Isolation of Tetracycline-Resistant Chlamydia suis from a Pig Herd Affected by Reproductive Disorders and Conjunctivitis
Abstract
:1. Introduction
2. Case Study
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schautteet, K.; Vanrompay, D. Chlamydiaceae infections in pig. Vet. Res. 2011, 42, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rogers, D.G.; Andersen, A.A. Conjunctivitis caused by a swine Chlamydia trachomatis-like organism in gnotobiotic pigs. J. Vet. Diagn. Investig. 1999, 11, 341–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Becker, A.; Lutz-Wohlgroth, L.; Brugnera, E.; Lu, Z.H.; Zimmermann, D.R.; Grimm, F.; Grosse Beilage, E.; Kaps, S.; Spiess, B.; Pospischil, A. Intensively kept pigs pre-disposed to chlamydial associated conjunctivitis. J. Vet. Med. Ser. A 2007, 54, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Reinhold, P.; Kirschvink, N.; Theegarten, D.; Berndt, A. An experimentally induced Chlamydia suis infection in pigs results in severe lung function disorders and pulmonary inflammation. Vet. Res. 2008, 39, 35. [Google Scholar] [CrossRef] [Green Version]
- Guscetti, F.; Schiller, I.; Sydler, T.; Heinen, E.; Pospischil, A. Experimental enteric infection of gnotobiotic piglets with Chlamydia suis strain S45. Vet. Microbiol. 2009, 135, 157–168. [Google Scholar] [CrossRef]
- Rogers, D.G.; Andersen, A.A. Intestinal lesions caused by a strain of Chlamydia suis in weanling pigs infected at 21 days of age. J. Vet. Diagn. Investig. 2000, 12, 233–239. [Google Scholar] [CrossRef] [Green Version]
- Turner, G.V.S. A microbiological study of polyarthritis in slaughter pigs. J. S. Afr. Vet. Assoc. 1982, 53, 99–101. [Google Scholar]
- Thoma, R.; Guscetti, F.; Schiller, I.; Schmeer, N.; Corboz, L.; Pospischil, A. Chlamydiae in porcine abortion. Vet. Pathol. 1997, 34, 467–469. [Google Scholar] [CrossRef]
- Woollen, N.; Daniels, E.K.; Yeary, T.; Leipold, H.W.; Phillips, R.M. Chlamydial infection and perinatal mortality in a swine herd. J. Am. Vet. Med. Assoc. 1990, 197, 600–601. [Google Scholar]
- Kauffold, J.; Melzer, F.; Berndt, A.; Hoffmann, G.; Hotzel, H.; Sachse, K. Chlamydiae in oviducts and uteri of repeat breeder pigs. Theriogenology 2006, 66, 1816–1823. [Google Scholar] [CrossRef]
- Camenisch, U.; Lu, Z.H.; Vaughan, L.; Corboz, L.; Zimmermann, D.R.; Wittenbrink, M.M.; Pospischil, A.; Sydler, T. Diagnostic investigation into the role of Chlamydiae in cases of increased rates of return to oestrus in pigs. Vet. Rec. 2004, 155, 593–596. [Google Scholar] [CrossRef] [PubMed]
- Eggemann, G.; Wendt, M.; Hoelzle, L.E.; Jäger, C.; Weiss, R.; Failing, K. Zum vorkommen von chlamydien-infektionen in zuchtsauenbeständen und deren bedeutung für das fruchtbarkeitsgeschehen. DTW Dtsch. Tierarztl. Wochenschr. 2000, 107, 3–10. [Google Scholar] [PubMed]
- Hoffmann, K.; Schott, F.; Donati, M.; Di Francesco, A.; Hässig, M.; Wanninger, S.; Sidler, X.; Borel, N. Prevalence of chlamydial infections in fattening pigs and their influencing factors. PLoS ONE 2015, 10, e0143576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borel, N.; Polkinghorne, A.; Pospischil, A. A review on chlamydial diseases in animals: Still a challenge for pathologists? Vet. Pathol. 2018, 55, 374–390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Broes, A.; Taylor, D.J.; Martineau, G.-P. Miscellaneous Bacterial Infections. In Diseases of Swine, 11th ed.; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2019; pp. 981–1001. ISBN 9781119350927. [Google Scholar]
- Angulo, F.J.; Collignon, P.; Powers, J.H.; Chiller, T.M.; Aidara-Kane, A.; Aarestrup, F.M. World Health Organization ranking of antimicrobials according to their importance in human medicine: A critical step for developing risk management strategies for the use of antimicrobials in food production animals. Clin. Infect. Dis. 2009, 49, 132–141. [Google Scholar] [CrossRef] [Green Version]
- Dugan, J.; Rockey, D.D.; Jones, L.; Andersen, A.A. Tetracycline resistance in Chlamydia suis mediated by genomic islands inserted into the chlamydial inv-like gene. Antimicrob. Agents Chemother. 2004, 48, 3989–3995. [Google Scholar] [CrossRef] [Green Version]
- Lenart, J.; Andersen, A.A.; Rockey, D.D. Growth and development of tetracycline-resistant Chlamydia suis. Antimicrob. Agents Chemother. 2001, 45, 2198–2203. [Google Scholar] [CrossRef] [Green Version]
- Marti, H.; Borel, N.; Dean, D.; Leonard, C.A. Evaluating the antibiotic susceptibility of Chlamydia—New approaches for in vitro assays. Front. Microbiol. 2018, 9, 1414. [Google Scholar] [CrossRef] [Green Version]
- Suchland, R.J.; Sandoz, K.M.; Jeffrey, B.M.; Stamm, W.E.; Rockey, D.D. Horizontal transfer of tetracycline resistance among Chlamydia spp. in vitro. Antimicrob. Agents Chemother. 2009, 53, 4604. [Google Scholar] [CrossRef] [Green Version]
- Dean, D.; Rothschild, J.; Ruettger, A.; Kandel, R.P.; Sachse, K. Zoonotic Chlamydiaceae species associated with trachoma, Nepal. Emerg. Infect. Dis. 2013, 19, 1948. [Google Scholar] [CrossRef] [Green Version]
- De Puysseleyr, K.; de Puysseleyr, L.; Dhondt, H.; Geens, T.; Braeckman, L.; Morré, S.A.; Cox, E.; Vanrompay, D. Evaluation of the presence and zoonotic transmission of Chlamydia suis in a pig slaughterhouse. BMC Infect. Dis. 2014, 14, 560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Puysseleyr, L.; de Puysseleyr, K.; Braeckman, L.; Morré, S.A.; Cox, E.; Vanrompay, D. Assessment of Chlamydia suis infection in pig farmers. Transbound. Emerg. Dis. 2017, 64, 826–833. [Google Scholar] [CrossRef] [PubMed]
- Reinhold, P.; Liebler-Tenorio, E.; Sattler, S.; Sachse, K. Recurrence of Chlamydia suis infection in pigs after short-term antimicrobial treatment. Vet. J. 2011, 187, 405–407. [Google Scholar] [CrossRef] [PubMed]
- Hogan, R.J.; Mathews, S.A.; Mukhopadhyay, S.; Summersgill, J.T.; Timms, P. Chlamydial persistence: Beyond the biphasic paradigm. Infect. Immun. 2004, 72, 1843–1855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lienard, J.; Croxatto, A.; Aeby, S.; Jaton, K.; Posfay-Barbe, K.; Gervaix, A.; Greub, G. Development of a new Chlamydiales-specific real-time PCR and its application to respiratory clinical samples. J. Clin. Microbiol. 2011, 49, 2637–2642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blumer, S.; Greub, G.; Waldvogel, A.; Hässig, M.; Thoma, R.; Tschuor, A.; Pospischil, A.; Borel, N. Waddlia, Parachlamydia and Chlamydiaceae in bovine abortion. Vet. Microbiol. 2011, 152, 385–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Jelocnik, M.; Yang, F.; Gong, J.; Kaltenboeck, B.; Polkinghorne, A.; Feng, Z.; Pannekoek, Y.; Borel, N.; Song, C.; et al. Asymptomatic infections with highly polymorphic Chlamydia suis are ubiquitous in pigs. BMC Vet. Res. 2017, 13, 370. [Google Scholar] [CrossRef] [Green Version]
- Borel, N.; Kempf, E.; Hotzel, H.; Schubert, E.; Torgerson, P.; Slickers, P.; Ehricht, R.; Tasara, T.; Pospischil, A.; Sachse, K. Direct identification of Chlamydiae from clinical samples using a DNA microarray assay: A validation study. Mol. Cell. Probes 2008, 22, 55–64. [Google Scholar] [CrossRef]
- Schnee, C.; Sachse, K. DNA microarray-based detection of multiple pathogens: Mycoplasma spp. and Chlamydia spp. Methods Mol. Biol. 2015, 1247, 193–208. [Google Scholar] [CrossRef]
- Schiller, I.; Schifferli, A.; Gysling, P.; Pospischil, A. Growth characteristics of porcine chlamydial strains in different cell culture systems and comparison with ovine and avian chlamydial strains. Vet. J. 2004, 168, 74–80. [Google Scholar] [CrossRef]
- Hartley, J.C.; Kaye, S.; Stevenson, S.; Bennett, J.; Ridgway, G. PCR detection and molecular identification of Chlamydiaceae Species. J. Clin. Microbiol. 2001, 39, 3072–3079. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sachse, K.; Grossmann, E.; Jäger, C.; Diller, R.; Hotzel, H. Detection of Chlamydia suis from clinical specimens: Comparison of PCR, antigen ELISA, and culture. J. Microbiol. Methods 2003, 54, 233–238. [Google Scholar] [CrossRef]
- Busch, M.; Thoma, R.; Schiller, I.; Corboz, L.; Pospischil, A. Occurrence of Chlamydiae in the genital tracts of sows at slaughter and their possible significance for reproductive failure. J. Vet. Med. Ser. B 2000, 47, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Donati, M.; Balboni, A.; Laroucau, K.; Aaziz, R.; Vorimore, F.; Borel, N.; Morandi, F.; Nepita, E.V.; Di Francesco, A. Tetracycline susceptibility in Chlamydia suis pig isolates. PLoS ONE 2016, 11, e0149914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wanninger, S.; Donati, M.; Di Francesco, A.; Hässig, M.; Hoffmann, K.; Seth-Smith, H.M.B.; Marti, H.; Borel, N. Selective pressure promotes tetracycline resistance of Chlamydia suis in fattening pigs. PLoS ONE 2016, 11, e0166917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Di Francesco, A.; Donati, M.; Rossi, M.; Pignanelli, S.; Shurdhi, A.; Baldelli, R.; Cevenini, R. Tetracycline-Resistant Chlamydia suis Isolates in Italy. Vet. Rec. 2008, 163, 251–252. [Google Scholar] [CrossRef]
- Chahota, R.; Ogawa, H.; Ohya, K.; Yamaguchi, T.; Everett, K.D.E.; Fukushi, H. Involvement of multiple Chlamydia suis genotypes in porcine conjunctivitis. Transbound. Emerg. Dis. 2018, 65, 272–277. [Google Scholar] [CrossRef]
- Seth-Smith, H.M.B.; Wanninger, S.; Bachmann, N.; Marti, H.; Qi, W.; Donati, M.; Di Francesco, A.; Polkinghorne, A.; Borel, N. The Chlamydia suis genome exhibits high levels of diversity, plasticity, and mobile antibiotic resistance: Comparative genomics of a recent livestock cohort shows influence of treatment regimes. Genome Biol. Evol. 2017, 9, 750–760. [Google Scholar] [CrossRef]
- Joseph, S.J.; Marti, H.; Didelot, X.; Read, T.D.; Dean, D. Tetracycline selective pressure and homologous recombination shape the evolution of Chlamydia suis: A recently identified zoonotic pathogen. Genome Biol. Evol. 2016, 8, 2613–2623. [Google Scholar] [CrossRef] [Green Version]
- Peisker, M.; Berens, C.; Schnee, C. Understanding tetracycline resistance in Chlamydia suis. In Proceedings of the 5th European Meeting on Animal Chlamydiosis and Zoonotic Implications (EMAC 5), Odessa, Ukraine, 3–5 October 2018; p. 23. [Google Scholar]
- Bryskier, A. Tetracyclines. In Antimicrobial Agents: Antibacterials and Antifungals; Bryskier, A., Ed.; ASM Press: Washington, DC, USA, 2005; pp. 642–651. ISBN 1555812376. [Google Scholar]
- Di Francesco, A.; Baldelli, R.; Cevenini, R.; Magnino, S.; Pignanelli, S.; Salvatore, D.; Galuppi, R.; Donati, M. Seroprevalence to Chlamydiae in Pigs in Italy. Vet. Res. 2006, 42, 29–38. [Google Scholar]
- Borel, N.; Regenscheit, N.; Di Francesco, A.; Donati, M.; Markov, J.; Masserey, Y.; Pospischil, A. Selection for tetracycline-resistant Chlamydia suis in treated pigs. Vet. Microbiol. 2012, 156, 143–146. [Google Scholar] [CrossRef] [Green Version]
- Schautteet, K.; de Clercq, E.; Miry, C.; van Groenweghe, F.; Delava, P.; Kalmar, I.; Vanrompay, D. Tetracycline-resistant Chlamydia suis in cases of reproductive failure on Belgian, Cypriote and Israeli pig production farms. J. Med. Microbiol. 2013, 62, 331–334. [Google Scholar] [CrossRef] [Green Version]
- Borel, N.; Leonard, C.; Slade, J.; Schoborg, R.V. Chlamydial antibiotic resistance and treatment failure in veterinary and human medicine. Curr. Clin. Microbiol. Rep. 2016, 3, 10–18. [Google Scholar] [CrossRef] [Green Version]
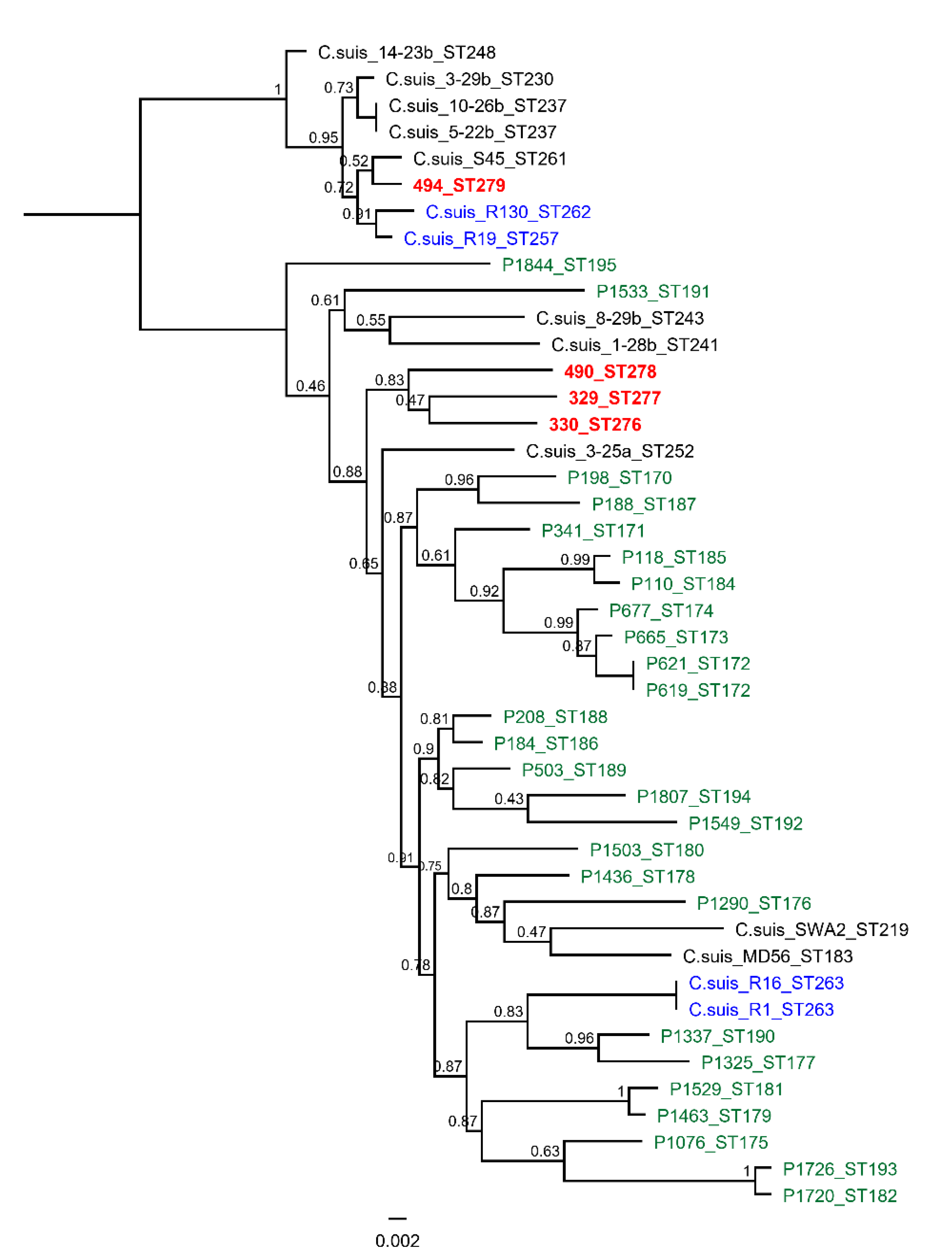

| Isolate 1 | Isolate 2 | Isolate 3 | Isolate 4 | |
|---|---|---|---|---|
| fattening ID | 494 MS | 490 MS | 329 MS | 330 MS |
| sequence type | ST279 | ST278 | ST277 | ST276 |
| phylogenetic clade | 1. clade | 2. clade | 2. clade | 2. clade |
| (a) | ||||
| MIC (µg/mL) | 329 MS | 330 MS | 490 MS | 494 MS |
| Initial phenotype | 2 to 4 | 0.125 | 0.125 | 4 |
| MIC (Suchland) | 4 | 0.06 | 0.125 | 4 |
| MIC (Donati) | 4 | 0.06 | 0.125 | 4 |
| MIC (consensus) | 4 | 0.06–0.125 | 0.125 | 4 |
| Interpretation | resistant | sensitive | sensitive | Resistant |
| (b) | ||||
| MIC (µg/mL) | 329 MS | 330 MS | 490 MS | 494 MS |
| Initial phenotype | 0.25 | 0.03–0.06 | 0.06 | 0.125 |
| MIC (Suchland) | 0.5 | 0.06 | 0.06 | 0.25 |
| MIC (Donati) | 0.25 | 0.06 | 0.06–0.125 | 0.25 |
| MIC (consensus) | 0.25–0.5 | 0.06 | 0.06 | 0.125–0.25 |
| Interpretation | Reduced susceptibility | sensitive | sensitive | Reduced susceptibility |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unterweger, C.; Schwarz, L.; Jelocnik, M.; Borel, N.; Brunthaler, R.; Inic-Kanada, A.; Marti, H. Isolation of Tetracycline-Resistant Chlamydia suis from a Pig Herd Affected by Reproductive Disorders and Conjunctivitis. Antibiotics 2020, 9, 187. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040187
Unterweger C, Schwarz L, Jelocnik M, Borel N, Brunthaler R, Inic-Kanada A, Marti H. Isolation of Tetracycline-Resistant Chlamydia suis from a Pig Herd Affected by Reproductive Disorders and Conjunctivitis. Antibiotics. 2020; 9(4):187. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040187
Chicago/Turabian StyleUnterweger, Christine, Lukas Schwarz, Martina Jelocnik, Nicole Borel, René Brunthaler, Aleksandra Inic-Kanada, and Hanna Marti. 2020. "Isolation of Tetracycline-Resistant Chlamydia suis from a Pig Herd Affected by Reproductive Disorders and Conjunctivitis" Antibiotics 9, no. 4: 187. https://0-doi-org.brum.beds.ac.uk/10.3390/antibiotics9040187







