Effects of Inflammation and Depression on Telomere Length in Young Adults in the United States
Abstract
:1. Introduction
2. Materials and Methods
2.1. Dataset
2.2. C-Reactive Protein
2.3. Depression
2.4. Telomere Length
2.5. Statistical Analyses
3. Discussion
Author Contributions
Conflicts of Interest
References
- Moyzis, R.K.; Buckingham, J.M.; Cram, L.S.; Dani, M.; Deaven, L.L.; Jones, M.D.; Meyne, J.; Ratliff, R.L.; Wu, J.R. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proc. Natl. Acad. Sci. USA 1988, 85, 6622–6626. [Google Scholar] [CrossRef]
- Verdun, R.E.; Karlseder, J. The DNA damage machinery and homologous recombination pathway act consecutively to protect human telomeres. Cell 2006, 127, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Rajaraman, S.; Choi, J.; Cheung, P.; Beaudry, V.; Moore, H.; Artandi, S.E. Telomere uncapping in progenitor cells with critical telomere shortening is coupled to S-phase progression in vivo. Proc. Natl. Acad. Sci. USA 2007, 104, 17747–17752. [Google Scholar] [CrossRef] [Green Version]
- Toupance, S.; Villemonais, D.; Germain, D.; Gegout-Petit, A.; Albuisson, E.; Benetos, A. The individual’s signature of telomere length distribution. Sci. Rep. 2019, 9, 685. [Google Scholar] [CrossRef] [PubMed]
- Harley, C.B.; Futcher, A.B.; Greider, C.W. Telomeres shorten during ageing of human fibroblasts. Nature 1990, 345, 458–460. [Google Scholar] [CrossRef]
- Butler, R.N.; Sprott, R.; Warner, H.; Bland, J.; Feuers, R.; Forster, M.; Fillit, H.; Harman, S.M.; Hewitt, M.; Hyman, M.; et al. Biomarkers of aging: From primitive organisms to humans. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, B560–B567. [Google Scholar] [CrossRef] [PubMed]
- Guralnik, J.M. Successful aging: Is it in our future? Arch. Intern. Med. 2008, 168, 131–132. [Google Scholar] [CrossRef]
- Koppelstaetter, C.; Kern, G.; Mayer, G. Biomarkers of aging with prognostic and predictive value in non-oncological diseases. Curr. Med. Chem. 2009, 16, 3469–3475. [Google Scholar] [CrossRef] [PubMed]
- von Zglinicki, T.; Serra, V.; Lorenz, M.; Saretzki, G.; Lenzen-Grossimlighaus, R.; Gessner, R.; Risch, A.; Steinhagen-Thiessen, E. Short telomeres in patients with vascular dementia: An indicator of low antioxidative capacity and a possible risk factor? Lab. Investig. 2000, 80, 1739–1747. [Google Scholar] [CrossRef]
- Zhu, H.; Belcher, M.; van der Harst, P. Healthy aging and disease: Role for telomere biology? Clin. Sci. 2011, 120, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Simon, N.M.; Smoller, J.W.; McNamara, K.L.; Maser, R.S.; Zalta, A.K.; Pollack, M.H.; Nierenberg, A.A.; Fava, M.; Wong, K.K. Telomere shortening and mood disorders: Preliminary support for a chronic stress model of accelerated aging. Biol. Psychiatry 2006, 60, 432–435. [Google Scholar] [CrossRef]
- Hartmann, N.; Boehner, M.; Groenen, F.; Kalb, R. Telomere length of patients with major depression is shortened but independent from therapy and severity of the disease. Depress. Anxiety 2010, 27, 1111–1116. [Google Scholar] [CrossRef]
- Hoen, P.W.; de Jonge, P.; Na, B.Y.; Farzaneh-Far, R.; Epel, E.; Lin, J.; Blackburn, E.; Whooley, M.A. Depression and leukocyte telomere length in patients with coronary heart disease: Data from the Heart and Soul Study. Psychosom. Med. 2011, 73, 541–547. [Google Scholar] [CrossRef]
- Wikgren, M.; Maripuu, M.; Karlsson, T.; Nordfjall, K.; Bergdahl, J.; Hultdin, J.; Del-Favero, J.; Roos, G.; Nilsson, L.G.; Adolfsson, R.; et al. Short telomeres in depression and the general population are associated with a hypocortisolemic state. Biol. Psychiatry 2012, 71, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Karabatsiakis, A.; Kolassa, I.T.; Kolassa, S.; Rudolph, K.L.; Dietrich, D.E. Telomere shortening in leukocyte subpopulations in depression. BMC Psychiatry 2014, 14, 192. [Google Scholar] [CrossRef] [PubMed]
- Ladwig, K.H.; Brockhaus, A.C.; Baumert, J.; Lukaschek, K.; Emeny, R.T.; Kruse, J.; Codd, V.; Hafner, S.; Albrecht, E.; Illig, T.; et al. Posttraumatic stress disorder and not depression is associated with shorter leukocyte telomere length: Findings from 3 000 participants in the population-based KORA F4 study. PLoS ONE 2013, 8, e64762. [Google Scholar] [CrossRef] [PubMed]
- Needham, B.L.; Mezuk, B.; Bareis, N.; Lin, J.; Blackburn, E.H.; Epel, E.S. Depression, anxiety and telomere length in young adults: Evidence from the National Health and Nutrition Examination Survey. Mol. Psychiatry 2015, 20, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, J.A.; Epel, E.; Kang, M.S.; Ye, S.; Schwartz, J.E.; Davidson, K.W.; Kirkland, S.; Honig, L.S.; Shimbo, D. Depressive symptoms are not associated with leukocyte telomere length: Findings from the Nova Scotia Health Survey (NSHS95), a population-based study. PLoS ONE 2012, 7, e48318. [Google Scholar] [CrossRef]
- Wium-Andersen, M.K.; Orsted, D.D.; Rode, L.; Bojesen, S.E.; Nordestgaard, B.G. Telomere length and depression: Prospective cohort study and Mendelian randomisation study in 67 306 individuals. Br. J. Psychiatry 2017, 210, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Pompili, M.; Venturini, P.; Lamis, D.A.; Giordano, G.; Serafini, G.; Belvederi Murri, M.; Amore, M.; Girardi, P. Suicide in stroke survivors: Epidemiology and prevention. Drugs Aging 2015, 32, 21–29. [Google Scholar] [CrossRef]
- O’Donovan, A.; Pantell, M.S.; Puterman, E.; Dhabhar, F.S.; Blackburn, E.H.; Yaffe, K.; Cawthon, R.M.; Opresko, P.L.; Hsueh, W.C.; Satterfield, S.; et al. Cumulative inflammatory load is associated with short leukocyte telomere length in the Health, Aging and Body Composition Study. PLoS ONE 2011, 6, e19687. [Google Scholar] [CrossRef]
- Shiels, P.G.; McGlynn, L.M.; MacIntyre, A.; Johnson, P.C.; Batty, G.D.; Burns, H.; Cavanagh, J.; Deans, K.A.; Ford, I.; McConnachie, A. Accelerated telomere attrition is associated with relative household income, diet and inflammation in the pSoBid cohort. PLoS ONE 2011, 6, e22521. [Google Scholar] [CrossRef]
- Wolkowitz, O.M.; Mellon, S.H.; Epel, E.S.; Lin, J.; Dhabhar, F.S.; Su, Y.; Reus, V.I.; Rosser, R.; Burke, H.M.; Kupferman, E.; et al. Leukocyte telomere length in major depression: Correlations with chronicity, inflammation and oxidative stress—Preliminary findings. PLoS ONE 2011, 6, e17837. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). NCHS Research Ethics Review Board (ERB) Approval. Available online: https://www.cdc.gov/nchs/nhanes/irba98.htm (accessed on 10 January 2019).
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey 1999–2000 Data Documentation, Codebook, and Frequencies: C-Reactive Protein (CRP) (LAB11). Available online: https://wwwn.cdc.gov/Nchs/Nhanes/1999-2000/LAB11.htm#Component_Description (accessed on 13 November 2018).
- World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines. 1992. Available online: https://apps.who.int/iris/handle/10665/37958 (accessed on 13 November 2018).
- Kessler, R.C.; Andrews, G.; Mroczek, D.; Ustun, B.; Wittchen, H.U. The World Health Organization Composite International Diagnostic Interview Short-Form (CIDI-SF). Int. J. Methods Psychiatr. Res. 1998, 7, 171–185. [Google Scholar] [CrossRef]
- Needham, B.L.; Adler, N.; Gregorich, S.; Rehkopf, D.; Lin, J.; Blackburn, E.H.; Epel, E.S. Socioeconomic status, health behavior, and leukocyte telomere length in the National Health and Nutrition Examination Survey, 1999–2002. Soc. Sci. Med. 2013, 85, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Cawthon, R.M. Telomere measurement by quantitative PCR. Nucleic Acids Res. 2002, 30, e47. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey 1999-2000 Data Documentation, Codebook, and Frequencies: Telomere Mean and Standard Deviation (Surplus) (TELO_A). Available online: https://wwwn.cdc.gov/nchs/nhanes/1999-2000/TELO_A.htm (accessed on 20 March 2019).
- Seeman, T.E.; McEwen, B.S.; Rowe, J.W.; Singer, B.H. Allostatic load as a marker of cumulative biological risk: MacArthur studies of successful aging. Proc. Natl. Acad. Sci. USA 2001, 98, 4770–4775. [Google Scholar] [CrossRef] [Green Version]
- Lavretsky, H.; Newhouse, P.A. Stress, inflammation, and aging. Am. J. Geriatr. Psychiatry 2012, 20, 729–733. [Google Scholar] [CrossRef] [PubMed]
- Bender, D.A. Biochemistry of tryptophan in health and disease. Mol. Aspects Med. 1983, 6, 101–197. [Google Scholar] [CrossRef]
- Myint, A.M.; Kim, Y.K.; Verkerk, R.; Scharpe, S.; Steinbusch, H.; Leonard, B. Kynurenine pathway in major depression: Evidence of impaired neuroprotection. J. Affect Disord. 2007, 98, 143–151. [Google Scholar] [CrossRef]
- Lovelace, M.D.; Varney, B.; Sundaram, G.; Lennon, M.J.; Lim, C.K.; Jacobs, K.; Guillemin, G.J.; Brew, B.J. Recent evidence for an expanded role of the kynurenine pathway of tryptophan metabolism in neurological diseases. Neuropharmacology 2017, 112, 373–388. [Google Scholar] [CrossRef]
- Braidy, N.; Guillemin, G.J.; Mansour, H.; Chan-Ling, T.; Grant, R. Changes in kynurenine pathway metabolism in the brain, liver and kidney of aged female Wistar rats. FEBS J. 2011, 278, 4425–4434. [Google Scholar] [CrossRef] [Green Version]
- Khansari, N.; Shakiba, Y.; Mahmoudi, M. Chronic inflammation and oxidative stress as a major cause of age-related diseases and cancer. Recent Pat. Inflamm. Allergy Drug Discov. 2009, 3, 73–80. [Google Scholar] [CrossRef]
- Alonso-Alvarez, C.; Bertrand, S.; Faivre, B.; Chastel, O.; Sorci, G. Testosterone and oxidative stress: The oxidation handicap hypothesis. Proc.Biol. Sci. 2007, 274, 819–825. [Google Scholar] [CrossRef]
- Kyo, S.; Takakura, M.; Kanaya, T.; Zhuo, W.; Fujimoto, K.; Nishio, Y.; Orimo, A.; Inoue, M. Estrogen activates telomerase. Cancer Res. 1999, 59, 5917–5921. [Google Scholar]
- Viña, J.; Sastre, J.; Pallardo, F.; Gambini, J.; Borras, C. Role of mitochondrial oxidative stress to explain the different longevity between genders: Protective effect of estrogens. Free Radic. Res. 2006, 40, 1359–1365. [Google Scholar] [CrossRef]
- Zalli, A.; Carvalho, L.A.; Lin, J.; Hamer, M.; Erusalimsky, J.D.; Blackburn, E.H.; Steptoe, A. Shorter telomeres with high telomerase activity are associated with raised allostatic load and impoverished psychosocial resources. Proc. Natl. Acad. Sci. USA 2014, 111, 4519–4524. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, P.J.; Ben Dor, R.; Martinez, P.E.; Guerrieri, G.M.; Harsh, V.L.; Thompson, K.; Koziol, D.E.; Nieman, L.K.; Rubinow, D.R. Effects of Estradiol Withdrawal on Mood in Women With Past Perimenopausal Depression: A Randomized Clinical Trial. JAMA Psychiatry 2015, 72, 714–726. [Google Scholar] [CrossRef]
- Shors, T.J.; Leuner, B. Estrogen-mediated effects on depression and memory formation in females. J. Affect. Disord. 2003, 74, 85–96. [Google Scholar] [CrossRef] [Green Version]
- Keyes, K.M.; Cheslack-Postava, K.; Westhoff, C.; Heim, C.M.; Haloossim, M.; Walsh, K.; Koenen, K. Association of hormonal contraceptive use with reduced levels of depressive symptoms: A national study of sexually active women in the United States. Am. J. Epidemiol. 2013, 178, 1378–1388. [Google Scholar] [CrossRef]
- Eaton, W.W.; Hall, A.L.; Macdonald, R.; Mckibben, J. Case identification in psychiatric epidemiology: A review. Int. Rev. Psychiatry 2007, 19, 497–507. [Google Scholar] [CrossRef] [PubMed]
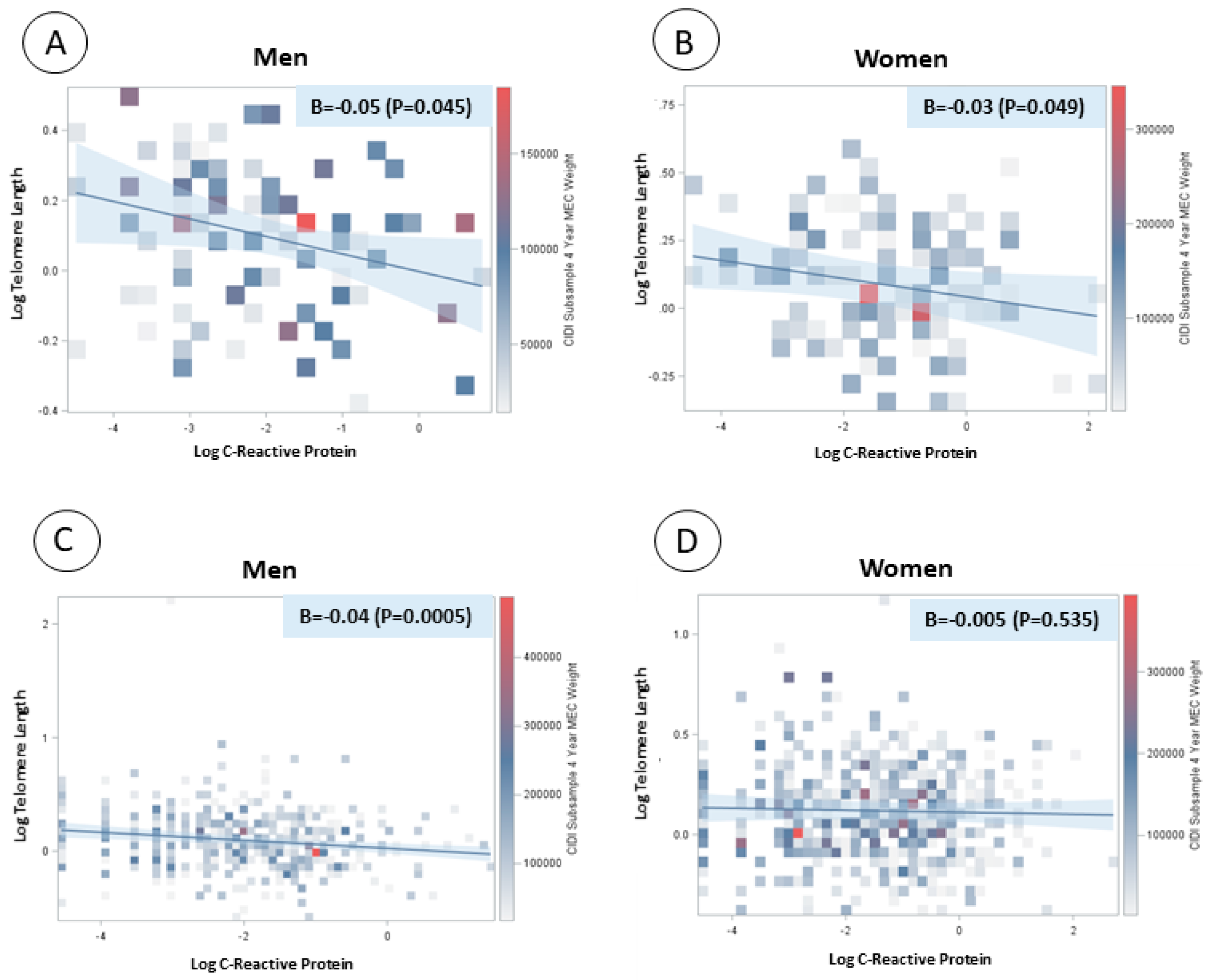
| Men | Women | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Telomere Length | Telomere Length | |||||||||||||
| Tertile 1 (n = 165) | Tertile 2 (n = 166) | Tertile 3 (n = 165) | Tertile 1 (n = 215) | Tertile 2 (n = 215) | Tertile 3 (n = 215) | |||||||||
| Mean | (SEM) | Mean | (SEM) | Mean | (SEM) | p Value | Mean | (SEM) | Mean | (SEM) | Mean | (SEM) | p Value | |
| Age | 31.8 | (0.5) | 29.2 | (0.6) | 28.5 | (0.6) | <0.0001 | 31.3 | (0.6) | 29.5 | (0.5) | 28.9 | (0.6) | 0.0062 |
| Family poverty income ratio | 2.9 | (0.2) | 2.9 | (0.2) | 2.7 | (0.1) | 0.0925 | 2.9 | (0.1) | 2.8 | (0.1) | 2.2 | (0.1) | 0.0006 |
| BMI (kg/m2) | 28.4 | (0.5) | 27.7 | (0.5) | 25.6 | (0.4) | <0.0001 | 27.5 | (0.7) | 26.6 | (0.6) | 27.2 | (0.5) | 0.7436 |
| C-reactive protein (mg/dL) | 0.33 | (0.04) | 0.29 | (0.04) | 0.20 | (0.03) | 0.027 | 0.46 | (0.07) | 0.43 | (0.03) | 0.41 | (0.05) | 0.5535 |
| n | (Wt’d %) | n | (Wt’d %) | n | (Wt’d %) | p value | n | (Wt’d %) | n | (Wt’d %) | n | (Wt’d %) | p value | |
| Race/Ethnicity | ||||||||||||||
| Mexican-American or other Hispanic | 66 | (19.4) | 56 | (16.1) | 47 | (13.2) | 0.5876 | 69 | (18.5) | 78 | (18.1) | 56 | (16.5) | 0.8552 |
| Non-Hispanic white | 72 | (68.2) | 75 | (68.0) | 83 | (73.0) | 103 | (68.0) | 98 | (67.8) | 101 | (65.3) | ||
| Non-Hispanic black | 21 | (7.0) | 29 | (9.9) | 30 | (9.7) | 34 | (9.9) | 32 | (10.6) | 48 | (14.9) | ||
| Other-including multi-racial | 6 | (5.5) | 6 | (6.0) | 5 | (4.1) | 9 | (3.6) | 7 | (3.5) | 10 | (3.3) | ||
| Education | ||||||||||||||
| ≤11th grade | 53 | (21.6) | 51 | (21.1) | 43 | (17.8) | 0.3973 | 54 | (15.7) | 50 | (17.0) | 49 | (16.8) | 0.9493 |
| High school grad/GED or equivalent | 43 | (28.5) | 38 | (25.6) | 54 | (35.2) | 49 | (27.4) | 53 | (24.3) | 50 | (23.3) | ||
| ≥ Some college or associate’s degree | 69 | (49.9) | 77 | (53.3) | 68 | (47.0) | 112 | (56.9) | 112 | (58.8) | 116 | (59.9) | ||
| Marital status | ||||||||||||||
| Never married | 38 | (23.2) | 75 | (44.4) | 74 | (43.8) | 0.0006 | 37 | (20.4) | 55 | (28.5) | 71 | (36.3) | 0.0623 |
| Married/living with partner | 114 | (69.4) | 78 | (47.3) | 82 | (50.7) | 147 | (65.5) | 141 | (61.3) | 121 | (51.5) | ||
| Widowed/divorced/separated | 13 | (7.4) | 13 | (8.2) | 9 | (5.5) | 31 | (14.1) | 19 | (10.2) | 23 | (12.2) | ||
| Depression | ||||||||||||||
| Major depression or depressed affect | 24 | (13.7) | 30 | (19.0) | 29 | (15.2) | 0.5171 | 40 | (19.9) | 35 | (17.8) | 37 | (17.9) | 0.924 |
| No | 141 | (86.3) | 136 | (81.0) | 136 | (84.8) | 175 | (80.1) | 180 | (82.2) | 178 | (82.1) | ||
| Physical activity | ||||||||||||||
| 0 MET | 64 | (32.0) | 55 | (29.1) | 49 | (29.4) | 0.5099 | 97 | (37.0) | 78 | (28.5) | 78 | (25.3) | 0.1379 |
| 0–500 MET | 34 | (24.3) | 40 | (22.1) | 32 | (17.2) | 46 | (22.4) | 53 | (23.5) | 57 | (30.3) | ||
| 500–1000 MET | 12 | (7.6) | 13 | (7.3) | 21 | (12.4) | 30 | (17.1) | 26 | (12.9) | 26 | (14.2) | ||
| >1000 MET | 55 | (36.2) | 58 | (41.5) | 63 | (41.0) | 42 | (23.5) | 58 | (35.1) | 54 | (30.2) | ||
| Smoking status | ||||||||||||||
| Never | 87 | (51.7) | 95 | (58.1) | 73 | (43.7) | 0.0637 | 135 | (52.7) | 143 | (61.9) | 135 | (59.4) | 0.4439 |
| Past | 25 | (14.9) | 11 | (7.1) | 29 | (16.9) | 41 | (18.2) | 24 | (11.8) | 25 | (9.8) | ||
| Current | 53 | (33.5) | 60 | (34.8) | 63 | (39.4) | 39 | (29.1) | 48 | (26.3) | 55 | (30.9) | ||
| Alcohol status (n = 464 for men and n = 576 for women) | ||||||||||||||
| Lifetime abstainer | 10 | (5.9) | 15 | (8.0) | 13 | (10.4) | NA | 31 | (11.5) | 48 | (17.5) | 53 | (26.8) | 0.0142 |
| Former drinker | 5 | (4.1) | 0 | (0.0) | 1 | (1.3) | 10 | (3.5) | 5 | (1.5) | 4 | (1.7) | ||
| Current drinker with moderate alcohol consumption | 59 | (40.5) | 55 | (38.0) | 48 | (30.4) | 60 | (29.4) | 45 | (22.5) | 44 | (21.9) | ||
| Current drinker with above moderate alcohol consumption | 75 | (49.5) | 89 | (54.0) | 94 | (57.9) | 95 | (55.6) | 92 | (58.5) | 89 | (49.6) | ||
| C-Reactive Protein (mg/dL) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | p-for-Trend | |||||
| Geometric mean telomere length | (SEM) | Geometric mean telomere length | (SEM) | Geometric mean telomere length | (SEM) | |||
| Men | No depression (n = 413) | 1.15 | (0.03) | 1.12 | (0.02) | 1.04 | (0.02) | <0.0001 |
| Major depression or depressed affect (n = 83) | 1.17 | (0.06) | 1.14 | (0.04) | 1.01 | (0.04) | <0.0001 | |
| Women | No depression (n = 533) | 1.14 | (0.04) | 1.12 | (0.02) | 1.12 | (0.03) | 0.0074 |
| Major depression or depressed affect (n = 112) | 1.15 | (0.05) | 1.06 | (0.06) | 1.07 | (0.05) | <0.0001 | |
| Men | Women | |||
| C-Reactive Protein | C-Reactive Protein | |||
| Short telomere length | ≤0.2 mg/dL | >0.2 mg/dL | ≤0.2 mg/dL | >0.2 mg/dL |
| Unadjusted model | 1.00 (Ref.) | 2.24 (1.39–3.59) | 1.00 (Ref.) | 1.16 (0.83–1.62) |
| Multivariable model * | 1.00 (Ref.) | 1.81 (1.12–2.91) | 1.00 (Ref.) | 1.02 (0.69–1.52) |
| Depression status | Depression status | |||
| Short telomere length | No depression | Major depression or depressed affect | No depression | Major depression or depressed affect |
| Unadjusted model | 1.00 (Ref.) | 1.09 (0.71–1.67) | 1.00 (Ref.) | 0.94 (0.48–1.82) |
| Multivariable model * | 1.00 (Ref.) | 1.10 (0.72–1.66) | 1.00 (Ref.) | 0.74 (0.41–1.34) |
| Short Telomere Length | Depression Status | |||
| Major Depression or Depressed Affect | No Depression | |||
| C-Reactive Protein | C-Reactive Protein | |||
| Men | ≤0.2 mg/dL | >0.2 mg/dL | ≤0.2 mg/dL | >0.2 mg/dL |
| Unadjusted model | 1.00 (Ref.) | 2.68 (0.81–8.84) | 1.00 (Ref.) | 2.16 (1.37–3.39) |
| Multivariable model * | 1.00 (Ref.) | 0.94 (0.22–4.01) | 1.00 (Ref.) | 1.77 (1.09–2.90) |
| Major depression or depressed affect | No depression | |||
| C-reactive protein | C-reactive protein | |||
| Women | ≤0.2 mg/dL | >0.2 mg/dL | ≤0.2 mg/dL | >0.2 mg/dL |
| Unadjusted model | 1.00 (Ref.) | 2.23 (0.97–5.11) | 1.00 (Ref.) | 1.01 (0.65–1.56) |
| Multivariable model * | 1.00 (Ref.) | 2.56 (0.76–8.61) | 1.00 (Ref.) | 0.87 (0.52–1.43) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, D.; Shin, J.; Lee, K.W. Effects of Inflammation and Depression on Telomere Length in Young Adults in the United States. J. Clin. Med. 2019, 8, 711. https://0-doi-org.brum.beds.ac.uk/10.3390/jcm8050711
Shin D, Shin J, Lee KW. Effects of Inflammation and Depression on Telomere Length in Young Adults in the United States. Journal of Clinical Medicine. 2019; 8(5):711. https://0-doi-org.brum.beds.ac.uk/10.3390/jcm8050711
Chicago/Turabian StyleShin, Dayeon, Jungwoon Shin, and Kyung Won Lee. 2019. "Effects of Inflammation and Depression on Telomere Length in Young Adults in the United States" Journal of Clinical Medicine 8, no. 5: 711. https://0-doi-org.brum.beds.ac.uk/10.3390/jcm8050711





