Facile In-Situ Fabrication of a Ternary ZnO/TiO2/Ag Nanocomposite for Enhanced Bactericidal and Biocompatibility Properties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Bioanalytical Characterisation of the Synthesised Nanomaterials
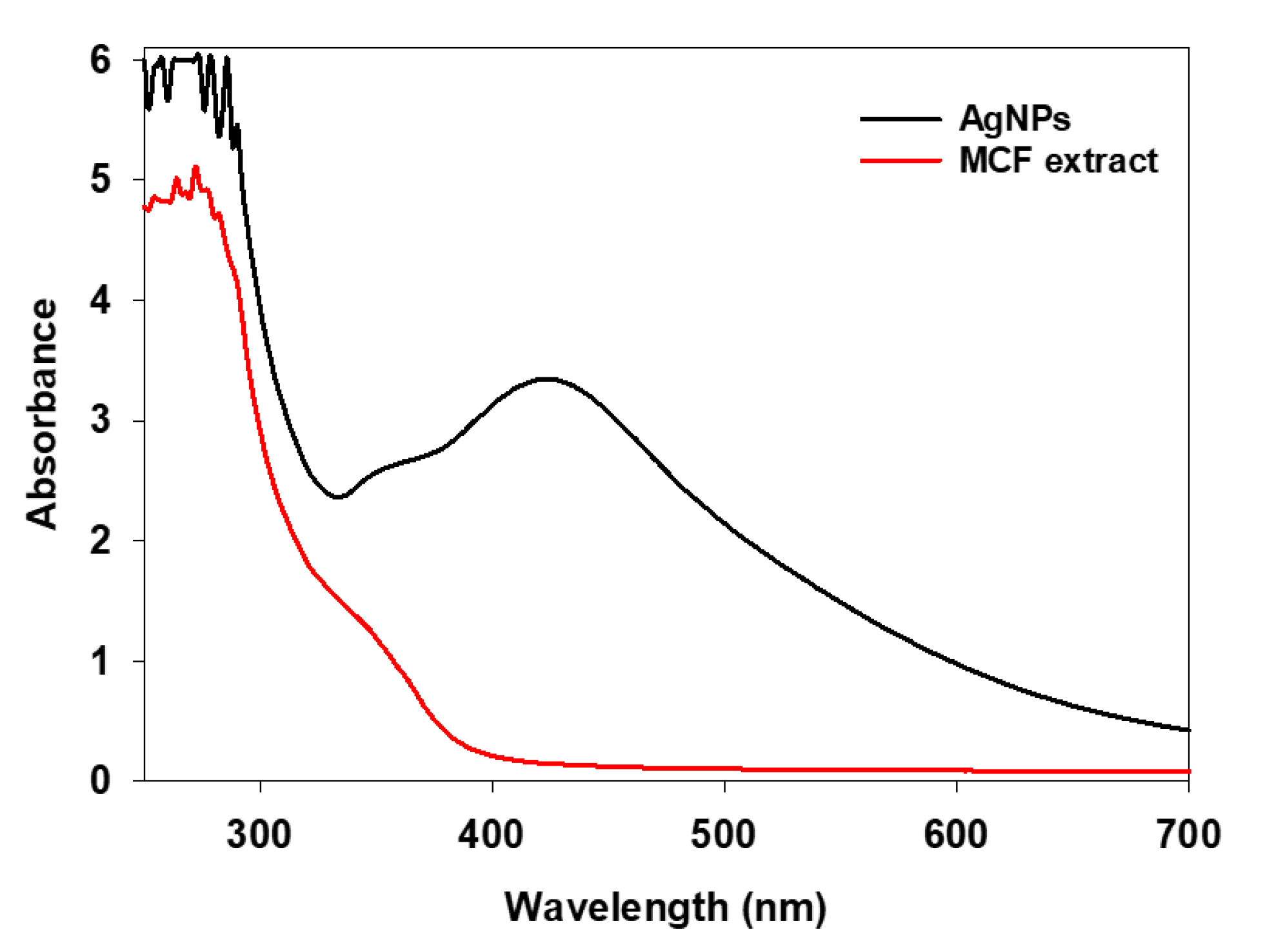
2.1.1. UV-Vis Analysis
2.1.2. Detection of the Functional Group Responsible for NPs Synthesis
2.1.3. XRD Analysis
2.1.4. FESEM, EDX and Elemental Mapping
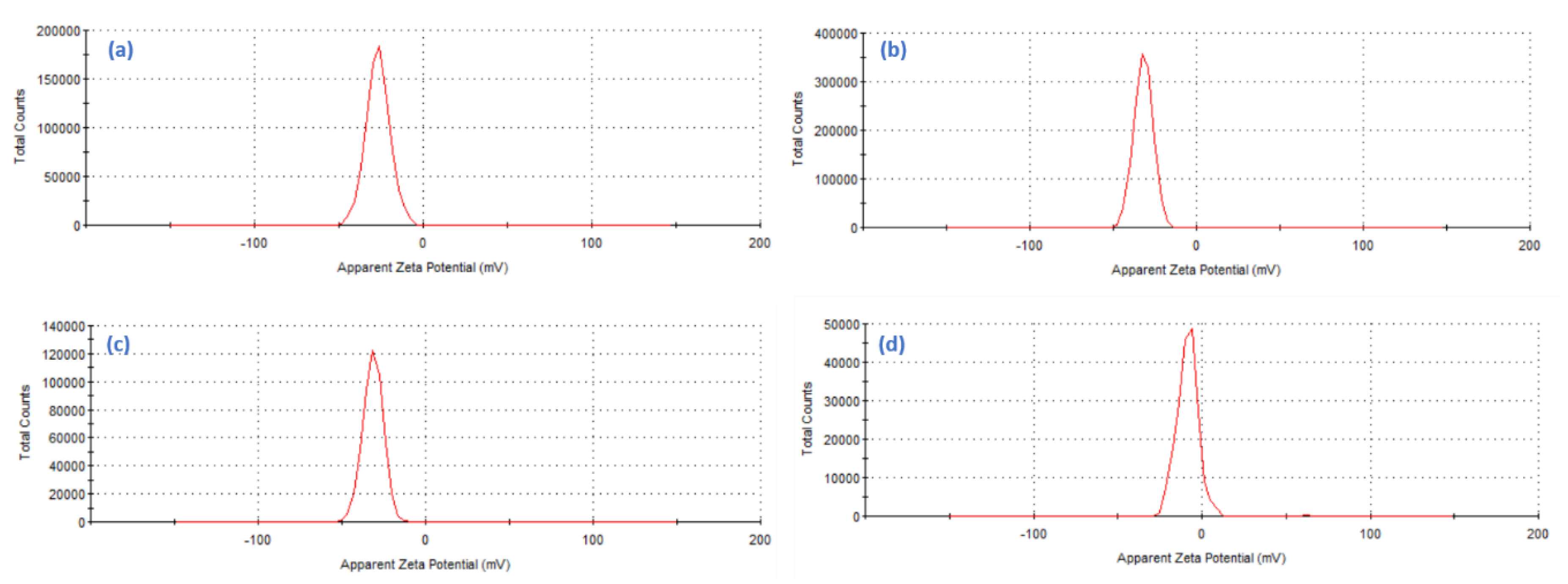
2.1.5. Zeta Potential Surface Charge Analysis
2.2. Antimicrobial Properties
2.2.1. Minimum Inhibitory Concentration (MIC)
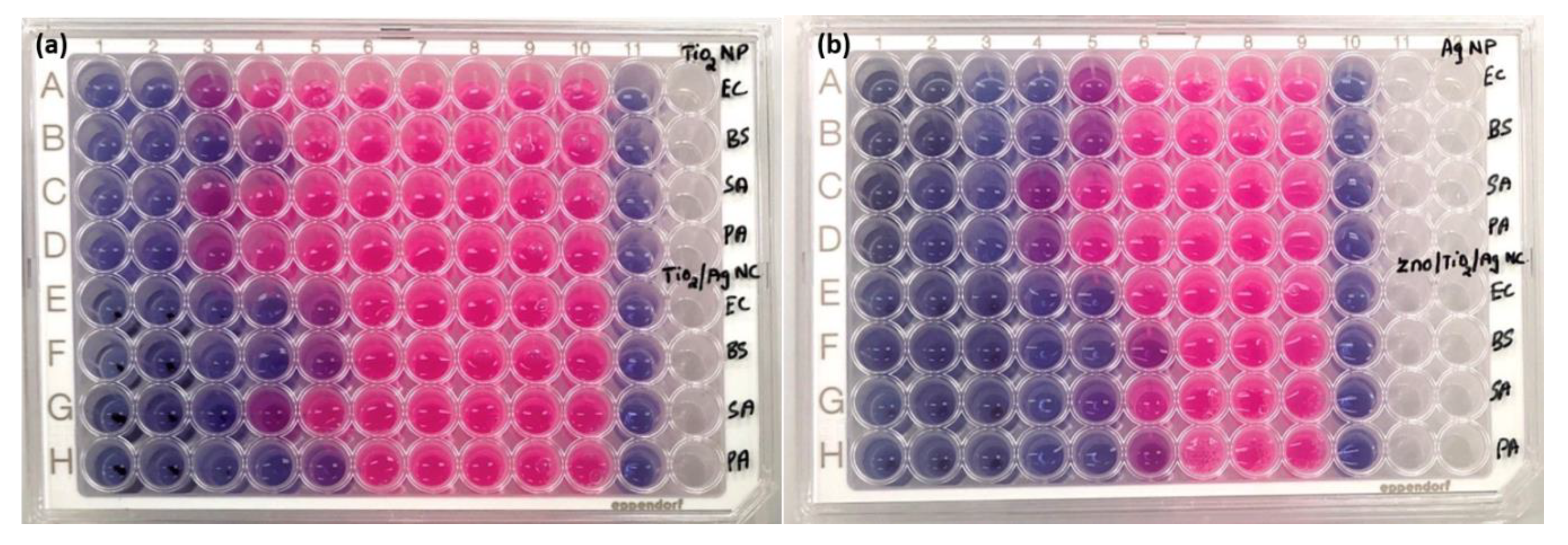
2.2.2. Bacterial Metabolic Activity
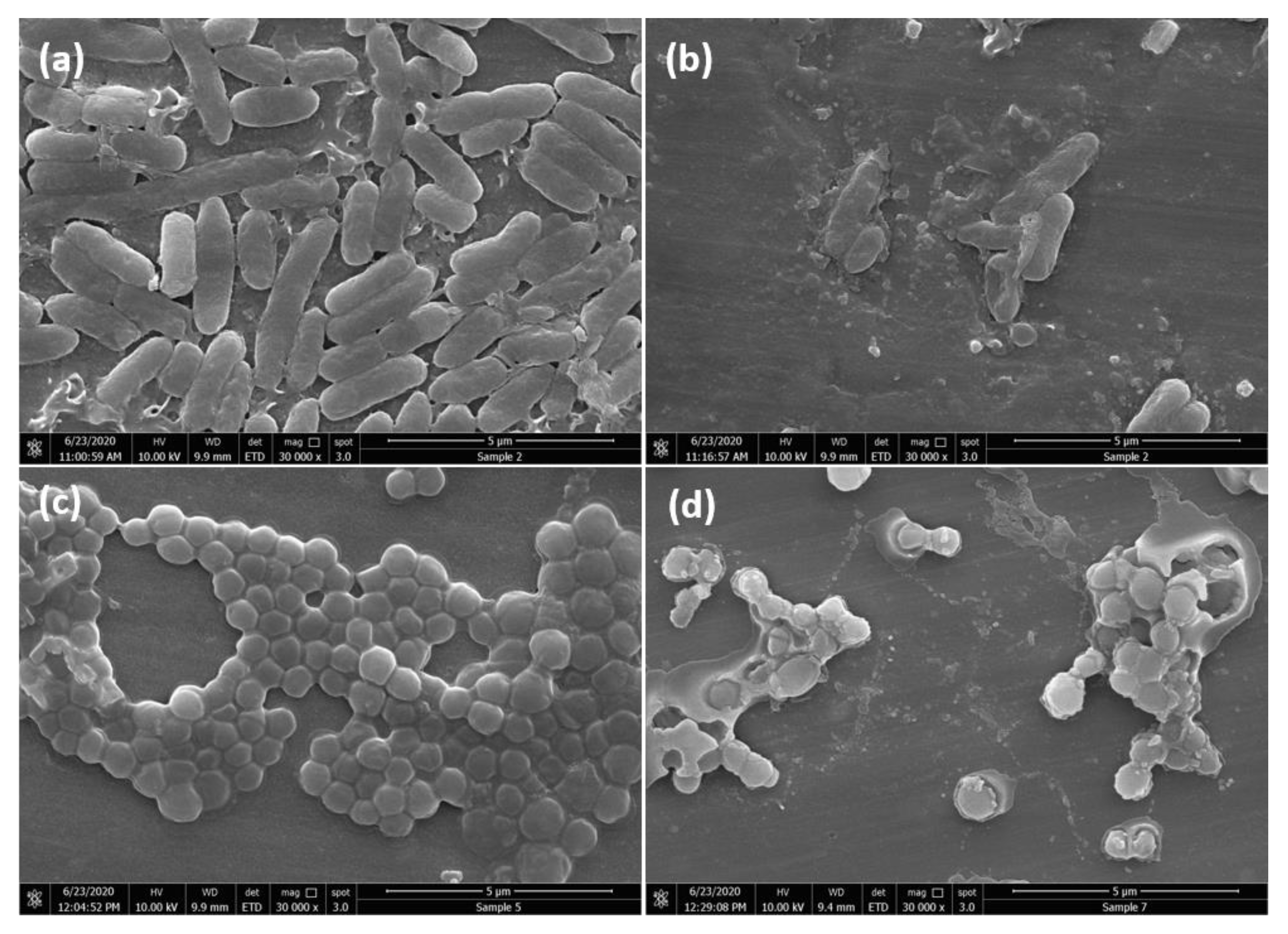
2.2.3. FESEM Analysis of Bacteria Morphological Changes
2.2.4. Confocal Visual Observation of Live/Dead Cells and ROS Generation
2.3. Cytotoxicity Analysis
2.3.1. MTT Cell Viability
2.3.2. Apoptosis
3. Conclusions
4. Materials and Methods
4.1. Chemicals and Media Components
4.2. Plant Extract Preparation and Optimisation
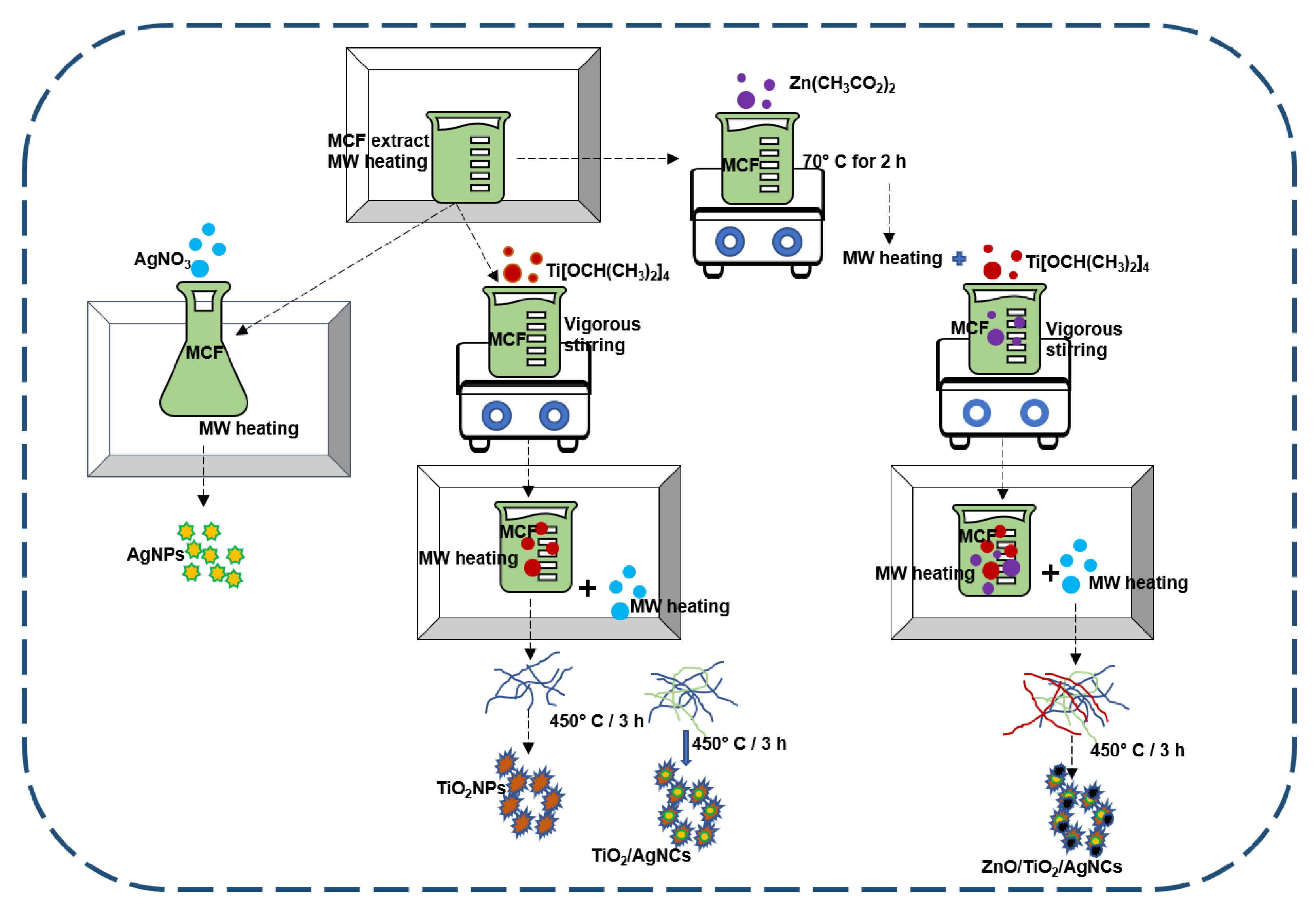
4.3. Nanoparticles Synthesis
4.4. Characterisation of Nanoparticles
4.5. Antimicrobial Activity of as-Synthesised Nanocomposites
4.5.1. Determination of Minimum Inhibitory Concentration (MIC)
4.5.2. Resazurin Assay
4.5.3. Bacterial Morphological Analysis upon Nanomaterials Exposure
4.5.4. Antibacterial Viability (Live/Dead Assay)
4.5.5. Detection of ROS Generation
4.6. Toxicity Assessment of Synthesised Materials
4.6.1. MTT Assay
4.6.2. Detection of Apoptosis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Amjadi, S.; Almasi, H.; Ghorbani, M.; Ramazani, S. Reinforced ZnONPs/rosemary essential oil-incorporated zein electrospun nanofibers by κ-carrageenan. Carbohydr. Polym. 2020, 232, 115800. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Abbasi, B.A.; Mahmood, T.; Kanwal, S.; Ahmad, R.; Ashraf, M. Plant-extract mediated green approach for the synthesis of ZnONPs: Characterization and evaluation of cytotoxic, antimicrobial and antioxidant potentials. J. Mol. Struct. 2019, 1189, 315–327. [Google Scholar] [CrossRef]
- Menazea, A.; Ahmed, M. Synthesis and antibacterial activity of graphene oxide decorated by silver and copper oxide nanoparticles. J. Mol. Struct. 2020, 128536. [Google Scholar] [CrossRef]
- Wang, C.Y.; Makvandi, P.; Zare, E.N.; Tay, F.R.; Niu, L.N. Advances in Antimicrobial Organic and Inorganic Nanocompounds in Biomedicine. Adv. Ther. 2020, 3, 2000024. [Google Scholar] [CrossRef]
- Karami, A.; Xie, Z.; Zhang, J.; Kabir, M.S.; Munroe, P.; Kidd, S.; Zhang, H. Insights into the antimicrobial mechanism of Ag and I incorporated ZnO nanoparticle derivatives under visible light. Mater. Sci. Eng. C 2020, 107, 110220. [Google Scholar] [CrossRef]
- Makvandi, P.; Wang, C.Y.; Zare, E.N.; Borzacchiello, A.; Niu, L.N.; Tay, F.R. Metal-based nanomaterials in biomedical applications: Antimicrobial activity and cytotoxicity aspects. Adv. Funct. Mater. 2020, 30, 1910021. [Google Scholar] [CrossRef]
- Das, B.; Khan, M.I.; Jayabalan, R.; Behera, S.K.; Yun, S.-I.; Tripathy, S.K.; Mishra, A. Understanding the antifungal mechanism of Ag@ ZnO core-shell nanocomposites against Candida krusei. Sci. Rep. 2016, 6, 36403. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.; Nawale, L.U.; Arkile, M.; Shedbalkar, U.U.; Wadhwani, S.A.; Sarkar, D.; Chopade, B.A. Chemical and biological metal nanoparticles as antimycobacterial agents: A comparative study. Int. J. Antimicrob. Agents 2015, 46, 183–188. [Google Scholar] [CrossRef]
- Ji, H.; Zhou, S.; Fu, Y.; Wang, Y.; Mi, J.; Lu, T.; Wang, X.; Lü, C. Size-controllable preparation and antibacterial mechanism of thermo-responsive copolymer-stabilized silver nanoparticles with high antimicrobial activity. Mater. Sci. Eng. C 2020, 110, 110735. [Google Scholar] [CrossRef]
- Zhan, Y.; Lan, J.; Shang, J.; Yang, L.; Guan, X.; Li, W.; Chen, S.; Qi, Y.; Lin, S. Durable ZIF-8/Ag/AgCl/TiO2 decorated PAN nanofibers with high visible light photocatalytic and antibacterial activities for degradation of dyes. J. Alloys Compd. 2020, 822, 153579. [Google Scholar] [CrossRef]
- Yan, X.-W.; Joharian, M.; Naghiloo, M.; Rasuli, R.; Hu, M.-L.; Morsali, A. Metal–organic framework derived porous 2D semiconductor C/ZnO nanocomposite with the high electrical conductivity. Mater. Lett. 2019, 252, 325–328. [Google Scholar] [CrossRef]
- Lee, M.; Shahbaz, H.M.; Kim, J.U.; Lee, H.; Lee, D.-U.; Park, J. Efficacy of UV-TiO2 photocatalysis technology for inactivation of Escherichia coli K12 on the surface of blueberries and a model agar matrix and the influence of surface characteristics. Food Microbiol. 2018, 76, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Hassanpour, M.; Salavati-Niasari, M.; Tafreshi, S.A.H.; Safardoust-Hojaghan, H.; Hassanpour, F. Synthesis, characterization and antibacterial activities of Ni/ZnO nanocomposites using bis (salicylaldehyde) complex precursor. J. Alloys Compd. 2019, 788, 383–390. [Google Scholar] [CrossRef]
- Noohpisheh, Z.; Amiri, H.; Farhadi, S.; Mohammadi-gholami, A. Green synthesis of Ag-ZnO nanocomposites using Trigonella foenum-graecum leaf extract and their antibacterial, antifungal, antioxidant and photocatalytic properties. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 240, 118595. [Google Scholar] [CrossRef]
- Elemike, E.E.; Onwudiwe, D.C.; Wei, L.; Chaogang, L.; Zhiwei, Z. Noble metal –semiconductor nanocomposites for optical, energy and electronics applications. Sol. Energy Mater. Sol. Cells 2019, 201, 110106. [Google Scholar] [CrossRef]
- Hajipour, P.; Bahrami, A.; Eslami, A.; Hosseini-Abari, A.; Hagh Ranjbar, H.R. Chemical bath synthesis of CuO-GO-Ag nanocomposites with enhanced antibacterial properties. J. Alloys Compd. 2020, 821, 153456. [Google Scholar] [CrossRef]
- Joe, A.; Park, S.-H.; Shim, K.-D.; Kim, D.-J.; Jhee, K.-H.; Lee, H.-W.; Heo, C.-H.; Kim, H.-M.; Jang, E.-S. Antibacterial mechanism of ZnO nanoparticles under dark conditions. J. Ind. Eng. Chem. 2017, 45, 430–439. [Google Scholar] [CrossRef]
- Kumar, R.; Umar, A.; Kumar, G.; Nalwa, H.S. Antimicrobial properties of ZnO nanomaterials: A review. Ceram. Int. 2017, 43, 3940–3961. [Google Scholar] [CrossRef]
- Priyadarshini, S.; Sonsudin, F.; Mainal, A.; Yahya, R.; Gopinath, V.; Vadivelu, J.; Alarjani, K.M.; Al Farraj, D.A.; Yehia, H.M. Phytosynthesis of biohybrid nano-silver anchors enhanced size dependent photocatalytic, antibacterial, anticancer properties and cytocompatibility. Process Biochem. 2021, 101, 59–71. [Google Scholar] [CrossRef]
- Priyadarshini, S.; Mainal, A.; Sonsudin, F.; Yahya, R.; Alyousef, A.A.; Mohammed, A. Biosynthesis of TiO2 nanoparticles and their superior antibacterial effect against human nosocomial bacterial pathogens. Res. Chem. Intermed. 2019, 46, 1077–1089. [Google Scholar] [CrossRef]
- Su, D.-L.; Li, P.-J.; Ning, M.; Li, G.-Y.; Shan, Y. Microwave assisted green synthesis of pectin based silver nanoparticles and their antibacterial and antifungal activities. Mater. Lett. 2019, 244, 35–38. [Google Scholar] [CrossRef]
- Song, P.; Lei, Y.; Hu, X.; Wang, C.; Wang, J.; Tang, Y. Rapid one-step synthesis of carbon-supported platinum–copper nanoparticles with enhanced electrocatalytic activity via microwave-assisted heating. J. Colloid Interface Sci. 2020, 574, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zhang, J.; Li, C.; Liu, S.; Wang, L. Morinda citrifolia L. leaves extracts obtained by traditional and eco-friendly extraction solvents: Relation between phenolic compositions and biological properties by multivariate analysis. Ind. Crop. Prod. 2020, 153, 112586. [Google Scholar] [CrossRef]
- Ranoszek-Soliwoda, K.; Tomaszewska, E.; Małek, K.; Celichowski, G.; Orlowski, P.; Krzyzowska, M.; Grobelny, J. The synthesis of monodisperse silver nanoparticles with plant extracts. Colloids Surf. B Biointerfaces 2019, 177, 19–24. [Google Scholar] [CrossRef]
- Kambale, E.K.; Nkanga, C.I.; Mutonkole, B.-P.I.; Bapolisi, A.M.; Tassa, D.O.; Liesse, J.-M.I.; Krause, R.W.; Memvanga, P.B. Green synthesis of antimicrobial silver nanoparticles using aqueous leaf extracts from three Congolese plant species (Brillantaisia patula, Crossopteryx febrifuga and Senna siamea). Heliyon 2020, 6, e04493. [Google Scholar] [CrossRef]
- Abou Assi, R.; Darwis, Y.; Abdulbaqi, I.M.; Vuanghao, L.; Laghari, M. Morinda citrifolia (Noni): A comprehensive review on its industrial uses, pharmacological activities, and clinical trials. Arab. J. Chem. 2017, 10, 691–707. [Google Scholar] [CrossRef]
- Morales-Lozoya, V.; Espinoza-Gómez, H.; Flores-López, Z.L.; Sotelo-Barrera, E.L.; Núñez-Rivera, A.; Cadena-Nava, R.D.; Alonso-Nuñez, G.; Rivero, I.A. Study of the effect of the different parts of Morinda citrifolia L. (noni) on the green synthesis of silver nanoparticles and their antibacterial activity. Appl. Surf. Sci. 2021, 537, 147855. [Google Scholar] [CrossRef]
- Roy, S.; Das, T.K.; Maiti, G.P.; Basu, U. Microbial biosynthesis of nontoxic gold nanoparticles. Mater. Sci. Eng. B 2016, 203, 41–51. [Google Scholar] [CrossRef]
- Alsammarraie, F.K.; Wang, W.; Zhou, P.; Mustapha, A.; Lin, M. Green synthesis of silver nanoparticles using turmeric extracts and investigation of their antibacterial activities. Colloids Surf. B Biointerfaces 2018, 171, 398–405. [Google Scholar] [CrossRef]
- Kumar, B.; Smita, K.; Debut, A.; Cumbal, L. Utilization of Persea americana (Avocado) oil for the synthesis of gold nanoparticles in sunlight and evaluation of antioxidant and photocatalytic activities. Environ. Nanotechnol. Monit. Manag. 2018, 10, 231–237. [Google Scholar] [CrossRef]
- Gole, A.; Dash, C.; Ramakrishnan, V.; Sainkar, S.; Mandale, A.; Rao, M.; Sastry, M. Pepsin− gold colloid conjugates: Preparation, characterization, and enzymatic activity. Langmuir 2001, 17, 1674–1679. [Google Scholar] [CrossRef]
- Xiu, Z.-M.; Zhang, Q.-B.; Puppala, H.L.; Colvin, V.L.; Alvarez, P.J. Negligible particle-specific antibacterial activity of silver nanoparticles. Nano Lett. 2012, 12, 4271–4275. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.G.; Biswas, S.; Roy, T.; Ghosh, S.; Majumder, D.; Basak, P.; Roy, S.; Dhara, S. Sonication assisted hierarchical decoration of Ag-NP on zinc oxide nanoflower impregnated eggshell membrane: Evaluation of antibacterial activity and in vitro cytocompatibility. ACS Sustain. Chem. Eng. 2019, 7, 13717–13733. [Google Scholar] [CrossRef]
- Al-Jumaili, A.; Mulvey, P.; Kumar, A.; Prasad, K.; Bazaka, K.; Warner, J.; Jacob, M.V. Eco-friendly nanocomposites derived from geranium oil and zinc oxide in one step approach. Sci. Rep. 2019, 9, 5973. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ding, Z.; Li, Y.; Miao, J.; Wang, W.; Nundlall, K.; Chen, S. Reactive oxygen species-sensitive thioketal-linked mesoporous silica nanoparticles as drug carrier for effective antibacterial activity. Mater. Des. 2020, 195, 109021. [Google Scholar] [CrossRef]
- Lv, Y.; Cai, G.; Zhang, X.; Fu, S.; Zhang, E.; Yang, L.; Xiao, J.; Dong, Z. Microstructural characterization and in vitro biological performances of Ag, Zn co-incorporated TiO2 coating. Ceram. Int. 2020, 46, 29160–29172. [Google Scholar] [CrossRef]
- Chen, M.; Yang, L.; Zhang, L.; Han, Y.; Lu, Z.; Qin, G.; Zhang, E. Effect of nano/micro-Ag compound particles on the bio-corrosion, antibacterial properties and cell biocompatibility of Ti-Ag alloys. Mater. Sci. Eng. C 2017, 75, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Ali, K.; Ahmed, B.; Ansari, S.M.; Saquib, Q.; Al-Khedhairy, A.A.; Dwivedi, S.; Alshaeri, M.; Khan, M.S.; Musarrat, J. Comparative in situ ROS mediated killing of bacteria with bulk analogue, Eucalyptus leaf extract (ELE)-capped and bare surface copper oxide nanoparticles. Mater. Sci. Eng. C 2019, 100, 747–758. [Google Scholar] [CrossRef]
- Zhang, L.; Gao, Q.; Han, Y. Zn and Ag co-doped anti-microbial TiO2 coatings on Ti by micro-arc oxidation. J. Mater. Sci. Technol. 2016, 32, 919–924. [Google Scholar] [CrossRef]
- He, T.; Peterson, T.E.; Holmuhamedov, E.L.; Terzic, A.; Caplice, N.M.; Oberley, L.W.; Katusic, Z.S. Human endothelial progenitor cells tolerate oxidative stress due to intrinsically high expression of manganese superoxide dismutase. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 2021–2027. [Google Scholar] [CrossRef] [Green Version]
- Hashim, N.; Paramasivam, M.; Tan, J.S.; Kernain, D.; Hussin, M.H.; Brosse, N.; Gambier, F.; Raja, P.B. Green mode synthesis of silver nanoparticles using Vitis vinifera’s tannin and screening its antimicrobial activity/apoptotic potential versus cancer cells. Mater. Today Commun. 2020, 25, 101511. [Google Scholar] [CrossRef]
- Zare, E.N.; Jamaledin, R.; Naserzadeh, P.; Afjeh-Dana, E.; Ashtari, B.; Hosseinzadeh, M.; Vecchione, R.; Wu, A.; Tay, F.R.; Borzacchiello, A. Metal-Based nanostructures/PLGA nanocomposites: Antimicrobial activity, cytotoxicity, and their biomedical applications. ACS Appl. Mater. Interfaces 2019, 12, 3279–3300. [Google Scholar] [CrossRef] [PubMed]
- Ghaemi, B.; Shaabani, E.; Najafi-Taher, R.; Jafari Nodooshan, S.; Sadeghpour, A.; Kharrazi, S.; Amani, A. Intracellular ROS induction by Ag@ ZnO core–shell nanoparticles: Frontiers of permanent optically active holes in breast cancer theranostic. ACS Appl. Mater. Interfaces 2018, 10, 24370–24381. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.-K.; Gurunathan, S.; Kang, M.-H.; Han, J.W.; Das, J.; Choi, Y.-J.; Kwon, D.-N.; Cho, S.-G.; Park, C.; Seo, H.G. Hypoxia-mediated autophagic flux inhibits silver nanoparticle-triggered apoptosis in human lung cancer cells. Sci. Rep. 2016, 6, 21688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarkar, A.; Roy, S.; Sanpui, P.; Jaiswal, A. Plasmonic Gold Nanorattle Impregnated Chitosan Nanocarrier for Stimulus Responsive Theranostics. ACS Appl. Bio Mater. 2019, 2, 4812–4825. [Google Scholar] [CrossRef]
- Xia, T.; Kovochich, M.; Brant, J.; Hotze, M.; Sempf, J.; Oberley, T.; Sioutas, C.; Yeh, J.I.; Wiesner, M.R.; Nel, A.E. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006, 6, 1794–1807. [Google Scholar] [CrossRef] [PubMed]
- Pageni, P.; Yang, P.; Bam, M.; Zhu, T.; Chen, Y.P.; Decho, A.W.; Nagarkatti, M.; Tang, C. Recyclable magnetic nanoparticles grafted with antimicrobial metallopolymer-antibiotic bioconjugates. Biomaterials 2018, 178, 363–372. [Google Scholar] [CrossRef]
- Das, S.; Diyali, S.; Vinothini, G.; Perumalsamy, B.; Balakrishnan, G.; Ramasamy, T.; Dharumadurai, D.; Biswas, B. Synthesis, morphological analysis, antibacterial activity of iron oxide nanoparticles and the cytotoxic effect on lung cancer cell line. Heliyon 2020, 6, e04953. [Google Scholar] [CrossRef]
- Gopinath, V.; Mubarak Ali, D.; Vadivelu, J.; Manjunath Kamath, S.; Syed, A.; Elgorban, A.M. Synthesis of biocompatible chitosan decorated silver nanoparticles biocomposites for enhanced antimicrobial and anticancer property. Process Biochem. 2020, 99, 348–356. [Google Scholar] [CrossRef]
- Hemalatha, T.; Prabu, P.; Gunadharini, D.N.; Gowthaman, M.K. Fabrication and characterization of dual acting oleyl chitosan functionalised iron oxide/gold hybrid nanoparticles for MRI and CT imaging. Int. J. Biol. Macromol. 2018, 112, 250–257. [Google Scholar] [CrossRef]






| Bacterial Strains | TiO2NPs (µg/mL) | AgNPs (µg/mL) | TiO2/AgNCs (µg/mL) | ZnO/TiO2/AgNCs (µg/mL) | ||||
|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| E. coli | 500 | 500 | 31.2 | 31.2 | 31.2 | 31.2 | 31.2 | 31.2 |
| P. aeruginosa | 500 | 500 | 62.5 | 62.5 | 31.2 | 31.2 | 15.6 | 31.2 |
| S. aureus | 500 | 500 | 62.5 | 62.5 | 62.5 | 62.5 | 31.2 | 31.2 |
| B. subtilis | 250 | 250 | 31.2 | 62.5 | 31.2 | 62.5 | 15.6 | 15.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakthi Mohan, P.; Sonsuddin, F.; Mainal, A.B.; Yahya, R.; Venkatraman, G.; Vadivelu, J.; Al-Farraj, D.A.; Al-Mohaimeed, A.M.; Alarjani, K.M. Facile In-Situ Fabrication of a Ternary ZnO/TiO2/Ag Nanocomposite for Enhanced Bactericidal and Biocompatibility Properties. Antibiotics 2021, 10, 86. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics10010086
Sakthi Mohan P, Sonsuddin F, Mainal AB, Yahya R, Venkatraman G, Vadivelu J, Al-Farraj DA, Al-Mohaimeed AM, Alarjani KM. Facile In-Situ Fabrication of a Ternary ZnO/TiO2/Ag Nanocomposite for Enhanced Bactericidal and Biocompatibility Properties. Antibiotics. 2021; 10(1):86. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics10010086
Chicago/Turabian StyleSakthi Mohan, Priyadarshini, Faridah Sonsuddin, Azizah Binti Mainal, Rosiyah Yahya, Gopinath Venkatraman, Jamuna Vadivelu, Dunia A. Al-Farraj, Amal M. Al-Mohaimeed, and Khaloud Mohammed Alarjani. 2021. "Facile In-Situ Fabrication of a Ternary ZnO/TiO2/Ag Nanocomposite for Enhanced Bactericidal and Biocompatibility Properties" Antibiotics 10, no. 1: 86. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics10010086





