Synthesis, Mass Spectroscopy Detection, and Density Functional Theory Investigations of the Gd Endohedral Complexes of C82 Fullerenols
Abstract
:1. Introduction
2. Experimental Procedures
3. Computational Details
4. Results and Discussion
4.1. The C82Ox(OH)y and Gd@C82Ox(OH)y Atomic Structures
4.2. The C82Ox(OH)y and Gd@C82Ox(OH)y Electronic Structures
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Popov, A.A.; Yang, S.; Dunsch, L. Endohedral Fullerenes. Chem. Rev. 2013, 113, 5989–6113. [Google Scholar] [CrossRef]
- Shinohara, H. Endohedral metallofullerenes. Rep. Prog. Phys. 2000, 63, 843–892. [Google Scholar] [CrossRef]
- Yang, S.; Wei, T.; Jin, F. When metal clusters meet carbon cages: Endohedral clusterfullerenes. Chem. Soc. Rev. 2017, 46, 5005–5058. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gao, X.; Zhao, Y. Mechanisms of Antioxidant Activities of Fullerenols from First-Principles Calculation. J. Phys. Chem. 2018, 122, 8183–8190. [Google Scholar] [CrossRef]
- Zhang, J.; Ye, Y.; Chen, Y.; Pregot, C.; Li, T.; Balasubramaniam, S.; Hobart, D.B.; Zhang, Y.; Wi, S.; Davis, R.M.; et al. Gd3N@C84(OH)x: A New Egg-Shaped Metallofullerene Magnetic Resonance Imaging Contrast Agent. J. Am. Chem. Soc. 2014, 136, 2630–2636. [Google Scholar] [CrossRef]
- Da Ross, T. Twenty Years of Promises: Fullerene in Medicinal Chemistry. In Medicinal Chemistry and Pharmacological Potential of Fullerenes and Carbon Nanotubes; Cataldo, F., Da Ros, T., Eds.; Carbon Materials: Chemistry and Physics; Springer: Dordrecht, The Netherlands, 2008; Volume 1. [Google Scholar] [CrossRef]
- Kang, S.G.; Huynh, T.; Zhou, R. Non-destructive Inhibition of Metallofullerenol Gd@C82(OH)22 on WW domain: Implication on Signal Transduction Pathway. Sci. Rep. 2012, 2, 957. [Google Scholar] [CrossRef] [Green Version]
- Kang, S.G.; Zhou, G.; Yang, P.; Liu, Y.; Sun, B.; Huynh, T.; Meng, H.; Zhao, L.; Xing, G.; Chen, C.; et al. Molecular mechanism of pancreatic tumor metastasis inhibition by Gd@C82(OH)22 and its implication for de novo design of nanomedicine. Proc. Natl. Acad. Sci. USA 2012, 109, 15431–15436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, J.; Liang, X.; Chen, X.; Zhao, Y. Biological characterizations of [Gd@C82(OH)22]n nanoparticles as fullerene derivatives for cancer therapy. Integr. Biol. 2013, 5, 43–47. [Google Scholar] [CrossRef] [Green Version]
- Churilov, G.N.; Kratschmer, W.; Osipova, I.V.; Glushenko, G.A.; Vnukova, N.G.; Kolonenko, A.L.; Dudnik, A.I. Sinthesis of fullerenes in a high-frequency arc plasma under elevated helium pressure. Carbon 2013, 62, 389–392. [Google Scholar] [CrossRef]
- Akiyama, K.; Hamano, T.; Nakanishi, Y.; Takeuchi, E.; Noda, S.; Wang, Z.; Kubuki, S.; Shinohara, H. Non-HPLC rapid separation of metallofullerenes and empty cages with TiCl4 Lewis acid. J. Am. Chem. Soc. 2012, 134, 9762–9767. [Google Scholar] [CrossRef] [PubMed]
- Chiang, L.Y.; Swirczewski, J.W.; Hsu, C.S.; Chowdhury, S.K.; Cameron, S.; Creegan, K. Multi-hydroxy additions onto C60 fullerene molecules. J. Chem. Soc. Chem. Commun. 1992, 114, 1791–1793. [Google Scholar] [CrossRef]
- Shilin, V.A.; Szhogina, A.A.; Suyasova, M.V.; Sedov, V.P.; Lebedev, V.T.; Kozlov, V.S. Fullerenes and fullerenols survival under irradiation. Nanosystems 2016, 7, 146–152. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.C.; Wang, H.W.; Tso, H.C.; Chen, T.L.; Chou, Y.M. Theoretical studies of C70(OH)n (n = 14, 16, 18 and 20) fullerols. J. Mol. Struct. THEOCHEM 2002, 581, 177–186. [Google Scholar] [CrossRef]
- Aschberger, K.; Johnston, H.J.; Stone, V.; Aitken, R.J.; Tran, C.L.; Hankin, S.M.; Peters, S.A.K.; Christensen, F.M. Review of fullerene toxicity and exposure—Appraisal of a human health risk assessment, based on open literature. Regul. Toxicol. Pharmacol. 2010, 58, 455–473. [Google Scholar] [CrossRef] [PubMed]
- Trpkovic, A.; Todorovic-Markovic, B.; Trajkovic, V. Toxicity of pristine versus functionalized fullerenes: Mechanisms of cell damage and the role of oxidative stress. Arch. Toxicol. 2012, 86, 1809–1827. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xing, G.; Wang, J.; Zhao, Y.; Li, B.; Tang, J.; Jia, G.; Wang, T.; Sun, J.; Xing, L. Multihydroxylated [Gd@C82(OH)22]n Nanoparticles: Antineoplastic Activity of High Efficiency and Low Toxicity. Nano Lett. 2005, 5, 2050–2057. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ma, L.; Liu, Y.; Chen, C. Applications of Functionalized Fullerenes in Tumor Theranostics. Theranostics 2012, 2, 238–250. [Google Scholar] [CrossRef] [Green Version]
- Keshri, S.; Tembe, B.L. Thermodynamics of hydration of fullerols [C60(OH)n] and hydrogen bond dynamics in their hydration shells. J. Chem. Phys. 2017, 146, 074501. [Google Scholar] [CrossRef]
- Djordjevic, A.; Srdjenovic, B.; Seke, M.; Petrovic, D.; Injac, R.; Mrdjanovic, J.J. Review of synthesis and antioxidant potential of fullerenol nanoparticles. J. Nanomater. 2015, 2015, 567073. [Google Scholar] [CrossRef] [Green Version]
- Eropkin, M.Y.; Melenevskaya, E.Y.; Nasonova, K.V.; Bryazzhikova, T.S.; Eropkina, E.M.; Danilenko, D.M.; Kiselev, O.I. Synthesis and biological activity of fullerenols with various contents of hydroxyl groups. Pharm. Chem. J. 2013, 47, 87–91. [Google Scholar] [CrossRef]
- Kovel, E.S.; Sachkova, A.S.; Vnukova, N.G.; Churilov, G.N.; Knyazeva, E.M.; Kudryasheva, N.S. Antioxidant activity and toxicity of fullerenols via bioluminescence signaling: Role of oxygen substituents. Int. J. Mol. Sci. 2019, 20, 2324. [Google Scholar] [CrossRef] [Green Version]
- Kudryasheva, N.S.; Kovel, E.S.; Sachkova, A.S.; Vorobeva, A.A.; Isakova, V.G.; Churilov, G.N. Bioluminescent enzymatic assay as a tool for studying antioxidant activity and toxicity of bioactive compounds. Photochem. Photobiol. 2017, 93, 536–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Guseynov, O.A.; Bondar, A.A.; Dubinina, I.A.; Kudryasheva, N.S. On mechanism of antioxidant effect of fullerenols. Biochem. Biophys. Rep. 2017, 9, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Sachkova, A.S.; Kovel, E.S.; Churilov, G.N.; Stom, D.I.; Kudryasheva, N.S. Biological activity of carbonic nano-structures—comparison via enzymatic bioassay. J. Soils Sediments 2019, 19, 2689–2696. [Google Scholar] [CrossRef] [Green Version]
- Maravilla, K.R.; San-Juan, D.; Kim, S.J.; Elizondo-Riojas, G.; Fink, J.R.; Escobar, W.; Bag, A.; Roberts, D.R.; Hao, J.; Pitrou, C.; et al. Comparison of Gadoterate Meglumine and Gadobutrol in the MRI Diagnosis of Primary Brain Tumors: A Double-Blind Randomized Controlled Intraindividual Crossover Study (the REMIND study). Am. J. Neuroradiol. 2017, 38, 1681–1688. [Google Scholar] [CrossRef] [Green Version]
- Ersoy, H.; Rybicki, F.J. Biochemical Safety Profiles of Gadolinium-Based Extracellular Contrast Agents and Nephrogenic Systemic Fibrosis. J. Magn. Reson. Imaging. 2007, 26, 1190–1197. [Google Scholar] [CrossRef] [Green Version]
- Guan, M.; Ge, J.; Wu, J.; Zhang, G.; Chen, D.; Zhang, W.; Zhang, Y.; Zou, T.; Zhen, M.; Wang, C.; et al. Fullerene/photosensitizer nanovesicles as highly efficient and clearable phototheranostics with enhanced tumor accumulation for cancer therapy. Biomaterials 2016, 103, 75–85. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez-Galvan, A.; Rivera, M.; Garcia-Lopez, P. Gadolinium-containing carbon nanomaterials for magnetic resonance imaging: Trends and challenges. J. Cell. Mol. Med. 2020, 24, 3779–3794. [Google Scholar] [CrossRef] [Green Version]
- Tang, J.; Zhang, R.; Guo, M. Gd-metallofullerenol drug delivery system mediated macrophage polarization enhaces the efficiency of chemotherapy. J. Control. Release 2020, 320, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Clavaguéra, C.; Sansot, E.; Calvo, F.; Dognon, J.P. Gd(III) polyaminocarboxylate chelate: Realistic many-body molecular dynamics simulations for molecular imaging applications. J. Phys. Chem. 2006, 110, 12848–12851. [Google Scholar] [CrossRef] [Green Version]
- Dudnik, A.I.; Vnukova, N.G.; Drokin, N.A.; Bondarev, V.S.; Shestakov, N.P.; Tomashevich, Y.V.; Churilov, G.N. Electrophysical properties of hydroxylated endohedral metallofullerene with gadolinium. J. Phys. Chem. Solids. 2019, 135, 109094. [Google Scholar] [CrossRef]
- Yu, H.S.; He, X.; Li, S.L.; Truhlar, D.G. MN15: A Kohn–Sham global-hybrid exchange–correlation density functional with broad accuracy for multi-reference and single-reference systems and noncovalent interactions. Chem. Sci. 2016, 7, 5032–5051. [Google Scholar] [CrossRef] [Green Version]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. 2005, 7, 3297. [Google Scholar] [CrossRef] [PubMed]
- Gulde, R.; Pollak, P.; Weigend, F. Error-Balanced Segmented Contracted Basis Sets of Double-ζ to Quadruple-ζ Valence Quality for the Lanthanides. J. Chem. Theory Comput. 2012, 8, 4062–4068. [Google Scholar] [CrossRef]
- Frisch, M.J. Gaussian 09, Revision A, 1st ed.; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal solvation model based on solute electron density and a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. 2009, 113, 6378–6396. [Google Scholar] [CrossRef] [PubMed]
- Dolg, M.; Stoll, H.; Preuss, H. Energy-adjusted ab initio pseudopotentials for the rare earth elements. J. Chem. Phys. 1989, 90, 1730–1734. [Google Scholar] [CrossRef]
- Pearson, R.G. Chemical Hardness: Applications from Molecules to Solids; Wiley: Weinheim, Germany, 1997. [Google Scholar]
- Nishibori, E.; Iwata, K.; Sakata, M.; Takata, M.; Tanaka, H.; Kato, H.; Shinohara, H. Anomalous endohedral structure of Gd@C82 metallofullerenes. Phys. Rev. 2004, 69, 113412. [Google Scholar] [CrossRef]
- Dai, X.; Gao, Y.; Xin, M.; Wang, Z.; Zhou, R. The ground state and electronic structure of Gd@C82: A systematic theoretical investigation of first principle density functional. J. Chem. Phys. 2014, 141, 244306. [Google Scholar] [CrossRef]
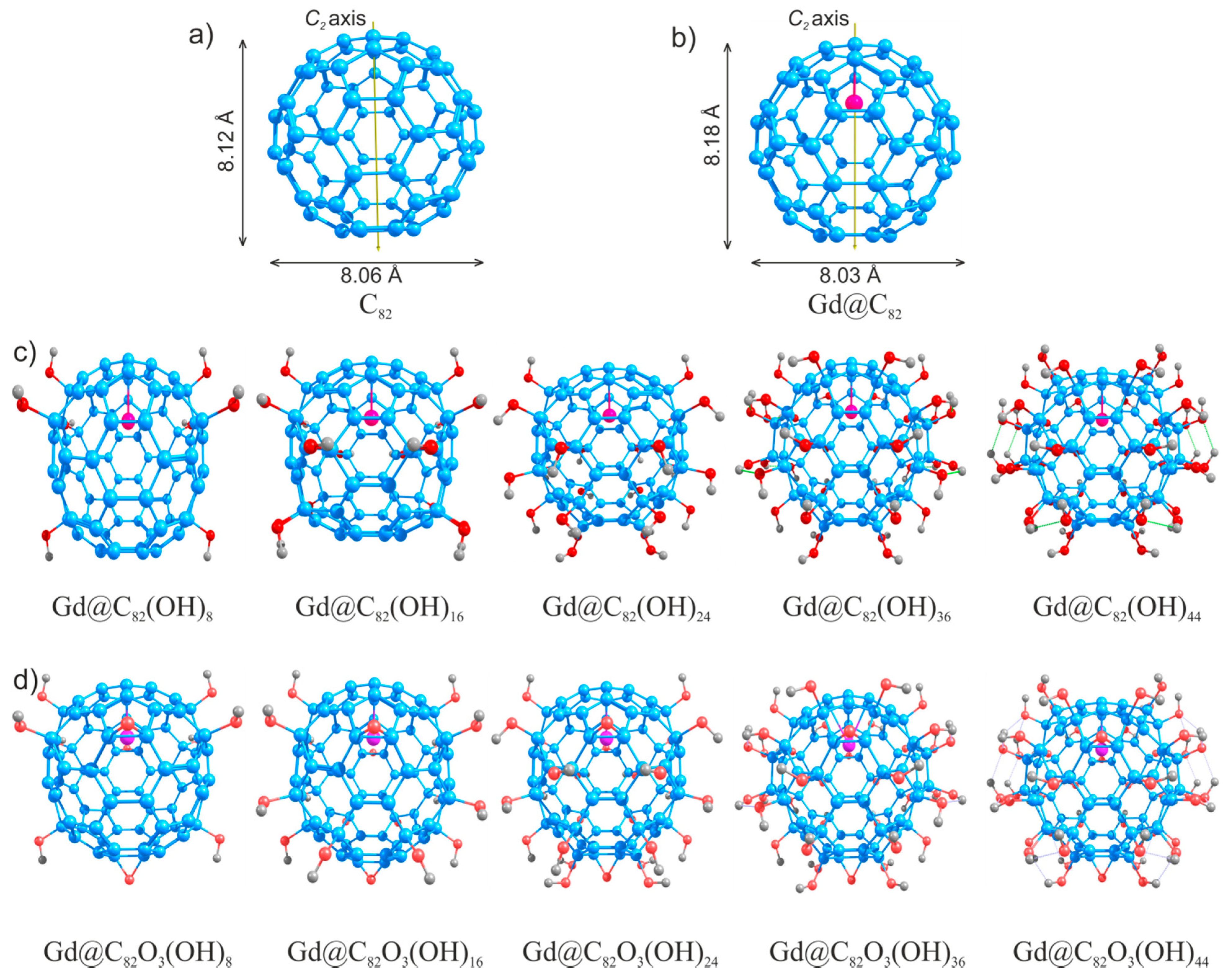

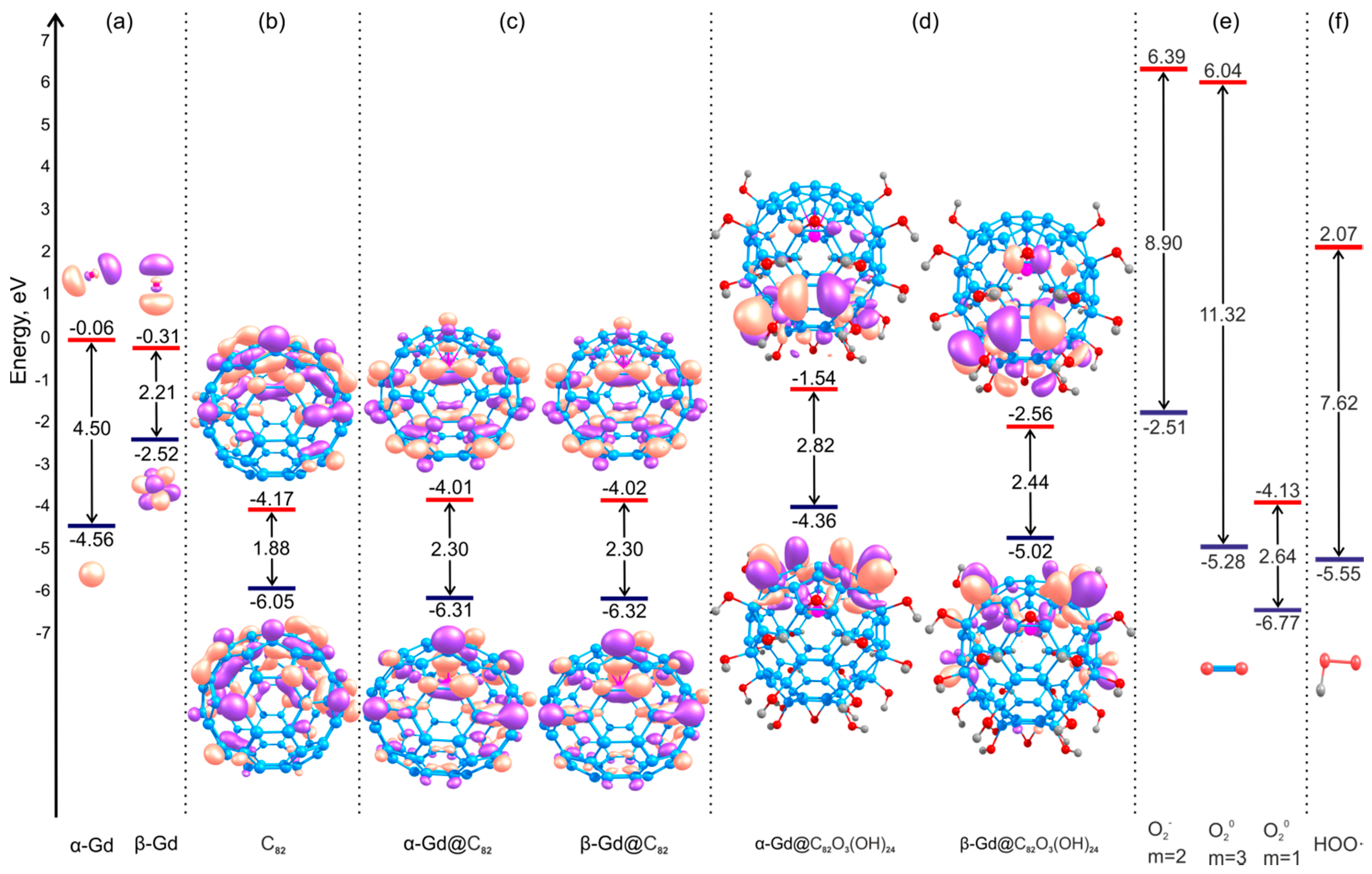
| Bond Lengths and Bond Angles | Complexes | ||||
| C82(OH)y | |||||
| C82(OH)8 | C82(OH)16 | C82(OH)24 | C82(OH)36 | C82(OH)44 | |
| l(C1-C2) | 1.42 | 1.41 | 1.42 | 1.53 | 1.53 |
| l(C2=C3) | 1.41 | 1.41 | 1.41 | 1.35 | 1.33 |
| ∠C1–2–3 | 121 | 121 | 121 | 124 | 123 |
| ∠C1–2–4 | 109 | 109 | 109 | 109 | 112 |
| ∠C3–2–4 | 122 | 122 | 121 | 118 | 122 |
| Gd@C82(OH)y | |||||
| Gd@C82(OH)8 | Gd@C82(OH)16 | Gd@C82(OH)24 | Gd@C82(OH)36 | Gd@C82(OH)44 | |
| l(C1-C2) | 1.42 | 1.41 | 1.41 | 1.52 | 1.51 |
| l(C2=C3) | 1.41 | 1.41 | 1.40 | 1.35 | 1.33 |
| ∠C1-2-3 | 119 | 120 | 120 | 121 | 122 |
| ∠C1-2-4 | 108 | 109 | 109 | 109 | 113 |
| ∠C3-2-4 | 125 | 121 | 121 | 119 | 122 |
| C82Ox(OH)y | |||||
| C82O3(OH)8 | C82O3(OH)16 | C82O3(OH)24 | C82O3(OH)36 | C82O3(OH)44 | |
| l(C1-C2) | 1.42 | 1.41 | 1.41 | 1.53 | 1.53 |
| l(C2=C3) | 1.41 | 1.41 | 1.41 | 1.35 | 1.33 |
| ∠C1-2-3 | 121 | 121 | 120 | 124 | 123 |
| ∠C1-2-4 | 109 | 109 | 109 | 109 | 113 |
| ∠C3-2-4 | 122 | 122 | 122 | 119 | 122 |
| Gd@C82Ox(OH)y | |||||
| Gd@C82O3(OH)8 | Gd@C82O3(OH)16 | Gd@C82O3(OH)24 | Gd@C82O3(OH)36 | Gd@C82O3(OH)44 | |
| l(C1-C2) | 1.42 | 1.42 | 1.41 | 1.53 | 1.52 |
| l(C2=C3) | 1.41 | 1.41 | 1.38 | 1.33 | 1.33 |
| ∠C1-2-3 | 120 | 120 | 120 | 123 | 122 |
| ∠C1-2-4 | 109 | 109 | 109 | 109 | 112 |
| ∠C3-2-4 | 123 | 123 | 122 | 118 | 121 |
| C82: l(C1-C2) = 1.43, l(C2=C3) = 1.42, ∠C1-2-3 = 121, ∠C1-2-4 = 108, ∠C3-2-4 = 121; Gd@C82: l(C1-C2) = 1.45, l(C2=C3) = 1.44, ∠C1-2-3 = 121, ∠C1-2-4 = 107, ∠C3-2-4 = 121 | |||||
| Indexes | Complexes | |||||
|---|---|---|---|---|---|---|
| C82Ox(OH)y | ||||||
| C82O3(OH)8 | C82O3(OH)16 | C82O3(OH)24 | ||||
| ω * | 7.3 | 5.2 | 11.6 | |||
| χ ** | −4.8 | −4.5 | −4.8 | |||
| Gd@C82Ox(OH)y | ||||||
| Gd@C82O3(OH)8 | Gd@C82O3(OH)16 | Gd@C82O3(OH)24 | ||||
| α | β | α | β | α | β | |
| ω | 13.0 | 6.5 | 5.7 | 5.8 | 3.1 | 5.8 |
| χ | −4.3 | −4.6 | −3.2 | −3.2 | −2.9 | −3.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakirova, A.A.; Tomilin, F.N.; Pomogaev, V.A.; Vnukova, N.G.; Churilov, G.N.; Kudryasheva, N.S.; Tchaikovskaya, O.N.; Ovchinnikov, S.G.; Avramov, P.V. Synthesis, Mass Spectroscopy Detection, and Density Functional Theory Investigations of the Gd Endohedral Complexes of C82 Fullerenols. Computation 2021, 9, 58. https://0-doi-org.brum.beds.ac.uk/10.3390/computation9050058
Shakirova AA, Tomilin FN, Pomogaev VA, Vnukova NG, Churilov GN, Kudryasheva NS, Tchaikovskaya ON, Ovchinnikov SG, Avramov PV. Synthesis, Mass Spectroscopy Detection, and Density Functional Theory Investigations of the Gd Endohedral Complexes of C82 Fullerenols. Computation. 2021; 9(5):58. https://0-doi-org.brum.beds.ac.uk/10.3390/computation9050058
Chicago/Turabian StyleShakirova, Anastasia A., Felix N. Tomilin, Vladimir A. Pomogaev, Natalia G. Vnukova, Grigory N. Churilov, Nadezhda S. Kudryasheva, Olga N. Tchaikovskaya, Sergey G. Ovchinnikov, and Pavel V. Avramov. 2021. "Synthesis, Mass Spectroscopy Detection, and Density Functional Theory Investigations of the Gd Endohedral Complexes of C82 Fullerenols" Computation 9, no. 5: 58. https://0-doi-org.brum.beds.ac.uk/10.3390/computation9050058









