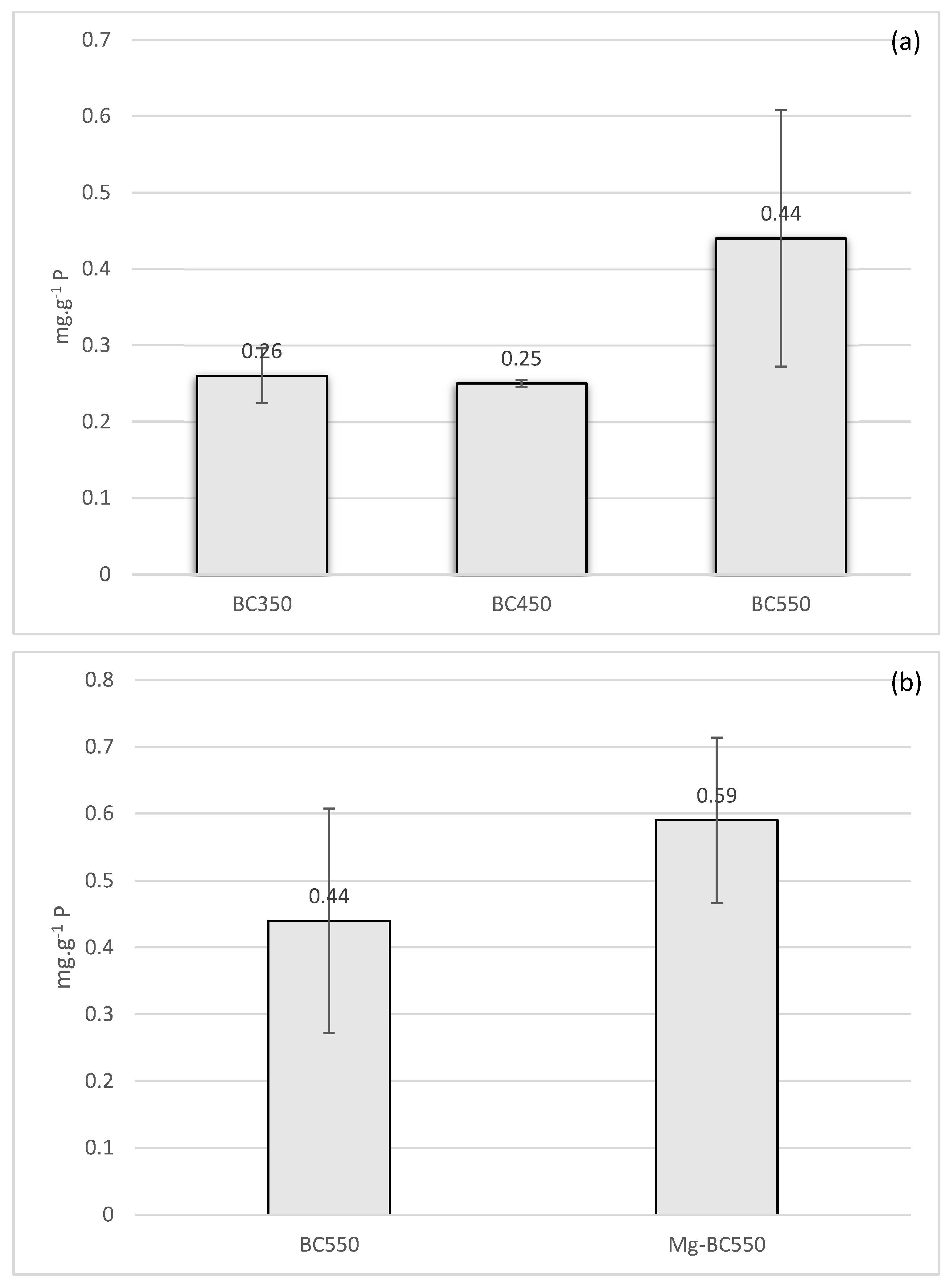
The phosphorus adsorption capacities of raw biochar (HW-biochar) and Mg modified biochar (Mg-biochar) are shown in
Figure 5. The raw biochar pyrolysed at 350 and 450 °C showed low adsorption capacity (0.26 and 0.25 mg-P·g
−1, respectively), as shown in
Figure 5a. On the other hand, raw biochar synthesised at 550 °C showed the highest adsorption of phosphorus with 0.44 mg-P·g
−1; although the ANOVA test failed to confirm that the results were significantly different (
p = 0.22), the BC550 adsorbed almost double the amount of phosphate compared to the other biochars. As pyrolysis temperature increases, biochar surface area and pore formation increase enhancing biochar adsorption capacity of phosphorus [
26,
51]. Similar results were found by Zhou and Xu [
52] and Yin and Liu [
25] with the increase in adsorption capacity of P for raw biochar with an increase in pyrolysis temperature. Because of its adsorption capacity, HW-biochar at 550 °C was chosen to be modified with Mg.
Biochar pyrolysed at 550 °C was selected to be modified with MgCl
2. Modified biochar with Mg showed an increase in the adsorption of P from 0.44 to 0.59 mg-P·g
−1 (
Figure 5). Although t-test failed to confirm the significance of the improvement (
p = 0.2), the change represents 34% improvement over unmodified biochar. Improvement in the performance of biochar by the addition of Mg has been presented in the literature. Fang and Zhang [
24] reported maximum adsorption of P of modified corn biochar was higher compared to raw biochar (239 and 225 mg·g
−1). Similar results were published by Yin and Liu [
25], describing biochar modified with Mg dramatically increased phosphate adsorption. However, the results reported in [
24,
25] were obtained using extremely high initial concentrations of phosphate solutions. The Mg-biochar developed in this study was also able to achieve 238 mg·g
−1 when it was subjected to PO
4-P solution with initial concentration of 30 g·L
−1. The dramatic increase may be explained by the increased driving force due to the extreme difference between the PO
4-P concentration in the solid and liquid phases. The modification with magnesium proved to be crucial for enhancing the adsorption capacity of raw biochar as it increased the positive surface charge on the biochar, thus increasing the tendency to attract anions. Mg modified biochar in the literature has shown a high affinity to anions due to the high pH
PZC (12), as this causes the surface to be positively charged [
45,
53]. In our study, the HW-biochar and Mg-biochar pH
PZC values were 7.0 and 7.9. As the pH
PZC was higher than pH from the solution (pH = 5), a positive surface in the Mg-biochar can be expected, which, due to electrostatic adsorption between the adsorbate and the biochar, can contribute to improving adsorption of PO
43− [
6,
25]. Moreover, the adsorption of phosphate increased 34% over the raw biochar to Mg-biochar despite the decrease in the SSA.
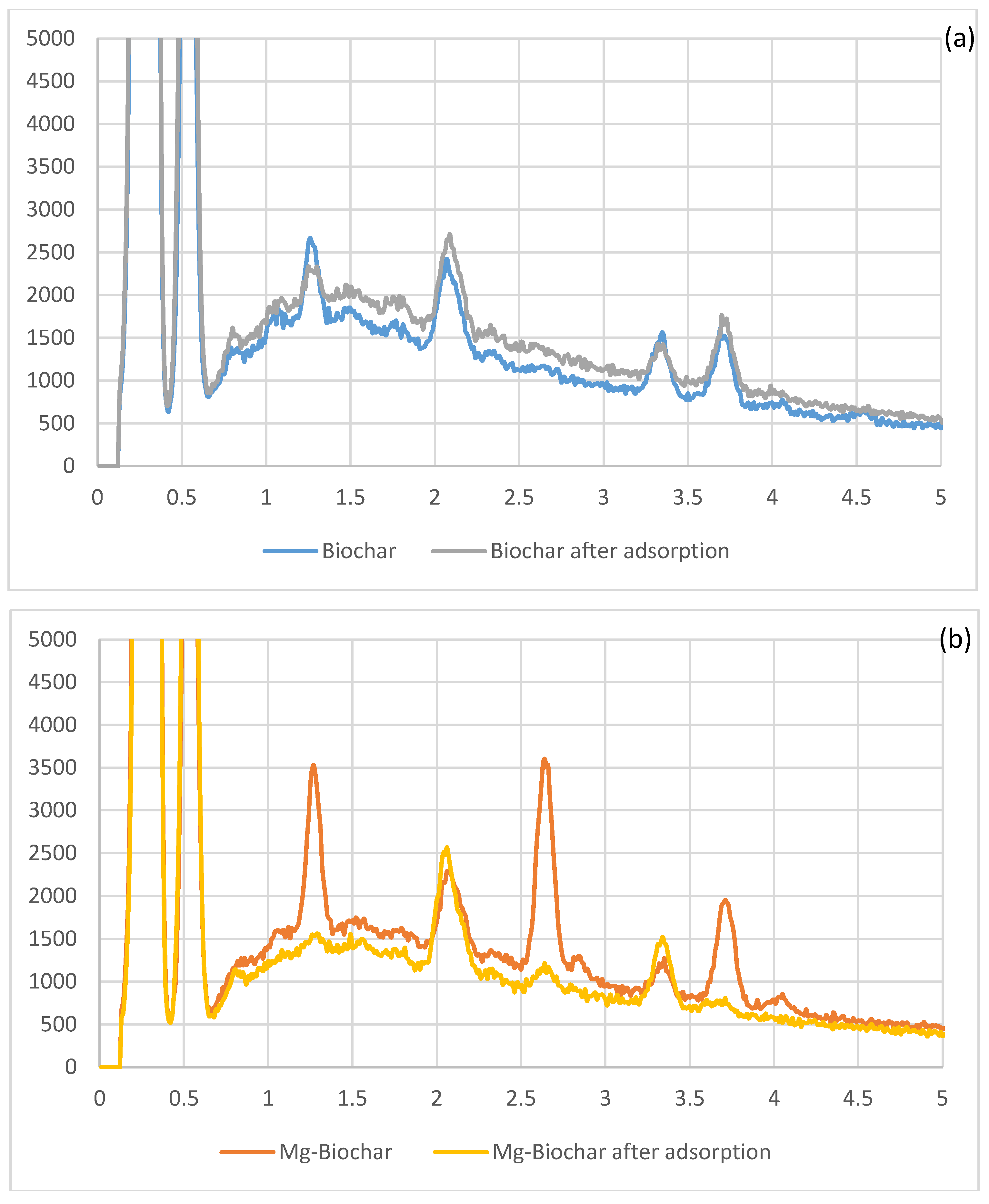
Figure 6 shows the EDS spectra of the biochar before and after adsorption. It can be seen that Mg has played a role in phosphate adsorption in the unmodified and modified biochar. However, Mg’s role can be easily seen in the modified biochar with the peak around 1.27 (corresponding to Mg) disappearing after adsorption. It can also be seen that the chloride (around 2.65) and calcium (3.73) have also have been involved in the adsorption of phosphate. Marshall and Morton [
54] proposed a multi-step mechanism for the formation of calcium phosphate on biochar in which Cl
− played an important role. We propose a similar mechanism for the formation of magnesium phosphate on biochar.
3.4. Adsorption Kinetics
The adsorption of phosphate by Mg-biochar as a function of time is presented in
Figure 8. It is clear that adsorption of PO
43− at the initial time is fast and then becomes relatively slow. Maximum adsorption was reached in 60 min. The fast initial adsorption of PO
43− can be caused by electrostatic forces between the positively charged Mg surface and the slow adsorption later might be due to intraparticle diffusion [
5,
53,
56]. The data from the kinetic experiment were further fitted using the pseudo-first- (PFO) and pseudo-second-order (PSO) kinetic models. The pseudo-first- and the pseudo-second-order models fitted the experimental data with correlation coefficients R
2 = 0.71 and 0.73, respectively. The fitted parameters are shown in
Table 6. The PSO model assumes the mechanism of P adsorption onto Mg-biochar can be determined by chemisorption interaction, which agrees with the isotherm results [
5,
53].
The kinetics data indicate a degree of overshooting behaviour with q
t oscillating as time progresses. One possibility to explain this phenomenon is that, as the biochar has more active positive surface due to the addition of Mg, P is attached to this surface; however, the bonding seems to be weak. When the surface of biochar reaches the critical charge density, the new surface charge of biochar is negative because of the phosphate, therefore it starts repelling the incoming phosphate (
Figure 9), which looks like there is a release instead of adsorption until equilibrium is reached.