Antimicrobial and Antivirulence Impacts of Phenolics on Salmonella Enterica Serovar Typhimurium
Abstract
:1. Introduction
2. Results
2.1. Antimicrobial Effect on Bacterial Growth
2.2. Evaluation of Resistance Development
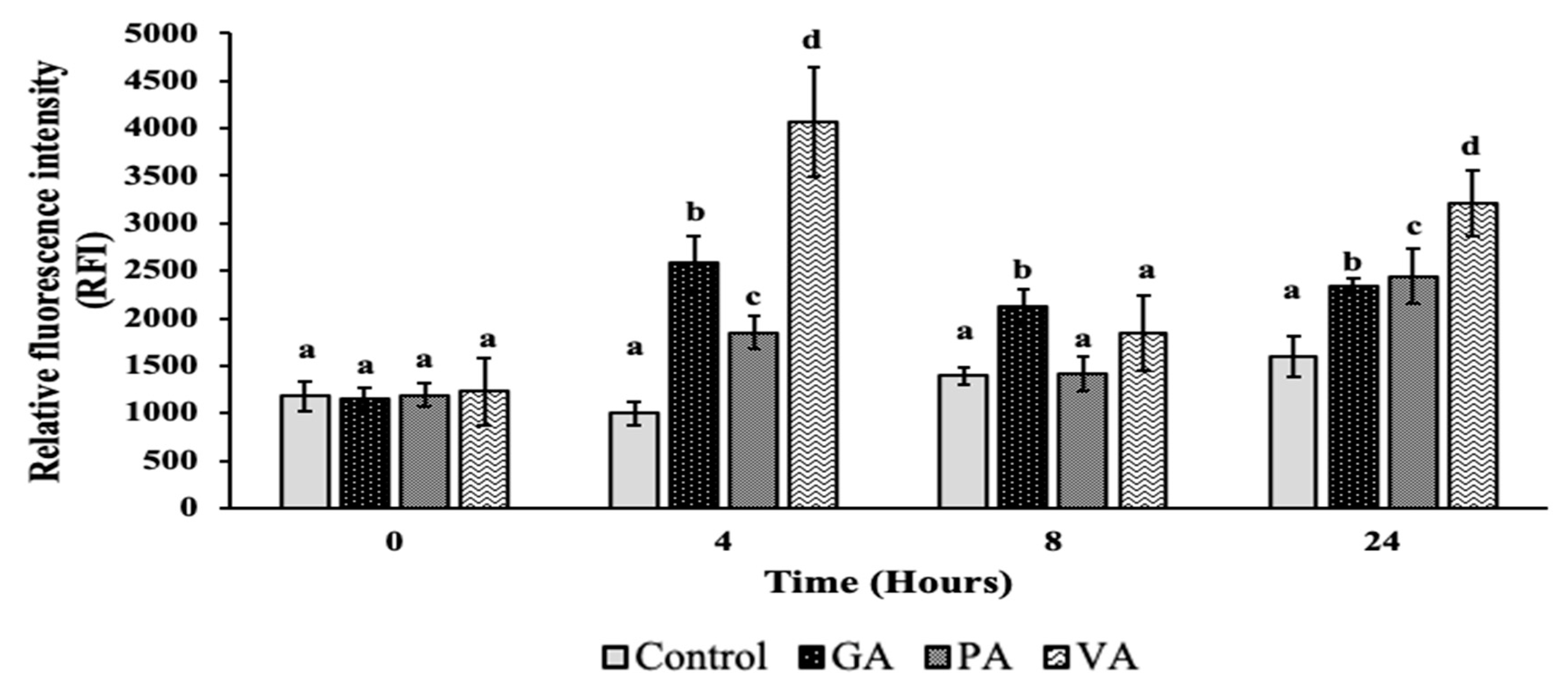
2.3. Plasma Membrane Permeability and Damage
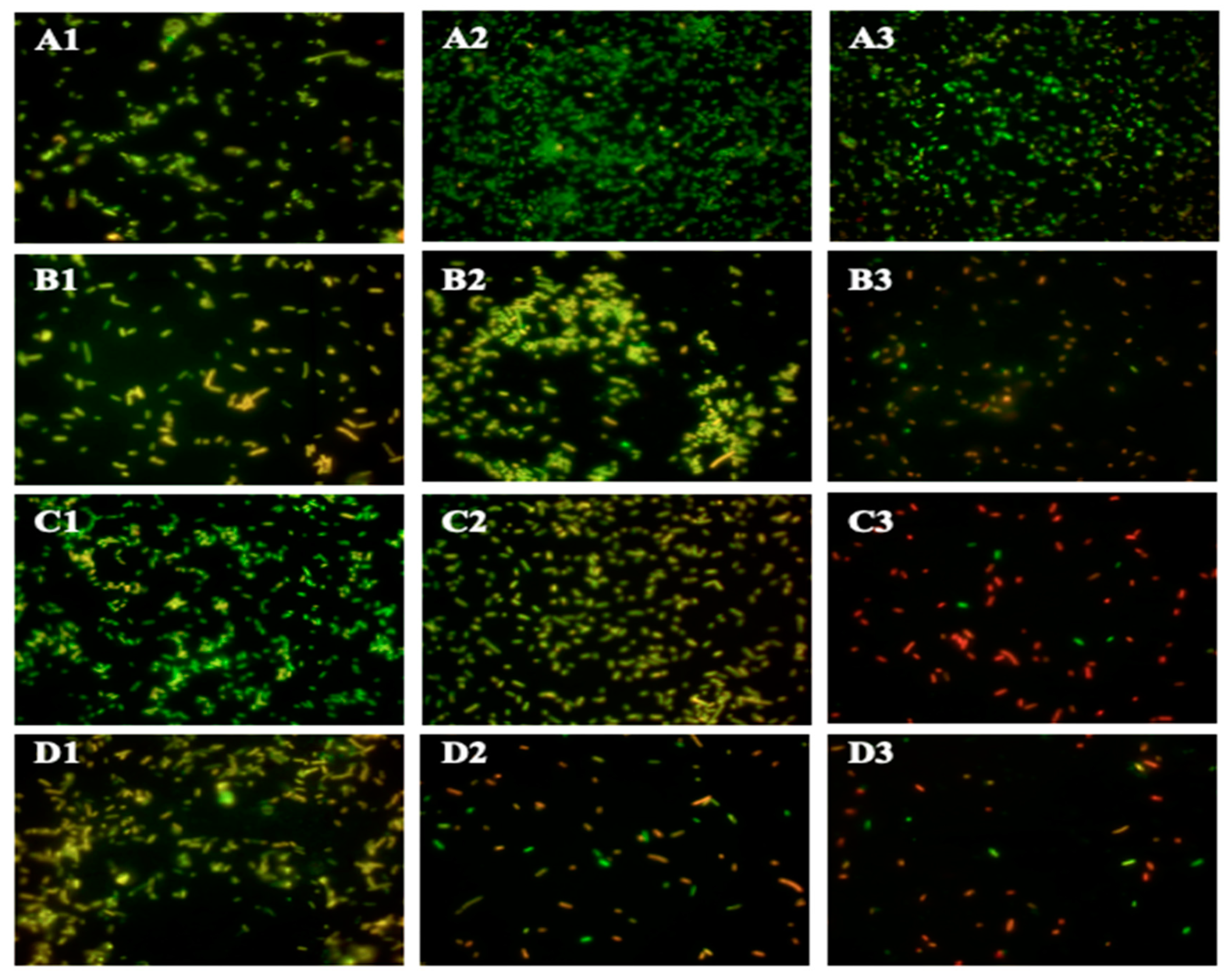
2.4. Morphological Alterations of ST
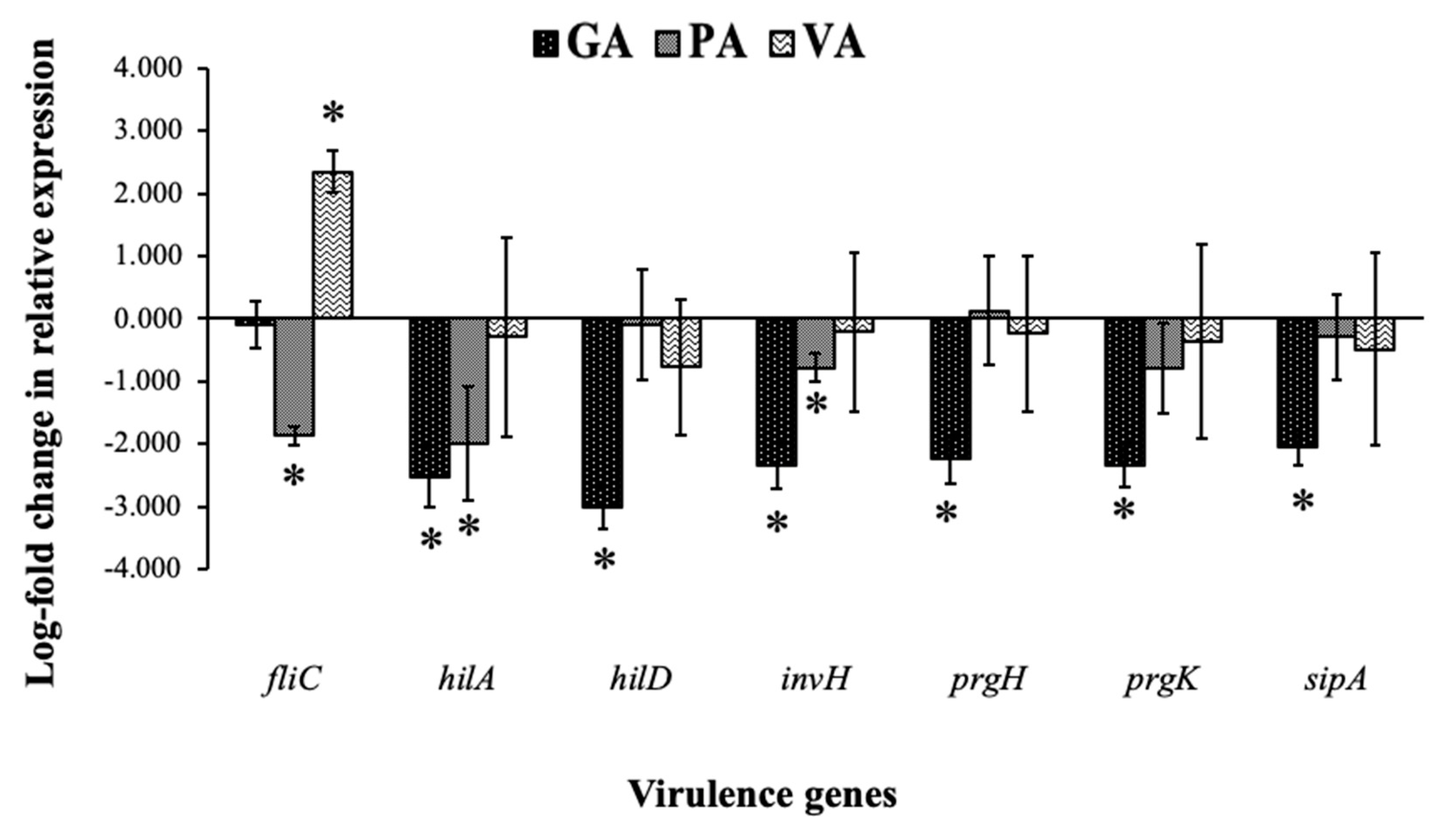
2.5. Relative Gene Expression of ST
3. Discussion
4. Materials and Methods
4.1. Bacterial Strain and Their Growth Conditions
4.2. Compounds and Stock Solution Preparation
4.3. Determining MIC and MBC
4.4. Resistance Development over Time
4.5. Measuring Membrane Damage and Permeability Using Fluorescent Dyes
4.6. Scanning Electron Microscopy
4.7. RNA Extraction and cDNA Synthesis
4.8. Quantitative RT-PCR Assay
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N. GBD 2017 Cause of Death Collaborators. Global, Regional, and National Age-Sex-Specific Mortality for 282 Causes of Death in 195 Countries and Territories, 1980–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef] [Green Version]
- Dewey-Mattia, D.; Manikonda, K.; Hall, A.J.; Wise, M.E.; Crowe, S.J. Surveillance for Foodborne Disease Outbreaks—United States, 2009-2015. Mmwr Surveill. Summ. 2018, 67, 1–11. [Google Scholar] [CrossRef]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne Illness Acquired in the United States-Major Pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Tack, D.M.; Ray, L.; Griffin, P.M.; Cieslak, P.R.; Dunn, J.; Rissman, T.; Jervis, R.; Lathrop, S.; Muse, A.; Duwell, M.; et al. Preliminary Incidence and Trends of Infections with Pathogens Transmitted Commonly Through Food—Foodborne Diseases Active Surveillance Network, 10 U.S. Sites, 2016-2019. MMWR. Morb. Mortal. Wkly. Rep. 2020, 69, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Fatica, M.K.; Schneider, K.R. Salmonella and Produce: Survival in the Plant Environment and Implications in Food Safety. Virulence 2011, 2, 573–579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carrasco, E.; Morales-Rueda, A.; García-Gimeno, R.M. Cross-Contamination and Recontamination by Salmonella in Foods: A Review. Food Res. Int. 2012, 45, 545–556. [Google Scholar] [CrossRef]
- CDC. Serotypes and the Importance of Serotyping Salmonella. Center for Disease Control and Prevention, 2020. Available online: https://www.cdc.gov/salmonella/reportspubs/salmonella-atlas/serotyping-importance.html#:~:text=Serotyping%20has%20been%20the%20core,and%20to%20detect%20more%20outbreaks (accessed on 17 April 2020).
- Crump, J.A.; Sjölund-Karlsson, M.; Gordon, M.A.; Parry, C.M. Epidemiology, Clinical Presentation, Laboratory Diagnosis, Antimicrobial Resistance, and Antimicrobial Management of Invasive Salmonella Infections. Clin. Microbiol. Rev. 2015, 28, 901–937. [Google Scholar] [CrossRef] [Green Version]
- Lou, L.; Zhang, P.; Piao, R.; Wang, Y. Salmonella Pathogenicity Island 1 (SPI-1) and Its Complex Regulatory Network. Front. Cell. Infect. Microbiol. 2019, 9, 270. [Google Scholar] [CrossRef] [Green Version]
- Jennings, E.; Thurston, T.L.M.; Holden, D.W. Salmonella SPI-2 Type III Secretion System Effectors: Molecular Mechanisms And Physiological Consequences. Cell Host Microbe 2017, 22, 217–231. [Google Scholar] [CrossRef]
- Jajere, S.M. A Review of Salmonella Enterica with Particular Focus on the Pathogenicity and Virulence Factors, Host Specificity and Adaptation and Antimicrobial Resistance Including Multidrug Resistance. Vet. World 2019, 12, 504–521. [Google Scholar] [CrossRef] [Green Version]
- Angelo, K.M.; Reynolds, J.; Karp, B.E.; Hoekstra, R.M.; Scheel, C.M.; Friedman, C. Antimicrobial Resistance among Nontyphoidal Salmonella Isolated from Blood in the United States, 2003-2013. J. Infect. Dis. 2016, 214, 1565–1570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fluit, A.C. Towards More Virulent and Antibiotic-Resistant Salmonella? FEMS Immunol. Med. Microbiol. 2005, 43, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). 2016-2017 NARMS Integrated Summary. U.S. Department of Health and Human Services: Rockville, MD, USA. Available online: https://www.fda.gov/animal-veterinary/national-antimicrobial-resistance-monitoring-system/2016-2017-narms-integrated-summary-interactive (accessed on 17 April 2020).
- Bythwood, T.N.; Soni, V.; Lyons, K.; Hurley-Bacon, A.; Lee, M.D.; Hofacre, C.; Sanchez, S.; Maurer, J.J. Antimicrobial Resistant Salmonella Enterica Typhimurium Colonizing Chickens: The Impact of Plasmids, Genotype, Bacterial Communities, and Antibiotic Administration on Resistance. Front. Sustain. Food Syst. 2019, 3, 20. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [Green Version]
- Alvarado-Martinez, Z.; Aditya, A.; Biswas, D. Plant Antioxidants, Extraction Strategies, and Their Application in Meat. In Meat Quality Analysis: Advanced Evaluation Methods, Techniques, and Technologies; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar]
- Goleniowski, M.; Bonfill, M.; Cusido, R.; Palazón, J. Phenolic Acids. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Li, W.; Beta, T. Food Sources of Phenolics Compounds. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Gyawali, R.; Ibrahim, S.A. Natural Products as Antimicrobial Agents. Food Control. 2014, 46, 412–429. [Google Scholar] [CrossRef]
- Salaheen, S.; Nguyen, C.; Hewes, D.; Biswas, D. Cheap Extraction of Antibacterial Compounds of Berry Pomace and Their Mode of Action against the Pathogen Campylobacter Jejuni. Food Control. 2014, 46, 174–181. [Google Scholar] [CrossRef]
- Salaheen, S.; Jaiswal, E.; Joo, J.; Peng, M.; Ho, R.; OConnor, D.; Adlerz, K.; Aranda-Espinoza, J.H.; Biswas, D. Bioactive Extracts from Berry Byproducts on the Pathogenicity of Salmonella Typhimurium. Int. J. Food Microbiol. 2016, 237, 128–135. [Google Scholar] [CrossRef]
- Lee, O.H.; Lee, B.Y. Antioxidant and Antimicrobial Activities of Individual and Combined Phenolics in Olea Europaea Leaf Extract. Bioresour. Technol. 2010, 101, 3751–3754. [Google Scholar] [CrossRef]
- Cueva, C.; Moreno-Arribas, M.V.; Martín-Álvarez, P.J.; Bills, G.; Vicente, M.F.; Basilio, A.; Rivas, C.L.; Requena, T.; Rodríguez, J.M.; Bartolomé, B. Antimicrobial Activity of Phenolic Acids against Commensal, Probiotic and Pathogenic Bacteria. Res. Microbiol. 2010, 161, 372–382. [Google Scholar] [CrossRef]
- Gutiérrez-Larraínzar, M.; Rúa, J.; Caro, I.; de Castro, C.; de Arriaga, D.; García-Armesto, M.R.; del Valle, P. Evaluation of Antimicrobial and Antioxidant Activities of Natural Phenolic Compounds against Foodborne Pathogens and Spoilage Bacteria. Food Control. 2012, 26, 555–563. [Google Scholar] [CrossRef]
- Slonczewski, J.L.; Fujisawa, M.; Dopson, M.; Krulwich, T.A. Cytoplasmic PH Measurement and Homeostasis in Bacteria and Archaea. Adv. Microb. Physiol. 2009, 55, 1–79. [Google Scholar] [PubMed]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial Activity and Mode of Action of Ferulic and Gallic Acids against Pathogenic Bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Kaurinovic, B.; Vastag, D. Flavonoids and Phenolic Acids as Potential Natural Antioxidants. In Antioxidants; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef] [Green Version]
- Ravichandran, M.; Hettiarachchy, N.S.; Ganesh, V.; Ricke, S.C.; Singh, S. Enhancement of Antimicrobial Activities of Naturally Occurring Phenolic Compounds by Nanoscale Delivery against Listeria Monocytogenes, Escherichia Coli O157:H7 and Salmonella Typhimurium in Broth and Chicken Meat System. J. Food Saf. 2011, 31, 462–471. [Google Scholar] [CrossRef]
- De Camargo, A.C.; Regitano-d’Arce, M.A.B.; Rasera, G.B.; Canniatti-Brazaca, S.G.; do Prado-Silva, L.; Alvarenga, V.O.; Sant’Ana, A.S.; Shahidi, F. Phenolic Acids and Flavonoids of Peanut By-Products: Antioxidant Capacity and Antimicrobial Effects. Food Chem. 2017, 237, 538–544. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Martins, C.; Pereira, C.; Silvestre, A.J.D.; Rocha, S.M. Current Challenges and Perspectives for the Use of Aqueous Plant Extracts in the Management of Bacterial Infections: The Case-Study of Salmonella Enterica Serovars. Int. J. Mol. Sci. 2019, 20, 940. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N.; Goel, N. Phenolic Acids: Natural Versatile Molecules with Promising Therapeutic Applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Aldulaimi, O.A. General Overview of Phenolics from Plant to Laboratory, Good Antibacterials or Not. Pharmacogn. Rev. 2017, 11, 123–127. [Google Scholar] [CrossRef] [Green Version]
- López-Romero, J.C.; Valenzuela-Melendres, M.; Juneja, V.K.; García-Dávila, J.; Camou, J.P.; Peña-Ramos, A.; González-Ríos, H. Effects and Interactions of Gallic Acid, Eugenol and Temperature on Thermal Inactivation of Salmonella Spp. in Ground Chicken. Food Res. Int. 2018, 103, 289–294. [Google Scholar] [CrossRef]
- Bernal-Mercado, A.T.; Gutierrez-Pacheco, M.M.; Encinas-Basurto, D.; Mata-Haro, V.; Lopez-Zavala, A.A.; Islas-Osuna, M.A.; Gonzalez-Aguilar, G.A.; Ayala-Zavala, J.F. Synergistic Mode of Action of Catechin, Vanillic and Protocatechuic Acids to Inhibit the Adhesion of Uropathogenic Escherichia Coli on Silicone Surfaces. J. Appl. Microbiol. 2020, 128, 387–400. [Google Scholar] [CrossRef]
- Torzewska, A.; Rozalski, A. Inhibition of Crystallization Caused by Proteus Mirabilis during the Development of Infectious Urolithiasis by Various Phenolic Substances. Microbiol. Res. 2014, 169, 579–584. [Google Scholar] [CrossRef]
- Liu, J.; Du, C.; Beaman, H.T.; Monroe, M.B.B. Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships. Pharmaceutics 2020, 12, 419. [Google Scholar] [CrossRef] [PubMed]
- Gootz, T.D. The Forgotten Gram-Negative Bacilli: What Genetic Determinants Are Telling Us about the Spread of Antibiotic Resistance. Biochem. Pharm. 2006, 71, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Yang, M.; Wang, T.; Sun, Z.; Liu, M.; Zhang, J.; Zeng, Q.; Cai, C.; Li, Y. Antibacterial Mechanism of Vanillic Acid on Physiological, Morphological, and Biofilm Properties of Carbapenem-Resistant Enterobacter Hormaechei. J. Food Prot. 2020, 83, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Wright, G.D. Bacterial Resistance to Antibiotics: Enzymatic Degradation and Modification. Adv. Drug Deliv. Rev. 2005, 57, 1451–1470. [Google Scholar] [CrossRef]
- Dcosta, V.M.; King, C.E.; Kalan, L.; Morar, M.; Sung, W.W.L.; Schwarz, C.; Froese, D.; Zazula, G.; Calmels, F.; Debruyne, R.; et al. Antibiotic Resistance Is Ancient. Nature 2011, 477, 457–461. [Google Scholar] [CrossRef]
- Sánchez-Maldonado, A.F.; Schieber, A.; Gänzle, M.G. Structure-Function Relationships of the Antibacterial Activity of Phenolic Acids and Their Metabolism by Lactic Acid Bacteria. J. Appl. Microbiol. 2011, 111, 1176–1184. [Google Scholar] [CrossRef]
- Cheng, Z.; Ren, J.; Li, Y.; Chang, W.; Chen, Z. Phenolic Antioxidants: Electrochemical Behavior and the Mechanistic Elements Underlying Their Anodic Oxidation Reaction. Redox Rep. 2002, 7, 395–402. [Google Scholar] [CrossRef]
- Page, M.G.P. The Role of the Outer Membrane of Gram-Negative Bacteria in Antibiotic Resistance: Ajax’ Shield or Achilles’ Heel? In Antibiotic Resistance; Springer: Berlin/Heidelberg, Germany, 2012; Volume 211, pp. 67–86. [Google Scholar]
- Berry, E.D. Development and Demonstration of RNA Isolation and RT-PCR Procedures to Detect Escherichia Coli O157:H7 Gene Expression on Beef Carcass Surfaces. Lett. Appl. Microbiol. 2000, 31, 265–269. [Google Scholar] [CrossRef]
- Jaishankar, J.; Srivastava, P. Molecular Basis of Stationary Phase Survival and Applications. Front. Microbiol. 2017, 8, 2000. [Google Scholar] [CrossRef] [Green Version]
- Poirel, L.; Jayol, A.; Nordmanna, P. Polymyxins: Antibacterial Activity, Susceptibility Testing, and Resistance Mechanisms Encoded by Plasmids or Chromosomes. Clin. Microbiol. Rev. 2017, 30, 557–596. [Google Scholar] [CrossRef] [Green Version]
- Krulwich, T.A.; Sachs, G.; Padan, E. Molecular Aspects of Bacterial PH Sensing and Homeostasis. Nat. Rev. Microbiol. 2011, 9, 330–343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirshfield, I.N.; Terzulli, S.; O’Byrne, C. Weak Organic Acids: A Panoply of Effects on Bacteria. Sci. Prog. 2003, 86, 245–270. [Google Scholar] [CrossRef] [PubMed]
- Varma, A.; Young, K.D. In Escherichia Coli, MreB and FtsZ Direct the Synthesis of Lateral Cell Wall via Independent Pathways That Require PBP 2. J. Bacteriol. 2009, 191, 3526–3533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bulmer, D.M.; Kharraz, L.; Grant, A.J.; Dean, P.; Morgan, F.J.E.; Karavolos, M.H.; Doble, A.C.; McGhie, E.J.; Koronakis, V.; Daniel, R.A.; et al. The Bacterial Cytoskeleton Modulates Motility, Type 3 Secretion, and Colonization in Salmonella. PLoS Pathog. 2012, 8, e1002500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doble, A.C.; Bulmer, D.M.; Kharraz, L.; Karavolos, M.H.; Khan, C.M.A. The Function of the Bacterial Cytoskeleton in Salmonella Pathogenesis. Virulence 2012, 3, 446–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fàbrega, A.; Vila, J. Salmonella Enterica Serovar Typhimurium Skills to Succeed in the Host: Virulence and Regulation. Clin. Microbiol. Rev. 2013, 26, 308–341. [Google Scholar]
- Spöring, I.; Felgner, S.; Preuße, M.; Eckweiler, D.; Rohde, M.; Häussler, S.; Weiss, S.; Erhardt, M. Regulation of Flagellum Biosynthesis in Response to Cell Envelope Stress in Salmonella Enterica Serovar Typhimurium. MBio 2018, 9. [Google Scholar] [CrossRef] [Green Version]
- Van Ampting, M.T.J.; Schonewille, A.J.; Vink, C.; Brummer, R.J.M.; van der Meer, R.; Bovee-Oudenhoven, I.M.J. Damage to the Intestinal Epithelial Barrier by Antibiotic Pretreatment of Salmonella-Infected Rats Is Lessened by Dietary Calcium or Tannic Acid. J. Nutr. 2010, 140, 2167–2172. [Google Scholar] [CrossRef]
- Gupta, P.D.; Birdi, T.J. Development of Botanicals to Combat Antibiotic Resistance. J. Ayurveda Integr. Med. 2017, 8, 266–275. [Google Scholar] [CrossRef]
- Salaheen, S.; Peng, M.; Joo, J.; Teramoto, H.; Biswas, D. Eradication and Sensitization of Methicillin Resistant Staphylococcus Aureus to Methicillin with Bioactive Extracts of Berry Pomace. Front. Microbiol. 2017, 8, 253. [Google Scholar] [CrossRef]
- Ejim, L.; Farha, M.A.; Falconer, S.B.; Wildenhain, J.; Coombes, B.K.; Tyers, M.; Brown, E.D.; Wright, G.D. Combinations of Antibiotics and Nonantibiotic Drugs Enhance Antimicrobial Efficacy. Nat. Chem. Biol. 2011, 6, 348–350. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing. In CLSI Supplement M100, 27th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2019. [Google Scholar]
- Ma, Z.; Kim, D.; Adesogan, A.T.; Ko, S.; Galvao, K.; Jeong, K.C. Chitosan Microparticles Exert Broad-Spectrum Antimicrobial Activity against Antibiotic-Resistant Micro-Organisms without Increasing Resistance. ACS Appl. Mater. Interfaces 2016, 8, 10700–10709. [Google Scholar] [CrossRef] [PubMed]
- Farkas, A.; Maróti, G.; Kereszt, A.; Kondorosi, É. Comparative Analysis of the Bacterial Membrane Disruption Effect of Two Natural Plant Antimicrobial Peptides. Front. Microbiol. 2017, 8, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, M.; Tabashsum, Z.; Patel, P.; Bernhardt, C.; Biswas, D. Linoleic Acids Overproducing Lactobacillus Casei Limits Growth, Survival, and Virulence of Salmonella Typhimurium and Enterohaemorrhagic Escherichia Coli. Front. Microbiol. 2018, 9, 2663. [Google Scholar] [CrossRef] [Green Version]
- Peng, M.; Reichmann, G.; Biswas, D. Lactobacillus Casei and Its Byproducts Alter the Virulence Factors of Foodborne Bacterial Pathogens. J. Funct. Foods 2015, 15, 418–428. [Google Scholar] [CrossRef]
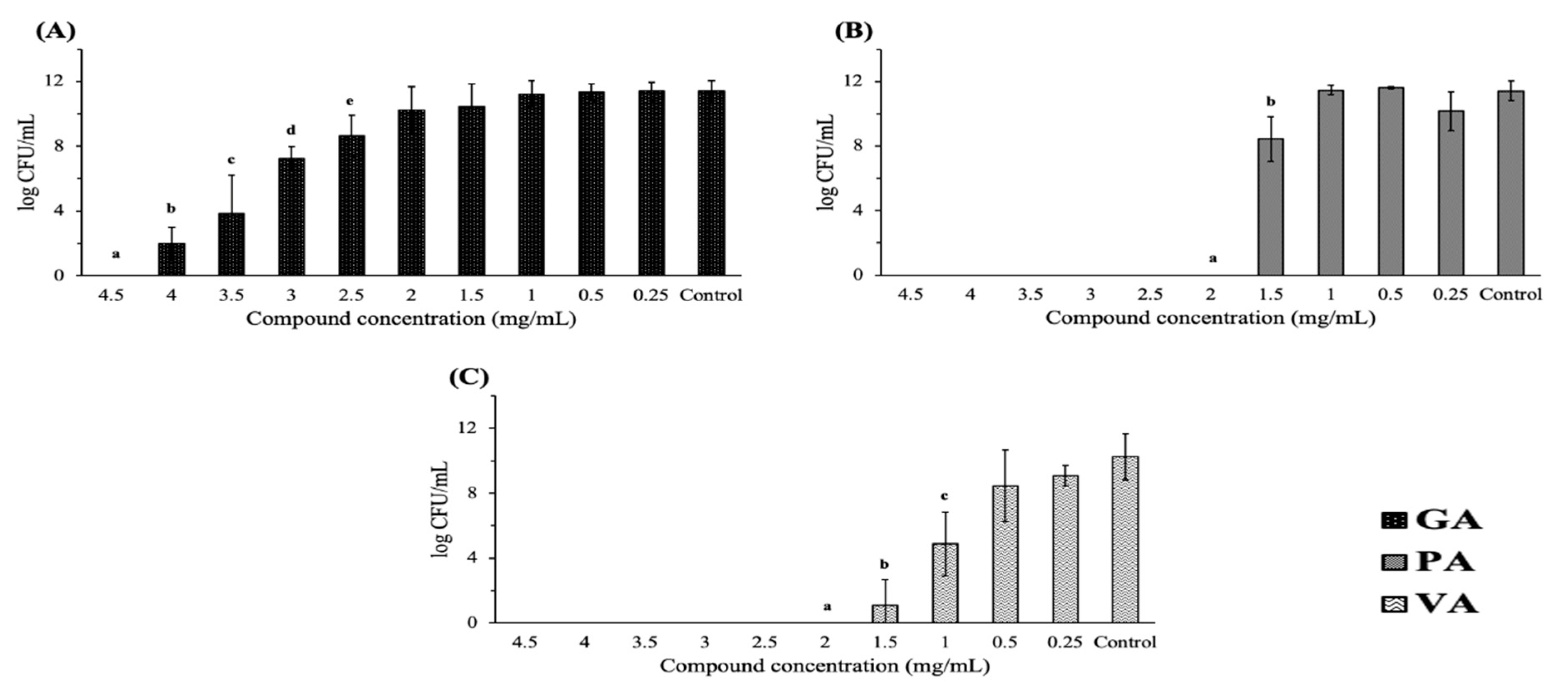


| Treatment | MIC (mg/mL) | MBC/MIC | Bactericidal/Bacteriostatic |
|---|---|---|---|
| Gallic acid | 3.5 | 1.28 | Bactericidal |
| Protocatechuic acid | 2 | 1 | Bactericidal |
| Vanillic acid | 1.5 | 1.33 | Bactericidal |
| Gene | Protein | Primer Sequence (5’-3’) |
|---|---|---|
| 16S rRNA | 16S ribosomal protein | F: GTAGTACGATGGCGAAACTGC R: CTTCTCGACCCGAGGGACTT |
| fliC | flagellum subunit | F: GCAGATGACGGTACATCCAA R: CCAGATCAGGCTGTGCTTTA |
| hilA | SPI-1 transcriptional regulator | F: AATGGTCACAGGCTGAGGTG R: ACATCGTCGCGACTTGTGAA |
| hilD | SPI-1 transcriptional regulator | F: CTCTGTGGGTACCGCCATTT R: TGCTTTCGGAGCGGTAAACT |
| invH | adherence and invasion | F: GGTGCCCCTCCCTTCCT R: TGCGTTGGCCAGTTGCT |
| prgH | T3SS needle support at membrane | F: TGAACGGCTGTGAGTTTCCA R: GCGCATCACTCTGACCTACCA |
| prgK | T3SS needle support at membrane | F: GGGTGGAAATAGCGCAGATG R: TCAGCTCGCGGAGACGATA |
| sipA | actin binding protein for cell invasion | F: CGTCTTCGCCTCAGGAGAAT R: TGCCGGGCTCTTTCGTT |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvarado-Martinez, Z.; Bravo, P.; Kennedy, N.-F.; Krishna, M.; Hussain, S.; Young, A.C.; Biswas, D. Antimicrobial and Antivirulence Impacts of Phenolics on Salmonella Enterica Serovar Typhimurium. Antibiotics 2020, 9, 668. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9100668
Alvarado-Martinez Z, Bravo P, Kennedy N-F, Krishna M, Hussain S, Young AC, Biswas D. Antimicrobial and Antivirulence Impacts of Phenolics on Salmonella Enterica Serovar Typhimurium. Antibiotics. 2020; 9(10):668. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9100668
Chicago/Turabian StyleAlvarado-Martinez, Zabdiel, Paulina Bravo, Nana-Frekua Kennedy, Mayur Krishna, Syed Hussain, Alana C. Young, and Debabrata Biswas. 2020. "Antimicrobial and Antivirulence Impacts of Phenolics on Salmonella Enterica Serovar Typhimurium" Antibiotics 9, no. 10: 668. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9100668





