Clavulanic Acid Production by Streptomyces clavuligerus: Insights from Systems Biology, Strain Engineering, and Downstream Processing
Abstract
:1. Introduction
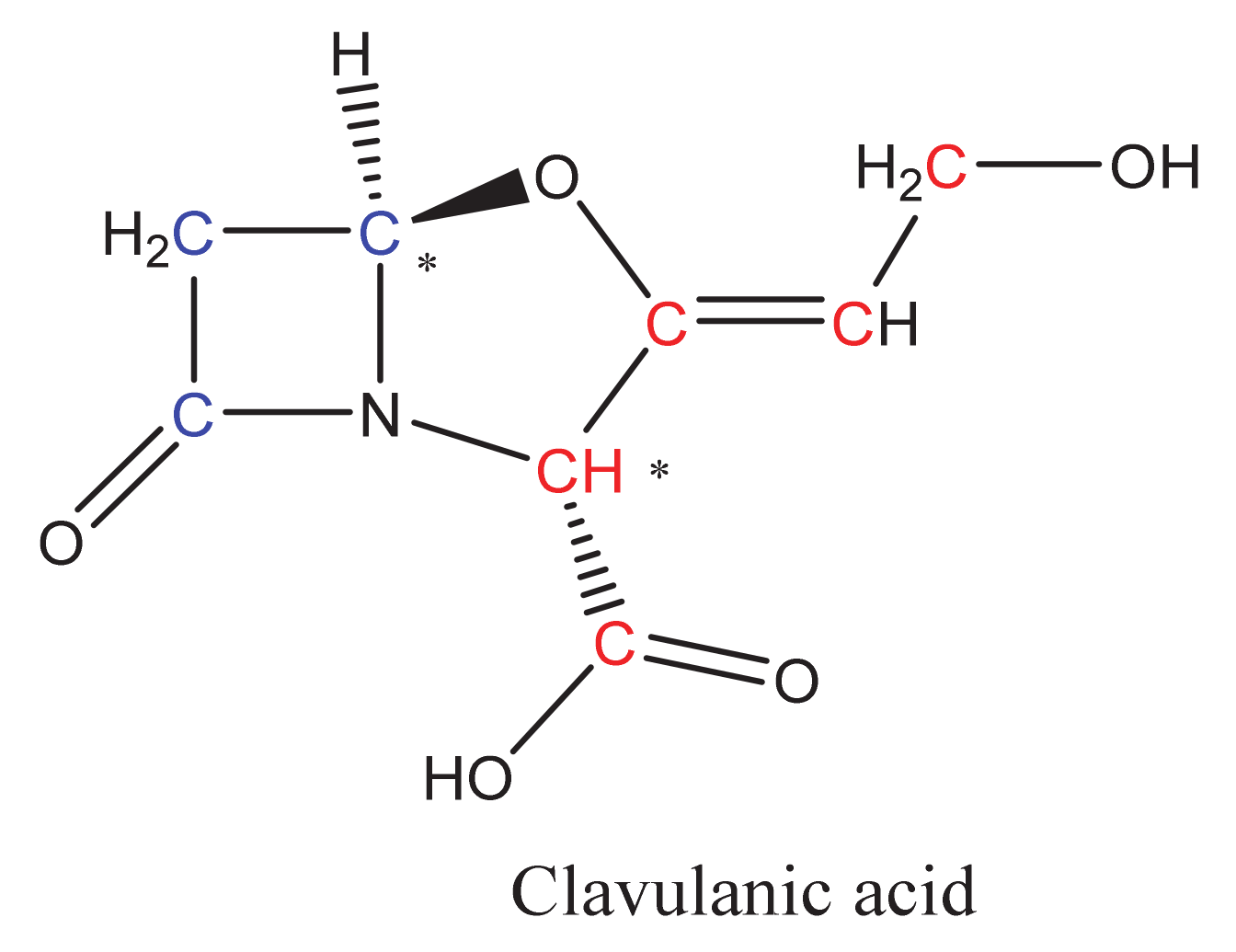
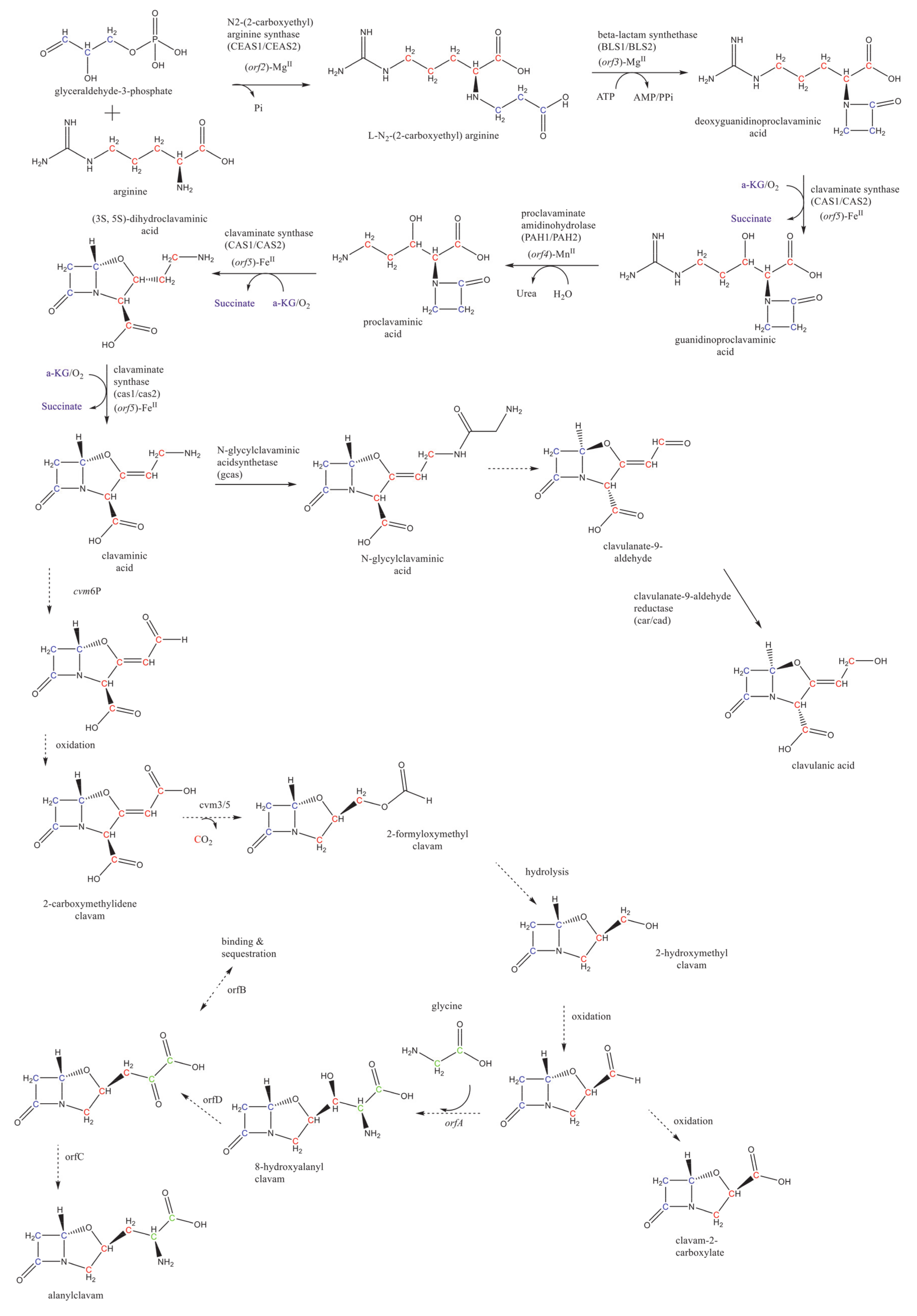
2. Overview of CA Biosynthesis in S. clavuligerus
3. CA Production in S. clavuligerus Submerged Cultivation
4. Regulation of the CA Production in S. clavuligerus
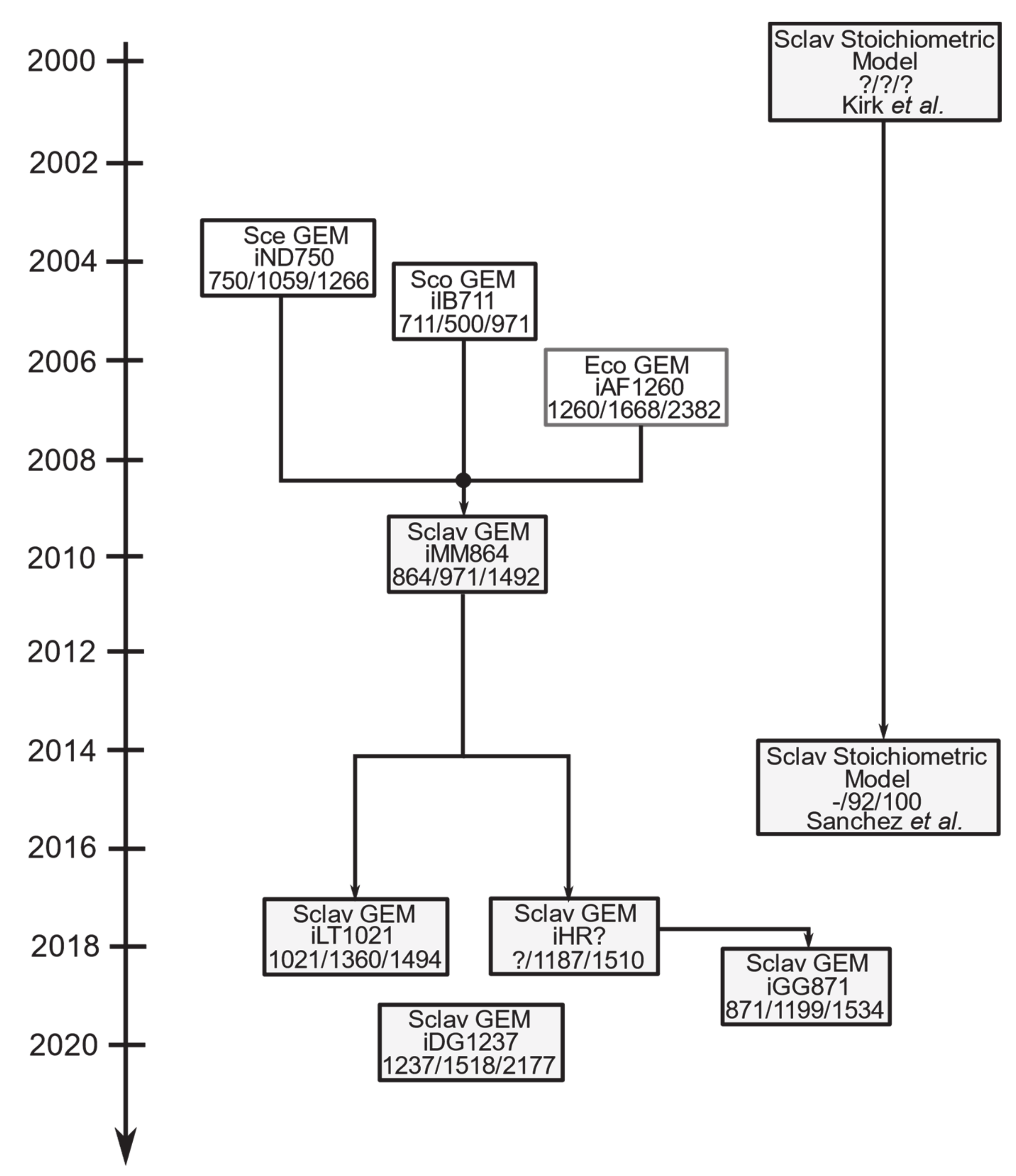
5. Advances in Systems Level Understanding of the CA Production in S. clavuligerus
6. Genome-Scale Modeling: A Suitable Platform for Improving CA Production
7. Downstream Processing of CA
8. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Gómez-Ríos, D.; Ramírez-Malule, H. Bibliometric analysis of recent research on multidrug and antibiotics resistance (2017–2018). J. Appl. Pharm. Sci. 2019, 9, 112–116. [Google Scholar] [CrossRef] [Green Version]
- Landecker, H. Antibiotic Resistance and the Biology of History. Body Soc. 2016, 22, 19–52. [Google Scholar] [CrossRef] [Green Version]
- Davies, J.; Davies, D. Origins and Evolution of Antibiotic Resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abraham, E.P.; Chain, E. An enzyme from bacteria able to destroy penicillin. Nature 1940, 146, 837. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. β-Lactams and β-Lactamase Inhibitors: An Overview. Cold Spring Harb. Perspect. Med. 2016, 6, a025247. [Google Scholar] [CrossRef] [PubMed]
- Saudagar, P.S.; Survase, S.A.; Singhal, R.S. Clavulanic acid: A review. Biotechnol. Adv. 2008, 26, 335–351. [Google Scholar] [CrossRef]
- Reading, C.; Cole, M. Clavulanic acid: A beta-lactamase-inhiting beta-lactam from Streptomyces clavuligerus. Antimicrob. Agents Chemother. 1977, 11, 852–857. [Google Scholar] [CrossRef] [Green Version]
- Huttner, A.; Bielicki, J.; Clements, M.N.; Frimodt-Møller, N.; Muller, A.E.; Paccaud, J.-P.; Mouton, J.W. Oral amoxicillin and amoxicillin–clavulanic acid: Properties, indications and usage. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Mutlu, A. Increasing Clavulanic Acid Production both in Wild Type and Industrial Streptomyces Clavuligerus Strains by Amplification of Positive Regulator claR Gene; Middle East Technical University: Çankaya/Ankara, Turkey, 2012. [Google Scholar]
- de Viana Marques, D.A.; Feitosa Machado, S.E.; Santos Ebinuma, V.C.; de Lima Duarte, C.A.; Converti, A.; Porto, A.L.F. Production of β -Lactamase Inhibitors by Streptomyces Species. Antibiotics 2018, 7, 61. [Google Scholar] [CrossRef] [Green Version]
- Nagarajan, R.; Boeck, L.D.; Gorman, M.; Hamill, R.L.; Higgens, C.E.; Hoehn, M.M.; Stark, W.M.; Whitney, J.G. β-Lactam antibiotics from Streptomyces. J. Am. Chem. Soc. 1971, 93, 2308–2310. [Google Scholar] [CrossRef]
- Higgens, C.E.; Kastner, R.E. Streptomyces Clavuligerus sp.nov. a B-Lactam Antibiotic Producer. Int. J. Syst. Bacteriol. 1971, 21, 326–331. [Google Scholar] [CrossRef] [Green Version]
- Howarth, T.T.; Brown, A.G.; King, T.J. Clavulanic acid, a novel β-lactam isolated from Streptomyces clavuligerus; X-ray crystal structure analysis. J. Chem. Soc. Chem. Commun. 1976, 266b–267b. [Google Scholar] [CrossRef]
- Clardy, J.; Fischbach, M.A.; Currie, C.R. The natural history of antibiotics. Curr. Biol. 2009, 19, R437–R441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watve, M.G.; Tickoo, R.; Jog, M.M.; Bhole, B.D. How many antibiotics are produced by the genus Streptomyces? Arch. Microbiol. 2001, 176, 386–390. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhang, J.; Zhuo, J.; Li, Y.; Tian, Y.; Tan, H. Activation and mechanism of a cryptic oviedomycin gene cluster via the disruption of a global regulatory gene, adpA, in Streptomyces ansochromogenes. J. Biol. Chem. 2017, 292, 19708–19720. [Google Scholar] [CrossRef] [Green Version]
- Peng, Q.; Gao, G.; Lü, J.; Long, Q.; Chen, X.; Zhang, F.; Xu, M.; Liu, K.; Wang, Y.; Deng, Z.; et al. Engineered streptomyces lividans strains for optimal identification and expression of cryptic biosynthetic gene clusters. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, H.T.; Pham, V.T.T.; Nguyen, C.T.; Pokhrel, A.R.; Kim, T.S.; Kim, D.H.; Na, K.; Yamaguchi, T.; Sohng, J.K. Exploration of cryptic organic photosensitive compound as Zincphyrin IV in Streptomyces venezuelae ATCC 15439. Appl. Microbiol. Biotechnol. 2020, 104, 713–724. [Google Scholar] [CrossRef]
- Gómez-Ríos, D.; López-Agudelo, V.A.; Ramírez-Malule, H.; Neubauer, P.; Junne, S.; Ochoa, S.; Ríos-Estepa, R. A Genome-Scale Insight into the Effect of Shear Stress During the Fed-Batch Production of Clavulanic Acid by Streptomyces Clavuligerus. Microorganisms 2020, 8, 1255. [Google Scholar] [CrossRef]
- Esnault, C.; Dulermo, T.; Smirnov, A.; Askora, A.; David, M.; Deniset-Besseau, A.; Holland, I.B.; Virolle, M.J. Strong antibiotic production is correlated with highly active oxidative metabolism in Streptomyces coelicolor M145. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Millan-Oropeza, A.; Henry, C.; Lejeune, C.; David, M.; Virolle, M.-J. Expression of genes of the Pho regulon is altered in Streptomyces coelicolor. Sci. Rep. 2020, 10, 8492. [Google Scholar] [CrossRef]
- Cho, H.; Uehara, T.; Bernhardt, T.G. Beta-Lactam Antibiotics Induce a Lethal Malfunctioning of the Bacterial Cell Wall Synthesis Machinery. Cell 2014, 159, 1300–1311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Virolle, M.-J. A Challenging View: Antibiotics Play a Role in the Regulation of the Energetic Metabolism of the Producing Bacteria. Antibiotics 2020, 9, 83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paradkar, A. Clavulanic acid production by Streptomyces clavuligerus: Biogenesis, regulation and strain improvement. J. Antibiot. 2013, 66, 411–420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jensen, S.E. Biosynthesis of clavam metabolites. J. Ind. Microbiol. Biotechnol. 2012, 39, 1407–1419. [Google Scholar] [CrossRef]
- Hamed, R.B.; Gomez-Castellanos, J.R.; Henry, L.; Ducho, C.; McDonough, M.A.; Schofield, C.J. The enzymes of β-lactam biosynthesis. Nat. Prod. Rep. 2013, 30, 21–107. [Google Scholar] [CrossRef]
- Bachmann, B.; Li, R.; Townsend, C. beta-Lactam synthetase: A new biosynthetic enzyme. Proc. Natl. Acad. Sci. USA 1998, 95, 9082–9086. [Google Scholar] [CrossRef] [Green Version]
- Bachmann, B.O.; Townsend, C.A. Kinetic mechanism of the β-lactam synthetase of streptomyces clavuligerus. Biochemistry 2000, 39, 11187–11193. [Google Scholar] [CrossRef]
- Tahlan, K.; Park, H.U.; Wong, A.; Beatty, P.H.; Jensen, S.E. Two Sets of Paralogous Genes Encode the Enzymes Involved in the Early Stages of Clavulanic Acid and Clavam Metabolite Biosynthesis in Streptomyces clavuligerus. Antimicrob. Agents Chemother. 2004, 48, 930–939. [Google Scholar] [CrossRef] [Green Version]
- Salowe, S.P.; Marsh, E.N.; Townsend, C.A. Purification and characterization of clavaminate synthase from Streptomyces clavuligerus: An unusual oxidative enzyme in natural product biosynthesis. Biochemistry 1990, 29, 6499–6508. [Google Scholar] [CrossRef]
- Busby, R.W.; Chang, M.D.-T.; Busby, R.C.; Wimp, J.; Townsend, C.A. Expression and Purification of Two Isozymes of Clavaminate Synthase and Initial Characterization of the Iron Binding Site. J. Biol. Chem. 1995, 270, 4262–4269. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Ren, J.S.; Harlos, K.; McKinnon, C.H.; Clifton, I.J.; Schofield, C.J. Crystal structure of a clavaminate synthase-Fe(II)-2-oxoglutarate-substrate-NO complex: Evidence for metal centered rearrangements. FEBS Lett. 2002, 517, 7–12. [Google Scholar] [CrossRef]
- Caines, M.E.C.; Elkins, J.M.; Hewitson, K.S.; Schofield, C.J. Crystal Structure and Mechanistic Implications of N 2-(2-Carboxyethyl)arginine Synthase, the First Enzyme in the Clavulanic Acid Biosynthesis Pathway. J. Biol. Chem. 2004, 279, 5685–5692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, T.K.; Busby, R.W.; Houston, T.A.; Mcilwaine, D.B.; Egan, L.A.; Townsend, C.A.; Wu, T.; Busby, R.W.; Houston, T.A.; Ilwaine, D.B.M.C.; et al. Identification, Cloning, Sequencing, and overexpression of the gene encoding proclavaminate amidino hydrolase and characterization of protein function in clavulanic acid biosynthesis. J. Bacteriol. 1995, 177, 3714–3720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shrestha, B.; Dhakal, D.; Darsandhari, S.; Pandey, R.P.; Pokhrel, A.R.; Jnawali, H.N.; Sohng, J.K. Heterologous production of clavulanic acid intermediates in Streptomyces venezuelae. Biotechnol. Bioprocess. Eng. 2017, 22, 359–365. [Google Scholar] [CrossRef]
- Ramirez-Malule, H.; Restrepo, A.; Cardona, W.; Junne, S.; Neubauer, P.; Rios-Estepa, R. Inversion of the stereochemical configuration (3S, 5S)-clavaminic acid into (3R, 5R)-clavulanic acid: A computationally-assisted approach based on experimental evidence. J. Theor. Biol. 2016, 395, 40–50. [Google Scholar] [CrossRef]
- Arulanantham, H.; Kershaw, N.J.; Hewitson, K.S.; Hughes, C.E.; Thirkettle, J.E.; Schofield, C.J. ORF17 from the clavulanic acid biosynthesis gene cluster catalyzes the ATP-dependent formation of N-glycyl-clavaminic acid. J. Biol. Chem. 2006, 281, 279–287. [Google Scholar] [CrossRef] [Green Version]
- MacKenzie, A.K.; Kershaw, N.J.; Hernandez, H.; Robinson, C.V.; Schofield, C.J.; Andersson, I. Clavulanic Acid Dehydrogenase: Structural and Biochemical Analysis of the Final Step in the Biosynthesis of the β-Lactamase Inhibitor Clavulanic Acid. Biochemistry 2007, 46, 1523–1533. [Google Scholar] [CrossRef]
- Gómez, S.; Ramírez-Malule, H.; Cardona-G, W.; Osorio, E.; Restrepo, A. Double-Ring Epimerization in the Biosynthesis of Clavulanic Acid. J. Phys. Chem. A 2020, 124, 9413–9426. [Google Scholar] [CrossRef]
- Zelyas, N.J.; Cai, H.; Kwong, T.; Jensen, S.E. Alanylclavam biosynthetic genes are clustered together with one group of clavulanic acid biosynthetic genes in Streptomyces clavuligerus. J. Bacteriol. 2008, 190, 7957–7965. [Google Scholar] [CrossRef] [Green Version]
- Nobary, S.G.; Jensen, S.E. A comparison of the clavam biosynthetic gene clusters in Streptomyces antibioticus Tü1718 and Streptomyces clavuligerus. Can. J. Microbiol. 2012, 58, 413–425. [Google Scholar] [CrossRef]
- Bushell, M.E.; Kirk, S.; Zhao, H.-J.; Avignone-Rossa, C.A. Manipulation of the physiology of clavulanic acid biosynthesis with the aid of metabolic flux analysis. Enzyme Microb. Technol. 2006, 39, 149–157. [Google Scholar] [CrossRef]
- Ramirez-Malule, H.; Junne, S.; Nicolás Cruz-Bournazou, M.; Neubauer, P.; Ríos-Estepa, R. Streptomyces clavuligerus shows a strong association between TCA cycle intermediate accumulation and clavulanic acid biosynthesis. Appl. Microbiol. Biotechnol. 2018, 102, 4009–4023. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, C.; Quintero, J.C.; Ochoa, S. Flux balance analysis in the production of clavulanic acid by Streptomyces clavuligerus. Biotechnol. Prog. 2015, 31, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Cavallieri, A.P.; Baptista, A.S.; Leite, C.A.; da Costa Araujo, M.L.G. A case study in flux balance analysis: Lysine, a cephamycin C precursor, can also increase clavulanic acid production. Biochem. Eng. J. 2016, 112, 42–53. [Google Scholar] [CrossRef]
- Kenig, M.; Reading, C. Holomycin and an antibiotic (mm 19290) related to tunicamycin, metabolites of streptomyces clavuligerus. J. Antibiot. 1979, 32, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Antonio, T.; Bellão, C.; Corrêa, T.; Cavallieri, A.P.; Badino, A.C.; da Costa Araujo, M.L.G. Evaluation of different media for the production of cephalosporins by streptomyces clavuligerus ATCC 27064. Braz. Arch. Biol. Technol. 2012, 55, 819–825. [Google Scholar] [CrossRef] [Green Version]
- Goo, K.S.; Chua, C.S.; Sim, T.S. Directed evolution and rational approaches to improving Streptomyces clavuligerus deacetoxycephalosporin C synthase for cephalosporin production. J. Ind. Microbiol. Biotechnol. 2009, 36, 619–633. [Google Scholar] [CrossRef]
- Öster, L.M.; Lester, D.R.; van Scheltinga, A.T.; Svenda, M.; van Lun, M.; Généreux, C.; Andersson, I. Insights into Cephamycin Biosynthesis: The Crystal Structure of CmcI from Streptomyces clavuligerus. J. Mol. Biol. 2006, 358, 546–558. [Google Scholar] [CrossRef]
- Liras, P. Holomycin, a dithiolopyrrolone compound produced by Streptomyces clavuligerus. Appl. Microbiol. Biotechnol. 2014, 98, 1023–1030. [Google Scholar] [CrossRef]
- Sim, J.; Sim, T.S. In vitro conversion of penicillin G and ampicillin by recombinant Streptomyces clavuligerus NRRL 3585 deacetoxycephalosporin C synthase. Enzyme Microb. Technol. 2001, 29, 240–245. [Google Scholar] [CrossRef]
- Law, K.H.; Tsang, M.W.; Wong, Y.K.; Tsang, M.S.; Lau, P.Y.; Wong, K.Y.; Ho, K.P.; Leung, Y.C. Efficient production of secretory streptomyces clavuligerus β-lactamase inhibitory protein (Blip) in pichia pastoris. AMB Express 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paradkar, A.S.; Petrich, A.K.; Leskiw, B.K.; Aidoo, K.A.; Jensen, S.E. Transcriptional analysis and heterologous expression of the gene encoding β-lactamase inhibitor protein (BLIP) from Streptomyces clavuligerus. Gene 1994, 144, 31–36. [Google Scholar] [CrossRef]
- Doran, J.L.; Leskiw, B.K.; Aippersbach, S.; Jensen, S.E. Isolation and Characterization of a β-Lactamase-Inhibitory Protein from Streptomyces Clavuligerus and Cloning and Analysis of the Corresponding Gene. J. Bacteriol. 1990, 172, 4909–4918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ser, H.-L.; Law, J.W.-F.; Chaiyakunapruk, N.; Jacob, S.A.; Palanisamy, U.D.; Chan, K.-G.; Goh, B.-H.; Lee, L.-H. Fermentation Conditions that Affect Clavulanic Acid Production in Streptomyces clavuligerus: A Systematic Review. Front. Microbiol. 2016, 7, 522. [Google Scholar] [CrossRef] [PubMed]
- Sanches Henao, C.P.; Gomez Grimaldos, N.A.; Quintero Diaz, J.C. Producción de acido clavulánico por fermentación de streptomyces clavuligerus: Evaluación de diferentes medios de cultivo y modelado matemático. Dyna 2012, 79, 158–165. [Google Scholar]
- Bellão, C.; Antonio, T.; Araujo, M.L.G.C.; Badino, A.C. Production of clavulanic acid and cephamycin c by streptomyces clavuligerus under different fed-batch conditions. Braz. J. Chem. Eng. 2013, 30, 257–266. [Google Scholar] [CrossRef] [Green Version]
- Saudagar, P.S.; Singhal, R.S. Optimization of nutritional requirements and feeding strategies for clavulanic acid production by Streptomyces clavuligerus. Bioresour. Technol. 2007, 98, 2010–2017. [Google Scholar] [CrossRef]
- Bussari, B.; Saudagar, P.S.; Shaligram, N.S.; Survase, S.A.; Singhal, R.S. Production of cephamycin C by Streptomyces clavuligerus NT4 using solid-state fermentation. J. Ind. Microbiol. Biotechnol. 2008, 35, 49–58. [Google Scholar] [CrossRef]
- da Silva Rodrigues, K.C.; de Souza, A.T.; Badino, A.C.; Pedrolli, D.B.; Cerri, M.O. Biotechnology and Industrial Microbiology Screening of medium constituents for clavulanic acid production by Streptomyces clavuligerus. Braz. J. Microbiol. 2018, 49, 832–839. [Google Scholar] [CrossRef]
- Neto, A.B.; Hirata, D.B.; Cassiano Filho, L.C.M.; Bellão, C.; Badino, A.C., Jr.; Hokka, C.O. A study on clavulanic acid production by Streptomyces clavuligerus in batch, FED-batch and continuous processes. Braz. J. Chem. Eng. 2005, 22, 557–563. [Google Scholar] [CrossRef]
- Teodoro, J.C.; Araujo, M.L.G.C.; Hokka, C.O.; Badino, A.C. Influence of glycerol and ornithine feeding on clavulanic acid production by Streptomyces clavuligerus. Braz. J. Chem. Eng. 2010, 27, 499–506. [Google Scholar] [CrossRef] [Green Version]
- Domingues, L.C.G.; Teodoro, J.C.; Hokka, C.O.; Badino, A.C.; Araujo, M.L.G.C. Optimisation of the glycerol-to-ornithine molar ratio in the feed medium for the continuous production of clavulanic acid by Streptomyces clavuligerus. Biochem. Eng. J. 2010, 53, 7–11. [Google Scholar] [CrossRef]
- Salem-Bekhit, M.M.; Alanazi, F.K.; Alsarra, I.A. Improvement and enhancement of clavulanic acid production in Streptomyces clavuligerus using vegetable oils. Afr. J. Biotechnol. 2010, 9, 6806–6812. [Google Scholar]
- Young, T.; Li, Y.; Efthimiou, G. Olive Pomace Oil can be Used as an Alternative Carbon Source for Clavulanic Acid Production by Streptomyces clavuligerus. Waste Biomass Valoriz. 2019. [Google Scholar] [CrossRef]
- Kim, S.-J.; Kim, J.-O.; Shin, C.-H.; Park, H.W.; Kim, C.-W. An approach to strain improvement and enhanced production of clavulanic acid in Streptomyces clavuligerus. Biosci. Biotechnol. Biochem. 2009, 73, 160–164. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, V.; Brandão, J.F.; Brandão, R.; Rangel-yagui, C.O.; Couto, J.A.; Converti, A.; Pessoa, A. Stability of clavulanic acid under variable pH, ionic strength and temperature conditions. A new kinetic approach. Biochem. Eng. J. 2009, 45, 89–93. [Google Scholar] [CrossRef] [Green Version]
- Brethauer, S.; Held, M.; Panke, S. Clavulanic Acid Decomposition Is Catalyzed by the Compound Itself and by Its Decomposition Products. J. Pharm. Sci. 2008, 97, 3451–3455. [Google Scholar] [CrossRef]
- Marques, D.A.V.; Oliveira, R.P.S.; Perego, P.; Porto, A.L.F.; Pessoa, A.; Converti, A. Kinetic and thermodynamic investigation on clavulanic acid formation and degradation during glycerol fermentation by Streptomyces DAUFPE 3060. Enzyme Microb. Technol. 2009, 45, 169–173. [Google Scholar] [CrossRef]
- Rodrigues, K.C.S.; Costa, C.L.L.; Badino, A.C.; Pedrolli, D.B.; Pereira, J.F.B.; Cerri, M.O. Application of Acid and Cold Stresses to Enhance the Production of Clavulanic Acid by Streptomyces clavuligerus. Appl. Biochem. Biotechnol. 2019, 188, 706–719. [Google Scholar] [CrossRef]
- Costa, C.L.L.; Badino, A.C. Production of clavulanic acid by Streptomyces clavuligerus in batch cultures without and with glycerol pulses under different temperature conditions. Biochem. Eng. J. 2012, 69, 1–7. [Google Scholar] [CrossRef]
- Yepes-García, J.; Caicedo-Montoya, C.; Pinilla, L.; Toro, L.F.; Ríos-Estepa, R. Morphological differentiation of streptomyces clavuligerus exposed to diverse environmental conditions and its relationship with clavulanic acid biosynthesis. Processes 2020, 8, 1038. [Google Scholar] [CrossRef]
- Yagüe, P.; López-García, M.T.; Rioseras, B.; Sánchez, J.; Manteca, Á. Pre-sporulation stages of Streptomyces differentiation: State-of-the-art and future perspectives. FEMS Microbiol. Lett. 2013, 342. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yagüe, P.; Lopez-Garcia, M.T.; Rioseras, B.; Sanchez, J.; Manteca, A. New insights on the development of Streptomyces and their relationships with secondary metabolite production. Curr. Trends Microbiol. 2012, 8, 65–73. [Google Scholar] [PubMed]
- Olmos, E.; Mehmood, N.; Haj Husein, L.; Goergen, J.L.; Fick, M.; Delaunay, S. Effects of bioreactor hydrodynamics on the physiology of Streptomyces. Bioprocess. Biosyst. Eng. 2013, 36, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Alok, S. Effect of Different Impellers and Baffles on Aerobic Stirred Tank Fermenter using Computational Fluid Dynamics. J. Bioprocess. Biotech. 2014, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Hristov, H.V.; Mann, R.; Lossev, V.; Vlaev, S.D. A simplified CFD for three-dimensional analysis of fluid mixing, mass transfer and bioreaction in a fermenter equipped with triple novel geometry impellers. Food Bioprod. Process. 2004, 82, 21–34. [Google Scholar] [CrossRef]
- Jin, B.; van Leeuwen, J.H.; Doelle, H.W.; Yu, Q.M. The influence of geometry on hydrodynamic and mass transfer characteristics in an external airlift reactor for the cultivation of filamentous fungi. World J. Microbiol. Biotechnol. 1999, 15, 83–90. [Google Scholar] [CrossRef]
- Gómez-Ríos, D.; Junne, S.; Neubauer, P.; Ochoa, S.; Ríos-Estepa, R.; Ramírez-Malule, H. Characterization of the Metabolic Response of Streptomyces clavuligerus to Shear Stress in Stirred Tanks and Single-Use 2D Rocking Motion Bioreactors for Clavulanic Acid Production. Antibiotics 2019, 8, 168. [Google Scholar] [CrossRef] [Green Version]
- Xia, X.; Lin, S.; Xia, X.X.; Cong, F.S.; Zhong, J.J. Significance of agitation-induced shear stress on mycelium morphology and lavendamycin production by engineered Streptomyces flocculus. Appl. Microbiol. Biotechnol. 2014, 98, 4399–4407. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Hwang, S.-C.J.; Gong, J.T.; Wu, J.-Y.; Chen, K.-C. Using Redox Potential to Detect Microbial Activities During Clavulanic Acid Biosynthesis in Streptomyces clavuligerus. Biotechnol. Lett. 2005, 27, 1791–1795. [Google Scholar] [CrossRef]
- Rosa, J.C.; Baptista Neto, A.; Hokka, C.O.; Badino, A.C. Influence of dissolved oxygen and shear conditions on clavulanic acid production by Streptomyces clavuligerus. Bioprocess. Biosyst. Eng. 2005, 27, 99–104. [Google Scholar] [CrossRef]
- Bentley, P.H.; Brooks, G.; Gilpin, M.L.; Hunt, E. A new total synthesis of (±)-clavulanic acid. Tetrahedron Lett. 1979, 20, 1889–1890. [Google Scholar] [CrossRef]
- Bentley, P.H.; Berry, P.D.; Brooks, G.; Gilpin, M.L.; Hunt, E.; Zomaya, I.I. Total synthesis of (±)-clavulanic acid. J. Chem. Soc. Chem. Commun. 1977, 748–749. [Google Scholar] [CrossRef]
- Liras, P.; Gomez-Escribano, J.P.; Santamarta, I. Regulatory mechanisms controlling antibiotic production in Streptomyces clavuligerus. J. Ind. Microbiol. Biotechnol. 2008, 35, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Shi, J.; Molle, V.; Sohlberg, B.; Weaver, D.; Bibb, M.J.; Karoonuthaisiri, N.; Lih, C.J.; Kao, C.M.; Buttner, M.J.; et al. Cross-regulation among disparate antibiotic biosynthetic pathways of Streptomyces coelicolor. Mol. Microbiol. 2005, 58, 1276–1287. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Mohsin, A.; Dai, Y.; Ali, M.F.; Chen, Z.; Zhuang, Y.; Chu, J.; Guo, M. Role of a Two-Component Signal Transduction System RspA1/A2 in Regulating the Biosynthesis of Salinomycin in Streptomyces albus. Appl. Biochem. Biotechnol. 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Hou, Y.; Zhang, H.; Chu, Y.; Xia, H.; Tian, Y. Regulatory genes and their roles for improvement of antibiotic biosynthesis in Streptomyces. 3 Biotech 2017, 7, 250. [Google Scholar] [CrossRef]
- Pérez-Redondo, R.; Rodríguez-García, A.; Martín, J.F.; Liras, P. The claR gene of Streptomyces clavuligerus, encoding a LysR-type regulatory protein controlling clavulanic acid biosynthesis, is linked to the clavulanate-9-aldehyde reductase (car) gene. Gene 1998, 211, 311–321. [Google Scholar] [CrossRef]
- Bignell, D.R.D.; Tahlan, K.; Colvin, K.R.; Jensen, S.E.; Leskiw, B.K. Expression of ccaR, Encoding the Positive Activator of Cephamycin C and Clavulanic Acid Production in Streptomyces clavuligerus, Is Dependent on bldG. Antimicrob. Agents Chemother. 2005, 49, 1529–1541. [Google Scholar] [CrossRef] [Green Version]
- Kurt, A.; Álvarez-Álvarez, R.; Liras, P.; Özcengiz, G. Role of the cmcH-ccaR intergenic region and ccaR overexpression in cephamycin C biosynthesis in Streptomyces clavuligerus. Appl. Microbiol. Biotechnol. 2013, 97, 5869–5880. [Google Scholar] [CrossRef]
- Pinilla, L.; Toro, L.F.; Laing, E.; Alzate, J.F.; Ríos-Estepa, R. Comparative transcriptome analysis of streptomyces clavuligerus in response to favorable and restrictive nutritional conditions. Antibiotics 2019, 8, 96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pérez-Llarena, F.J.; Liras, P.; Rodríguez-García, A.; Martín, J.F. A regulatory gene (ccaR) required for cephamycin and clavulanic acid production in Streptomyces clavuligerus: Amplification results in overproduction of both beta-lactam Compounds. J. Bacteriol. 1997, 179, 2053–2059. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexander, D.C.; Jensen, S.E. Investigation of the Streptomyces clavuligerus Cephamycin C Gene Cluster and Its Regulation by the CcaR Protein. J. Bacteriol. 1998, 180, 4068–4079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurt Kizildoğan, A.; Vanli Jaccard, G.; Mutlu, A.; Sertdemir, İ.; Özcengiz, G. Genetic engineering of an industrial strain of Streptomyces clavuligerus for further enhancement of clavulanic acid production. Turk. J. Biol. 2017, 41, 342–353. [Google Scholar] [CrossRef]
- Qin, R.; Zhong, C.; Zong, G.; Fu, J.; Pang, X.; Cao, G. Improvement of clavulanic acid production in Streptomyces clavuligerus F613-1 by using a claR-neo reporter strategy. Electron. J. Biotechnol. 2017, 28, 41–46. [Google Scholar] [CrossRef]
- Cho, H.S.; Jo, J.C.; Shin, C.-H.; Lee, N.; Choi, J.-S.; Cho, B.-K.; Roe, J.-H.; Kim, C.-W.; Kwon, H.J.; Yoon, Y.J. Improved production of clavulanic acid by reverse engineering and overexpression of the regulatory genes in an industrial Streptomyces clavuligerus strain. J. Ind. Microbiol. Biotechnol. 2019, 46, 1205–1215. [Google Scholar] [CrossRef]
- Santamarta, I.; López-García, M.T.; Pérez-Redondo, R.; Koekman, B.; Martín, J.F.; Liras, P. Connecting primary and secondary metabolism: AreB, an IclR-like protein, binds the AREccaR sequence of S. clavuligerus and modulates leucine biosynthesis and cephamycin C and clavulanic acid production. Mol. Microbiol. 2007, 66, 511–524. [Google Scholar] [CrossRef]
- López-García, M.T.; Santamarta, I.; Liras, P. Morphological differentiation and clavulanic acid formation are affected in a Streptomyces clavuligerus adpA-deleted mutant. Microbiology 2010, 156, 2354–2365. [Google Scholar] [CrossRef] [Green Version]
- Mascher, T.; Helmann, J.D.; Unden, G. Stimulus Perception in Bacterial Signal-Transducing Histidine Kinases. Microbiol. Mol. Biol. Rev. 2006, 70, 910–938. [Google Scholar] [CrossRef] [Green Version]
- Papon, N.; Stock, A.M. Two-component systems. Curr. Biol. 2019, 29, R724–R725. [Google Scholar] [CrossRef]
- Fu, J.; Qin, R.; Zong, G.; Liu, C.; Kang, N.; Zhong, C.; Cao, G. The CagRS Two-Component System Regulates Clavulanic Acid Metabolism via Multiple Pathways in Streptomyces clavuligerus F613-1. Front. Microbiol. 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Jnawali, H.N.; Oh, T.J.; Liou, K.; Park, B.C.; Sohng, J.K. A two-component regulatory system involved in clavulanic acid production. J. Antibiot. 2008, 61, 651–659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwong, T.; Zelyas, N.J.; Cai, H.; Tahlan, K.; Wong, A.; Jensen, S.E. 5S clavam biosynthesis is controlled by an atypical two-component regulatory system in Streptomyces clavuligerus. Antimicrob. Agents Chemother. 2012, 56, 4845–4855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos-Beneit, F. The Pho regulon: A huge regulatory network in bacteria. Front. Microbiol. 2015, 6, 402. [Google Scholar] [CrossRef] [Green Version]
- Salehghamari, E.; Hamedi, J.; Elahi, E.; Sepehrizadeh, Z.; Sadeghi, M.; Muth, G. Prediction of the pho regulon in Streptomyces clavuligerus DSM 738. New Microbiol. 2012, 35, 447–457. [Google Scholar]
- Horinouchi, S.; Hara, O.; Beppu, T. Cloning of a pleiotropic gene that positively controls biosynthesis of A-factor, actinorhodin, and prodigiosin in Streptomyces coelicolor A3(2) and Streptomyces lividans. J. Bacteriol. 1983, 155, 1238–1248. [Google Scholar] [CrossRef] [Green Version]
- Parajuli, N.; Hung, T.V.; Ishida, K.; Hang, T.T.; Hei, C.L.; Liou, K.; Jae, K.S. Identification and characterization of the afsR homologue regulatory gene from Streptomyces peucetius ATCC 27952. Res. Microbiol. 2005, 156, 707–712. [Google Scholar] [CrossRef]
- Fu, J.; Qin, R.; Zong, G.; Zhong, C.; Zhang, P.; Kang, N.; Qi, X.; Cao, G. The two-component system CepRS regulates the cephamycin C biosynthesis in Streptomyces clavuligerus F613-1. AMB Express 2019, 9. [Google Scholar] [CrossRef] [Green Version]
- Sun, D.; Liu, C.; Zhu, J.; Liu, W. Connecting metabolic pathways: Sigma factors in Streptomyces spp. Front. Microbiol. 2017, 8, 2546. [Google Scholar] [CrossRef]
- Jnawali, H.N.; Liou, K.; Sohng, J.K. Role of σ-factor (orf21) in clavulanic acid production in Streptomyces clavuligerus NRRL3585. Microbiol. Res. 2011, 166, 369–379. [Google Scholar] [CrossRef]
- Baş, L.; Otur, Ç.; Kurt-Kızıldoğan, A. Enhanced Tunicamycin Biosynthesis in BldG Overexpressed Streptomyces clavuligerus. Appl. Biochem. Microbiol. 2020, 56, 412–419. [Google Scholar] [CrossRef]
- Chatterji, D.; Kumar Ojha, A. Revisiting the stringent response, ppGpp and starvation signaling. Curr. Opin. Microbiol. 2001, 4, 160–165. [Google Scholar] [CrossRef]
- Jin, W.; Ryu, Y.G.; Kang, S.G.; Kim, S.K.; Saito, N.; Ochi, K.; Lee, S.H.; Lee, K.J. Two relAlspoT homologous genes are involved in the morphological and physiological differentiation of Streptomyces clavuligerus. Microbiology 2004, 150, 1485–1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Escribano, J.P.; Martín, J.F.; Hesketh, A.; Bibb, M.J.; Liras, P. Streptomyces clavuligerus relA-null mutants overproduce clavulanic acid and cephamycin C: Negative regulation of secondary metabolism by (p)ppGpp. Microbiology 2008, 154, 744–755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, R.; Townsend, C.A. Rational strain improvement for enhanced clavulanic acid production by genetic engineering of the glycolytic pathway in Streptomyces clavuligerus. Metab. Eng. 2006, 8, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Jnawali, H.N.; Lee, H.C.; Sohng, J.K. Enhancement of clavulanic acid production by expressing regulatory genes in gap gene deletion mutant of Streptomyces clavuligerus NRRL3585. J. Microbiol. Biotechnol. 2010, 20, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Medema, M.H.; Alam, M.T.; Heijne, W.H.M.; Van Den Berg, M.A.; Müller, U.; Trefzer, A.; Bovenberg, R.A.L.; Breitling, R.; Takano, E. Genome-wide gene expression changes in an industrial clavulanic acid overproduction strain of Streptomyces clavuligerus. Microb. Biotechnol. 2011, 4, 300–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Said, I.G.; Abdelwahed, N.A.M.; Awad, H.M.; Shallan, M.A.; El-Shahed, K.Y.; Abdel-Rahim, E.A. Enhancement of clavulanic acid production by Streptomyces sp MU-NRC77 via mutation and medium optimization. Trop. J. Pharm. Res. 2017, 16, 31–42. [Google Scholar] [CrossRef]
- Cruz-Hernández, I.L.; Vasconcelos, E.D.S.; Teodoro, J.C.; De Baptista-Neto, A.; Da Costa Araujo, M.L.G.; Badino, A.C. Exploring the Optimization of UV Mutants of Streptomyces clavuligerus for Clavulanic Acid Production. Microbiol. Res. J. Int. 2019, 26, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Song, J.Y.; Jeong, H.; Yu, D.S.; Fischbach, M.A.; Park, H.-S.; Kim, J.J.; Seo, J.-S.; Jensen, S.E.; Oh, T.K.; Lee, K.J.; et al. Draft genome sequence of Streptomyces clavuligerus NRRL 3585, a producer of diverse secondary metabolites. J. Bacteriol. 2010, 192, 6317–6318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medema, M.H.; Trefzer, A.; Kovalchuk, A.; van den Berg, M.; Müller, U.; Heijne, W.; Wu, L.; Alam, M.T.; Ronning, C.M.; Nierman, W.C.; et al. The sequence of a 1.8-mb bacterial linear plasmid reveals a rich evolutionary reservoir of secondary metabolic pathways. Genome Biol. Evol. 2010, 2, 212–224. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Zhong, C.; Zong, G.; Fu, J.; Liu, Z.; Zhang, G.; Qin, R. Complete Genome Sequence of Streptomyces clavuligerus F613-1, an Industrial Producer of Clavulanic Acid. Genome Announc. 2016, 4, 4–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, S.; Lee, N.; Jeong, Y.; Lee, Y.; Kim, W.; Cho, S.; Palsson, B.O.; Cho, B.-K. Primary transcriptome and translatome analysis determines transcriptional and translational regulatory elements encoded in the Streptomyces clavuligerus genome. Nucleic Acids Res. 2019, 47, 6114–6129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, N.; Kim, W.; Hwang, S.; Lee, Y.; Cho, S.; Palsson, B.; Cho, B.K. Thirty complete Streptomyces genome sequences for mining novel secondary metabolite biosynthetic gene clusters. Sci. Data 2020, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Hwang, S.; Lee, N.; Lee, Y.; Cho, S.; Palsson, B.; Cho, B.K. Transcriptome and translatome profiles of Streptomyces species in different growth phases. Sci. Data 2020, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Álvarez, R.; Rodríguez-García, A.; Santamarta, I.; Pérez-Redondo, R.; Prieto-Domínguez, A.; Martínez-Burgo, Y.; Liras, P. Transcriptomic analysis of S treptomyces clavuligerus Δ ccaR: Tsr: Effects of the cephamycin C-clavulanic acid cluster regulator CcaR on global regulation. Microb. Biotechnol. 2014, 7, 221–231. [Google Scholar] [CrossRef]
- Santamarta, I.; López-García, M.T.; Kurt, A.; Nárdiz, N.; Álvarez-Álvarez, R.; Pérez-Redondo, R.; Martín, J.F.; Liras, P. Characterization of DNA-binding sequences for CcaR in the cephamycin-clavulanic acid supercluster of Streptomyces clavuligerus. Mol. Microbiol. 2011, 81, 968–981. [Google Scholar] [CrossRef]
- Robles-Reglero, V.; Santamarta, I.; Álvarez-álvarez, R.; Martín, J.F.; Liras, P. Transcriptional analysis and proteomics of the holomycin gene cluster in overproducer mutants of Streptomyces clavuligerus. J. Biotechnol. 2013, 163, 69–76. [Google Scholar] [CrossRef]
- Martínez-Burgo, Y.; Álvarez-Álvarez, R.; Rodríguez-García, A.; Liras, P. The pathway-specific regulator ClaR of Streptomyces clavuligerus has a global effect on the expression of genes for secondary metabolism and differentiation. Appl. Environ. Microbiol. 2015, 81, 6637–6648. [Google Scholar] [CrossRef] [Green Version]
- Álvarez-Álvarez, R.; Rodríguez-García, A.; Martínez-Burgo, Y.; Martín, J.F.; Liras, P. Transcriptional studies on a Streptomyces clavuligerus oppA2 deletion mutant: Nacetylglycyl- clavaminic acid is an intermediate of clavulanic acid biosynthesis. Appl. Environ. Microbiol. 2018, 84. [Google Scholar] [CrossRef] [Green Version]
- Nárdiz, N.; Santamarta, I.; Lorenzana, L.M.; Martín, J.F.; Liras, P. A rhodanese-like protein is highly overrepresented in the mutant S. clavuligerus oppA2::aph: Effect on holomycin and other secondary metabolites production. Microb. Biotechnol. 2011, 4, 216–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martínez-Burgo, Y.; Santos-Aberturas, J.; Rodríguez-García, A.; Barreales, E.G.; Tormo, J.R.; Truman, A.W.; Reyes, F.; Aparicio, J.F.; Liras, P. Activation of Secondary Metabolite Gene Clusters in Streptomyces clavuligerus by the PimM Regulator of Streptomyces natalensis. Front. Microbiol. 2019, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, N.L.; Peña-Castillo, L.; Moore, M.A.; Bignell, D.R.D.; Tahlan, K. Proteomics analysis of global regulatory cascades involved in clavulanic acid production and morphological development in Streptomyces clavuligerus. J. Ind. Microbiol. Biotechnol. 2016, 43, 537–555. [Google Scholar] [CrossRef] [PubMed]
- Ünsaldı, E.; Kurt-Kızıldoğan, A.; Voigt, B.; Becher, D.; Özcengiz, G. Proteome-wide alterations in an industrial clavulanic acid producing strain of Streptomyces clavuligerus. Synth. Syst. Biotechnol. 2017, 2, 39–48. [Google Scholar] [CrossRef] [Green Version]
- Tahlan, K.; Anders, C.; Wong, A.; Mosher, R.H.; Beatty, P.H.; Brumlik, M.J.; Griffin, A.; Hughes, C.; Griffin, J.; Barton, B.; et al. 5S clavam biosynthetic genes are located in both the clavam and paralog gene clusters in Streptomyces clavuligerus. Chem. Biol. 2007, 14, 131–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Busche, T.; Tsolis, K.C.; Koepff, J.; Rebets, Y.; Rückert, C.; Hamed, M.B.; Bleidt, A.; Wiechert, W.; Lopatniuk, M.; Yousra, A.; et al. Multi-omics and targeted approaches to determine the role of cellular proteases in streptomyces protein secretion. Front. Microbiol. 2018, 9, 1174. [Google Scholar] [CrossRef] [PubMed]
- Sulheim, S.; Kumelj, T.; van Dissel, D.; Salehzadeh-Yazdi, A.; Du, C.; Van Wezel, G.P.; Nieselt, K.; Almaas, E.; Wentzel, A.; Kerkhoven, E.J. Enzyme-constrained models and omics analysis of Streptomyces coelicolor reveal metabolic changes that enhance heterologous production. Iscience 2020, 23, 101525. [Google Scholar] [CrossRef]
- Wu, Q.; Ni, M.; Liu, W.C.; Ren, J.H.; Rao, Y.H.; Chen, J.; Lu, C.G. Omics for understanding the mechanisms of Streptomyces lydicus A01 promoting the growth of tomato seedlings. Plant. Soil 2018, 431, 129–141. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, N.; Hwang, S.; Kim, K.; Kim, W.; Kim, J.; Cho, S.; Palsson, B.O.; Cho, B.K. System-level understanding of gene expression and regulation for engineering secondary metabolite production in Streptomyces. J. Ind. Microbiol. Biotechnol. 2020, 1–14. [Google Scholar] [CrossRef]
- Kim, S.H.; Herazo-Maya, J.D.; Kang, D.D.; Juan-Guardela, B.M.; Tedrow, J.; Martinez, F.J.; Sciurba, F.C.; Tseng, G.C.; Kaminski, N. Integrative phenotyping framework (iPF): Integrative clustering of multiple omics data identifies novel lung disease subphenotypes. BMC Genom. 2015, 16, 924. [Google Scholar] [CrossRef] [Green Version]
- Yan, J.; Risacher, S.L.; Shen, L.; Saykin, A.J. Network approaches to systems biology analysis of complex disease: Integrative methods for multi-omics data. Brief. Bioinform. 2017, 19, 1370–1381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krishnan, K.C.; Kurt, Z.; Barrere-Cain, R.; Sabir, S.; Das, A.; Floyd, R.; Vergnes, L.; Zhao, Y.; Che, N.; Charugundla, S. Integration of multi-omics data from mouse diversity panel highlights mitochondrial dysfunction in non-alcoholic fatty liver disease. Cell Syst. 2018, 6, 103–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinu, F.R.; Beale, D.J.; Paten, A.M.; Kouremenos, K.; Swarup, S.; Schirra, H.J.; Wishart, D. Systems biology and multi-omics integration: Viewpoints from the metabolomics research community. Metabolites 2019, 9, 76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumelj, T.; Sulheim, S.; Wentzel, A.; Almaas, E. Predicting Strain Engineering Strategies Using iKS1317: A Genome-Scale Metabolic Model of Streptomyces coelicolor. Biotechnol. J. 2019, 14, 1800180. [Google Scholar] [CrossRef] [Green Version]
- Scobie, D.; Hjorleifsson, G.; Herron, P.; Rogers, S.; Duncan, K. The Missing Link: Developing a pipeline for accelerated antibiotic discovery from Streptomyces through linking ‘omics data. Access Microbiol. 2020, 2, 753. [Google Scholar] [CrossRef]
- Jamil, I.N.; Remali, J.; Azizan, K.A.; Nor Muhammad, N.A.; Arita, M.; Goh, H.H.; Aizat, W.M. Systematic Multi-Omics Integration (MOI) Approach in Plant Systems Biology. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- Gutierrez, J.M.; Feizi, A.; Li, S.; Kallehauge, T.B.; Hefzi, H.; Grav, L.M.; Ley, D.; Baycin Hizal, D.; Betenbaugh, M.J.; Voldborg, B.; et al. Genome-scale reconstructions of the mammalian secretory pathway predict metabolic costs and limitations of protein secretion. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef] [Green Version]
- López-Agudelo, V.A.; Baena, A.; Ramirez-Malule, H.; Ochoa, S.; Barrera, L.F.; Ríos-Estepa, R. Metabolic adaptation of two in silico mutants of Mycobacterium tuberculosis during infection. BMC Syst. Biol. 2017, 11, 107. [Google Scholar] [CrossRef] [Green Version]
- Baloni, P.; Funk, C.C.; Yan, J.; Yurkovich, J.T.; Kueider-Paisley, A.; Nho, K.; Heinken, A.; Jia, W.; Mahmoudiandehkordi, S.; Louie, G.; et al. Metabolic Network Analysis Reveals Altered Bile Acid Synthesis and Cholesterol Metabolism in Alzheimer’s Disease. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Greenhalgh, K.; Ramiro-Garcia, J.; Heinken, A.; Ullmann, P.; Bintener, T.; Pacheco, M.P.; Baginska, J.; Shah, P.; Frachet, A.; Halder, R.; et al. Integrated In Vitro and In Silico Modeling Delineates the Molecular Effects of a Synbiotic Regimen on Colorectal-Cancer-Derived Cells. Cell Rep. 2019, 27, 1621–1632.e9. [Google Scholar] [CrossRef] [Green Version]
- Peñalver Bernabé, B.; Thiele, I.; Galdones, E.; Siletz, A.; Chandrasekaran, S.; Woodruff, T.K.; Broadbelt, L.J.; Shea, L.D. Dynamic genome-scale cell-specific metabolic models reveal novel inter-cellular and intra-cellular metabolic communications during ovarian follicle development. BMC Bioinform. 2019, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Mccloskey, D.; Gangoiti, J.A.; King, Z.A.; Naviaux, R.K.; Barshop, B.A.; Palsson, B.O.; Feist, A.M. A model-driven quantitative metabolomics analysis of aerobic and anaerobic metabolism in E. coli K-12 MG1655 that is biochemically and thermodynamically consistent. Biotechnol. Bioeng. 2014, 111, 803–815. [Google Scholar] [CrossRef] [PubMed]
- Kleessen, S.; Irgang, S.; Klie, S.; Giavalisco, P.; Nikoloski, Z. Integration of transcriptomics and metabolomics data specifies the metabolic response of Chlamydomonas to rapamycin treatment. Plant J. 2015, 81, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, M.P.; Bintener, T.; Ternes, D.; Kulms, D.; Haan, S.; Letellier, E.; Sauter, T. Identifying and targeting cancer-specific metabolism with network-based drug target prediction. EBioMedicine 2019, 43, 98–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hastings, J.; Mains, A.; Virk, B.; Rodriguez, N.; Murdoch, S.; Pearce, J.; Bergmann, S.; Le Novère, N.; Casanueva, O. Multi-omics and genome-scale modeling reveal a metabolic shift during C. elegans aging. Front. Mol. Biosci. 2019, 6, 2. [Google Scholar] [CrossRef]
- Bordbar, A.; Monk, J.M.; King, Z.A.; Palsson, B.O. Constraint-based models predict metabolic and associated cellular functions. Nat. Rev. Genet. 2014, 15, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Feist, A.M.; Palsson, B.O. The biomass objective function. Curr. Opin. Microbiol. 2010, 13, 344–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thiele, I.; Palsson, B.Ø. A protocol for generating a high-quality genome-scale metabolic reconstruction. Nat. Protoc. 2010, 5, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orth, J.D.; Thiele, I.; Palsson, B.O. What is flux balance analysis? Nat. Biotechnol. 2010, 28, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Gianchandani, E.P.; Chavali, A.K.; Papin, J.A. The application of flux balance analysis in systems biology. Wiley Interdiscip. Rev. Syst. Biol. Med. 2010, 2, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Razmilic, V.; Castro, J.F.; Andrews, B.; Asenjo, J.A. Analysis of metabolic networks of Streptomyces leeuwenhoekii C34 by means of a genome scale model: Prediction of modifications that enhance the production of specialized metabolites. Biotechnol. Bioeng. 2018, 115, 1815–1828. [Google Scholar] [CrossRef] [PubMed]
- Amara, A.; Takano, E.; Breitling, R. Development and validation of an updated computational model of Streptomyces coelicolor primary and secondary metabolism. BMC Genom. 2018, 19, 519. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, J.; Liu, H.; Wang, J.; Wen, J. A genome-scale dynamic flux balance analysis model of Streptomyces tsukubaensis NRRL18488 to predict the targets for increasing FK506 production. Biochem. Eng. J. 2017, 123, 45–56. [Google Scholar] [CrossRef]
- Toro, L.; Pinilla, L.; Avignone-Rossa, C.; Ríos-Estepa, R. An enhanced genome-scale metabolic reconstruction of Streptomyces clavuligerus identifies novel strain improvement strategies. Bioprocess. Biosyst. Eng. 2018, 41, 657–669. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Cerón, S.; Galindo-Betancur, D.; Ramírez-Malule, H. Data set of in silico simulation for the production of clavulanic acid and cephamycin C by Streptomyces clavuligerus using a genome scale metabolic model. Data Br. 2019, 24, 103992. [Google Scholar] [CrossRef] [PubMed]
- Kirk, S.; Avignone-rossa, C.A.; Bushell, M.E. Growth limiting substrate affects antibiotic production and associated metabolic fluxes in Streptomyces clavuligerus. Biotechnol. Lett. 2000, 22, 1803–1809. [Google Scholar] [CrossRef]
- Gómez-Ríos, D.; Ramírez-Malule, H.; Neubauer, P.; Junne, S.; Ríos-Estepa, R. Data of clavulanic acid and clavulanate-imidazole stability at low temperatures. Data Br. 2019, 23, 103775. [Google Scholar] [CrossRef]
- Bersanetti, P.A.; Almeida, R.M.R.G.; Barboza, M.; Araújo, M.L.G.C.; Hokka, C.O. Kinetic studies on clavulanic acid degradation. Biochem. Eng. J. 2005, 23, 31–36. [Google Scholar] [CrossRef]
- Roubos, J.A.; Krabben, P.; De Laat, W.; Heijnen, J.J. Clavulanic Acid Degradation in Streptomyces clavuligerus Fed-Batch Cultivations. Biotechnol. Prog. 2002, 18, 451–457. [Google Scholar] [CrossRef]
- Ramirez-Malule, H.; Junne, S.; López, C.; Zapata, J.; Sáez, A.; Neubauer, P.; Rios-Estepa, R. An improved HPLC-DAD method for clavulanic acid quantification in fermentation broths of Streptomyces clavuligerus. J. Pharm. Biomed. Anal. 2016, 120, 241–247. [Google Scholar] [CrossRef]
- Vahdat, L.; Sunderland, V.B. Kinetics of amoxicillin and clavulanate degradation alone and in combination in aqueous solution under frozen conditions. Int. J. Pharm. 2007, 342, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Peace, N.; Olubukola, O.; Moshood, A. Stability of reconstituted amoxicillin clavulanate potassium under simulated in-home storage conditions. J. Appl. Pharm. Sci. 2012, 2, 28–31. [Google Scholar]
- Nur, A.O.; Hassan, A.A.; Gadkariem, E.A.; Osman, Z. Stability of Co-Amoxiclav Reconstituted Injectable Solution. Eur. J. Pharm. Med. Res. 2015, 2, 109–123. [Google Scholar]
- Jerzsele, Á.; Nagy, G. The stability of amoxicillin trihydrate and potassium clavulanate combination in aqueous solutions. Acta Vet. Hung. 2009, 57, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ríos, D.; Ramírez-Malule, H.; Neubauer, P.; Junne, S.; Ríos-Estepa, R. Degradation Kinetics of Clavulanic Acid in Fermentation Broths at Low Temperatures. Antibiotics 2019, 8, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, X.; Cao, G.; Zhang, X.; Chen, Y.; Wang, L.; Zhong, C. Studies on the formation and synthetic mechanism of related substance G in potassium clavulanate production. Braz. J. Pharm. Sci. 2015, 51, 77–83. [Google Scholar] [CrossRef] [Green Version]
- Forte, M.B.S.; Rodrigues, M.I.; Filho, F.M. Clavulanic Acid Adsorption Studies in Zeolites. Adsorpt. Sci. Technol. 2011, 29, 391–403. [Google Scholar] [CrossRef]
- Forte, M.B.S.; Mazutti, M.A.; Filho, F.M.; Rodrigues, M.I. Evaluation of kinetic and mass transfer parameters for adsorption of clavulanic acid into natural and synthetic zeolites. Biotechnol. Bioprocess. Eng. 2011, 16, 1223–1230. [Google Scholar] [CrossRef]
- Barboza, M.; Almeida, R.M.R.G.; Hokka, C.O. Kinetic studies of clavulanic acid recovery by ion exchange chromatography. Bioseparation 2001, 10, 221–227. [Google Scholar] [CrossRef]
- Barboza, M.; Almeida, R.M.R.G.; Hokka, C.O. Influence of temperature on the kinetics of adsorption and desorption of clavulanic acid by ionic exchange. Biochem. Eng. J. 2003, 14, 19–26. [Google Scholar] [CrossRef]
- Almeida, R.M.R.G.; Barboza, M.; Hokka, C.O. Continuous Clavulanic Acid Adsorption Process. In Biotechnology for Fuels and Chemicals; Davison, B.H., Lee, J.W., Finkelstein, M., McMillan, J.D., Eds.; Humana Press: Totowa, NJ, USA, 2003; pp. 867–879. ISBN 978-1-4612-0057-4. [Google Scholar]
- Cuel, M.; Barboza, M.; Hokka, C.; Kwong, W. Heterogeneous Model of the Process of Clavulanic Acid Purification by Ionic Exchange in a Fixed-Bed Column. Chem. Prod. Process. Model. 2011, 6. [Google Scholar] [CrossRef]
- Forte, M.B.S.; Elias, É.C.L.; Pastore, H.O.; Filho, F.M.; Rodrigues, M.I. Evaluation of Clavulanic Acid Adsorption in MgAl-Layered Double Hydroxides: Kinetic, Equilibrium and Thermodynamic Studies. Adsorpt. Sci. Technol. 2012, 30, 65–80. [Google Scholar] [CrossRef] [Green Version]
- Forte, M.B.S.; Taviot-Guého, C.; Leroux, F.; Rodrigues, M.I.; Maugeri Filho, F. Development and characterization of a new adsorbent for biomolecule separation: Intercalation and adsorption of clavulanic acid in layered double hydroxides. J. Chem. Technol. Biotechnol. 2016, 91, 1709–1719. [Google Scholar] [CrossRef]
- Forte, M.B.S.; Taviot-Guého, C.; Leroux, F.; Rodrigues, M.I.; Maugeri Filho, F. Clavulanic acid separation on fixed bed columns of layered double hydroxides: Optimization of operating parameters using breakthrough curves. Process. Biochem. 2016, 51, 509–516. [Google Scholar] [CrossRef]
- Carneiro-da-Cunha, M.N.; Souza, K.P.S.; Mota, A.M.O.; Teixeira, J.A.; Porto, C.S.; Porto, T.S.; Porto, A.L.F. Stability of clavulanic acid in PEG/citrate and liquid-liquid extraction in aqueous two-phase system. Fluid Phase Equilib. 2014, 375, 104–109. [Google Scholar] [CrossRef] [Green Version]
- Videira, M.; Aires-Barros, M.R. Liquid-liquid extraction of clavulanic acid using an aqueous two-phase system of polyethylene glycol and potassium phosphate. J. Chromatogr. A 1994, 668, 237–240. [Google Scholar] [CrossRef]
- Silva, C.S.; Bovarotti, E.; Rodrigues, M.I.; Hokka, C.O.; Barboza, M. Evaluation of the effects of the parameters involved in the purification of clavulanic acid from fermentation broth by aqueous two-phase systems. Bioprocess. Biosyst. Eng. 2009, 32, 625–632. [Google Scholar] [CrossRef]
- Pereira, J.F.B.; Santos, V.C.; Johansson, H.O.; Teixeira, J.A.C.; Pessoa, A. A stable liquid-liquid extraction system for clavulanic acid using polymer-based aqueous two-phase systems. Sep. Purif. Technol. 2012, 98, 441–450. [Google Scholar] [CrossRef] [Green Version]
- Panas, P.; Lopes, C.; Cerri, M.O.; Ventura, S.P.M.; Santos-Ebinuma, V.C.; Pereira, J.F.B. Purification of clavulanic acid produced by Streptomyces clavuligerus via submerged fermentation using polyethylene glycol/cholinium chloride aqueous two-phase systems. Fluid Phase Equilib. 2017, 450, 42–50. [Google Scholar] [CrossRef] [Green Version]
- da Silva, C.S.; Cuel, M.F.; Barreto, V.O.; Kwong, W.H.; Hokka, C.O.; Barboza, M. Separation of clavulanic acid from fermented broth of amino acids by an aqueous two-phase system and ion-exchange adsorption. New Biotechnol. 2012, 29, 428–431. [Google Scholar] [CrossRef]
- Viana Marques, D.A.; Pessoa-Júnior, A.; Lima-Filho, J.L.; Converti, A.; Perego, P.; Porto, A.L.F. Extractive fermentation of clavulanic acid by Streptomyces DAUFPE 3060 using aqueous two-phase system. Biotechnol. Prog. 2011, 27, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Marques, D.A.V.; Santos-Ebinuma, V.C.; Pessoa-Junior, A.; Porto, A.L.F.; Rivas Torres, B.; Concerti, A. Effect of Aeration and Agitation on Extractive Fermentation of Clavulanic Acid by Using Aqueous Two-phase System. Biotechnol. Prog. 2016, 32, 1444–1452. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.L.L.; Badino, A.C. Overproduction of clavulanic acid by extractive fermentation. Electron. J. Biotechnol. 2015, 18, 154–160. [Google Scholar] [CrossRef] [Green Version]
- Guedes De Andrade, E.; De Siqueira Cardoso Silva, M.; Brandão Haga, R.; Carvalho Santos, V.; Pessoa, A., Jr.; Rangel-Yagui, C.D.O. Extraction of clavulanic acid using aqueous two-phase micellar system. Biotechnol. Appl. Biochem. 2011, 58, 103–108. [Google Scholar] [CrossRef]
- Santos, V.C.; Hasmann, F.A.; Converti, A.; Pessoa, A. Liquid-liquid extraction by mixed micellar systems: A new approach for clavulanic acid recovery from fermented broth. Biochem. Eng. J. 2011, 56, 75–83. [Google Scholar] [CrossRef]
- De Siqueira Cardoso Silva, M.; de Carvalho Santos-Ebinuma, V.; Lopes, A.M.; de Oliveira Rangel-Yagui, C. Dextran sulfate/Triton X two-phase micellar systems as an alternative first purification step for clavulanic acid. Fluid Phase Equilib. 2015, 399, 80–86. [Google Scholar] [CrossRef]
- Brites, L.M.; Oliveira, J.H.; Barboza, M.; Hokka, C.O. Effect of physicochemical properties of solvents on clavulanic acid extraction from fermentation broth. Lat. Am. Appl. Res. 2012, 42, 65–70. [Google Scholar]
- de Mancilha, M.A.; Guimarães, G.C.; de Nardi, J.C.S.C.; de Oliveira, J.H.H.L.; Hirata, D.B. Optimization of Liquid-Liquid Extraction Step for Clavulanic Acid From Fermentation Broth Using Solvent Mixtures. Quim. Nova 2014, 37, 1335–1341. [Google Scholar] [CrossRef]
- Hirata, D.B.; Oliveira, J.H.H.L.; Leao, K.V.; Rodrigues, M.I.; Ferreira, A.G.; Giulietti, M.; Barboza, M.; Hokka, C.O. Optimization of the precipitation of clavulanic acid from fermented broth using T-octylamine as intermediate. Braz. J. Chem. Eng. 2013, 30, 231–244. [Google Scholar] [CrossRef]
- Hirata, D.B.; Oliveira, J.H.H.L.; Leão, K.V.; Rodrigues, M.I.; Ferreira, A.G.; Giulietti, M.; Barboza, M.; Hokka, C.O. Precipitation of clavulanic acid from fermentation broth with potassium 2-ethyl hexanoate salt. Sep. Purif. Technol. 2009, 66, 598–605. [Google Scholar] [CrossRef]

| Strain | Operation Mode | Strain Intervention | Supplement | Titers (g∙L−1) | Ref. |
|---|---|---|---|---|---|
| S. clavuligerus IDG3 | Fed-batch | Overexpression: cas2, ccaR, claR | Glycerol trioleate | 6.690 | [95] |
| S. clavuligerus M3-19 | Batch | Reporter gene neo fused downstream of claR | - | 4.330 | [96] |
| S. clavuligerus NEO | Batch | Reporter gene neo fused downstream of claR | - | 3.260 | [96] |
| Streptomyces sp. MU-NRC77 | Fed-batch/Batch | UV mutagenesis | H2O2/Activated animal charcoal | 0.649/0.683 | [119] |
| S. clavuligerus 70 | Batch | UV mutagenesis | - | 0.500 | [120] |
| S. clavuligerus OR | Batch | Random mutagenesis and cas1, ccaR and claR overexpression | - | 5.520 (OR/pCAS1) 6.010 (OR/pCCAR-CLAR) | [97] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Agudelo, V.A.; Gómez-Ríos, D.; Ramirez-Malule, H. Clavulanic Acid Production by Streptomyces clavuligerus: Insights from Systems Biology, Strain Engineering, and Downstream Processing. Antibiotics 2021, 10, 84. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics10010084
López-Agudelo VA, Gómez-Ríos D, Ramirez-Malule H. Clavulanic Acid Production by Streptomyces clavuligerus: Insights from Systems Biology, Strain Engineering, and Downstream Processing. Antibiotics. 2021; 10(1):84. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics10010084
Chicago/Turabian StyleLópez-Agudelo, Víctor A., David Gómez-Ríos, and Howard Ramirez-Malule. 2021. "Clavulanic Acid Production by Streptomyces clavuligerus: Insights from Systems Biology, Strain Engineering, and Downstream Processing" Antibiotics 10, no. 1: 84. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics10010084





