Assessing the Role of Cannabis Use on Cortical Surface Structure in Adolescents and Young Adults: Exploring Gender and Aerobic Fitness as Potential Moderators
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedures
2.3. Measures
2.3.1. Detailed Phone Screen
2.3.2. Session Measures
2.4. Statistical Analysis
3. Results
3.1. Demographic Data
3.2. Primary Analyses
3.2.1. Cannabis Results
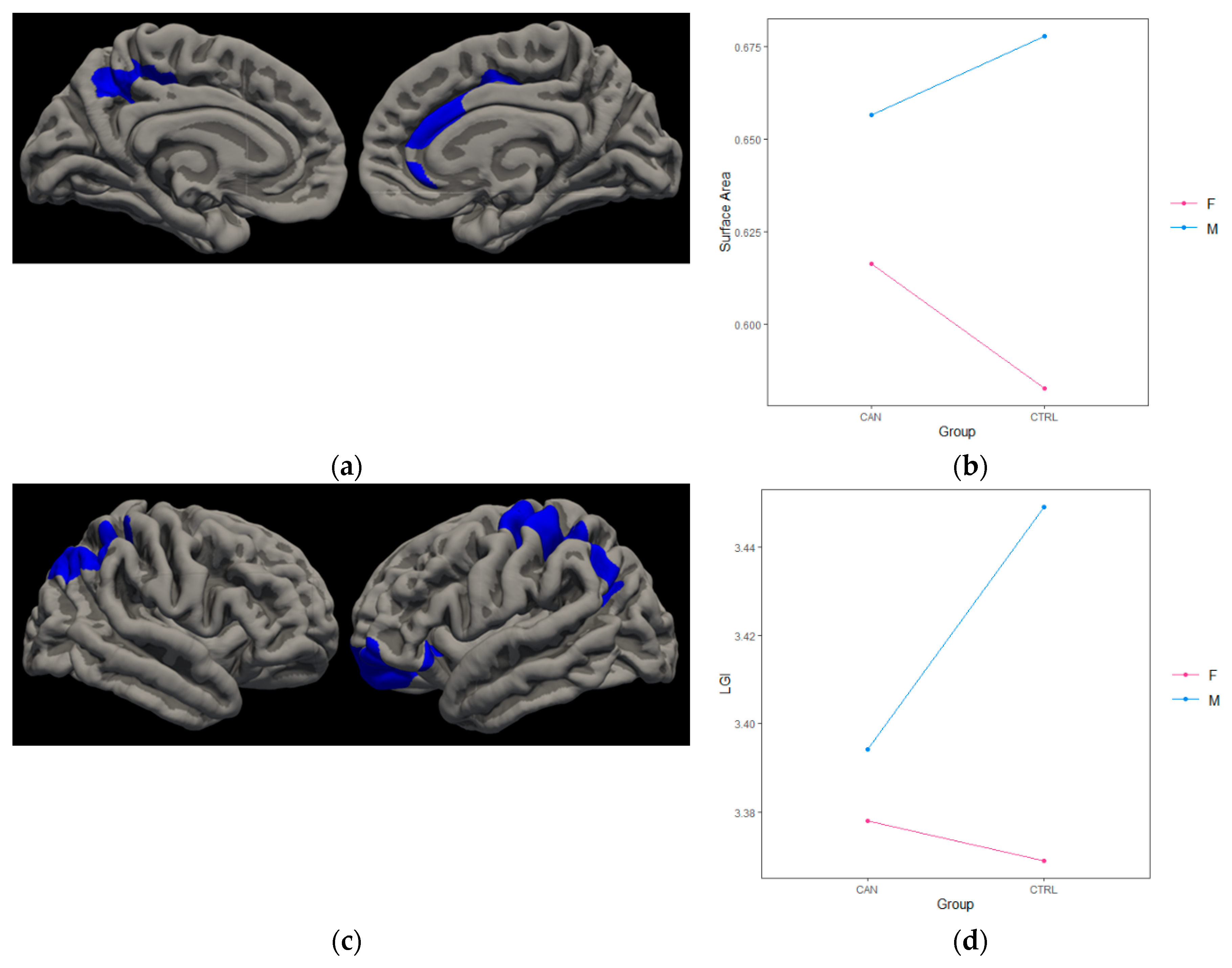
3.2.2. Cannabis*Gender
3.2.3. VO2 Results
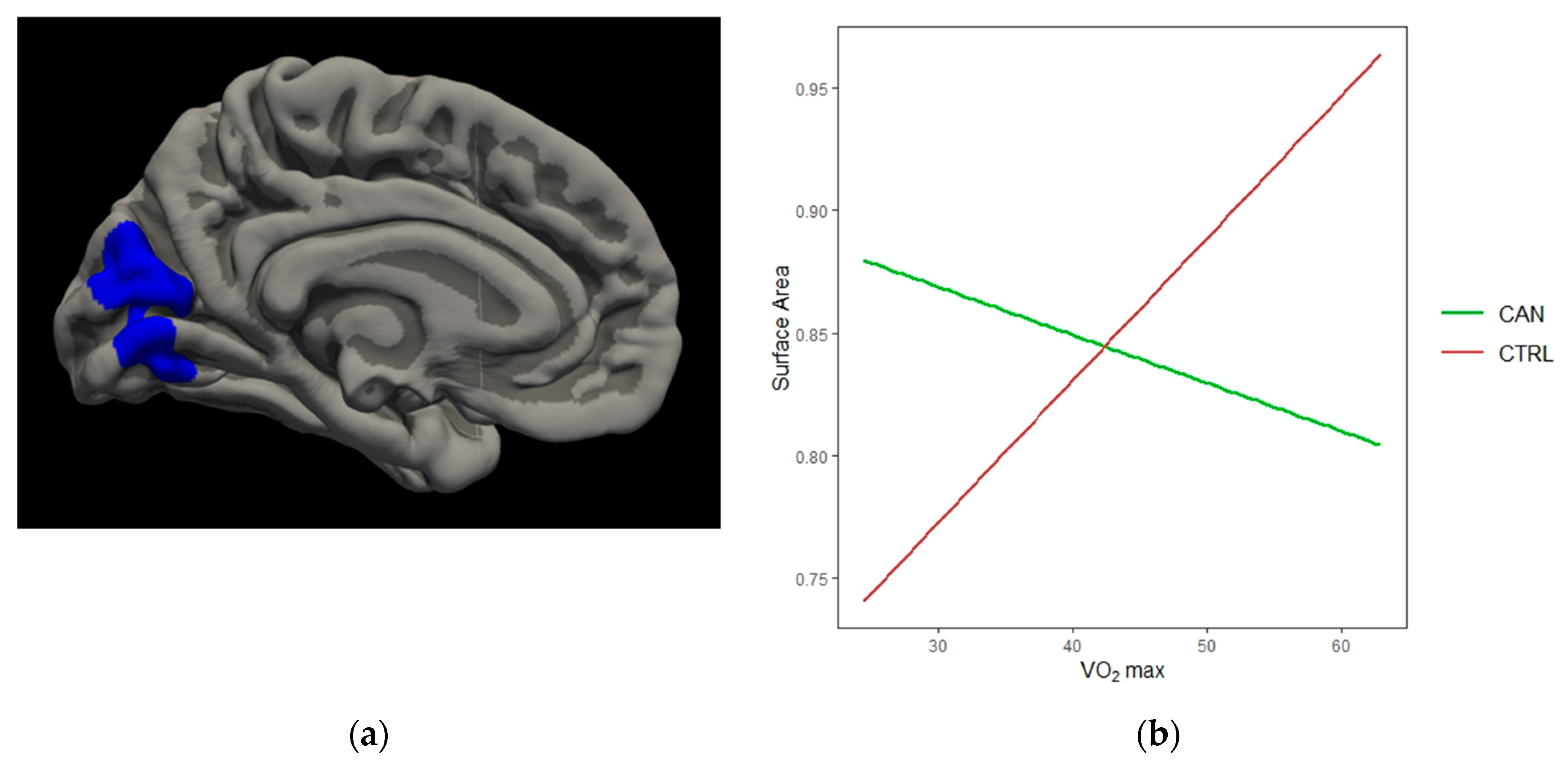
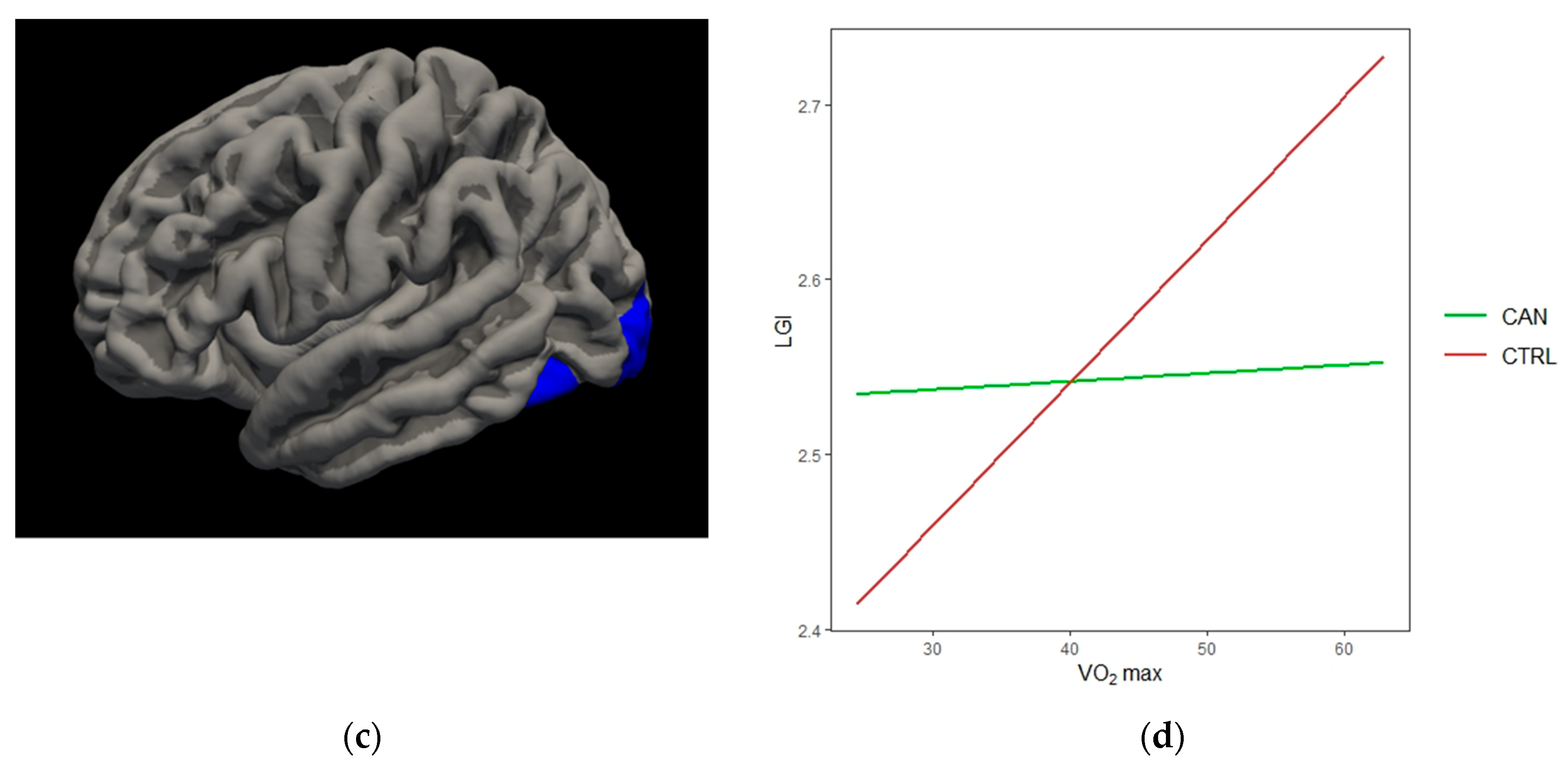
3.2.4. Cannabis*VO2
3.2.5. Cannabis*VO2*Gender
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Degenhardt, L.; Bharat, C.; Glantz, M.D.; Sampson, N.A.; Al-Hamzawi, A.; Alonso, J.; Andrade, L.H.; Bunting, B.; Cia, A.; de Girolamo, G.; et al. Association of Cohort and Individual Substance Use with Risk of Transitioning to Drug Use, Drug Use Disorder, and Remission from Disorder: Findings From the World Mental Health Surveys. JAMA Psychiatry 2019, 76, 708–720. [Google Scholar] [CrossRef]
- Schulenberg, J.E.; Johnston, L.D.; O’Malley, P.M.; Bachman, J.G.; Miech, R.A.; Patrick, M.E. Monitoring the Future National Survey Results on Drug Use, 1975–2018: Volume II, College Students and Adults Ages 19–60; Institute for Social Research: Ann Arbor, MI, USA, 2019. [Google Scholar] [CrossRef]
- Johnston, L.D.; Miech, R.A.; O’Malley, P.M.; Bachman, J.G.; Schulenberg, J.E.; Patrick, M.E. Monitoring the Future National Survey Results on Drug Use, 1975–2018: Overview, Key Findings on Adolescent Drug Use; Institute for Social Research: Ann Arbor, MI, USA, 2019. [Google Scholar] [CrossRef]
- Han, B.; Compton, W.M.; Blanco, C.; Jones, C.M. Time since first cannabis use and 12-month prevalence of cannabis use disorder among youth and emerging adults in the United States. Addiction 2019, 114, 698–707. [Google Scholar] [CrossRef]
- Carliner, H.; Brown, Q.L.; Sarvet, A.L.; Hasin, D.S. Cannabis use, attitudes, and legal status in the U.S.: A review. Prev. Med. 2017, 104, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Hasin, D.S.; Kerridge, B.T.; Saha, T.D.; Huang, B.; Pickering, R.; Smith, S.M.; Jung, J.; Zhang, H.; Grant, B.F. Prevalence and correlates of DSM-5 cannabis use disorder, 2012–2013: Findings from the National Epidemiologic Survey on Alcohol and Related Conditions-III. Am. J. Psychiatry 2016, 173, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Lisdahl, K.M.; Wright, N.E.; Kirchner-Medina, C.; Maple, K.E.; Shollenbarger, S. Considering Cannabis: The Effects of Regular Cannabis Use on Neurocognition in Adolescents and Young Adults. Curr. Addict. Rep. 2014, 1, 144–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorenzetti, V.; Solowij, N.; Fornito, A.; Lubman, D.I.; Yucel, M. The association between regular cannabis exposure and alterations of human brain morphology: An updated review of the literature. Curr. Pharm. Des. 2014, 20, 2138–2167. [Google Scholar] [CrossRef] [PubMed]
- Mechoulam, R.; Parker, L.A. The endocannabinoid system and the brain. Annu. Rev. Psychol. 2013, 64, 21–47. [Google Scholar] [CrossRef] [Green Version]
- Villares, J. Chronic use of marijuana decreases cannabinoid receptor binding and mRNA expression in the human brain. Neuroscience 2007, 145, 323–334. [Google Scholar] [CrossRef]
- Hirvonen, J.; Goodwin, R.S.; Li, C.T.; Terry, G.E.; Zoghbi, S.S.; Morse, C.; Pike, V.W.; Volkow, N.D.; Huestis, M.A.; Innis, R.B. Reversible and regionally selective downregulation of brain cannabinoid CB1 receptors in chronic daily cannabis smokers. Mol. Psychiatry 2012, 17, 642–649. [Google Scholar] [CrossRef] [Green Version]
- Bloomfield, M.A.P.; Hindocha, C.; Green, S.F.; Wall, M.B.; Lees, R.; Petrilli, K.; Costello, H.; Ogunbiyi, M.O.; Bossong, M.G.; Freeman, T.P. The neuropsychopharmacology of cannabis: A review of human imaging studies. Pharmacol. Ther. 2019, 195, 132–161. [Google Scholar] [CrossRef]
- Batalla, A.; Bhattacharyya, S.; Yucel, M.; Fusar-Poli, P.; Crippa, J.A.; Nogue, S.; Torrens, M.; Pujol, J.; Farre, M.; Martin-Santos, R. Structural and functional imaging studies in chronic cannabis users: A systematic review of adolescent and adult findings. PLoS ONE 2013, 8, e55821. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, M.; Crescini, A.; Borgwardt, S.; Caverzasi, E.; Politi, P.; Atakan, Z.; Fusar-Poli, P. Is cannabis neurotoxic for the healthy brain? A meta-analytical review of structural brain alterations in non-psychotic users. Psychiatry Clin. Neurosci. 2013, 67, 483–492. [Google Scholar] [CrossRef]
- Ashtari, M.; Avants, B.; Cyckowski, L.; Cervellione, K.L.; Roofeh, D.; Cook, P.; Gee, J.; Sevy, S.; Kumra, S. Medial temporal structures and memory functions in adolescents with heavy cannabis use. J. Psychiatr. Res. 2011, 45, 1055–1066. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medina, K.L.; Schweinsburg, A.D.; Cohen-Zion, M.; Nagel, B.J.; Tapert, S.F. Effects of alcohol and combined marijuana and alcohol use during adolescence on hippocampal volume and asymmetry. Neurotoxicol. Teratol. 2007, 29, 141–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medina, K.L.; McQueeny, T.; Nagel, B.J.; Hanson, K.L.; Yang, T.T.; Tapert, S.F. Prefrontal cortex morphometry in abstinent adolescent marijuana users: Subtle gender effects. Addict. Biol. 2009, 14, 457–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yucel, M.; Solowij, N.; Respondek, C.; Whittle, S.; Fornito, A.; Pantelis, C.; Lubman, D.I. Regional brain abnormalities associated with long-term heavy cannabis use. Arch. Gen. Psychiatry 2008, 65, 694–701. [Google Scholar] [CrossRef]
- McQueeny, T.; Padula, C.B.; Price, J.; Medina, K.L.; Logan, P.; Tapert, S.F. Gender effects on amygdala morphometry in adolescent marijuana users. Behav. Brain Res. 2011, 224, 128–134. [Google Scholar] [CrossRef] [Green Version]
- Mashhoon, Y.; Sava, S.; Sneider, J.T.; Nickerson, L.D.; Silveri, M.M. Cortical thinness and volume differences associated with marijuana abuse in emerging adults. Drug Alcohol Depend. 2015, 155, 275–283. [Google Scholar] [CrossRef] [Green Version]
- Price, J.S.; McQueeny, T.; Shollenbarger, S.; Browning, E.L.; Wieser, J.; Lisdahl, K.M. Effects of marijuana use on prefrontal and parietal volumes and cognition in emerging adults. Psychopharmacology 2015, 232, 2939–2950. [Google Scholar] [CrossRef] [Green Version]
- Filbey, F.M.; Aslan, S.; Calhoun, V.D.; Spence, J.S.; Damaraju, E.; Caprihan, A.; Segall, J. Long-term effects of marijuana use on the brain. Proc. Natl. Acad. Sci. USA 2014, 111, 16913–16918. [Google Scholar] [CrossRef] [Green Version]
- Lisdahl, K.M.; Tamm, L.; Epstein, J.N.; Jernigan, T.; Molina, B.S.; Hinshaw, S.P.; Swanson, J.M.; Newman, E.; Kelly, C.; Bjork, J.M.; et al. The impact of ADHD persistence, recent cannabis use, and age of regular cannabis use onset on subcortical volume and cortical thickness in young adults. Drug Alcohol Depend. 2016, 161, 135–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maple, K.E.; Thomas, A.M.; Kangiser, M.M.; Lisdahl, K.M. Anterior cingulate volume reductions in abstinent adolescent and young adult cannabis users: Association with affective processing deficits. Psychiatry Res. Neuroimaging 2019, 288, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Matochik, J.A.; Eldreth, D.A.; Cadet, J.L.; Bolla, K.I. Altered brain tissue composition in heavy marijuana users. Drug Alcohol Depend. 2005, 77, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Medina, K.L.; Nagel, B.J.; Tapert, S.F. Abnormal cerebellar morphometry in abstinent adolescent marijuana users. Psychiatry Res. 2010, 182, 152–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorenzetti, V.; Chye, Y.; Silva, P.; Solowij, N.; Roberts, C.A. Does regular cannabis use affect neuroanatomy? An updated systematic review and meta-analysis of structural neuroimaging studies. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 59–71. [Google Scholar] [CrossRef] [PubMed]
- White, T.; Su, S.; Schmidt, M.; Kao, C.Y.; Sapiro, G. The development of gyrification in childhood and adolescence. Brain Cognit. 2010, 72, 36–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hogstrom, L.J.; Westlye, L.T.; Walhovd, K.B.; Fjell, A.M. The structure of the cerebral cortex across adult life: Age-related patterns of surface area, thickness, and gyrification. Cereb. Cortex 2013, 23, 2521–2530. [Google Scholar] [CrossRef] [Green Version]
- Infante, M.A.; Moore, E.M.; Bischoff-Grethe, A.; Migliorini, R.; Mattson, S.N.; Riley, E.P. Atypical cortical gyrification in adolescents with histories of heavy prenatal alcohol exposure. Brain Res. 2015, 1624, 446–454. [Google Scholar] [CrossRef] [Green Version]
- Jednorog, K.; Altarelli, I.; Monzalvo, K.; Fluss, J.; Dubois, J.; Billard, C.; Dehaene-Lambertz, G.; Ramus, F. The influence of socioeconomic status on children’s brain structure. PLoS ONE 2012, 7, e42486. [Google Scholar] [CrossRef]
- Lohmann, G.; von Cramon, D.Y.; Steinmetz, H. Sulcal variability of twins. Cereb. Cortex 1999, 9, 754–763. [Google Scholar] [CrossRef] [Green Version]
- Kuhn, S.; Witt, C.; Banaschewski, T.; Barbot, A.; Barker, G.J.; Buchel, C.; Conrod, P.J.; Flor, H.; Garavan, H.; Ittermann, B.; et al. From mother to child: Orbitofrontal cortex gyrification and changes of drinking behaviour during adolescence. Addict. Biol. 2016, 21, 700–708. [Google Scholar] [CrossRef]
- Schnack, H.G.; van Haren, N.E.; Brouwer, R.M.; Evans, A.; Durston, S.; Boomsma, D.I.; Kahn, R.S.; Hulshoff Pol, H.E. Changes in thickness and surface area of the human cortex and their relationship with intelligence. Cereb. Cortex 2015, 25, 1608–1617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panizzon, M.S.; Fennema-Notestine, C.; Eyler, L.T.; Jernigan, T.L.; Prom-Wormley, E.; Neale, M.; Jacobson, K.; Lyons, M.J.; Grant, M.D.; Franz, C.E.; et al. Distinct genetic influences on cortical surface area and cortical thickness. Cereb. Cortex 2009, 19, 2728–2735. [Google Scholar] [CrossRef] [PubMed]
- Mata, I.; Perez-Iglesias, R.; Roiz-Santianez, R.; Tordesillas-Gutierrez, D.; Pazos, A.; Gutierrez, A.; Vazquez-Barquero, J.L.; Crespo-Facorro, B. Gyrification brain abnormalities associated with adolescence and early-adulthood cannabis use. Brain Res. 2010, 1317, 297–304. [Google Scholar] [CrossRef] [Green Version]
- Shollenbarger, S.G.; Price, J.; Wieser, J.; Lisdahl, K. Impact of cannabis use on prefrontal and parietal cortex gyrification and surface area in adolescents and emerging adults. Dev. Cognit. Neurosci. 2015, 16, 46–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filbey, F.M.; McQueeny, T.; DeWitt, S.J.; Mishra, V. Preliminary findings demonstrating latent effects of early adolescent marijuana use onset on cortical architecture. Dev. Cognit. Neurosci. 2015, 16, 16–22. [Google Scholar] [CrossRef] [Green Version]
- Infante, M.A.; Courtney, K.E.; Castro, N.; Squeglia, L.M.; Jacobus, J. Adolescent Brain Surface Area Pre- and Post-Cannabis and Alcohol Initiation. J. Stud. Alcohol Drugs 2018, 79, 835–843. [Google Scholar] [CrossRef]
- Chye, Y.; Suo, C.; Lorenzetti, V.; Batalla, A.; Cousijn, J.; Goudriaan, A.E.; Martin-Santos, R.; Whittle, S.; Solowij, N.; Yucel, M. Cortical surface morphology in long-term cannabis users: A multi-site MRI study. Eur. Neuropsychopharmacol. 2019, 29, 257–265. [Google Scholar] [CrossRef]
- Sim, Y.J.; Kim, H.; Shin, M.S.; Chang, H.K.; Shin, M.C.; Ko, I.G.; Kim, K.J.; Kim, T.S.; Kim, B.K.; Rhim, Y.T.; et al. Effect of postnatal treadmill exercise on c-Fos expression in the hippocampus of rat pups born from the alcohol-intoxicated mothers. Brain Dev. 2008, 30, 118–125. [Google Scholar] [CrossRef]
- Huang, T.; Larsen, K.T.; Ried-Larsen, M.; Moller, N.C.; Andersen, L.B. The effects of physical activity and exercise on brain-derived neurotrophic factor in healthy humans: A review. Scand. J. Med. Sci. Sports 2014, 24, 1–10. [Google Scholar] [CrossRef]
- Hill, M.N.; Titterness, A.K.; Morrish, A.C.; Carrier, E.J.; Lee, T.T.; Gil-Mohapel, J.; Gorzalka, B.B.; Hillard, C.J.; Christie, B.R. Endogenous cannabinoid signaling is required for voluntary exercise-induced enhancement of progenitor cell proliferation in the hippocampus. Hippocampus 2010, 20, 513–523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakurai, T.; Izawa, T.; Kizaki, T.; Ogasawara, J.E.; Shirato, K.; Imaizumi, K.; Takahashi, K.; Ishida, H.; Ohno, H. Exercise training decreases expression of inflammation-related adipokines through reduction of oxidative stress in rat white adipose tissue. Biochem. Biophys. Res. Commun. 2009, 379, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Radak, Z.; Kumagai, S.; Taylor, A.W.; Naito, H.; Goto, S. Effects of exercise on brain function: Role of free radicals. Appl. Physiol. Nutr. Metab. 2007, 32, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Leasure, J.L.; Jones, M. Forced and voluntary exercise differentially affect brain and behavior. Neuroscience 2008, 156, 456–465. [Google Scholar] [CrossRef] [Green Version]
- Helfer, J.L.; Goodlett, C.R.; Greenough, W.T.; Klintsova, A.Y. The effects of exercise on adolescent hippocampal neurogenesis in a rat model of binge alcohol exposure during the brain growth spurt. Brain Res. 2009, 1294, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Thomas, A.G.; Dennis, A.; Bandettini, P.A.; Johansen-Berg, H. The effects of aerobic activity on brain structure. Front. Psychol. 2012, 3, 86. [Google Scholar] [CrossRef] [Green Version]
- Smith, P.J.; Blumenthal, J.A.; Hoffman, B.M.; Cooper, H.; Strauman, T.A.; Welsh-Bohmer, K.; Browndyke, J.N.; Sherwood, A. Aerobic exercise and neurocognitive performance: A meta-analytic review of randomized controlled trials. Psychosom. Med. 2010, 72, 239–252. [Google Scholar] [CrossRef]
- Bherer, L.; Erickson, K.I.; Liu-Ambrose, T. A review of the effects of physical activity and exercise on cognitive and brain functions in older adults. J. Aging Res. 2013, 2013, 657508. [Google Scholar] [CrossRef] [Green Version]
- Herting, M.M.; Chu, X. Exercise, cognition, and the adolescent brain. Birth Defects Res. 2017, 109, 1672–1679. [Google Scholar] [CrossRef]
- Wengaard, E.; Kristoffersen, M.; Harris, A.; Gundersen, H. Cardiorespiratory Fitness Is Associated with Selective Attention in Healthy Male High-School Students. Front. Hum. Neurosci. 2017, 11, 330. [Google Scholar] [CrossRef] [Green Version]
- Wade, N.E.; Wallace, A.L.; Swartz, A.M.; Lisdahl, K.M. Aerobic Fitness Level Moderates the Association Between Cannabis Use and Executive Functioning and Psychomotor Speed Following Abstinence in Adolescents and Young Adults. J. Int. Neuropsychol. Soc. 2019, 25, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Castelli, D.M.; Gonzalez-Lima, F. The positive cognitive impact of aerobic fitness is associated with peripheral inflammatory and brain-derived neurotrophic biomarkers in young adults. Physiol. Behav. 2017, 179, 75–89. [Google Scholar] [CrossRef] [PubMed]
- Chaddock, L.; Pontifex, M.B.; Hillman, C.H.; Kramer, A.F. A review of the relation of aerobic fitness and physical activity to brain structure and function in children. J. Int. Neuropsychol. Soc. 2011, 17, 975–985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwarb, H.; Johnson, C.L.; Daugherty, A.M.; Hillman, C.H.; Kramer, A.F.; Cohen, N.J.; Barbey, A.K. Aerobic fitness, hippocampal viscoelasticity, and relational memory performance. NeuroImage 2017, 153, 179–188. [Google Scholar] [CrossRef]
- Whiteman, A.S.; Young, D.E.; Budson, A.E.; Stern, C.E.; Schon, K. Entorhinal volume, aerobic fitness, and recognition memory in healthy young adults: A voxel-based morphometry study. NeuroImage 2016, 126, 229–238. [Google Scholar] [CrossRef] [Green Version]
- Herting, M.M.; Nagel, B.J. Differences in brain activity during a verbal associative memory encoding task in high- and low-fit adolescents. J. Cognit. Neurosci. 2013, 25, 595–612. [Google Scholar] [CrossRef] [Green Version]
- Koltyn, K.F.; Brellenthin, A.G.; Cook, D.B.; Sehgal, N.; Hillard, C. Mechanisms of exercise-induced hypoalgesia. J. Pain 2014, 15, 1294–1304. [Google Scholar] [CrossRef] [Green Version]
- Lisdahl, K.M.; Gilbart, E.R.; Wright, N.E.; Shollenbarger, S. Dare to delay? The impacts of adolescent alcohol and marijuana use onset on cognition, brain structure, and function. Front. Psychiatry 2013, 4, 53. [Google Scholar] [CrossRef] [Green Version]
- Buchowski, M.S.; Meade, N.N.; Charboneau, E.; Park, S.; Dietrich, M.S.; Cowan, R.L.; Martin, P.R. Aerobic exercise training reduces cannabis craving and use in non-treatment seeking cannabis-dependent adults. PLoS ONE 2011, 6, e17465. [Google Scholar] [CrossRef] [Green Version]
- Schlienz, N.J.; Budney, A.J.; Lee, D.C.; Vandrey, R. Cannabis Withdrawal: A Review of Neurobiological Mechanisms and Sex Differences. Curr. Addict. Rep. 2017, 4, 75–81. [Google Scholar] [CrossRef]
- Khan, S.S.; Secades-Villa, R.; Okuda, M.; Wang, S.; Perez-Fuentes, G.; Kerridge, B.T.; Blanco, C. Gender differences in cannabis use disorders: Results from the National Epidemiologic Survey of Alcohol and Related Conditions. Drug Alcohol Depend. 2013, 130, 101–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fogel, J.S.; Kelly, T.H.; Westgate, P.M.; Lile, J.A. Sex differences in the subjective effects of oral Delta(9)-THC in cannabis users. Pharm. Biochem. Behav. 2017, 152, 44–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cooper, Z.D.; Haney, M. Investigation of sex-dependent effects of cannabis in daily cannabis smokers. Drug Alcohol Depend. 2014, 136, 85–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lisdahl, K.M.; Price, J.S. Increased marijuana use and gender predict poorer cognitive functioning in adolescents and emerging adults. J. Int. Neuropsychol. Soc. 2012, 18, 678–688. [Google Scholar] [CrossRef] [Green Version]
- Luders, E.; Narr, K.L.; Thompson, P.M.; Rex, D.E.; Jancke, L.; Steinmetz, H.; Toga, A.W. Gender differences in cortical complexity. Nat. Neurosci. 2004, 7, 799–800. [Google Scholar] [CrossRef]
- Luders, E.; Thompson, P.M.; Narr, K.L.; Toga, A.W.; Jancke, L.; Gaser, C. A curvature-based approach to estimate local gyrification on the cortical surface. NeuroImage 2006, 29, 1224–1230. [Google Scholar] [CrossRef]
- Salinas, J.; Mills, E.D.; Conrad, A.L.; Koscik, T.; Andreasen, N.C.; Nopoulos, P. Sex differences in parietal lobe structure and development. Gend. Med. 2012, 9, 44–55. [Google Scholar] [CrossRef] [Green Version]
- Wierenga, L.M.; Langen, M.; Oranje, B.; Durston, S. Unique developmental trajectories of cortical thickness and surface area. NeuroImage 2014, 87, 120–126. [Google Scholar] [CrossRef]
- Raznahan, A.; Shaw, P.; Lalonde, F.; Stockman, M.; Wallace, G.L.; Greenstein, D.; Clasen, L.; Gogtay, N.; Giedd, J.N. How does your cortex grow? J. Neurosci. 2011, 31, 7174–7177. [Google Scholar] [CrossRef]
- Dupuy, O.; Gauthier, C.J.; Fraser, S.A.; Desjardins-Crepeau, L.; Desjardins, M.; Mekary, S.; Lesage, F.; Hoge, R.D.; Pouliot, P.; Bherer, L. Higher levels of cardiovascular fitness are associated with better executive function and prefrontal oxygenation in younger and older women. Front. Hum. Neurosci. 2015, 9, 66. [Google Scholar] [CrossRef] [Green Version]
- Killgore, W.D.; Schwab, Z.J. Sex differences in the association between physical exercise and IQ. Percept. Mot. Ski. 2012, 115, 605–617. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Reading, J.; Shephard, R.J. Revision of the Physical Activity Readiness Questionnaire (PAR-Q). Can. J. Sport Sci. 1992, 17, 338–345. [Google Scholar] [PubMed]
- Fogelholm, M.; Malmberg, J.; Suni, J.; Santtila, M.; Kyrolainen, H.; Mantysaari, M.; Oja, P. International Physical Activity Questionnaire: Validity against fitness. Med. Sci. Sports Exerc 2006, 38, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.L.; Wade, N.E.; Hatcher, K.F.; Lisdahl, K.M. Effects of Cannabis Use and Subclinical ADHD Symptomology on Attention Based Tasks in Adolescents and Young Adults. Arch. Clin. Neuropsychol. 2018, 34, 700–705. [Google Scholar] [CrossRef] [PubMed]
- Meier, M.H.; Caspi, A.; Ambler, A.; Harrington, H.; Houts, R.; Keefe, R.S.; McDonald, K.; Ward, A.; Poulton, R.; Moffitt, T.E. Persistent cannabis users show neuropsychological decline from childhood to midlife. Proc. Natl. Acad. Sci. USA 2012, 109, E2657–E2664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sobell, L.C.; Sobell, M.B. Timeline Follow-Back. In Measuring Alcohol Consumption; Litten, R.Z., Allen, J.P., Eds.; Humana Press: Totowa, NJ, USA, 1992; pp. 41–72. [Google Scholar] [CrossRef]
- Pescatello, L.S. ACSM’s Guidelines for Exercise Testing and Prescription, 9th ed.; Wolters Kluwer/Lippincott Williams & Wilkins Health: Philadelphia, PA, USA, 2014. [Google Scholar]
- Fitness Ranking by the KU Alzheimers Disease Center. Available online: http://www.kumc.edu/fitness-ranking.html (accessed on 10 November 2019).
- Wallace, A.L.; Wade, N.E.; Lisdahl, K.M. Impact of Two-Weeks of Monitored Abstinence on Cognition in Adolescent and Young Adult Cannabis Users. J. Int. Neuropsychol. Soc. 2020, in press. [Google Scholar]
- Brown, S.A.; Myers, M.G.; Lippke, L.; Tapert, S.F.; Stewart, D.G.; Vik, P.W. Psychometric evaluation of the Customary Drinking and Drug Use Record (CDDR): A measure of adolescent alcohol and drug involvement. J. Stud. Alcohol 1998, 59, 427–438. [Google Scholar] [CrossRef]
- Sheehan, D.V.; Lecrubier, Y.; Sheehan, K.H.; Amorim, P.; Janavs, J.; Weiller, E.; Hergueta, T.; Baker, R.; Dunbar, G.C. The Mini-International Neuropsychiatric Interview (M.I.N.I.): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 1998, 59, 22–23. [Google Scholar]
- Sheehan, D.V.; Sheehan, K.H.; Shytle, R.D.; Janavs, J.; Bannon, Y.; Rogers, J.E.; Milo, K.M.; Stock, S.L.; Wilkinson, B. Reliability and validity of the Mini International Neuropsychiatric Interview for Children and Adolescents (MINI-KID). J. Clin. Psychiatry 2010, 71, 313–326. [Google Scholar] [CrossRef]
- Howley, E.T.; Bassett, D.R., Jr.; Welch, H.G. Criteria for maximal oxygen uptake: Review and commentary. Med. Sci. Sports Exerc. 1995, 27, 1292–1301. [Google Scholar] [CrossRef]
- Schaer, M.; Cuadra, M.B.; Schmansky, N.; Fischl, B.; Thiran, J.P.; Eliez, S. How to measure cortical folding from MR images: A step-by-step tutorial to compute local gyrification index. J. Vis. Exp. 2012, e3417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing, 3.5.2; R Found. for Statistical Computing: Vienna, Austria, 2010; Available online: https://www.R-project.org/ (accessed on 22 February 2020).
- Dale, A.M.; Fischl, B.; Sereno, M.I. Cortical surface-based analysis. I. Segmentation and surface reconstruction. NeuroImage 1999, 9, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Schaer, M.; Cuadra, M.B.; Tamarit, L.; Lazeyras, F.; Eliez, S.; Thiran, J.P. A surface-based approach to quantify local cortical gyrification. IEEE Trans. Med. Imaging 2008, 27, 161–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desikan, R.S.; Segonne, F.; Fischl, B.; Quinn, B.T.; Dickerson, B.C.; Blacker, D.; Buckner, R.L.; Dale, A.M.; Maguire, R.P.; Hyman, B.T.; et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage 2006, 31, 968–980. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, L.; Chiu, W.T.; Sampson, N.; Kessler, R.C.; Anthony, J.C.; Angermeyer, M.; Bruffaerts, R.; de Girolamo, G.; Gureje, O.; Huang, Y.; et al. Toward a global view of alcohol, tobacco, cannabis, and cocaine use: Findings from the WHO World Mental Health Surveys. PLoS Med. 2008, 5, e141. [Google Scholar] [CrossRef] [Green Version]
- Brellenthin, A.G.; Koltyn, K.F. Exercise as an adjunctive treatment for cannabis use disorder. Am. J. Drug Alcohol Abus. 2016, 42, 481–489. [Google Scholar] [CrossRef]
- Tomasi, D.; Volkow, N.D. Association between functional connectivity hubs and brain networks. Cereb. Cortex 2011, 21, 2003–2013. [Google Scholar] [CrossRef] [Green Version]
- Eggan, S.M.; Lewis, D.A. Immunocytochemical distribution of the cannabinoid CB1 receptor in the primate neocortex: A regional and laminar analysis. Cereb. Cortex 2007, 17, 175–191. [Google Scholar] [CrossRef]
- Vijayakumar, N.; Allen, N.B.; Youssef, G.; Dennison, M.; Yucel, M.; Simmons, J.G.; Whittle, S. Brain development during adolescence: A mixed-longitudinal investigation of cortical thickness, surface area, and volume. Hum. Brain Mapp. 2016, 37, 2027–2038. [Google Scholar] [CrossRef]
- Tervo-Clemmens, B.; Simmonds, D.; Calabro, F.J.; Montez, D.F.; Lekht, J.A.; Day, N.L.; Richardson, G.A.; Luna, B. Early Cannabis Use and Neurocognitive Risk: A Prospective Functional Neuroimaging Study. Biol. Psychiatry Cognit. Neurosci. Neuroimaging 2018, 3, 713–725. [Google Scholar] [CrossRef]
- Martin-Santos, R.; Fagundo, A.B.; Crippa, J.A.; Atakan, Z.; Bhattacharyya, S.; Allen, P.; Fusar-Poli, P.; Borgwardt, S.; Seal, M.; Busatto, G.F.; et al. Neuroimaging in cannabis use: A systematic review of the literature. Psychol. Med. 2010, 40, 383–398. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.H.; Abraham, T.T.; Darwin, W.D.; Herning, R.; Cadet, J.L.; Huestis, M.A. Extended urinary Δ9-tetrahydrocannabinol excretion in chronic cannabis users precludes use as a biomarker of new drug exposure. Drug Alcohol Depend. 2009, 105, 24–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Battistella, G.; Fornari, E.; Annoni, J.M.; Chtioui, H.; Dao, K.; Fabritius, M.; Favrat, B.; Mall, J.F.; Maeder, P.; Giroud, C. Long-term effects of cannabis on brain structure. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2014, 39, 2041–2048. [Google Scholar] [CrossRef] [PubMed]
- Weiland, B.J.; Thayer, R.E.; Depue, B.E.; Sabbineni, A.; Bryan, A.D.; Hutchison, K.E. Daily marijuana use is not associated with brain morphometric measures in adolescents or adults. J. Neurosci. 2015, 35, 1505–1512. [Google Scholar] [CrossRef] [Green Version]
- Lenroot, R.K.; Giedd, J.N. Sex differences in the adolescent brain. Brain Cognit. 2010, 72, 46–55. [Google Scholar] [CrossRef] [Green Version]
- Scott, E.P.; Brennan, E.; Benitez, A. A Systematic Review of the Neurocognitive Effects of Cannabis Use in Older Adults. Curr. Addict. Rep. 2019. [Google Scholar] [CrossRef]
- Gonzalez, R.; Pacheco-Colon, I.; Duperrouzel, J.C.; Hawes, S.W. Does Cannabis Use Cause Declines in Neuropsychological Functioning? A Review of Longitudinal Studies. J. Int. Neuropsychol. Soc. 2017, 23, 893–902. [Google Scholar] [CrossRef] [Green Version]
- Cuttler, C.; Mischley, L.K.; Sexton, M. Sex Differences in Cannabis Use and Effects: A Cross-Sectional Survey of Cannabis Users. Cannabis Cannabinoid Res. 2016, 1, 166–175. [Google Scholar] [CrossRef] [Green Version]
- Burston, J.J.; Wiley, J.L.; Craig, A.A.; Selley, D.E.; Sim-Selley, L.J. Regional enhancement of cannabinoid CB1 receptor desensitization in female adolescent rats following repeated Delta-tetrahydrocannabinol exposure. Br. J. Pharmacol. 2010, 161, 103–112. [Google Scholar] [CrossRef] [Green Version]
- Rubino, T.; Vigano, D.; Realini, N.; Guidali, C.; Braida, D.; Capurro, V.; Castiglioni, C.; Cherubino, F.; Romualdi, P.; Candeletti, S.; et al. Chronic delta 9-tetrahydrocannabinol during adolescence provokes sex-dependent changes in the emotional profile in adult rats: Behavioral and biochemical correlates. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2008, 33, 2760–2771. [Google Scholar] [CrossRef]
- Lenroot, R.K.; Gogtay, N.; Greenstein, D.K.; Wells, E.M.; Wallace, G.L.; Clasen, L.S.; Blumenthal, J.D.; Lerch, J.; Zijdenbos, A.P.; Evans, A.C.; et al. Sexual dimorphism of brain developmental trajectories during childhood and adolescence. NeuroImage 2007, 36, 1065–1073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giedd, J.N.; Blumenthal, J.; Jeffries, N.O.; Castellanos, F.X.; Liu, H.; Zijdenbos, A.; Paus, T.; Evans, A.C.; Rapoport, J.L. Brain development during childhood and adolescence: A longitudinal MRI study. Nat. Neurosci. 1999, 2, 861–863. [Google Scholar] [CrossRef] [PubMed]
- Wittfeld, K.; Jochem, C.; Dorr, M.; Schminke, U.; Glaser, S.; Bahls, M.; Markus, M.R.P.; Felix, S.B.; Leitzmann, M.F.; Ewert, R.; et al. Cardiorespiratory Fitness and Gray Matter Volume in the Temporal, Frontal, and Cerebellar Regions in the General Population. Mayo Clin. Proc. 2020, 95, 44–56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herting, M.M.; Keenan, M.F. Exercise and the Developing Brain in Children and Adolescents. In Physical Activity and the Aging Brain; Watson, R.R., Ed.; Academic Press: London, UK, 2017; pp. 13–19. [Google Scholar] [CrossRef]
- Haeger, A.; Costa, A.S.; Schulz, J.B.; Reetz, K. Cerebral changes improved by physical activity during cognitive decline: A systematic review on MRI studies. NeuroImage Clin. 2019, 23, 101933. [Google Scholar] [CrossRef]
- Fleenor, B.S.; Marshall, K.D.; Durrant, J.R.; Lesniewski, L.A.; Seals, D.R. Arterial stiffening with ageing is associated with transforming growth factor-beta1-related changes in adventitial collagen: Reversal by aerobic exercise. J. Physiol. 2010, 588, 3971–3982. [Google Scholar] [CrossRef]
- Lopez-Lopez, C.; LeRoith, D.; Torres-Aleman, I. Insulin-like growth factor I is required for vessel remodeling in the adult brain. Proc. Natl. Acad. Sci. USA 2004, 101, 9833–9838. [Google Scholar] [CrossRef] [Green Version]
- Nokia, M.S.; Lensu, S.; Ahtiainen, J.P.; Johansson, P.P.; Koch, L.G.; Britton, S.L.; Kainulainen, H. Physical exercise increases adult hippocampal neurogenesis in male rats provided it is aerobic and sustained. J. Physiol. 2016, 594, 1855–1873. [Google Scholar] [CrossRef]
- Waters, R.P.; Emerson, A.J.; Watt, M.J.; Forster, G.L.; Swallow, J.G.; Summers, C.H. Stress induces rapid changes in central catecholaminergic activity in Anolis carolinensis: Restraint and forced physical activity. Brain Res. Bull. 2005, 67, 210–218. [Google Scholar] [CrossRef]
- Frodl, T.; Strehl, K.; Carballedo, A.; Tozzi, L.; Doyle, M.; Amico, F.; Gormley, J.; Lavelle, G.; O’Keane, V. Aerobic exercise increases hippocampal subfield volumes in younger adults and prevents volume decline in the elderly. Brain Imaging Behav. 2019. [Google Scholar] [CrossRef]
- Henchoz, Y.; Dupuis, M.; Deline, S.; Studer, J.; Baggio, S.; N’Goran, A.A.; Daeppen, J.B.; Gmel, G. Associations of physical activity and sport and exercise with at-risk substance use in young men: A longitudinal study. Prev. Med. 2014, 64, 27–31. [Google Scholar] [CrossRef] [Green Version]
- Westin, A.A.; Mjones, G.; Burchardt, O.; Fuskevag, O.M.; Slordal, L. Can physical exercise or food deprivation cause release of fat-stored cannabinoids? Basic Clin. Pharm. Toxicol. 2014, 115, 467–471. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.; Montebello, M.E.; Norberg, M.M.; Rooney, K.; Lintzeris, N.; Bruno, R.; Booth, J.; Arnold, J.C.; McGregor, I.S. Exercise increases plasma THC concentrations in regular cannabis users. Drug Alcohol Depend. 2013, 133, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.D.; Crombie, K.M.; Cook, D.B.; Hillard, C.J.; Koltyn, K.F. Serum Endocannabinoid and Mood Changes after Exercise in Major Depressive Disorder. Med. Sci. Sports Exerc. 2019, 51, 1909–1917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watkins, B.A. Endocannabinoids, exercise, pain, and a path to health with aging. Mol. Asp. Med. 2018, 64, 68–78. [Google Scholar] [CrossRef]
- Hillard, C.J. Circulating Endocannabinoids: From Whence Do They Come and where are They Going? Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2018, 43, 155–172. [Google Scholar] [CrossRef]
- Gao, Y.; Riklin-Raviv, T.; Bouix, S. Shape analysis, a field in need of careful validation. Hum. Brain Mapp. 2014, 35, 4965–4978. [Google Scholar] [CrossRef]
- Lee, J.K.; Lee, J.M.; Kim, J.S.; Kim, I.Y.; Evans, A.C.; Kim, S.I. A novel quantitative cross-validation of different cortical surface reconstruction algorithms using MRI phantom. NeuroImage 2006, 31, 572–584. [Google Scholar] [CrossRef]
- Segonne, F.; Pacheco, J.; Fischl, B. Geometrically accurate topology-correction of cortical surfaces using nonseparating loops. IEEE Trans. Med. Imaging 2007, 26, 518–529. [Google Scholar] [CrossRef]
- Verweij, K.J.; Zietsch, B.P.; Lynskey, M.T.; Medland, S.E.; Neale, M.C.; Martin, N.G.; Boomsma, D.I.; Vink, J.M. Genetic and environmental influences on cannabis use initiation and problematic use: A meta-analysis of twin studies. Addiction 2010, 105, 417–430. [Google Scholar] [CrossRef]
- Shollenbarger, S.G.; Price, J.; Wieser, J.; Lisdahl, K. Poorer frontolimbic white matter integrity is associated with chronic cannabis use, FAAH genotype, and increased depressive and apathy symptoms in adolescents and young adults. NeuroImage Clin. 2015, 8, 117–125. [Google Scholar] [CrossRef] [Green Version]
- Filbey, F.M.; Schacht, J.P.; Myers, U.S.; Chavez, R.S.; Hutchison, K.E. Individual and additive effects of the CNR1 and FAAH genes on brain response to marijuana cues. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2010, 35, 967–975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moore, T.H.M.; Zammit, S.; Lingford-Hughes, A.; Barnes, T.R.E.; Jones, P.B.; Burke, M.; Lewis, G. Cannabis use and risk of psychotic or affective mental health outcomes: A systematic review. Lancet 2007, 370, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Crippa, J.A.; Zuardi, A.W.; Martin-Santos, R.; Bhattacharyya, S.; Atakan, Z.; McGuire, P.; Fusar-Poli, P. Cannabis and anxiety: A critical review of the evidence. Hum. Psychopharmacol. 2009, 24, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Lev-Ran, S.; Roerecke, M.; Le Foll, B.; George, T.P.; McKenzie, K.; Rehm, J. The association between cannabis use and depression: A systematic review and meta-analysis of longitudinal studies. Psychol. Med. 2014, 44, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Lisdahl, K.M.; Sher, K.J.; Conway, K.P.; Gonzalez, R.; Feldstein Ewing, S.W.; Nixon, S.J.; Tapert, S.; Bartsch, H.; Goldstein, R.Z.; Heitzeg, M. Adolescent brain cognitive development (ABCD) study: Overview of substance use assessment methods. Dev. Cognit. Neurosci. 2018, 32, 80–96. [Google Scholar] [CrossRef] [PubMed]


| Cannabis Users | Non-Using Controls | |||||
|---|---|---|---|---|---|---|
| All | Male | Female | All | Male | Female | |
| N | 36 | 23 | 13 | 38 | 18 | 20 |
| M (SD) or % | ||||||
| Age (years) | 21.4 (2.3) | 21.4 (2.4) | 21.4 (2.0) | 20.8 (2.8) | 20.5 (3.1) | 21.0 (2.6) |
| Race (% Caucasian) | 58.3% | 65.2% | 46.2% | 71.1% | 72.2% | 70.0% |
| Ethnicity (% Non-Hisp) | 77.8% | 78.3% | 76.9% | 86.8% | 94.4% | 80.0% |
| Educational Attainment (years) | 14.0 (1.6) | 13.9 (1.8) | 14.1 (1.3) | 14.1 (2.4) | 14.0 (2.9) | 14.2 (1.9) |
| Past yr Alcohol Use a,* | 338.7 (300.8) | 376.6 (306.2) | 271.6 (290.5) | 100.6 (173.6) | 141.8 (225.1) | 63.5 (101.5) |
| Past yr Tobacco Use a,* | 214.6 (483.7) | 311.8 (585.1) | 42.8 (68.1) | 0.5 (1.97) | 0.2 (0.43) | 0.7 (2.7) |
| Cotinine Level b,* | 2.0 (1.8) | 2.3 (2.1) | 1.5 (1.0) | 1.1 (0.6) | 1.1 (0.6) | 1.1 (0.6) |
| Past yr Cannabis Use a,* | 428.2 (440.4) | 499.9 (510.7) | 301.5 (245.4) | 0.36 (1.2) | 0.7 (1.6) | 0.1 (0.22) |
| Lifetime Cannabis Use a,* | 1189.6 (1372.3) | 1419.7 (1621.6) | 782.5 (625.0) | 1.5 (2.9) | 1.2 (2.3) | 1.8 (3.5) |
| Age at Regular Cannabis Use Onset (years) | 17.5 (1.7) | 17.4 (1.9) | 17.8 (1.3) | − c | − c | − c |
| Cannabis Abstinence Length (days) d | 31.1 (22.9) | 34.3 (27.9) | 25.5 (6.5) | − c | − c | − c |
| VO2 maximum e,^ | 43.7 (9.0) | 47.9 (6.6) | 36.1 (7.7) | 41.4 (9.8) | 47.9 (8.8) | 35.5 (6.3) |
| VO2 maximum (%ile) f | − c | 69.4% | 37.2% | − c | 68.9% | 33.9% |
| Body Fat (%) g,^ | 19.1% (8.5) | 15.6% (6.9) | 25.3% (7.7) | 21.6% (10.0) | 13.6% (6.1) | 28.7% (7.0) |
| t | Size (mm2) | x | y | z | cwp | |
|---|---|---|---|---|---|---|
| Cannabis | ||||||
| Left Cuneus | 2.639 | 1706.64 | −4.1 | −78.6 | 19.1 | 0.006 |
| Cannabis*Gender | ||||||
| Left Precuneus | −3.306 | 1718.12 | −10.3 | −54.6 | 46.7 | 0.006 |
| Left Rostral Middle Frontal | −2.299 | 2348.85 | −44.7 | 27 | 31.4 | 0.0006 |
| Right Superior Frontal | −3.491 | 1819.72 | 11.5 | 9 | 36.9 | 0.003 |
| Right Superior Frontal | −2.248 | 2007.88 | 23.1 | 0.4 | 61.6 | 0.002 |
| VO2 | ||||||
| Left Superior Parietal | 4.654 | 1673.98 | −28.4 | −64.4 | 39.9 | 0.007 |
| Left Inferior Parietal | 4.236 | 2535.22 | −45.1 | −63.9 | 10 | 0.0001 |
| Right Inferior Parietal | 3.894 | 3235.39 | 47.3 | −59.7 | 29.4 | 0.0001 |
| Right Inferior Temporal | 3.268 | 2877.28 | 51.4 | −55.2 | −15.1 | 0.0001 |
| Cannabis*VO2 | ||||||
| Left Cuneus | −3.724 | 2736.75 | −4.4 | −77.1 | 21.6 | 0.0001 |
| t | Size (mm2) | x | y | z | cwp | |
|---|---|---|---|---|---|---|
| Cannabis*Gender | ||||||
| Left Precentral | −2.894 | 4993.5 | −37.6 | −12.2 | 62.1 | 0.0001 |
| Left Lateral Orbitofrontal | −2.533 | 3240.22 | −18.8 | 51.8 | −13.8 | 0.0004 |
| Right Supramarginal | −3.784 | 4763.28 | 49.6 | −41.1 | 40.9 | 0.0001 |
| VO2 | ||||||
| Left Superior Temporal | 5.174 | 10682.85 | −64.5 | −25.2 | 4.2 | 0.0001 |
| Right Lateral Orbitofrontal | 3.272 | 13062.8 | 39 | 27.3 | −9 | 0.0001 |
| Right Inferior Parietal | 2.78 | 2718.55 | 34.2 | −73.4 | 37.6 | 0.0015 |
| Cannabis*VO2 | ||||||
| Left Lateral Occipital | −3.712 | 3297.29 | −28.2 | −95.1 | −12.7 | 0.0004 |
| Cannabis*VO2*Gender | ||||||
| Right Supramarginal | 2.572 | 2207.65 | 48.8 | −40.5 | 40.3 | 0.009 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sullivan, R.M.; Wallace, A.L.; Wade, N.E.; Swartz, A.M.; Lisdahl, K.M. Assessing the Role of Cannabis Use on Cortical Surface Structure in Adolescents and Young Adults: Exploring Gender and Aerobic Fitness as Potential Moderators. Brain Sci. 2020, 10, 117. https://0-doi-org.brum.beds.ac.uk/10.3390/brainsci10020117
Sullivan RM, Wallace AL, Wade NE, Swartz AM, Lisdahl KM. Assessing the Role of Cannabis Use on Cortical Surface Structure in Adolescents and Young Adults: Exploring Gender and Aerobic Fitness as Potential Moderators. Brain Sciences. 2020; 10(2):117. https://0-doi-org.brum.beds.ac.uk/10.3390/brainsci10020117
Chicago/Turabian StyleSullivan, Ryan M., Alexander L. Wallace, Natasha E. Wade, Ann M. Swartz, and Krista M. Lisdahl. 2020. "Assessing the Role of Cannabis Use on Cortical Surface Structure in Adolescents and Young Adults: Exploring Gender and Aerobic Fitness as Potential Moderators" Brain Sciences 10, no. 2: 117. https://0-doi-org.brum.beds.ac.uk/10.3390/brainsci10020117





