Role of Diffusion Tensor Imaging in Prognostication and Treatment Monitoring in Niemann-Pick Disease Type C1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Image Acquisition and Analysis
2.3. Severity Score, DTI and Volume Measurements
2.4. Statistical Analyses
3. Results
3.1. Sample Demographics
3.2. Cerebellar Fractional Anisotropy (FA)
3.3. Cerebellar Volume
3.4. Cerebellar Mean Diffusivity (MD)
4. Discussion
Study Limitations
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| NPC | Niemann-Pick Disease, type C |
| NPC1 | Niemann-Pick Disease, type C1 |
| NPC2 | Niemann-Pick Disease, type C2 |
| DTI | diffusion tensor imaging |
| FA | fractional anisotropy |
| MD | mean diffusivity |
| NNSS | NIH NPC severity score |
| MRI | magnetic resonance imaging |
| MPRAGE | spatially matched axial magnetization-prepared rapid-gradient echo |
| T2WI | T2-weighted imaging |
| LDDMM | Large deformation diffeomorphic metric mapping |
| ANCOVA | Analysis of covariance |
References
- Kemp, W.L.; Burns, D.K.; Travis, G.; Brown, T.G. Chapter 6: Genetic Disorders—Lysosomal Storage Disorders. In Pathology: The Big Picture; McGraw-Hill Education: New York, NY, USA, 2008. [Google Scholar]
- Vanier, M.T.; Latour, P. Laboratory diagnosis of Niemann-Pick disease type C: The filipin staining test. Methods Cell Biol. 2015, 126, 357–375. [Google Scholar] [PubMed]
- Wassif, C.A.; Cross, J.L.; Iben, J.; Sanchez-Pulido, L.; Cougnoux, A.; Platt, F.M.; Ory, D.S.; Ponting, C.P.; Bailey-Wilson, J.E.; Biesecker, L.G.; et al. High incidence of unrecognized visceral/neurological late-onset Niemann-Pick disease, type C1, predicted by analysis of massively parallel sequencing data sets. Genet. Med. 2015, 18, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Patterson, M.C. Overview of Niemann-Pick Disease. Available online: http://www.uptodate.com/contents/overview-of-niemann-pick-disease (accessed on 1 July 2014).
- Zimran, A.; Elstein, D. Lipid Storage Diseases. In Williams Hematology, 8th ed.; McGraw-Hill Education: New York, NY, USA, 2010. [Google Scholar]
- Cluzeau, C.V.; Watkins-Chow, D.E.; Fu, R.; Borate, B.; Yanjanin, N.; Dail, M.K.; Davidson, C.D.; Walkley, S.U.; Ory, D.S.; Wassif, C.A.; et al. Microarray expression analysis and identificantion of serum biomarkers for Niemann-Pick disease type C1. Hum. Mol. Gen. 2012, 202, 3632–3646. [Google Scholar] [CrossRef] [PubMed]
- Porter, F.D.; Scherrer, D.E.; Lanier, M.H.; Langmade, S.J.; Molugu, V.; Gale, S.E.; Olzeski, D.; Sidhu, R.; Dietzen, D.J.; Fu, R.; et al. Cholesterol oxidation products are sensitive and specific blood-based biomarkers for Niemann-Pick C1 disease. Sci. Transl. Med. 2010, 2, 56–81. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Apkarian, K.; Jung, E.S.; Yanjanin, N.; Yoshida, S.; Mori, S.; Park, J.; Gropman, A.; Baker, E.H.; Porter, F.D. Corpus Callosum Diffusion Tensor Imaging and Volume Measures are Associated with Disease Severity in Pediatric Niemann-Pick Disease Type C1. Pediatr. Neurol. 2014, 51, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Walterfang, M.; Abel, L.A.; Desmond, P.; Fahey, M.C.; Bowman, E.A.; Velakoulis, D. Cerebellar volume correlates with saccadic gain and ataxia in adult Niemann-Pick type C. Mol. Gen. Metab. 2013, 108, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Walterfang, M.; Fahey, M.; Desmond, P.; Wood, A.; Seal, M.L.; Steward, C.; Adamson, C.; Kokkinos, C.; Fietz, M.; Velakoulis, D. White and gray matter alterations in adults with Niemann-Pick disease type CA cross-sectional study. Neurology 2010, 75, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Walterfang, M.; Fahey, M.; Abel, L.; Fietz, M.; Wood, A.; Bowman, E.; Reutens, D.; Velakoulis, D. Size and shape of the corpus callosum in adult Niemann-Pick type C reflects state and trait illness variables. Am. J. Neuroradiol. 2011, 32, 1340–1346. [Google Scholar] [CrossRef] [PubMed]
- Trouard, T.P.; Heidenreich, R.A.; Seeger, J.F.; Erickson, R.P. Diffusion tensor imaging in Niemann-Pick Type C disease. Pediatr. Neurol. 2005, 33, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Landman, B.A.; Farrell, J.A.; Jones, C.K.; Smith, S.A.; Prince, J.L.; Mori, S. Effects of diffusion weighting schemes on the reproducibility of DTI-derived fractional anisotropy, mean diffusivity, and principal eigenvector measurements at 1.5T. Neuroimaging 2007, 36, 1123–1138. [Google Scholar] [CrossRef] [PubMed]
- Woods, R.P.; Grafton, S.T.; Holmes, C.J.; Cherry, S.R.; Mazziotta, J.C. Automated image registration: I. General methods and intrasubject, intramodality validation. J. Comput. Assist. Tomogr. 1998, 22, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Ceritoglu, C.; Li, X.; Qiu, A.; Miller, M.I.; van Zijl, P.C.; Mori, S. Correction of B0 susceptibility induced distortion in diffusion-weighted images using large deformation diffeomorphic metric mapping. Magn. Reson. Imaging 2008, 26, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.I.; Beg, M.F.; Ceritoglu, C.; Stark, C. Increasing the power of functional maps of the medial temporal lobe by using large deformation diffeomorphic metric mapping. Proc. Natl. Acad. Sci. USA 2005, 102, 9685–9690. [Google Scholar] [CrossRef] [PubMed]
- Beg, M.F.; Miller, M.I.; Trouvé, A.; Younes, L. Computing large deformation metric mappings via geodesic flows of diffeomorphisms. Int. J. Comput. Vis. 2005, 61, 139–157. [Google Scholar] [CrossRef]
- Oishi, K.; Faria, A.; Jiang, H.; Li, X.; Akhter, K.; Zhang, J.; Hsu, J.T.; Miller, M.I.; van Zijl, P.C.; Albert, M.; et al. Atlas-based whole brain white matter analysis using large deformation diffeomorphic metric mapping: Application to normal elderly and Alzheimer’s disease participants. Neuroimage 2009, 46, 485–499. [Google Scholar] [CrossRef]
- Faria, A.V.; Zhang, J.; Oishi, K.; Li, X.; Jiang, H.; Akhter, K.; Hermoye, L.; Lee, S.K.; Hoon, A.; Stashinko, E.; et al. Atlas-based analysis of neurodevelopment from infancy to adulthood using diffusion tensor imaging and applications for automated abnormality detection. Neuroimage 2010, 52, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.V.; Hoon, A.; Stashinko, E.; Li, X.; Jiang, H.; Mashayekh, A.; Akhter, K.; Hsu, J.; Oishi, K.; Zhang, J.; et al. Quantitative analysis of brain pathology based on MRI and brain atlases—Applications for cerebral palsy. Neuroimage 2011, 54, 1854–1861. [Google Scholar] [CrossRef] [PubMed]
- Yanjanin, N.M.; Vélez, J.I.; Gropman, A.; King, K.; Bianconi, S.E.; Conley, S.K.; Brewer, C.C.; Solomon, B.; Pavan, W.J.; Arcos-Burgos, M.; et al. Linear clinical progression, independent of age of onset, in Niemann-Pick disease. Am. J. Med. Genet. 2010, 153, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.E.; LeBlanc, V.G.; Mailman, T.M.; Fice, D.; Burton, I.; Karakach, T.K.; Karten, B. Pre-symptomatic activation of antioxidant responses and alterations inglucose and pyruvate metabolism in Niemann-Pick Type C1-deficient murine brain. PloS ONE 2013, 8, e82685. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.; Maldonado, C.; Vargas, L.M.; Gonzalez, M.; Robledo, F.; de Arce, K.P.; Muñoz, F.J.; Hetz, C.; Alvarez, A.R.; Zanlungo, S. Oxidative stress activates the c-Abl/p73 proapoptotic pathway in Niemann-Pick type C neurons. Neurobiol. Dis. 2011, 41, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Ulatowski, L.; Parker, R.; Warrier, G.; Sultana, R.; Butterfield, D.A.; Manor, D. Vitamin E is essential for Purkinje neuron integrity. Neuroscience 2014, 260, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Byun, K.; Kim, D.; Bayarsaikhan, E.; Oh, J.; Kim, J.; Kwak, G.; Jeong, G.B.; Jo, S.M.; Lee, B. Changes of calcium binding proteins, c-Fos, and COX in hippocampal formation and cerebellum of Niemann-Pick type C mouse. J. Chem. Neuroanat. 2013, 52, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.L.; Su, L.D.; Li, Q.; Wang, X.X.; Shen, Y. Cerebellar long-term depression is deficient in Niemann-Pick type C disease mice. Cerebellum 2011, 10, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Elrick, M.J.; Pacheco, C.D.; Yu, T.; Dadgar, N.; Shakkottai, V.G.; Ware, C.; Paulson, H.L.; Lieberman, A.P. Conditional Niemann-Pick C mice demonstrate cell autonomous Purkinje cell neurodegeneration. Hum. Mol. Gen. 2010, 19, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Zaaraoui, W.; Crespy, L.; Rico, A.; Faivre, A.; Soulier, E.; Confort-Gouny, S.; Cozzone, P.J.; Pelletier, J.; Ranjeva, J.P.; Kaphan, E.; et al. In vivo quantification of brain injury in adult Niemann-Pick Disease Type C. Mol. Gen. Metab. 2011, 103, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Mancall, E. Cerebellum. In Gray’s Clinical Neuroanatomy: The Anatomic Basis for Clinical Neuroscience; Saunders: Philadelphia, PA, USA, 2011. [Google Scholar]
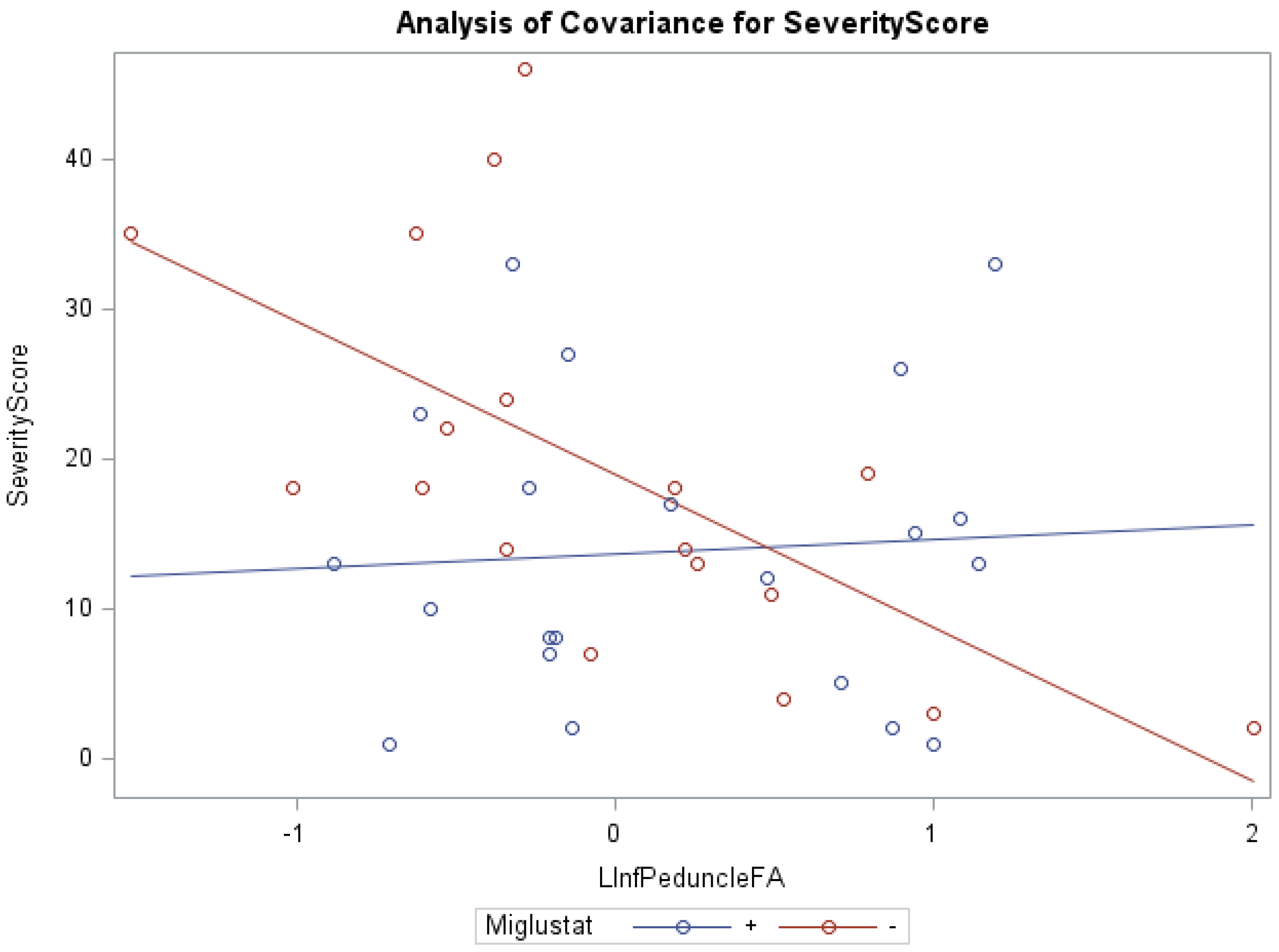
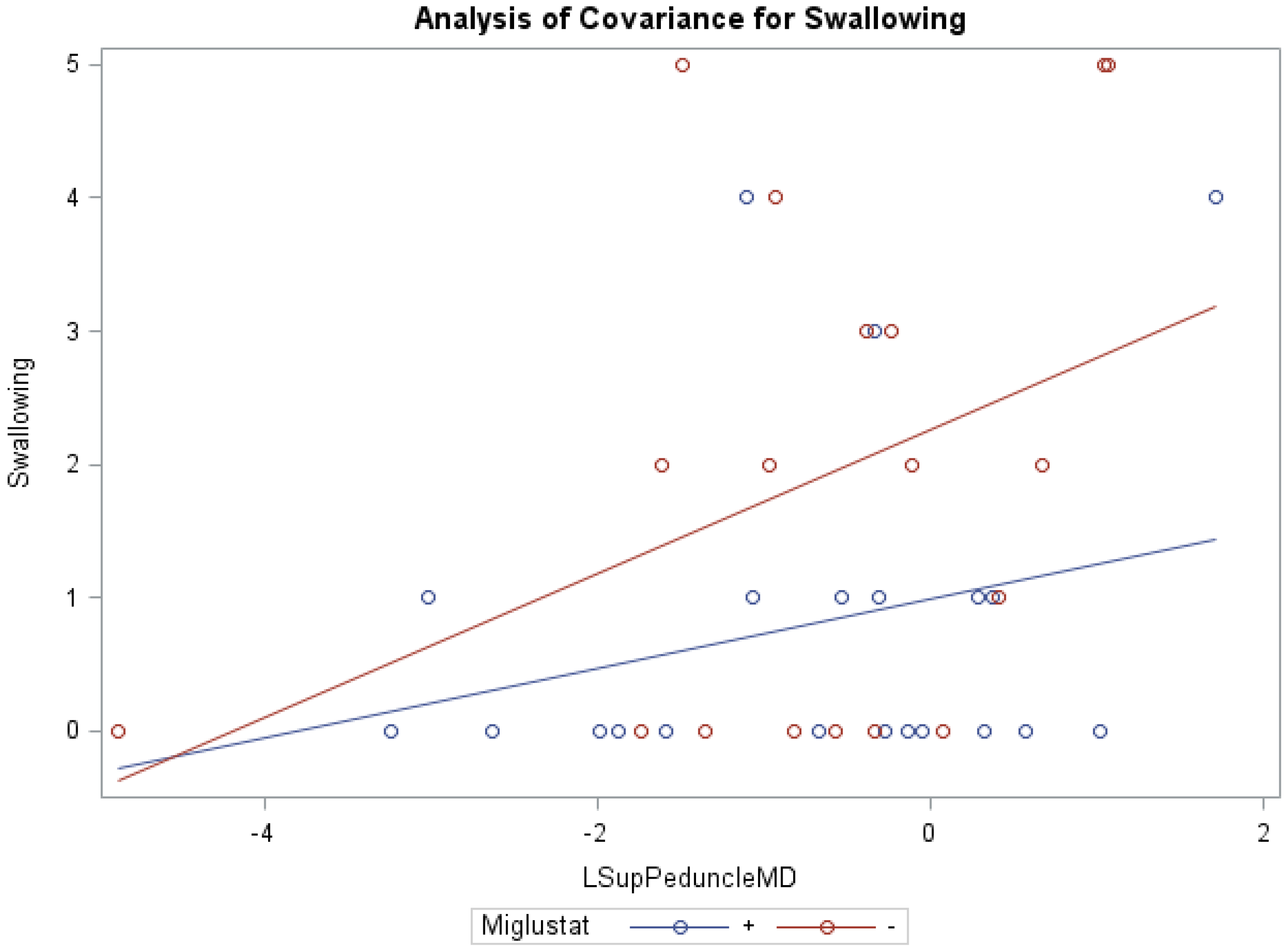
| Acquisition Parameter | T2WI | DTI |
|---|---|---|
| Repetition Time (TR) | 5400 ms | 6401 ms |
| Echo Time (TE) | 100 ms | 60 ms |
| Field of View (FOV) | 220 × 165 mm | 224 × 224 mm |
| Acquisition Matrix | 384 × 227 | 112 × 112 |
| Reconstruction Matrix | 512 × 512 | 128 × 128 |
| In-Plane Resolution | 0.43 × 0.43 mm/pixel | 1.75 × 1.75 mm/pixel |
| Acquisition Duration | 2:25 | 2:27 |
| Slice Thickness | 5 mm | 2 mm |
| Number of Axial Slices | 28 | 70 |
| Scan Parameters | Axial MPRGE < 2010 | Axial MPRGE > 2010 (Upgrade) |
|---|---|---|
| Repetition Time (TR) | 8.2 ms | 11.4 ms |
| Echo Time (TE) | 3.8 ms | 6.5 ms |
| FA | 8° | 6° |
| Field of View (FOV) | 256 mm | 220 mm |
| Acquisition Matrix | 256 × 256 | 256 × 131 |
| Reconstruction Matrix | 256 × 256 | 256 × 256 |
| Slice Thickness | 1.0 mm | 1.0 mm |
| Number of Excitations | 1 | 2 |
| Subject # | Gender | Age (years) | Total NNSS | Presenting Neurologic Symptom | Age at First Neurologic Symptom (years) | Duration of Neurologic Symptoms (years) | Miglustat Status |
|---|---|---|---|---|---|---|---|
| 1 | F | 21.2 | 35 | VGSP | 9 | 12.2 | − |
| 2 | M | 7.7 | 5 | Fine Motor Limitation | 5 | 2.7 | + |
| 3 | F | 13.1 | 33 | Clumsy, possible VGSP | 2 | 11.1 | + |
| 4 | M | 5 | 11 | Clumsy, Dysarthria | 3 | 2.0 | − |
| 5 | M | 10 | 14 | Fine Motor Limitation | 3 | 7.0 | − |
| 6 | M | 16.3 | 18 | Dysarthria | 3 | 13.3 | − |
| 7 | F | 11.8 | 22 | Clumsy | 1.5 | 10.3 | − |
| 8 | F | 4 | 26 | Clumsy, VGSP | 2 | 2.0 | + |
| 9 | M | 7.7 | 7 | Abnormal Gait | 2 | 5.7 | + |
| 10 | M | 11.8 | 18 | Fine Motor, Coordination Deficit | 6 | 5.8 | + |
| 11 | M | 5.4 | 8 | Clumsy, Speech Delay | 2 | 3.4 | + |
| 12 | F | 5.2 | 13 | Clumsy | 1 | 4.2 | + |
| 13 | M | 4.7 | 2 | None Reported | N/A | N/A | + |
| 14 | M | 6.1 | 7 | Clumsy | 2 | 4.1 | − |
| 15 | F | 3.8 | 3 | Clumsy | 2 | 1.7 | − |
| 16 | F | 21.5 | 35 | VGSP | 5 | 16.5 | − |
| 17 | F | 6.7 | 40 | Abnormal gait, fine motor limitation | 3 | 3.7 | − |
| 18 | F | 15.7 | 1 | None Reported | N/A | N/A | + |
| 19 | F | 6.8 | 27 | Gross motor delay | 1.5 | 5.3 | + |
| 20 | F | 12.6 | 24 | VGSP | 5 | 7.6 | − |
| 21 | F | 16.8 | 23 | Learning difficulty | 8 | 8.8 | + |
| 22 | M | 8.4 | 46 | Clumsy | 1.5 | 6.9 | − |
| 23 | M | 1 | 1 | None Reported | N/A | N/A | + |
| 24 | M | 1.6 | 4 | None Reported | N/A | N/A | − |
| 25 | M | 17.2 | 10 | Abnormal Gait | 12 | 5.2 | + |
| 26 | F | 8.1 | 8 | Tremor, fine motor limitation | 7 | 1.1 | + |
| 27 | M | 20.3 | 14 | VGSP, slurred speech | 13 | 7.3 | − |
| 28 | M | 6.6 | 13 | VGSP | 3.5 | 3.1 | − |
| 29 | M | 4 | 2 | None Reported | N/A | N/A | − |
| 30 | F | 12.7 | 17 | Learning disability | 8 | 4.7 | + |
| 31 | F | 21 | 18 | School problems | 5 | 16.0 | − |
| 32 | F | 15.6 | 12 | Gross motor delay | 1.2 | 14.4 | + |
| 33 | M | 17.2 | 19 | Learning disability | 6 | 11.2 | − |
| 34 | F | 21.9 | 18 | Learning disability | 11 | 10.9 | − |
| 35 | F | 17.3 | 15 | Clumsy | 7 | 10.2 | + |
| 36 | F | 13.3 | 13 | Learning disability | 7 | 6.3 | + |
| 37 | M | 15.2 | 33 | Seizures | 6 | 9.2 | + |
| 38 | F | 11.8 | 16 | VGSP | 3 | 8.7 | + |
| 39 | M | 3.8 | 2 | Gross motor delay | 2 | 1.7 | + |
| Cerebellar Measure | FA | Volume | MD | |||
|---|---|---|---|---|---|---|
| r | p | r | p | r | p | |
| R inferior peduncle | −0.38 | 0.02 | −0.35 | 0.03 | ||
| L inferior peduncle | −0.35 | 0.03 | ||||
| R superior peduncle | −0.35 | 0.03 | −0.46 | 0.003 | ||
| L superior peduncle | −0.41 | 0.01 | −0.49 | 0.001 | ||
| R middle peduncle | 0.35 | 0.03 | ||||
| R white matter | −0.33 | 0.04 | 0.33 | 0.04 | ||
| R cerebellum | −0.42 | 0.01 | 0.37 | 0.02 | ||
| L cerebellum | −0.35 | 0.03 | 0.35 | 0.03 | ||
| Whole cerebellum | −0.40 | 0.01 | 0.37 | 0.02 | ||
| Parameter | t-Value | p< | Model f | Model p< | R2 | |
|---|---|---|---|---|---|---|
| FA | ||||||
| R inferior peduncle | −78.01 | −2.14 | 0.05 | 6.01 | 0.01 | 0.25 |
| Age at scan | 1.16 | 3.47 | 0.01 | |||
| R white matter | −202.26 | −2.48 | 0.05 | 6.94 | 0.01 | 0.28 |
| Age at scan | 1.16 | 3.67 | 0.001 | |||
| L white matter | −214.00 | −2.62 | 0.05 | 7.38 | 0.01 | 0.29 |
| Age at scan | 1.16 | 3.76 | 0.001 | |||
| Volume | ||||||
| R inferior peduncle | −0.03 | −2.48 | 0.05 | 6.95 | 0.01 | 0.28 |
| Age at scan | 1.02 | 3.52 | 0.01 | |||
| L inferior peduncle | −0.03 | −2.32 | 0.05 | 6.48 | 0.01 | 0.26 |
| Age at scan | 0.94 | 3.33 | 0.01 | |||
| R superior peduncle | −0.03 | −3.12 | 0.01 | 9.08 | 0.001 | 0.34 |
| Age at scan | 0.88 | 3.38 | 0.01 | |||
| L superior peduncle | −0.03 | −3.07 | 0.01 | 8.87 | 0.001 | 0.33 |
| Age at scan | 0.85 | 3.27 | 0.01 | |||
| R middle peduncle | −0.01 | −2.29 | 0.05 | 6.42 | 0.01 | 0.26 |
| Age at scan | 1.07 | 3.50 | 0.01 | |||
| L middle peduncle | −0.003 | −2.42 | 0.05 | 6.78 | 0.01 | 0.27 |
| Age at scan | 1.05 | 3.54 | 0.01 | |||
| R white matter | −0.003 | −2.66 | 0.05 | 7.50 | 0.01 | 0.29 |
| Age at scan | 0.94 | 3.43 | 0.01 | |||
| L white matter | −0.003 | −3.21 | 0.01 | 9.42 | 0.001 | 0.34 |
| Age at scan | 1.01 | 3.78 | 0.01 | |||
| MD | ||||||
| R middle peduncle | 16112 | 2.25 | 0.05 | 6.31 | 0.01 | 0.26 |
| Age at scan | 0.93 | 3.29 | 0.01 | |||
| L middle peduncle | 16918 | 2.40 | 0.05 | 6.71 | 0.01 | 0.27 |
| Age at scan | 0.95 | 3.37 | 0.01 | |||
| R white matter | 12378 | 2.18 | 0.05 | 6.12 | 0.01 | 0.25 |
| Age at scan | 0.89 | 3.20 | 0.01 | |||
| R cerebellum | 5248.47 | 2.10 | 0.05 | 5.92 | 0.01 | 0.25 |
| Age at scan | 0.58 | 2.03 | 0.05 | |||
| L cerebellum | 5035.14 | 2.08 | 0.05 | 5.86 | 0.01 | 0.25 |
| Age at scan | 0.60 | 2.12 | 0.05 | |||
| Whole cerebellum | 5182.60 | 2.10 | 0.05 | 5.92 | 0.01 | 0.25 |
| Age at scan | 0.58 | 2.07 | 0.05 |
| Type I NNSS | f-value | p< | Type III NNSS | f -value | p< | Model f value | Model p< | Model R2 | |
|---|---|---|---|---|---|---|---|---|---|
| FA | |||||||||
| Miglustat | 266.74 | 2.58 | 0.12 | 197.29 | 1.91 | 0.18 | 4.63 | 0.01 | 0.28 |
| R inferior peduncle | 600.57 | 5.82 | 0.05 | 503.01 | 4.87 | 0.05 | |||
| Age at scan | 565.80 | 5.48 | 0.05 | ||||||
| Miglustat | 266.74 | 2.59 | 0.12 | 263.45 | 2.56 | 0.12 | 4.66 | 0.01 | 0.29 |
| L inferior peduncle | 530.46 | 5.15 | 0.05 | 442.57 | 4.30 | 0.05 | |||
| Age at scan | 642.40 | 6.24 | 0.05 | ||||||
| Miglustat | 266.74 | 2.37 | 0.14 | 122.76 | 1.09 | 0.31 | 3.24 | 0.05 | 0.22 |
| L superior peduncle | 700.03 | 6.21 | 0.05 | 724.33 | 6.42 | 0.05 | |||
| Age at scan | 130.67 | 1.16 | 0.29 | ||||||
| Volume | |||||||||
| Miglustat | 266.74 | 2.73 | 0.11 | 165.04 | 1.69 | 0.21 | 5.57 | 0.01 | 0.32 |
| R superior peduncle | 1299.66 | 13.32 | 0.001 | 1363.60 | 13.98 | 0.00 | |||
| Age at scan | 63.98 | 0.66 | 0.43 | 1 | |||||
| Miglustat | 266.74 | 2.48 | 0.13 | 85.78 | 0.80 | 0.38 | 3.97 | 0.05 | 0.25 |
| L superior peduncle | 1011.34 | 9.40 | 0.01 | 846.23 | 7.87 | 0.01 | |||
| Age at scan | 2.12 | 0.02 | 0.89 | ||||||
| MD | |||||||||
| Miglustat | 266.74 | 2.51 | 0.13 | 463.12 | 4.35 | 0.05 | 4.14 | 0.05 | 0.26 |
| R middle peduncle | 719.61 | 6.76 | 0.05 | 1001.38 | 9.41 | 0.01 | |||
| Age at scan | 334.83 | 3.15 | 0.09 | ||||||
| Miglustat | 266.74 | 2.44 | 0.13 | 345.29 | 3.16 | 0.09 | 3.73 | 0.05 | 0.24 |
| L middle peduncle | 715.91 | 6.55 | 0.05 | 920.75 | 8.43 | 0.01 | |||
| Age at scan | 238.39 | 2.18 | 0.15 | ||||||
| Miglustat | 266.74 | 2.33 | 0.14 | 238.23 | 2.09 | 0.16 | 3.05 | 0.05 | 0.21 |
| R white matter | 608.32 | 5.32 | 0.05 | 729.96 | 6.39 | 0.05 | |||
| Age at scan | 171.50 | 1.50 | 0.23 | ||||||
| Miglustat | 266.74 | 2.31 | 0.14 | 175.21 | 1.52 | 0.23 | 2.90 | 0.05 | 0.20 |
| R cerebellum | 711.98 | 6.17 | 0.05 | 718.60 | 6.23 | 0.05 | |||
| Age at scan | 26.07 | 0.23 | 0.64 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lau, M.W.; Lee, R.W.; Miyamoto, R.; Jung, E.S.; Yanjanin Farhat, N.; Yoshida, S.; Mori, S.; Gropman, A.; Baker, E.H.; Porter, F.D. Role of Diffusion Tensor Imaging in Prognostication and Treatment Monitoring in Niemann-Pick Disease Type C1. Diseases 2016, 4, 29. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases4030029
Lau MW, Lee RW, Miyamoto R, Jung ES, Yanjanin Farhat N, Yoshida S, Mori S, Gropman A, Baker EH, Porter FD. Role of Diffusion Tensor Imaging in Prognostication and Treatment Monitoring in Niemann-Pick Disease Type C1. Diseases. 2016; 4(3):29. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases4030029
Chicago/Turabian StyleLau, Meghann W., Ryan W. Lee, Robin Miyamoto, Eun Sol Jung, Nicole Yanjanin Farhat, Shoko Yoshida, Susumu Mori, Andrea Gropman, Eva H. Baker, and Forbes D. Porter. 2016. "Role of Diffusion Tensor Imaging in Prognostication and Treatment Monitoring in Niemann-Pick Disease Type C1" Diseases 4, no. 3: 29. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases4030029






