Empowering Melatonin Therapeutics with Drosophila Models
Abstract
:1. Introduction
2. Melatonin Receptors
2.1. Melatonin Receptor Functions
2.2. Molecular Mechanisms of Melatonin-Receptor Signaling
3. Receptor-Independent Melatonin Functions
3.1. Melatonin and Longevity
Oxidative Damage
3.2. Drosophila in Longevity Studies
4. Melatonin and Neurological Disease
4.1. Alzheimer’s Disease
4.2. Huntington’s Disease
4.3. Parkinson’s Disease
5. Melatonin Life Extension in Drosophila Models of Aging and Disease
5.1. Melatonin Treatments in Drosophila Models of Neurological Disease
5.1.1. Huntington’s Disease
5.1.2. Parkinson’s Disease
5.1.3. Alzheimer’s Disease
5.2. Melatonin Treatment in a Novel Drosophila Model of Polycystic Kidney Disease
6. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chowdhury, I.; Sengupta, A.; Maitra, S.K. Melatonin: Fifty years of scientific journey from the discovery in bovine pineal gland to delineation of functions in human. Ind. J. Biochem. Biophys. 2008, 45, 289–304. [Google Scholar]
- Zhao, D.; Yu, Y.; Shen, Y.; Liu, Q.; Zhao, Z.; Sharma, R.; Reiter, R.J. Melatonin synthesis and function: Evolutionary history in animals and plants. Front. Endocrinol. 2019, 10, 249. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.B.; Case, J.D.; Takahashi, Y.; Lee, T.H.; Mori, W. Isolation of melatonin, the pineal gland factor that lightens melanocytes. J. Am. Chem. Soc. 1958, 80, 2587. [Google Scholar] [CrossRef]
- Dubbels, R.; Reiter, R.J.; Klenke, E.; Goebel, A.; Schnakenberg, E.; Ehlers, C.; Schiwara, H.W.; Schloot, W. Melatonin in edible plants identified by radioimmunoassay and by high performance liquid chromatography-mass spectrometry. J. Pineal Res. 1995, 18, 28–31. [Google Scholar] [CrossRef]
- Hattori, A.; Migitaka, H.; Iigo, M.; Itoh, M.; Yamamoto, K.; Ohtani-Kaneko, R.; Hara, M.; Suzuki, T.; Reiter, R.J. Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates. Biochem. Mol. Biol. Int. 1995, 35, 627–634. [Google Scholar] [PubMed]
- Manchester, L.C.; Tan, D.X.; Reiter, R.J.; Qi, W.; Karbownik, M.; Calvo, J.R. High levels of melatonin in edible seeds: Possible function in germ cell protection. Life Sci. 2000, 67, 3023–3029. [Google Scholar] [CrossRef]
- Reiter, R.J. Melatonin: Clinical relevance. Best Pract. Res. Clin. Endocrinol. Metab. 2003, 17, 273–285. [Google Scholar] [CrossRef]
- Masters, A.; Pandi-Perumal, S.R.; Seixas, A.; Girardin, J.L.; McFarlane, S.I. Melatonin, the hormone of darkness: From sleep promotion to Ebola treatment. Brain Disord. Ther. 2014, 4, 1000151. [Google Scholar] [CrossRef] [Green Version]
- Reiter, R.J.; Tan, D.X.; Galano, A. Melatonin: Exceeding expectations. Physiology 2014, 29, 325–333. [Google Scholar] [CrossRef] [Green Version]
- Shi, H.; Chen, K.; Wei, Y.; He, C. Fundamental issues of melatonin-mediated stress signaling in plants. Front. Plant Sci. 2016, 7, 1124. [Google Scholar] [CrossRef] [Green Version]
- Tan, D.X.; Hardeland, R.; Back, K.; Manchester, L.C.; Alatorre-Jimenez, M.A.; Reiter, R.J. On the significance of an alternate pathway of melatonin synthesis via 5-methoxytryptamine: Comparisons across species. J. Pineal Res. 2016, 61, 27–40. [Google Scholar] [CrossRef] [Green Version]
- Cecon, E.; Oishi, A.; Jockers, R. Melatonin receptors: Molecular pharmacology and signalling in the context of system bias. Br. J. Pharmacol. 2018, 175, 3263–3280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiter, R.J. Melatonin: The chemical expression of darkness. Mol. Cell. Endocrinol. 1991, 79, C153–C158. [Google Scholar] [CrossRef]
- Reiter, R.J. Pineal melatonin: Cell biology of its synthesis and of its physiological interactions. Endocr. Rev. 1991, 12, 151–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erren, T.C.; Reiter, R.J.; Piekarski, C. Chronodisruption and melatonin: The need for sensible exposure metrics in epidemiological studies. J. Pineal Res. 2008, 45, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R. Melatonin, hormone of darkness and more: Occurrence, control mechanisms, actions and bioactive metabolites. Cell Mol. Life Sci. 2008, 65, 2001–2018. [Google Scholar] [CrossRef] [Green Version]
- Jenwitheesuk, A.; Nopparat, C.; Mukda, S.; Wongchitrat, P.; Govitrapong, P. Melatonin regulates aging and neurodegeneration through energy metabolism, epigenetics, autophagy and circadian rhythm pathways. Int. J. Mol. Sci. 2014, 15, 16848–16884. [Google Scholar] [CrossRef] [Green Version]
- Venegas, C.; García, J.A.; Escames, G.; Ortiz, F.; López, A.; Doerrier, C.; García-Corzo, L.; López, L.C.; Reiter, R.J.; Acuña-Castroviejo, D. Extrapineal melatonin: Analysis of its subcellular distribution and daily fluctuations. J. Pineal Res. 2012, 52, 217–227. [Google Scholar] [CrossRef]
- Suofu, Y.; Li, W.; Jean-Alphonse, F.G.; Jia, J.; Khattar, N.K.; Li, J.; Baranov, S.V.; Leronni, D.; Mihalik, A.C.; He, Y.; et al. Dual role of mitochondria in producing melatonin and driving GPCR signaling to block cytochrome c release. Proc. Natl. Acad. Sci. USA 2017, 114, E7997–E8006. [Google Scholar] [CrossRef] [Green Version]
- Tan, D.X.; Reiter, R.J. Mitochondria: The birth place, battle ground and the site of melatonin metabolism in cells. Melatonin Res. 2019, 2, 1. [Google Scholar] [CrossRef]
- Macchi, M.M.; Bruce, J.N. Human pineal physiology and functional significance of melatonin. Front. Neuroendocrinol. 2004, 25, 177–195. [Google Scholar] [CrossRef]
- Dubocovich, M.L.; Markowska, M. Functional MT1 and MT2 melatonin receptors in mammals. Endocrine 2005, 27, 101–110. [Google Scholar] [CrossRef]
- Reiter, R.J.; Melchiorri, D.; Sewerynek, E.; Poeggeler, B.; Barlow-Walden, L.; Chuang, J.; Ortiz, G.G.; Acuña-Castroviejo, D. A review of the evidence supporting melatonin’s role as an antioxidant. J. Pineal Res. 1995, 18, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with reactive oxygen and nitrogen species? J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; Reiter, R.J.; Menéndez-Peláez, A.; Pablos, M.I.; Burgos, A. Characterization of high-affinity melatonin binding sites in purified cell nuclei of rat liver. J. Pineal Res. 1994, 16, 100–112. [Google Scholar] [CrossRef]
- Emet, M.; Ozcan, H.; Ozel, L.; Yayla, M.; Halici, Z.; Hacimuftuoglu, A. A review of melatonin, its receptors and drugs. Eurasian J. Med. 2016, 48, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Zheng, X.; Kong, J.; Manchester, L.C.; Hardeland, R.; Kim, S.J.; Xu, X.; Reiter, R.J. Fundamental issues related to the origin of melatonin and melatonin isomers during evolution: Relation to their biological functions. Int. J. Mol. Sci. 2014, 15, 15858–15890. [Google Scholar] [CrossRef] [Green Version]
- Poeggeler, B.; Saarela, S.; Reiter, R.J.; Tan, D.X.; Chen, L.D.; Manchester, L.C.; Barlow-Walden, L.R. Melatonin—A highly potent endogenous radical scavenger and electron donor: New aspects of the oxidation chemistry of this indole accessed in vitro. Ann. N. Y. Acad. Sci. 1994, 738, 419–420. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Reiter, R.J.; Plummer, B.F.; Hardies, L.J.; Weintraub, S.T.; Vijayalaxmi; Shepherd, A.M. A novel melatonin metabolite, cyclic 3-hydroxymelatonin: A biomarker of in vivo hydroxyl radical generation. Biochem. Biophys. Res. Commun. 1998, 253, 614–620. [Google Scholar] [CrossRef]
- Margulis, L. Symbiotic theory of the origin of eukaryotic organelles; criteria for proof. Symp. Soc. Exp. Biol. 1975, 29, 21–38. [Google Scholar]
- Reiter, R.J.; Rosales-Corral, S.; Tan, D.X.; Jou, M.J.; Galano, A.; Xu, B. Melatonin as a mitochondria-targeted antioxidant: One of evolution’s best ideas. Cell. Mol. Life Sci. 2017, 74, 3863–3881. [Google Scholar] [CrossRef]
- Cecon, E.; Liu, L.; Jockers, R. Melatonin receptor structures shed new light on melatonin research. J. Pineal Res. 2019, 67, e12606. [Google Scholar] [CrossRef]
- Barrett, P.; Conway, S.; Morgan, P.J. Digging deep—Structure-function relationships in the melatonin receptor family. J. Pineal Res. 2003, 35, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Pandi-Perumal, S.R.; Trakht, I.; Srinivasan, V.; Spence, D.W.; Maestroni, G.J.M.; Zisapel, N.; Cardinali, D.P. Physiological effects of melatonin: Role of melatonin receptors and signal transduction pathways. Prog. Neurobiol. 2008, 85, 335–353. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.H.; Ursinus, J.; Zhou, J.N.; Scheer, F.A.; Ai-Min, B.; Jockers, R.; van Heerikhuize, J.; Swaab, D.F. Alterations of melatonin receptors MT1 and MT2 in the hypothalamic suprachiasmatic nucleus during depression. J. Affect. Disord. 2013, 148, 357–367. [Google Scholar] [CrossRef]
- Benleulmi-Chaachoua, A.; Chen, L.; Sokolina, K.; Wong, V.; Jurisica, I.; Emerit, M.B.; Darmon, M.; Espin, A.; Stagljar, I.; Tafelmeyer, P.; et al. Protein interactome mining defines melatonin MT1 receptors as integral component of presynaptic protein complexes of neurons. J. Pineal Res. 2016, 60, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Jockers, R.; Delagrange, P.; Dubocovich, M.L.; Markus, R.P.; Renault, N.; Tosini, G.; Cecon, E.; Zlotos, D.P. Update on melatonin receptors: IUPHAR review 20. Br. J. Pharmacol. 2016, 173, 2702–2725. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Clough, S.J.; Hutchinson, A.J.; Adamah-Biassi, E.B.; Popovska-Gorevski, M.; Dubocovich, M.L. MT1 and MT2 melatonin receptors: A therapeutic perspective. Annu. Rev. Pharmacol. Toxicol. 2016, 56, 361–383. [Google Scholar] [CrossRef] [Green Version]
- Nosjean, O.; Ferro, M.; Coge, F.; Beauverger, P.; Henlin, J.M.; Lefoulon, F.; Fauchere, J.L.; Delagrange, P.; Canet, E.; Boutin, J.A. Identification of the melatonin-binding site MT3 as the quinone reductase 2. J. Biol. Chem. 2000, 275, 31311–31317. [Google Scholar] [CrossRef] [Green Version]
- Ekmekcioglu, C. Melatonin receptors in humans: Biological role and clinical relevance. Biomed. Pharmacother. 2006, 60, 97–108. [Google Scholar] [CrossRef]
- Dubocovich, M.L.; Delagrange, P.; Krause, D.N.; Sugden, D.; Cardinali, D.P.; Olcese, J. International Union of Basic and Clinical Pharmacology. LXXV. Nomenclature, classification, and pharmacology of G protein-coupled melatonin receptors. Pharmacol. Rev. 2010, 62, 343–380. [Google Scholar] [CrossRef] [Green Version]
- Li, D.Y.; Smith, D.G.; Hardeland, R.; Yang, M.Y.; Xu, H.L.; Zhang, L.; Yin, H.D.; Zhu, Q. Melatonin receptor genes in vertebrates. Int. J. Mol. Sci. 2013, 14, 11208–11223. [Google Scholar] [CrossRef] [Green Version]
- Stauch, B.; Johansson, L.C.; Cherezov, V. Structural insights into melatonin receptors. FEBS J. 2020, 287, 1496–1510. [Google Scholar] [CrossRef] [Green Version]
- Levoye, A.; Jockers, R.; Ayoub, M.A.; Delagrange, P.; Savaskan, E.; Guillaume, J.L. Are G protein-coupled receptor heterodimers of physiological relevance? Focus on melatonin receptors. Chronobiol. Int. 2006, 23, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, M.A.; Couturier, C.; Lucas-Meunier, E.; Angers, S.; Fossier, P.; Bouvier, M.; Jockers, R. Monitoring of ligand-independent dimerization and ligand-induced conformational changes of melatonin receptors in living cells by bioluminescence resonance energy transfer. J. Biol. Chem. 2002, 277, 21522–21528. [Google Scholar] [CrossRef] [Green Version]
- Oishi, A.; Cecon, E.; Jockers, R. Melatonin receptor signaling: Impact of receptor oligomerization on receptor function. Int. Rev. Cell Mol. Biol. 2018, 338, 59–77. [Google Scholar] [CrossRef] [PubMed]
- Luchetti, F.; Canonico, B.; Betti, M.; Arcangeletti, M.; Pilolli, F.; Piroddi, M.; Canesi, L.; Papa, S.; Galli, F. Melatonin signaling and cell protection function. FASEB J. 2010, 24, 3603–3624. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.Y.; Leong, M.K.; Liang, H.; Paxinos, G. Melatonin receptors: Distribution in mammalian brain and their respective putative functions. Brain Struct. Funct. 2017, 222, 2921–2939. [Google Scholar] [CrossRef] [PubMed]
- Jockers, R.; Maurice, P.; Boutin, J.A.; Delagrange, P. Melatonin receptors, heterodimerization, signal transduction and binding sites: What’s new? Br. J. Pharmacol. 2008, 154, 1182–1195. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Lv, Y.; Bian, C.; You, X.; Shi, Q. Molecular evolution of melatonin receptor genes (mtnr) in vertebrates and its shedding light on mtnr1c. Gene 2021, 769, 145256. [Google Scholar] [CrossRef]
- Guyomard, R.; Boussaha, M.; Krieg, F.; Hervet, C.; Quillet, E. A synthetic rainbow trout linkage map provides new insights into the salmonid whole genome duplication and the conservation of synteny among teleosts. BMC Genet. 2012, 13, 15. [Google Scholar] [CrossRef]
- Levoye, A.; Dam, J.; Ayoub, M.A.; Guillaume, J.L.; Couturier, C.; Delagrange, P.; Jockers, R. The orphan GPR50 receptor specifically inhibits MT1 melatonin receptor function through heterodimerization. EMBO J. 2006, 25, 3012–3023. [Google Scholar] [CrossRef]
- Ma, H.; Kang, J.; Fan, W.; He, H.; Huang, F. ROR: Nuclear receptor for melatonin or not? Molecules 2021, 26, 2693. [Google Scholar] [CrossRef]
- Pace-Schott, E.F.; Hobson, J.A. The neurobiology of sleep: Genetics, cellular physiology and subcortical networks. Nat. Rev. Neurosci. 2002, 3, 591–605. [Google Scholar] [CrossRef]
- Gobbi, G.; Comai, S. Sleep well. Untangling the role of melatonin MT1 and MT2 receptors in sleep. J. Pineal Res. 2019, 66, e12544. [Google Scholar] [CrossRef]
- Dubocovich, M.L. Melatonin receptors: Role on sleep and circadian rhythm regulation. Sleep Med. 2007, 8, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Scher, J.; Wankiewicz, E.; Brown, G.M.; Fujieda, H. MT1 melatonin receptor in the human retina: Expression and localization. Investig. Ophthalmol. Vis. Sci. 2002, 43, 889–897. [Google Scholar]
- Rada, J.A.; Wiechmann, A.F. Melatonin receptors in chick ocular tissues: Implications for a role of melatonin in ocular growth regulation. Invest. Ophthalmol. Vis. Sci. 2006, 47, 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arangino, S.; Cagnacci, A.; Angiolucci, M.; Vacca, A.M.; Longu, G.; Volpe, A.; Melis, G.B. Effects of melatonin on vascular reactivity, catecholamine levels, and blood pressure in healthy men. Am. J. Cardiol. 1999, 83, 1417–1419. [Google Scholar] [CrossRef]
- Srinivasan, V.; Maestroni, G.J.M.; Cardinali, D.P.; Esquifino, A.I.; Pandi-Perumal, S.R.; Miller, S.C. Melatonin, immune function and aging. Immun. Ageing 2005, 2, 17. [Google Scholar] [CrossRef] [Green Version]
- Carrillo-Vico, A.; Reiter, R.J.; Lardone, P.J.; Herrera, J.L.; Fernandez-Montesinos, R.; Guerrero, J.M.; Pozo, D. The modulatory role of melatonin on immune responsiveness. Curr. Opin. Investig. Drugs 2006, 7, 423–431. [Google Scholar]
- Slominski, A.; Pisarchik, A.; Wortsman, J. Expression of genes coding melatonin and serotonin receptors in rodent skin. Biochim. Biophys. Acta 2004, 1680, 67–70. [Google Scholar] [CrossRef]
- Weaver, D.R.; Liu, C.; Reppert, S.M. Nature’s knockout: The Mel1b receptor is not necessary for reproductive and circadian responses to melatonin in Siberian hamsters. Mol. Endocrinol. 1996, 10, 1478–1487. [Google Scholar] [CrossRef] [Green Version]
- Adamah-Biassi, E.B.; Zhang, Y.; Jung, H.; Vissapragada, S.; Miller, R.J.; Dubocovich, M.I. Distribution of MT1 melatonin receptor promoter-driven RFP expression in the brains of BAC C3H/HeN transgenic mice. J. Histochem. Cytochem. 2014, 62, 70–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, J.E.; McKellar, S.; Klein, D.C. Melatonin inhibition of the in vivo pituitary response to luteinizing hormone-releasing hormone in the neonatal rat. Neuroendocrinology 1980, 31, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Vanecek, J.; Vollrath, L. Developmental changes and daily rhythm in melatonin-induced inhibition of 3′,5′-cyclic AMP accumulation in the rat pituitary. Endocrinology 1990, 126, 1509–1513. [Google Scholar] [CrossRef]
- Vanecek, J.; Klein, D.C. Melatonin inhibition of GnRH-induced LH release from neonatal rat gonadotroph: Involvement of Ca2+ not cAMP. Am. J. Physiol. 1995, 269, E85–E90. [Google Scholar] [CrossRef] [PubMed]
- von Gall, C.; Garabette, M.L.; Kell, C.A.; Frenzel, S.; Dehghani, F.; Schumm-Draeger, P.M.; Weaver, D.R.; Korf, H.W.; Hastings, M.H.; Stehle, J.H. Rhythmic gene expression in pituitary depends on heterologous sensitization by the neurohormone melatonin. Nat. Neurosci. 2002, 5, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Frungieri, M.B.; Mayerhofer, A.; Zitta, K.; Pignataro, O.P.; Calandra, R.S.; Gonzalez-Calvar, S.I. Direct effect of melatonin on Syrian hamster testes: Melatonin subtype 1a receptors, inhibition of androgen production, and interaction with the local corticotropin-releasing hormone system. Endocrinology 2005, 146, 1541–1552. [Google Scholar] [CrossRef] [PubMed]
- Torres-Farfan, C.; Richter, H.G.; Rojas-Garcia, P.; Vergara, M.; Forcelledo, M.L.; Valladares, L.E.; Torrealba, F.; Valenzuela, G.J.; Seron-Ferre, M. MT1 melatonin receptor in the primate adrenal gland: Inhibition of adrenocorticotropin-stimulated cortisol production by melatonin. J. Clin. Endocrinol. Metab. 2003, 88, 450–458. [Google Scholar] [CrossRef] [Green Version]
- Torres-Farfan, C.; Richter, H.G.; Germain, A.M.; Valenzuela, G.J.; Campino, C.; Rojas-Garcia, P.; Forcelledo, M.L.; Torrealba, F.; Seron-Ferre, M. Maternal melatonin selectively inhibits cortisol production in the primate fetal adrenal gland. J. Physiol. 2004, 554, 841–856. [Google Scholar] [CrossRef]
- Alonso-Vale, M.I.C.; Andreotti, S.; Peres, S.B.; Anhe, G.F.; Borges-Silva, C.N.; Neto, J.C.; Lima, F.B. Melatonin enhances leptin expression by rat adipocytes in the presence of insulin. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E805–E812. [Google Scholar] [CrossRef] [Green Version]
- Kemp, D.M.; Ubeda, M.; Habener, J.F. Identification and functional characterization of melatonin Mel 1a receptors in pancreatic beta cells: Potential role in incretin-mediated cell function by sensitization of cAMP signaling. Mol. Cell. Endocrinol. 2002, 191, 157–166. [Google Scholar] [CrossRef]
- Nelson, M.T.; Quayle, J.M. Physiological roles and properties of potassium channels in arterial smooth muscle. Am. J. Physiol. 1995, 268, C799–C822. [Google Scholar] [CrossRef] [PubMed]
- Doolen, S.; Krause, D.N.; Dubocovich, M.L.; Duckles, S.P. Melatonin mediates two distinct responses in vascular smooth muscle. Eur. J. Pharmacol. 1998, 345, 67–69. [Google Scholar] [CrossRef]
- Geary, G.G.; Duckles, S.P.; Krause, D.N. Effect of melatonin in the rat tail artery: Role of K+ channels and endothelial factors. Br. J. Pharmacol. 1998, 123, 1533–1540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masana, M.I.; Doolen, S.; Ersahin, C.; Al-Ghoul, W.M.; Duckles, S.P.; Dubocovich, M.L.; Krause, D.N. MT2 melatonin receptors are present and functional in rat caudal artery. J. Pharmacol. Exp. Ther. 2002, 302, 1295–1302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, T.; Zhang, H.; Jin, C.; Qiu, F.; Wu, Y.; Shi, L. Melatonin mediates vasodilation through both direct and indirect activation of BKCa channels. J. Mol. Endocrinol. 2017, 59, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Shiu, S.Y.; Li, L.; Xu, J.N.; Pang, C.S.; Wong, J.T.; Pang, S.F. Melatonin-induced inhibition of proliferation and G1/S cell cycle transition delay of human choriocarcinoma Jar cells: Possible involvement of MT2 (MEL1B) receptor. J. Pineal Res. 1999, 27, 183–192. [Google Scholar] [CrossRef]
- Collins, A.; Yuan, L.; Kiefer, T.L.; Cheng, Q.; Lai, L.; Hill, S.M. Overexpression of the MT1 melatonin receptor in MCF-7 human breast cancer cells inhibits mammary tumor formation in nude mice. Cancer Lett. 2003, 189, 49–57. [Google Scholar] [CrossRef]
- Drazen, D.L.; Nelson, R.J. Melatonin receptor subtype MT2 (Mel 1b) and not MT1 (Mel 1a) is associated with melatonin-induced enhancement of cell-mediated and humoral immunity. Neuroendocrinology 2001, 74, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Lotufo, C.M.; Lopes, C.; Dubocovich, M.L.; Farsky, S.H.; Markus, R.P. Melatonin and N-acetylserotonin inhibit leukocyte rolling and adhesion to rat microcirculation. Eur. J. Pharmacol. 2001, 430, 351–357. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; Garcia-Maurino, S.; Calvo, J.R.; Guerrero, J.M. Melatonin counteracts the inhibitory effect of PGE2 on IL-2 production in human lymphocytes via its mt1 membrane receptor. FASEB J. 2003, 17, 755–757. [Google Scholar] [CrossRef]
- Kadekaro, A.L.; Andrade, L.N.; Floeter-Winter, L.M.; Rollag, M.D.; Virador, V.; Vieira, W.; Castrucci, A.M.D.L. MT-1 melatonin receptor expression increases the antiproliferative effect of melatonin on S-91 murine melanoma cells. J. Pineal Res. 2004, 36, 204–211. [Google Scholar] [CrossRef]
- Carrillo-Vico, A.; Lardone, P.J.; Fernandez-Santos, J.M.; Martin-Lacave, I.; Calvo, J.R.; Karasek, M.; Guerrero, J.M. Human lymphocyte-synthesized melatonin is involved in the regulation of the interleukin-2/interleukin-2 receptor system. J. Clin. Endocrinol. Metab. 2005, 90, 992–1000. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morgan, P.J.; Barrett, P.; Howell, H.E.; Helliwell, R. Melatonin receptors: Localization, molecular pharmacology and physiological significance. Neurochem. Int. 1994, 24, 101–146. [Google Scholar] [CrossRef]
- Comai, S.; Ochoa-Sanchez, R.; Gobbi, G. Sleep-wake characterization of double MT1/MT2 receptor knockout mice and comparison with MT1 and MT2 receptor knockout mice. Behav. Brain Res. 2013, 243, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Boutin, J.A.; Audinot, V.; Ferry, G.; Delagrange, P. Molecular tools to study melatonin pathways and actions. Trends Pharmacol. Sci. 2005, 26, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Sugden, D.; McArthur, A.J.; Ajpru, S.; Duniec, K.; Piggins, H.D. Expression of mt1 melatonin receptor subtype mRNA in the entrained rat suprachiasmatic nucleus: A quantitative RT-PCR study across the diurnal cycle. Brain Res. Mol. Brain Res. 1999, 72, 176–182. [Google Scholar] [CrossRef]
- Lacoste, B.; Angeloni, D.; Dominguez-Lopez, S.; Calderoni, S.; Mauro, A.; Faschini, F.; Descarries, L.; Gobbi, G. Anatomical and cellular localization of melatonin MT1 and MT2 receptors in the adult rat brain. J. Pineal Res. 2015, 58, 397–417. [Google Scholar] [CrossRef] [PubMed]
- Waly, N.E.; Hallworth, R. Circadian pattern of melatonin MT1 and MT2 receptor localization in the rat suprachiasmatic nucleus. J. Circadian Rhythms 2015, 13, 1. [Google Scholar] [CrossRef] [Green Version]
- Klosen, P.; Lapmanee, S.; Schuster, C.; Guardiola, B.; Hicks, D.; Pevet, P.; Felder-Schmittbuhl, M.P. MT1 and MT2 melatonin receptors are expressed in nonoverlapping neuronal populations. J. Pineal Res. 2019, 67, e12575. [Google Scholar] [CrossRef]
- Baba, K.; Benleulmi-Chaachoua, A.; Journé, A.S.; Kamal, M.; Guillaume, J.L.; Dussaud, S.; Gbahou, F.; Yettou, K.; Liu, C.; Contreras-Alcantara, S.; et al. Heteromeric MT1/MT2 melatonin receptors modulate photoreceptor function. Sci. Signal. 2013, 6, ra89. [Google Scholar] [CrossRef] [Green Version]
- Perreau-Lenz, S.; Kalsbeek, A.; Garidou, M.L.; Wortel, J.; Van Der Vliet, J.; Van Heijningen, C.; Simonneaux, V.; Pevet, P.; Buijs, R.M. Suprachiasmatic control of melatonin synthesis in rats: Inhibitory and stimulatory mechanisms. Eur. J. Neurosci. 2003, 17, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, W.B.; Gillin, J.C.; Dawson, S.D.; Lewy, A.J.; Wyatt, R.J. Effects of melatonin on propranolol on sleep of the rat. Brain Res. 1980, 201, 240–244. [Google Scholar] [CrossRef]
- Zhdanova, I.V.; Wurtman, R.J.; Morabito, C.; Piotrovska, V.R.; Lynch, H.J. Effects of low oral doses of melatonin, given 2-4 hours before habitual bedtime, on sleep in normal young humans. Sleep 1996, 19, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Huber, R.; Deboer, T.; Schwierin, B.; Tobler, I. Effect of melatonin on sleep and brain temperature in the Djungarian hamster and the rat. Physiol Behav. 1998, 65, 77–82. [Google Scholar] [CrossRef]
- Cipolla-Neto, J.; do Amaral, F.G. Melatonin as a hormone: New physiological and clinical insights. Endocr. Rev. 2018, 39, 990–1028. [Google Scholar] [CrossRef] [Green Version]
- Kalsbeek, A.; Buijs, R.M. Output pathways of the mammalian suprachiasmatic nucleus: Coding circadian time by transmitter selection and specific targeting. Cell Tissue Res. 2002, 309, 109–118. [Google Scholar] [CrossRef]
- Borjigin, J.; Zhang, L.S.; Calinescu, A.A. Circadian regulation of pineal gland rhythmicity. Mol. Cell Endocrinol. 2012, 349, 13–19. [Google Scholar] [CrossRef] [Green Version]
- Pinato, L.; Ramos, D.; Hataka, A.; Rossignoli, P.S.; Granado, M.D.J.; Mazzetto, M.C. Day/night expression of MT1 and MT2 receptors in hypothalamic nuclei of the primate Sapajus apella. J. Chem. Neuroanat. 2017, 81, 10–17. [Google Scholar] [CrossRef] [Green Version]
- James, F.O.; Cermakian, N.; Boivin, D.B. Circadian rhythms of melatonin, cortisol, and clock gene expression during simulated night shift work. Sleep 2007, 30, 1427–1436. [Google Scholar] [CrossRef] [Green Version]
- Jan, J.E.; Reiter, R.J.; Wong, P.K.H.; Bax, M.C.O.; Ribary, U.; Wasdell, M.B. Melatonin has membrane receptor-independent hypnotic action on neurons: An hypothesis. J. Pineal Res. 2010, 50, 233–240. [Google Scholar] [CrossRef]
- Fisher, S.P.; Sugden, D. Sleep-promoting action of IIK7, a selective MT2 melatonin receptor agonist in the rat. Neurosci. Lett. 2009, 457, 93–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisher, S.P.; Sugden, D. Endogenous melatonin is not obligatory for the regulation of the rat sleep-wake cycle. Sleep 2010, 33, 833–840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Witt-Enderby, P.A.; Bennett, J.; Jarzynka, M.J.; Firestine, S.; Melan, M.A. Melatonin receptors and their regulation: Biochemical and structural mechanisms. Life Sci. 2003, 72, 2183–2198. [Google Scholar] [CrossRef]
- Gerdin, M.J.; Masana, M.I.; Dubocovich, M.L. Melatonin-mediated regulation of human MT1 melatonin receptors expressed in mammalian cells. Biochem. Pharmacol. 2004, 67, 2023–2030. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, G.; Comai, S. Differential function of melatonin MT1 and MT2 receptors in REM and NREM sleep. Front. Endocrinol. 2019, 10, 87. [Google Scholar] [CrossRef] [Green Version]
- Grossini, E.; Molinari, C.; Uberti, F.; Mary, D.A.S.G.; Vacca, G.; Caimmi, P.P. Intracoronary melatonin increases coronary blood flow and cardiac function through β-adrenoreceptors, MT1/MT2 receptors, and nitric oxide in anesthetized pigs. J. Pineal Res. 2011, 51, 246–257. [Google Scholar] [CrossRef]
- Ersahin, C.; Masana, M.I.; Dubocovich, M.L. Constitutively active melatonin MT1 receptors in male rat caudal arteries. Eur. J. Pharmacol. 2002, 439, 171–172. [Google Scholar] [CrossRef]
- Capsoni, S.; Viswanathan, M.; De Oliveira, A.M.; Saavedra, J.M. Characterization of melatonin receptors and signal transduction system in rat arteries forming the circle of Willis. Endocrinology 1994, 135, 373–378. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Korkmaz, A. The circadian melatonin rhythm and its modulation: Possible impact on hypertension. J. Hypertens. Suppl. 2009, 27, S17–S20. [Google Scholar] [CrossRef]
- Weekley, L.B. Effects of melatonin on pulmonary and coronary vessels are exerted through perivascular nerves. Clin. Auton. Res. 1993, 3, 45–47. [Google Scholar] [CrossRef]
- Satake, N.; Shibata, S.; Takagi, T. The inhibitory action of melatonin on the contractile response to 5-hydroxytryptamine in various isolated vascular smooth muscles. Gen. Pharmacol. 1986, 17, 553–558. [Google Scholar] [CrossRef]
- Shibata, S.; Satake, N.; Takagi, T.; Usui, H. Vasorelaxing action of melatonin in rabbit basilar artery. Gen. Pharmacol. 1989, 20, 677–680. [Google Scholar] [CrossRef]
- Satake, N.; Oe, H.; Sawada, T.; Shibata, S. The mode of vasorelaxing action of melatonin in rabbit aorta. Gen. Pharmacol. 1991, 22, 219–221. [Google Scholar] [CrossRef]
- Satake, N.; Oe, H.; Shibata, S. Vasorelaxing action of melatonin in rat isolated aorta; possible endothelium dependent relaxation. Gen. Pharmacol. 1991, 22, 1127–1133. [Google Scholar] [CrossRef]
- Capsoni, S.; Stankov, B.M.; Fraschini, F. Reduction of regional cerebral blood flow by melatonin in young rats. Neuroreport 1995, 6, 1346–1348. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, C.; Giummelly, P.; Atkinson, J.; Delagrange, P.; Scalbert, E.; Capdeville-Atkinson, C. Melatonin potentiates NE-induced vasoconstriction without augmenting cytosolic calcium concentration. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H420–H425. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.S.; Sauder, C.L.; Ray, C.A. Melatonin differentially affects vascular blood flow in humans. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H670–H674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ting, N.; Thambyraja, A.; Sugden, D.; Scalbert, E.; Delagrange, P.; Wilson, V.G. Pharmacological studies on the inhibitory action of melatonin and putative melatonin analogues on porcine vascular smooth muscle. Naunyn Schmiedebergs Arch. Pharmacol. 2000, 36, 327–333. [Google Scholar] [CrossRef]
- Okatani, Y.; Wakatsuki, A.; Kaneda, C. Melatonin increases activities of glutathione peroxidase and superoxide dismutase in fetal rat brain. J. Pineal Res. 2000, 28, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Terron, M.P.; Flores, L.J.; Czarnocki, Z. Melatonin and its metabolites: New findings regarding their production and their radical scavenging actions. Acta. Biochim. Pol. 2007, 54, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.C.; Lai, C.J.; Tsai, M.H.; Wu, Y.C.; Chen, K.T.; Jou, M.J.; Fu, P.I.; Wu, C.H.; Wei, I.H. Effects of melatonin on the nitric oxide system and protein nitration in the hypobaric hypoxic rat hippocampus. BMC Neurosci. 2015, 16, 61. [Google Scholar] [CrossRef] [Green Version]
- Drazen, D.L.; Bilu, D.; Bilbo, S.D.; Nelson, R.J. Melatonin enhancement of splenocyte proliferation is attenuated by luzindole, a melatonin receptor antagonist. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 280, R1476–R1482. [Google Scholar] [CrossRef] [Green Version]
- Swaminathan, A.; Lucas, R.M.; Dear, K.; McMichael, A.J. Keyhole limpet haemocyanin-a model antigen for human immunotoxicological studies. Br. J. Clin. Pharmacol. 2014, 78, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.E.; Wiechmann, A.F.; Hu, D.N. Melatonin receptors in human uveal melanocytes and melanoma cells. J. Pineal Res. 2000, 28, 165–171. [Google Scholar] [CrossRef]
- Tarocco, A.; Caroccia, N.; Morciano, G.; Wieckowski, M.R.; Ancora, G.; Garani, G.; Pinton, P. Melatonin as a master regulator of cell death and inflammation: Molecular mechanisms and clinical implications for newborn care. Cell Death Dis. 2019, 10, 317. [Google Scholar] [CrossRef] [Green Version]
- Ko, C.H.; Takahashi, J.S. Molecular components of the mammalian circadian clock. Hum. Mol. Genet. 2006, 15, R271–R277. [Google Scholar] [CrossRef]
- Jetten, A.M. Retinoid-related orphan receptors (RORs): Critical roles in development, immunity, circadian rhythm, and cellular metabolism. Nucl. Recept. Signal. 2009, 7, e003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lardone, P.J.; Guerrero, J.M.; Fernández-Santos, J.M.; Rubio, A.; Martín-Lacave, I.; Carrillo-Vico, A. Melatonin synthesized by T lymphocytes as a ligand of the retinoic acid-related orphan receptor. J. Pineal Res. 2011, 51, 454–462. [Google Scholar] [CrossRef]
- Ram, P.T.; Dai, J.; Yuan, L.; Dong, C.; Kiefer, T.L.; Lai, L.; Hill, S.M. Involvement of the mt1 melatonin receptor in human breast cancer. Cancer Lett. 2002, 179, 141–150. [Google Scholar] [CrossRef]
- Chang, H.C.; Guarente, L. SIRT1 mediates central circadian control in the SCN by a mechanism that decays with aging. Cell 2013, 153, 1448–1460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, M.; Feng, N.; Tang, D.; Feng, J.; Li, Z.; Jia, M.; Liu, Z.; Gu, X.; Wang, Y.; Fu, F.; et al. Melatonin prevents Drp1-mediated mitochondrial fission in diabetic hearts through SIRT1-PGC1a pathway. J. Pineal Res. 2018, 65, e12491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hardeland, R. Melatonin and inflammation-Story of a double-edged blade. J. Pineal Res. 2018, 65, e12525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiter, R.J.; Tan, D.X.; Rosales-Corral, S.; Galano, A.; Jou, M.J.; Acuna-Castroviejo, D. Melatonin mitigates mitochondrial meltdown: Interactions with SIRT3. Int. J. Mol. Sci. 2018, 19, 2439. [Google Scholar] [CrossRef] [Green Version]
- Sánchez, D.I.; González-Fernández, B.; Crespo, I.; San-Miguel, B.; Álvarez, M.; González-Gallego, J.; Tuñón, M.J. Melatonin modulates dysregulated circadian clocks in mice with diethylnitrosamine-induced hepatocellular carcinoma. J. Pineal Res. 2018, 65, e12506. [Google Scholar] [CrossRef]
- Han, D.; Wang, Y.; Chen, J.; Zhang, J.; Yu, P.; Zhang, R.; Li, S.; Tao, B.; Wang, Y.; Qiu, Y.; et al. Activation of melatonin receptor 2 but not melatonin receptor 1 mediates melatonin-conferred cardioprotection against myocardial ischemia/reperfusion injury. J. Pineal Res. 2019, 67, e12571. [Google Scholar] [CrossRef]
- Merlo, S.; Luaces, J.P.; Spampinato, S.F.; Toro-Urrego, N.; Caruso, G.I.; D’Amico, F.; Capani, F.; Sortino, M.A. SIRT1 mediates melatonin’s effects on microglial activation in hypoxia: In vitro and in vivo evidence. Biomolecules 2020, 10, 364. [Google Scholar] [CrossRef] [Green Version]
- Guerrero, H.Y.; Gauer, F.; Pevet, P.; Masson-Pevet, M. Daily and circadian expression patterns of mt1 melatonin receptor mRNA in the rat pars tuberalis. Adv. Exp. Med. Biol. 1999, 460, 175–179. [Google Scholar] [CrossRef]
- Gubitz, A.K.; Reppert, S.M. Assignment of the melatonin-related receptor to human chromosome X (GPR50) and mouse chromosome X (Gpr50). Genomics 1999, 55, 248–251. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, I.R.; Mazuruk, K.; Schoen, T.J.; Chader, G.J. Structural analysis of the human hydroxyindole-O-methyltransferase gene. Presence of two distinct promoters. J. Biol. Chem. 1994, 269, 31969–31977. [Google Scholar] [CrossRef]
- Estrada-Rodgers, L.; Levy, G.N.; Weber, W.W. Characterization of a hormone response element in the mouse N-acetyltransferase 2 (Nat2*) promoter. Gene Expr. 1998, 7, 13–24. [Google Scholar]
- Muxel, S.M.; Laranjeira-Silva, M.F.; Carvalho-Sousa, C.E.; Floeter-Winter, L.M.; Markus, R.P. The RelA/cRel nuclear factor-κB (NF-κB) dimer, crucial for inflammation resolution, mediates the transcription of the key enzyme in melatonin synthesis in RAW 264.7 macrophages. J. Pineal Res. 2016, 60, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Boveris, A. Mitochondrial production of superoxide radical and hydrogen peroxide. Adv. Exp. Med. Biol. 1977, 78, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Sohal, R.S.; Brunk, U.T. Mitochondrial production of pro-oxidants and cellular senescence. Mutat. Res. 1992, 275, 295–304. [Google Scholar] [CrossRef]
- Martin, M.; Macias, M.; Escames, G.; Leon, J.; Acuna-Castroviejo, D. Melatonin but not vitamins C and E maintains glutathione homeostasis in t-butyl hydroperoxide-induced mitochondrial oxidative stress. FASEB J. 2000, 14, 1677–1679. [Google Scholar] [CrossRef]
- Huo, X.; Wang, C.; Yu, Z.; Peng, Y.; Wang, S.; Feng, S.; Zhang, S.; Tian, X.; Sun, C.; Liu, K.; et al. Human transporters, PEPT1/2, facilitate melatonin transportation into mitochondria of cancer cells: An implication of the therapeutic potential. J. Pineal Res. 2017, 62, e12390. [Google Scholar] [CrossRef]
- Hevia, D.; González-Menéndez, P.; Quiros-González, I.; Miar, A.; Rodríguez-García, A.; Tan, D.X.; Reiter, R.J.; Mayo, J.C.; Sainz, R.M. Melatonin uptake through glucose transporters: A new target for melatonin inhibition of cancer. J. Pineal Res. 2015, 58, 234–250. [Google Scholar] [CrossRef]
- Reiter, R.J.; Ma, Q.; Sharma, R. Melatonin in mitochondria: Mitigating clear and present dangers. Physiology 2020, 35, 86–95. [Google Scholar] [CrossRef]
- Acuña-Castroviejo, D.; López, L.C.; Escames, G.; López, A.; García, J.A.; Reiter, R.J. Melatonin-mitochondria interplay in health and disease. Curr. Top. Med. Chem. 2011, 11, 221–240. [Google Scholar] [CrossRef]
- Fridovich, I. Oxygen: How do we stand it? Med. Princ. Pract. 2013, 22, 131–137. [Google Scholar] [CrossRef]
- Han, L.; Wang, H.; Li, L.; Li, X.; Ge, J.; Reiter, R.J.; Wang, Q. Melatonin protects against maternal obesity-associated oxidative stress and meiotic defects in oocytes via the SIRT3-SOD2-dependent pathway. J. Pineal Res. 2017, 63, e12431. [Google Scholar] [CrossRef]
- Zhai, M.; Li, B.; Duan, W.; Jing, L.; Zhang, B.; Zhang, M.; Yu, L.; Liu, Z.; Yu, B.; Ren, K.; et al. Melatonin ameliorates myocardial ischemia reperfusion injury through SIRT3-dependent regulation of oxidative stress and apoptosis. J. Pineal Res. 2017, 63, e12419. [Google Scholar] [CrossRef]
- Naaz, S.; Mishra, S.; Pal, P.K.; Chattopadhyay, A.; Das, A.R.; Bandyopadhyay, D. Activation of SIRT1/PGC 1α/SIRT3 pathway by melatonin provides protection against mitochondrial dysfunction in isoproterenol induced myocardial injury. Heliyon 2020, 6, e05159. [Google Scholar] [CrossRef]
- Klein, D.C. Arylalkylamine N-acetyltransferase: “The Timezyme”. J. Biol. Chem. 2007, 282, 4233–4237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okatani, Y.; Wakatsuki, A.; Reiter, R.J. Melatonin protects hepatic mitochondrial respiratory chain activity in senescence-accelerated mice. J. Pineal Res. 2002, 32, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Okatani, Y.; Wakatsuki, A.; Reiter, R.J.; Enzan, H.; Miyahara, Y. Protective effect of melatonin against mitochondrial injury induced by ischemia and reperfusion of rat liver. Eur. J. Pharmacol. 2003, 469, 145–152. [Google Scholar] [CrossRef]
- Jou, M.J.; Peng, T.I.; Reiter, R.J.; Jou, S.B.; Wu, H.Y.; Wen, S.T. Visualization of the antioxidative effects of melatonin at the mitochondrial level during oxidative stress-induced apoptosis of rat brain astrocytes. J. Pineal Res. 2004, 37, 55–70. [Google Scholar] [CrossRef]
- Paradies, G.; Paradies, V.; Ruggiero, F.M.; Petrosillo, G. Protective role of melatonin in mitochondrial dysfunction and related disorders. Arch. Toxicol. 2015, 89, 923–939. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sirianni, A.; Pei, Z.; Cormier, K.; Smith, K.; Jiang, J.; Zhou, S.; Wang, H.; Zhao, R.; Yano, H.; et al. The melatonin MT1 receptor axis modulates mutant Huntingtin-mediated toxicity. J. Neurosci. 2011, 31, 14496–14507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahluwalia, A.; Brzozowska, I.M.; Hoa, N.; Jones, M.K.; Tarnawski, A.S. Melatonin signaling in mitochondria extends beyond neurons and neuroprotection: Implications for angiogenesis and cardio/gastroprotection. Proc. Natl. Acad. Sci. USA 2018, 115, E1942–E1943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Adams, J.M.; White, M. Biological ageing: A fundamental, biological link between socio-economic status and health? Eur. J. Public Health 2004, 14, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Kenyon, C.J. The genetics of ageing. Nature 2010, 464, 504–512. [Google Scholar] [CrossRef]
- Talens, R.P.; Christensen, K.; Putter, H.; Willemsen, G.; Christiansen, L.; Kremer, D.; Suchiman, H.E.D.; Slagboom, P.E.; Boomsma, D.I.; Heijmans, B.T. Epigenetic variation during the adult lifespan: Cross-sectional and longitudinal data on monozygotic twin pairs. Aging Cell 2012, 11, 694–703. [Google Scholar] [CrossRef]
- Gurland, B.J.; Page, W.F.; Plassman, B.L. A twin study of the genetic contribution to age-related functional impairment. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 859–863. [Google Scholar] [CrossRef] [Green Version]
- Reiter, R.J.; Craft, C.M.; Johnson, J.E., Jr.; King, T.S.; Richardson, B.A.; Vaughan, G.M.; Vaughan, M.K. Age-associated reduction in nocturnal pineal melatonin levels in female rats. Endocrinology 1981, 109, 1295–1297. [Google Scholar] [CrossRef]
- Zisapel, N. New perspectives on the role of melatonin in human sleep, circadian rhythms and their regulation. Br. J. Pharmacol. 2018, 175, 3190–3199. [Google Scholar] [CrossRef]
- Lopez-Otin, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [Green Version]
- Barardo, D.; Thornton, D.; Thoppil, H.; Walsh, M.; Sharifi, S.; Ferreira, S.; Anzic, A.; Fernandes, M.; Monteiro, P.; Grum, T.; et al. The DrugAge database of aging-related drugs. Aging Cell 2017, 16, 594–597. [Google Scholar] [CrossRef] [Green Version]
- DrugAge Database of Aging-Related Drugs. Available online: http://genomics.senescence.info/drugs/index.php (accessed on 24 June 2021).
- Anisimov, V.N.; Arutjunyan, A.V.; Khavinson, V.K. Effects of pineal peptide preparation epithalamin on free-radical processes in humans and animals. Neuro. Endocrinol. Lett. 2001, 22, 9–18. [Google Scholar]
- Khavinson, V.; Goncharova, N.; Lapin, B. Synthetic tetrapeptide epitalon restores disturbed neuroendocrine regulation in senescent monkeys. Neuro. Endocrinol. Lett. 2001, 22, 251–254. [Google Scholar]
- Kossoy, G.; Anisimov, V.N.; Ben-Hur, H.; Kossoy, N.; Zusman, I. Effect of the synthetic pineal peptide epitalon on spontaneous carcinogenesis in female C3H/He mice. In Vivo 2006, 20, 253–257. [Google Scholar] [PubMed]
- Vinogradova, I.A. Comparative study of the effects of melatonin and epitalon on the protracted memory under the shuttle labyrinth test conditions in rats in the course of aging. Eksp. Klin. Farmakol. 2006, 69, 13–16. [Google Scholar] [PubMed]
- Anisimov, V.N.; Bondarenko, L.A.; Khavinson, V. Effect of pineal peptide preparation (epithalamin) on life span and pineal and serum melatonin level in old rats. Ann. N. Y. Acad. Sci. 1992, 673, 53–57. [Google Scholar] [CrossRef]
- Anisimov, V.N.; Khavinson, V.K.; Popovich, I.G.; Zabezhinski, M.A. Inhibitory effect of peptide Epitalon on colon carcinogenesis induced by 1,2-dimethylhydrazine in rats. Cancer Lett. 2002, 183, 1–8. [Google Scholar] [CrossRef]
- Anisimov, V.N.; Khavinson, V.K.; Provinciali, M.; Alimova, I.N.; Baturin, D.A.; Popovich, I.G.; Zabezhinski, M.A.; Imyanitov, E.N.; Mancini, R.; Franceschi, C. Inhibitory effect of the peptide epitalon on the development of spontaneous mammary tumors in HER-2/neu transgenic mice. Int. J. Cancer 2002, 101, 7–10. [Google Scholar] [CrossRef]
- Khavinson, V.; Razumovsky, M.; Trofimova, S.; Grigorian, R.; Razumovskaya, A. Pineal-regulating tetrapeptide epitalon improves eye retina condition in retinitis pigmentosa. Neuroendocrinol. Lett. 2002, 23, 365–368. [Google Scholar]
- Khavinson, V.K.; Korneva, E.A.; Malinin, V.V.; Rybakina, E.G.; Pivanovich, I.Y.; Shanin, S.N. Effect of epitalon on interleukin-1beta signal transduction and the reaction of thymocyte blast transformation under stress. Neuro. Endocrinol. Lett. 2002, 23, 411–416. [Google Scholar]
- Anisimov, V.N.; Khavinson, V.K.; Popovich, I.G.; Zabezhinski, M.A.; Alimova, I.N.; Rosenfeld, S.V.; Zavarzina, N.Y.; Semenchenko, A.V.; Yashin, A.I. Effect of Epitalon on biomarkers of aging, life span and spontaneous tumor incidence in female Swiss-derived SHR mice. Biogerontology 2003, 4, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Sibarov, D.A.; Kovalenko, R.I.; Malinin, V.V.; Khavinson, V. Epitalon influences pineal secretion in stress-exposed rats in the daytime. Neuro Endocrinol. Lett. 2002, 23, 452–454. [Google Scholar] [PubMed]
- Kossoy, G.; Zandbank, J.; Tendler, E.; Anisimov, V.; Khavinson, V.; Popovich, I.; Zabezhinski, M.; Zusman, I.; Ben-Hur, H. Epitalon and colon carcinogenesis in rats: Proliferative activity and apoptosis in colon tumors and mucosa. Int. J. Mol. Med. 2003, 12, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Popovich, I.G.; Zabezhinski, M.A.; Rosenfeld, S.V.; Khavinson, V.K.; Semenchenko, A.V.; Yashin, A.I. Effect of epitalon and melatonin on life span and spontaneous carcinogenesis in senescence accelerated mice (SAM). Vopr. Onkol. 2005, 51, 93–98. [Google Scholar] [PubMed]
- Svechkina, E.B.; Tiutiunnik, N.N.; Vinogradova, I.A. Influence of light regimens, melatonin, and epitalon on amylase activity in the pancreas and small intestine in rats of different age. Adv. Gerontol. 2006, 19, 66–71. [Google Scholar] [PubMed]
- Vinogradova, I.A. Effect of preparations melatonin and epitalon on the age-related dynamics of thyrotrophic activity of the hypophysis and thyroid gland function in different light regimes. Adv. Gerontol. 2009, 22, 631–638. [Google Scholar]
- Goncharova, N.D.; Khavinson, B.K.; Lapin, B.A. Regulatory effect of Epithalon on production of melatonin and cortisol in old monkeys. Bull. Exp. Biol. Med. 2001, 131, 394–396. [Google Scholar] [CrossRef]
- Djeridane, Y.; Khavinson, V.K.; Anisimov, V.N.; Touitou, Y. Effect of a synthetic pineal tetrapeptide (Ala-Glu-Asp-GLy) on melatonin secretion by the pineal gland of young and old rats. J. Endocrinol. Invest. 2003, 26, 211–215. [Google Scholar] [CrossRef]
- Korkushko, O.V.; Lapin, B.A.; Goncharova, N.D.; Khavinson, V.K.; Shatilo, V.B.; Vengerin, A.A.; Antoniuk-Shcheglova, I.A.; Magdich, L.V. Normalizing effect of the pineal gland peptides on the daily melatonin rhythm in old monkeys and elderly people. Adv. Gerontol. 2007, 20, 74–85. [Google Scholar]
- Dilman, V.M.; Anisimov, V.N.; Ostroumova, M.N.; Khavinson, V.K.; Morozov, V.G. Increase in lifespan of rats following polypeptide pineal extract treatment. Exp. Pathol. 1979, 17, 539–545. [Google Scholar] [CrossRef]
- Dilman, V.M.; Anisimov, V.N.; Ostroumova, M.N.; Morosov, V.G.; Khavinson, V.K.; Azarova, M.A. Study of the anti-tumor effect of polypeptide pineal extract. Oncology 1979, 36, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Khavinson, V.; Morozov, V.G. Twenty years of study on effects of pineal peptide preparation: Epithalamin in experimental gerontology and oncology. Ann. N. Y. Acad. Sci. 1994, 719, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S. “I think that the small peptides are the best for healthy ageing...”, an interview with Vladimir Khavinson. Biogerontology 2013, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Khavinson, V.K.; Pronyaeva, V.E.; Linkova, N.S.; Trofimova, S.V. Peptidegic regulation of differentiation of embryonic retinal cells. Bull. Exp. Biol. Med. 2013, 155, 172–174. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Mylnikov, S.V.; Oparina, T.I.; Khavinson, V.K. Effect of melatonin and pineal peptide preparation epithalamin on life span and free radical oxidation in Drosophila melanogaster. Mech. Ageing Dev. 1997, 97, 81–91. [Google Scholar] [CrossRef]
- Anisimov, V.N.; Zavarzina, N.Y.; Zabezhinski, M.A.; Popovich, I.G.; Zimina, O.A.; Shtylick, A.V.; Arutjunyan, A.V.; Oparina, T.I.; Prokopenko, V.M.; Mikhalski, A.I.; et al. Melatonin increases both life span and tumor incidence in female CBA mice. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, B311–B323. [Google Scholar] [CrossRef] [Green Version]
- Anisimov, V.N. Effect of melatonin on life span and longevity. In Melatonin: Biological Basis of Its Function in Health and Disease, 1st ed.; Pandi-Perumal, S.R., Cardinali, D.P., Eds.; Eurekah.com/Landes Biosciences: Georgetown, TX, USA, 2006; pp. 45–59. [Google Scholar]
- Izmaylov, D.M.; Obukhova, L.K. Geroprotector effectiveness of melatonin: Investigation of lifespan of Drosophila melanogaster. Mech. Ageing Dev. 1999, 106, 233–240. [Google Scholar] [CrossRef]
- Bonilla, E.; Medina-Leendertz, S.; Díaz, S. Extension of life span and stress resistance of Drosophila melanogaster by long-term supplementation with melatonin. Exp. Gerontol. 2002, 37, 629–638. [Google Scholar] [CrossRef]
- Karbownik, M.; Reiter, R.J.; Qi, W.; Garcia, J.J.; Tan, D.X.; Manchester, L.C. Protective effects of melatonin against oxidation of guanine bases in DNA and decreased microsomal membrane fluidity in rat liver induced by whole body ionizing radiation. Mol. Cell. Biochem. 2000, 211, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Lee, R.Y.N.; Fraser, A.G.; Kamath, R.S.; Ahringer, J.; Ruvkun, G. A systematic RNAi screen identifies a critical role for mitochondria in C. elegans longevity. Nat. Genet. 2003, 33, 40–48. [Google Scholar] [CrossRef]
- Squier, T.C. Oxidative stress and protein aggregation during biological aging. Exp. Gerontol. 2001, 36, 1539–1550. [Google Scholar] [CrossRef]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef]
- Krumova, K.; Cosa, G. Chapter 1: Overview of reactive oxygen species. In Singlet Oxygen: Applications in Biosciences and Nanosciences; Nonell, S., Flors, C., Eds.; Royal Society of Chemistry: London, UK, 2016; Volume 1, pp. 1–21. [Google Scholar] [CrossRef]
- Salvioli, S.; Bonafe, M.; Capri, M.; Monti, D.; Franceschi, C. Mitochondria, aging and longevity—A new perspective. FEBS Lett. 2001, 492, 9–13. [Google Scholar] [CrossRef] [Green Version]
- Schroeder, E.A.; Raimundo, N.; Shadel, G.S. Epigenetic silencing mediates mitochondria stress-induced longevity. Cell Metab. 2013, 17, 954–964. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bornstein, R.; Gonzalez, B.; Johnson, S.C. Mitochondrial pathways in human health and aging. Mitochondrion 2020, 54, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. The free radical theory of aging. Antioxid. Redox Signal 2003, 5, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Pavelescu, L.A. On reactive oxygen species measurement in living systems. J. Med. Life 2015, 8, 38–42. [Google Scholar]
- Acuña-Castroviejo, D.; Escames, G.L.C.; Lopez, L.C.; Ortiz, F.; Lopez, A.; García, J.A. Evidencias de la utilidad de la melatonina frente al envejecimiento y los procesos neurodegenerativos. Psicogeriatría 2009, 1, 3–21. [Google Scholar]
- Tan, D.X.; Chen, L.D.; Poeggeler, B.; Manchester, L.C.; Reiter, R.J. Melatonin: A potent endogenous hydroxyl radical scavenger. Endocrine J. 1993, 1, 57–60. [Google Scholar]
- Reiter, R.J. Lowering the high price of free radicals. News Physiol. Sci. 2000, 15, 246–250. [Google Scholar] [CrossRef]
- Hardeland, R. Antioxidative protection by melatonin: Multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 2005, 27, 119–130. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Qi, W. Biochemical reactivity of melatonin with reactive oxygen and nitrogen species. Cell. Biochem. Biophys. 2001, 34, 237–256. [Google Scholar] [CrossRef]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Qin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Karasek, M. Does melatonin play a role in aging processes? J. Physiol. Pharmacol. 2007, 58, 105–113. [Google Scholar] [PubMed]
- Marshall, K.A.; Reiter, R.J.; Poeggeler, B.; Aruoma, O.I.; Halliwell, B. Evaluation of the antioxidant activity of melatonin in vitro. Free Radic. Biol. Med. 1996, 21, 307–315. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Burkhardt, S.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Shohami, E.; Huo, Y.S.; Hardeland, R.; Reiter, R.J. N1-acetyl-N2-formyl-5-methoxykynuramine, a biogenic amine and melatonin metabolite, functions as a potent antioxidant. FASEB J. 2001, 15, 2294–2296. [Google Scholar] [CrossRef]
- Tan, D.X.; Reiter, R.J.; Manchester, L.C.; Yan, M.T.; El-Sawi, M.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Allegra, M.; Hardeland, R. Chemical and physical properties and potential mechanisms: Melatonin as a broad spectrum antioxidant and free radical scavenger. Curr. Top Med. Chem. 2002, 2, 181–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ressmeyer, A.R.; Mayo, J.C.; Zelosko, V.; Sáinz, R.M.; Tan, D.X.; Poeggeler, B.; Antolín, I.; Zsizsik, B.K.; Reiter, R.J.; Hardeland, R. Antioxidant properties of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK): Scavenging of free radicals and prevention of protein destruction. Redox Rep. 2003, 8, 205–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guenther, A.L.; Schmidt, S.I.; Laatsch, H.; Fotso, S.; Ness, H.; Ressmeyer, A.R.; Poeggeler, B.; Hardeland, R. Reactions of the melatonin metabolite AMK (N1-acetyl-5-methoxykynuramine) with reactive nitrogen species: Formation of novel compounds, 3-acetamidomethyl-6-methoxycinnolinone and 3-nitro-AMK. J. Pineal Res. 2005, 39, 251–260. [Google Scholar] [CrossRef]
- Onuki, J.; Almeida, E.A.; Medeiros, M.H.; Di Mascio, P. Inhibition of 5-aminolevulinic acid-induced DNA damage by melatonin, N1-acetyl-N2-formyl-5-methoxykynuramine, quercetin or resveratrol. J. Pineal Res. 2005, 38, 107–115. [Google Scholar] [CrossRef]
- López-Lluch, G.; Irusta, P.M.; Navas, P.; de Cabo, R. Mitochondrial biogenesis and healthy aging. Exp. Gerontol. 2008, 43, 813–819. [Google Scholar] [CrossRef] [Green Version]
- Acuña-Castroviejo, D.; Carretero, M.; Doerrier, C.; López, L.C.; García-Corzo, L.; Tresguerres, J.A.; Escames, G. Melatonin protects lung mitochondria from aging. AGE 2012, 34, 681–692. [Google Scholar] [CrossRef] [Green Version]
- Reiter, L.T.; Potocki, L.; Chien, S.; Gribskov, M.; Bier, E. A systematic analysis of human disease-associated gene sequences in Drosophila melanogaster. Genome Res. 2001, 11, 1114–1125. [Google Scholar] [CrossRef] [Green Version]
- Pandey, U.B.; Nichols, C.D. Human disease models in Drosophila melanogaster and the role of the fly in therapeutic drug discovery. Pharmacol. Rev. 2011, 63, 411–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toivonen, J.M.; Partridge, L. Endocrine regulation of aging and reproduction in Drosophila. Mol. Cell. Endocrinol. 2009, 299, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Partridge, L. Promoting health and longevity through diet: From model organisms to humans. Cell 2015, 161, 106–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bjedov, I.; Toivonen, J.M.; Kerr, F.; Slack, C.; Jacobson, J.; Foley, A.; Partridge, L. Mechanisms of life span extension by rapamycin in the fruit fly Drosophila melanogaster. Cell Metab. 2010, 11, 35–46. [Google Scholar] [CrossRef] [Green Version]
- Gamberi, C.; Hipfner, D.R.; Trudel, M.; Lubell, W.D. Bicaudal C mutation causes myc and TOR pathway up-regulation and polycystic kidney disease-like phenotypes in Drosophila. PLoS Genet. 2017, 13, e1006694. [Google Scholar] [CrossRef] [Green Version]
- Grönke, S.; Clarke, D.F.; Broughton, S.; Andrews, T.D.; Partridge, L. Molecular evolution and functional characterization of Drosophila insulin-like peptides. PLoS Genet. 2010, 6, e1000857. [Google Scholar] [CrossRef] [Green Version]
- Weigelt, C.M.; Sehgal, R.; Tain, L.S.; Cheng, J.; Eßer, J.; Pahl, A.; Dieterich, C.; Grönke, S.; Partridge, L. An insulin-sensitive circular RNA that regulates lifespan in Drosophila. Mol. Cell. 2020, 79, 268–279.e5. [Google Scholar] [CrossRef]
- Mair, W.; Piper, M.D.; Partridge, L. Calories do not explain extension of life span by dietary restriction in Drosophila. PLoS Biol. 2005, 3, e223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piper, M.D.W.; Partridge, L. Drosophila as a model for ageing. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 2707–2717. [Google Scholar] [CrossRef] [PubMed]
- Khyati; Malik, I.; Agrawal, N.; Kumar, V. Melatonin and curcumin reestablish disturbed circadian gene expressions and restore locomotion ability and eclosion behavior in Drosophila model of Huntington’s disease. Chronobiol. Int. 2021, 38, 61–78. [Google Scholar] [CrossRef]
- Musiek, E.S.; Holtzman, D.M. Mechanisms linking circadian clocks, sleep, and neurodegeneration. Science 2016, 354, 1004–1008. [Google Scholar] [CrossRef] [Green Version]
- Dabbeni-Sala, F.; Di Santo, S.; Franceschini, D.; Skaper, S.D.; Giusti, P. Melatonin protects against 6-OHDA-induced neurotoxicity in rats: A role for mitochondrial complex I activity. FASEB J. 2001, 15, 164–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antolín, I.; Mayo, J.C.; Sainz, R.M.; del Brío, M.A.; Herrera, F.; Martín, V.; Rodríguez, C. Protective effect of melatonin in a chronic experimental model of Parkinson’s disease. Brain Res. 2002, 943, 163–173. [Google Scholar] [CrossRef]
- Polimeni, G.; Esposito, E.; Bevelacqua, V.; Guarneri, C.; Cuzzocrea, S. Role of melatonin supplementation in neurodegenerative disorders. Front. Biosci. 2014, 19, 429–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cabral-Costa, J.V.; Kowaltowski, A.J. Neurological disorders and mitochondria. Mol. Aspects Med. 2020, 71, 100826. [Google Scholar] [CrossRef] [PubMed]
- Sarantseva, S.; Timoshenko, S.; Bolshakova, O.; Karaseva, E.; Rodin, D.; Schwarzman, A.L.; Vitek, M.P. Apolipoprotein E-mimetics inhibit neurodegeneration and restore cognitive functions in a transgenic Drosophila model of Alzheimer’s disease. PLoS ONE 2009, 4, e8191. [Google Scholar] [CrossRef]
- Khatoon, R.; Rasheed, M.Z.; Rawat, M.; Alam, M.M.; Tabassum, H.; Parvez, S. Effect of melatonin on Aβ42 induced changes in the mitochondrial function related to Alzheimer’s disease in Drosophila melanogaster. Neurosci. Lett. 2019, 711, 134376. [Google Scholar] [CrossRef]
- Hardy, J.; Selkoe, D.J. The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 2002, 297, 353–356. [Google Scholar] [CrossRef] [Green Version]
- Parker, W.D., Jr.; Parks, J.; Filley, C.M.; Kleinschmidt-DeMasters, B.K. Electron transport chain defects in Alzheimer’s disease brain. Neurology 1994, 44, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Pappolla, M.; Chyan, Y.J.; Poeggeler, B.; Frangione, B.; Wilson, G.; Ghiso, J.; Reiter, R.J. An assessment of the antioxidant and the antiamyloidogenic properties of melatonin: Implications for Alzheimer’s disease. J. Neural Transm. 2000, 107, 203–231. [Google Scholar] [CrossRef]
- Savaskan, E.; Wirz-Justice, A.; Olivieri, G.; Pache, M.; Kräuchi, K.; Brydon, L.; Jockers, R.; Müller-Spahn, F.; Meyer, P. Distribution of melatonin MT1 receptor immunoreactivity in human retina. J. Histochem. Cytochem. 2002, 50, 519–526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brunner, P.; Sözer-Topcular, N.; Jockers, R.; Ravid, R.; Angeloni, D.; Fraschini, F.; Eckert, A.; Müller-Spahn, F.; Savaskan, E. Pineal and cortical melatonin receptors MT1 and MT2 are decreased in Alzheimer’s disease. Eur. J. Histochem. 2006, 50, 311–316. [Google Scholar]
- Roos, R.A. Huntington’s disease: A clinical review. Orphanet. J. Rare Dis. 2010, 5, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ismailoglu, I.; Chen, Q.; Popowski, M.; Yang, L.; Gross, S.S.; Brivanlou, A.H. Huntingtin protein is essential for mitochondrial metabolism, bioenergetics and structure in murine embryonic stem cells. Dev. Biol. 2014, 391, 230–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panov, A.V.; Gutekunst, C.A.; Leavitt, B.R.; Hayden, M.R.; Burke, J.R.; Strittmatter, W.J.; Greenamyre, J.T. Early mitochondrial calcium defects in Huntington’s disease are a direct effect of polyglutamines. Nat. Neurosci. 2002, 5, 731–736. [Google Scholar] [CrossRef]
- Panchal, K.; Tiwari, A.K. Mitochondrial dynamics, a key executioner in neurodegenerative diseases. Mitochondrion 2019, 47, 151–173. [Google Scholar] [CrossRef]
- Kalliolia, E.; Silajdžić, E.; Nambron, R.; Hill, N.R.; Doshi, A.; Frost, C.; Watt, H.; Hindmarsh, P.; Bjorkqvist, M.; Warner, T.T. Plasma melatonin is reduced in Huntington’s disease. Mov. Disord. 2014, 29, 1511–1515. [Google Scholar] [CrossRef] [Green Version]
- Túnez, I.; del Carmen Muñoz, M.; Feijóo, M.; Muñoz-Castañeda, J.R.; Bujalance, I.; Valdelvira, M.E.; López, P.M. Protective melatonin effect on oxidative stress induced by okadaic acid into rat brain. J. Pineal Res. 2003, 34, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Greggio, E.; Cookson, M.R. Leucine-rich repeat kinase 2 mutations and Parkinson’s disease: Three questions. ASN Neuro 2009, 1, e00002. [Google Scholar] [CrossRef] [Green Version]
- Cookson, M.R. The role of leucine-rich repeat kinase 2 (LRRK2) in Parkinson’s disease. Nat. Rev. Neurosci. 2010, 11, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Ran, D.; Zhao, X.; Huang, Y.; Long, S.; Liang, F.; Guo, W.; Nucifora, F.C., Jr.; Gu, H.; Lu, X.; et al. Melatonin attenuates hLRRK2-induced sleep disturbances and synaptic dysfunction in a Drosophila model of Parkinson’s disease. Mol. Med. Rep. 2016, 13, 3936–3944. [Google Scholar] [CrossRef] [PubMed]
- Berg, D.; Schweitzer, K.J.; Leitner, P.; Zimprich, A.; Lichtner, P.; Belcredi, P.; Brussel, T.; Schulte, C.; Maass, S.; Nagele, T.; et al. Type and frequency of mutations in the LRRK2 gene in familial and sporadic Parkinson’s disease. Brain 2005, 128, 3000–3011. [Google Scholar] [CrossRef] [Green Version]
- Parker, W.D., Jr.; Boyson, S.J.; Parks, J.K. Abnormalities of the electron transport chain in idiopathic Parkinson’s disease. Ann. Neurol. 1989, 26, 719–723. [Google Scholar] [CrossRef]
- Alam, Z.I.; Jenner, A.; Daniel, S.E.; Lees, A.J.; Cairns, N.; Marsden, C.D.; Jenner, P.; Halliwell, B. Oxidative DNA damage in the parkinsonian brain: An apparent selective increase in 8-hydroxyguanine levels in substantia nigra. J. Neurochem. 1997, 69, 1196–1203. [Google Scholar] [CrossRef]
- Sheehan, J.P.; Swerdlow, R.H.; Parker, W.D.; Miller, S.W.; Davis, R.E.; Tuttle, J.B. Altered calcium homeostasis in cells transformed by mitochondria from individuals with Parkinson’s disease. J. Neurochem. 1997, 68, 1221–1233. [Google Scholar] [CrossRef]
- Chinopoulos, C.; Adam-Vizi, V. Mitochondria deficient in complex I activity are depolarized by hydrogen peroxide in nerve terminals: Relevance to Parkinson’s disease. J. Neurochem. 2001, 76, 302–306. [Google Scholar] [CrossRef]
- Kim, I.; Rodriguez-Enriquez, S.; Lemasters, J.J. Selective degradation of mitochondria by mitophagy. Arch. Biochem. Biophys. 2007, 462, 245–253. [Google Scholar] [CrossRef] [Green Version]
- Gorman, G.S.; Chinnery, P.F.; DiMauro, S.; Hirano, M.; Koga, Y.; McFarland, R.; Suomalainen, A.; Thorburn, D.R.; Zeviani, M.; Turnbull, D.M. Mitochondrial diseases. Nat. Rev. Dis. Primers 2016, 2, 16080. [Google Scholar] [CrossRef]
- Craven, L.; Alston, C.L.; Taylor, R.W.; Turnbull, D.M. Recent advances in mitochondrial disease. Annu. Rev. Genomics Hum. Genet. 2017, 18, 257–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Finocchiaro, L.; Callebert, J.; Launay, J.M.; Jallon, J.M. Melatonin biosynthesis in Drosophila: Its nature and its effects. J. Neurochem. 1988, 50, 382–387. [Google Scholar] [CrossRef]
- Khavinson, V.K.; Izmaylov, D.M.; Obukhova, L.K.; Malinin, V.V. Effect of epitalon on the lifespan increase in Drosophila melanogaster. Mech. Ageing Dev. 2000, 120, 141–149. [Google Scholar] [CrossRef]
- Khavinson, V.K.; Myl’nikov, S.V. Effect of epithalone on the age-specific changes in the time course of lipid peroxidation in Drosophila melanogaster. Bull. Exp. Biol. Med. 2000, 130, 1116–1119. [Google Scholar] [CrossRef]
- Khavinson, V.K.; Myl’nikov, S.V.; Oparina, T.I.; Arutyunyan, A.V. Effects of peptides on generation of reactive oxygen species in subcellular fractions of Drosophila melanogaster. Bull. Exp. Biol. Med. 2001, 132, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Khavinson, V.K.; Lezhava, T.A.; Monaselidze, J.R.; Jokhadze, T.A.; Dvalishvili, N.A.; Bablishvili, N.K.; Trofimova, S.V. Peptide Epitalon activates chromatin at the old age. Neuro. Endocrinol. Lett. 2003, 24, 329–333. [Google Scholar]
- Terán, R.; Bonilla, E.; Medina-Leendertz, S.; Mora, M.; Villalobos, V.; Paz, M.; Arcaya, J.L. The life span of Drosophila melanogaster is affected by melatonin and thioctic acid. Investig. Clín. 2012, 53, 250–261. [Google Scholar]
- Gaweł, S.; Wardas, M.; Niedworok, E.; Wardas, P. Malondialdehyde (MDA) as a lipid peroxidation marker. Wiad. Lek. 2004, 57, 453–455. [Google Scholar]
- Vasiliou, V.; Ross, D.; Nebert, D.W. Update of the NAD(P)H:quinone oxidoreductase (NQO) gene family. Hum. Genom. 2006, 2, 329–335. [Google Scholar] [CrossRef] [Green Version]
- Hall, B.L.; Thummel, C.S. The RXR homolog ultraspiracle is an essential component of the Drosophila ecdysone receptor. Development 1998, 125, 4709–4717. [Google Scholar] [CrossRef]
- Ghbeish, N.; Tsai, C.C.; Schubiger, M.; Zhou, J.Y.; Evans, R.M.; McKeown, M. The dual role of ultraspiracle, the Drosophila retinoid X receptor, in the ecdysone response. Proc. Natl. Acad. Sci. USA 2001, 98, 3867–3872. [Google Scholar] [CrossRef] [Green Version]
- Picking, W.L.; Chen, D.M.; Lee, R.D.; Vogt, M.E.; Polizzi, J.L.; Marietta, R.G.; Stark, W.S. Control of Drosophila opsin gene expression by carotenoids and retinoic acid: Northern and western analyses. Exp. Eye. Res. 1996, 63, 493–500. [Google Scholar] [CrossRef]
- Arshavsky, V.Y. Vision: The retinoid cycle in Drosophila. Curr. Biol. 2010, 20, R96–R98. [Google Scholar] [CrossRef] [Green Version]
- Ashton, A.; Stoney, P.N.; Ransom, J.; McCaffery, P. Rhythmic diurnal synthesis and signaling of retinoic acid in the rat pineal gland and its action to rapidly downregulate ERK phosphorylation. Mol. Neurobiol. 2018, 55, 8219–8235. [Google Scholar] [CrossRef] [Green Version]
- Mackay, T.F.; Anholt, R.R. Of flies and man: Drosophila as a model for human complex traits. Annu. Rev. Genomics Hum. Genet. 2006, 7, 339–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koh, K.; Evans, J.M.; Hendricks, J.C.; Sehgal, A. A Drosophila model for age-associated changes in sleep: Wake cycles. Proc. Natl. Acad. Sci. USA 2006, 103, 13843–13847. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, V.; Sharma, A. Common features of circadian timekeeping in diverse organisms. Curr. Opin. Physiol. 2018, 5, 58–67. [Google Scholar] [CrossRef]
- Konopka, R.J.; Benzer, S. Clock mutants of Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 1971, 68, 2112–2116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bargiello, T.A.; Young, M.W. Molecular genetics of a biological clock in Drosophila. Proc. Natl. Acad. Sci. USA 1984, 81, 2142–2146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, P.; Zehring, W.A.; Wheeler, D.A.; Pirrotta, V.; Hadfield, C.; Hall, J.C.; Rosbash, M. Molecular analysis of the period locus in Drosophila melanogaster and identification of a transcript involved in biological rhythms. Cell 1984, 38, 701–710. [Google Scholar] [CrossRef]
- Sehgal, A.; Price, J.L.; Man, B.; Young, M.W. Loss of circadian behavioral rhythms and per RNA oscillations in the Drosophila mutant timeless. Science 1994, 263, 1603–1606. [Google Scholar] [CrossRef]
- Kaneko, M.; Park, J.H.; Cheng, Y.; Hardin, P.E.; Hall, J.C. Disruption of synaptic transmission or clock-gene-product oscillations in circadian pacemaker cells of Drosophila cause abnormal behavioral rhythms. J. Neurobiol. 2000, 43, 207–233. [Google Scholar] [CrossRef]
- Rosbash, M.; Bradley, S.; Kadener, S.; Li, Y.; Luo, W.; Menet, J.S.; Nagoshi, E.; Palm, K.; Schoer, R.; Shang, Y.; et al. Transcriptional feedback and definition of the circadian pacemaker in Drosophila and animals. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 75–83. [Google Scholar] [CrossRef] [Green Version]
- Rosbash, M. Circadian rhythms and the transcriptional feedback loop (Nobel lecture). Angew. Chem. 2021, 60, 8650–8666. [Google Scholar] [CrossRef]
- Takahashi, J.S. Transcriptional architecture of the mammalian circadian clock. Nat. Rev. Genet. 2017, 18, 164–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zordan, M.A.; Sandrelli, F. Circadian clock dysfunction and psychiatric disease: Could fruit flies have a say? Front. Neurol. 2015, 6, 80. [Google Scholar] [CrossRef] [Green Version]
- Ferrandon, D.; Imler, J.L.; Hetru, C.; Ho_mann, J.A. The Drosophila systemic immune response: Sensing and signaling during bacterial and fungal infections. Nat. Rev. Immunol. 2007, 7, 862–874. [Google Scholar] [CrossRef] [PubMed]
- Buchon, N.; Broderick, N.A.; Chakrabarti, S.; Lemaitre, B. Invasive and indigenous microbiota impact intestinal stem cell activity through multiple pathways in Drosophila. Genes Dev. 2009, 23, 2333–2344. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, T.; Nioi, P.; Pickett, C.B. The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J. Biol. Chem. 2009, 284, 13291–13295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, S.C.; Liao, C.W.; Pan, R.L.; Juang, J.L. Infection-induced intestinal oxidative stress triggers organ-to-organ immunological communication in Drosophila. Cell Host Microbe 2012, 11, 410–417. [Google Scholar] [CrossRef] [Green Version]
- Bostantjopoulou, S.; Kyriazis, G.; Katsarou, Z.; Kiosseoglou, G.; Kazis, A.; Mentenopoulos, G. Superoxide dismutase activity in early and advanced Parkinson’s disease. Funct. Neurol. 1997, 12, 63–68. [Google Scholar] [PubMed]
- Bahadorani, S.; Hilliker, A.J. Antioxidants cannot suppress the lethal phenotype of a Drosophila melanogaster model of Huntington’s disease. Genome 2008, 51, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.; Rudym, D.; Chandra, P.; Miskulin, D.; Perrone, R.; Sarnak, M. Inflammation, oxidative stress, and insulin resistance in polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 2011, 6, 7–13. [Google Scholar] [CrossRef] [Green Version]
- Kish, S.J.; Morito, C.; Hornykiewicz, O. Glutathione peroxidase activity in Parkinson’s disease brain. Neurosci. Lett. 1985, 58, 343–346. [Google Scholar] [CrossRef]
- Maser, R.L.; Vassmer, D.; Magenheimer, B.S.; Clavet, J.P. Oxidant stress and reduced antioxidant enzyme protection in polycystic kidney disease. J. Am. Soc. Nephrol. 2002, 13, 991–999. [Google Scholar] [CrossRef]
- Nikam, S.; Nikam, P.; Ahaley, S.K.; Sontakke, A.V. Oxidative stress in Parkinson’s disease. Indian J. Clin. Biochem. 2009, 24, 98–101. [Google Scholar] [CrossRef] [Green Version]
- Mason, R.P.; Casu, M.; Butler, N.; Breda, C.; Campesan, S.; Clapp, J.; Green, E.W.; Dhulkhed, D.; Kyriacou, C.P.; Giorgini, F. Glutathione peroxidase activity is neuroprotective in models of Huntington’s disease. Nat. Genet. 2013, 45, 1249–1254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zima, T.; Crkovska, J.; Merta, M.; Stípek, S.; Nĕmecek, K.; Tesar, V. Activity of the antioxidant enzyme, glutathione peroxidase, on autosomal dominant polycystic kidney disease patients. Biochem. Mol. Biol. Int. 1995, 35, 699–704. [Google Scholar]
- del Hoyo, P.; García-Redondo, A.; de Bustos, F.; Molina, J.A.; Sayed, Y.; Alonso-Navarro, H.; Caballero, L.; Arenas, J.; Jiménez-Jiménez, F.J. Oxidative stress in skin fibroblasts cultures of patients with Huntington’s disease. Neurochem. Res. 2006, 31, 1103–1109. [Google Scholar] [CrossRef]
- Nandi, A.; Yan, L.J.; Jana, C.K.; Das, N. Role of catalase in oxidative stress-and age-associated degenerative diseases. Oxid. Med. Cell. Longev. 2019, 2019, 9613090. [Google Scholar] [CrossRef] [Green Version]
- Torres, V.E.; Bengal, R.J.; Litwiller, R.D.; Wilson, D.M. Aggravation of polycystic kidney disease in Han: SPRD rats by buthionine sulfoximine. J. Am. Soc. Nephrol. 1997, 8, 1283–1291. [Google Scholar] [CrossRef]
- Ilic, T.V.; Jovanovic, M.; Jovicic, A.; Tomovic, M. Oxidative stress indicators are elevated in de novo Parkinson’s disease patients. Funct. Neurol. 1999, 14, 141–147. [Google Scholar]
- Martin, H.L.; Teismann, P. Glutathione—A review on its role and significance in Parkinson’s disease. FASEB J. 2009, 23, 3263–3272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peña-Sánchez, M.; Riverón-Forment, G.; Zaldívar-Vaillant, T.; Soto-Lavastida, A.; Borrero-Sánchez, J.; Lara-Fernández, G.; Esteban-Hernandez, E.M.; Hernandez-Diaz, Z.; Gonzalez-Quevedo, A.; Fernandez-Almirall, I.; et al. Association of status redox with demographic, clinical and imaging parameters in patients with Huntington’s disease. Clin. Biochem. 2015, 48, 1258–1263. [Google Scholar] [CrossRef] [PubMed]
- Darvesh, A.S.; Carroll, R.T.; Bishayee, A.; Novotny, N.A.; Geldenhuys, W.J.; Van der Schyf, C.J. Curcumin and neurodegenerative diseases: A perspective. Expert Opin. Investig. Drugs. 2012, 21, 1123–1140. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, N.; Rakshit, K.; Chow, E.S.; Wentzell, J.S.; Kretzschmar, D.; Giebultowicz, J.M. Loss of circadian clock accelerates aging in neurodegeneration-prone mutants. Neurobiol. Dis. 2012, 45, 1129–1135. [Google Scholar] [CrossRef] [Green Version]
- Mattis, J.; Sehgal, A. Circadian rhythms, sleep, and disorders of aging. Trends Endocrinol. Metab. 2016, 27, 192–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hickey, M.A.; Zhu, C.; Medvedeva, V.; Lerner, R.P.; Patassini, S.; Franich, N.R.; Maiti, P.; Frautschy, S.A.; Zeitlin, S.; Levine, M.S.; et al. Improvement of neuropathology and transcriptional deficits in CAG 140 knock-in mice supports a beneficial effect of dietary curcumin in Huntington’s disease. Mol. Neurodegener. 2012, 7, 12. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.B.; Kim, W.; Lee, S.; Chung, J. Loss of LRRK2/PARK8 induces degeneration of dopaminergic neurons in Drosophila. Biochem. Biophys. Res. Commun. 2007, 358, 534–539. [Google Scholar] [CrossRef]
- Ambani, L.M.; Van Woert, M.H.; Murphy, S. Brain peroxidase and catalase in Parkinson disease. Arch. Neurol. 1975, 32, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Ran, D.; Xie, B.; Gan, Z.; Sun, X.; Gu, H.; Yang, J. Melatonin attenuates hLRRK2-induced long-term memory deficit in a Drosophila model of Parkinson’s disease. Biomed. Rep. 2018, 9, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Hely, M.A.; Morris, J.G.; Reid, W.G.; Trafficante, R. Sydney multicenter study of Parkinson’s disease: Non-L-dopa–responsive problems dominate at 15 years. Mov. Disord. 2005, 20, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Spuch, C.; Ortolano, S.; Navarro, C. New insights in the amyloid-Beta interaction with mitochondria. J. Aging Res. 2012, 2012, 324968. [Google Scholar] [CrossRef] [Green Version]
- He, H.; Dong, W.; Huang, F. Anti-amyloidogenic and anti-apoptotic role of melatonin in Alzheimer disease. Curr. Neuropharmacol. 2010, 8, 211–217. [Google Scholar] [CrossRef] [Green Version]
- Millet-Boureima, C.; Rozencwaig, R.; Polyak, F.; Gamberi, C. Cyst reduction by melatonin in a novel Drosophila model of polycystic kidney disease. Molecules 2020, 25, 5477. [Google Scholar] [CrossRef]
- Bergmann, C.; Guay-Woodford, L.M.; Harris, P.C.; Horie, S.; Peters, D.J.M.; Torres, V.E. Polycystic kidney disease. Nat. Rev. Dis. Primers 2018, 4, 50. [Google Scholar] [CrossRef]
- Millet-Boureima, C.; He, S.; Le, T.B.U.; Gamberi, C. Modeling neoplastic growth in renal cell carcinoma and polycystic kidney disease. Int. J. Mol. Sci. 2021, 22, 3918. [Google Scholar] [CrossRef]
- Gamberi, C.; Lasko, P. The Bic-C family of developmental translational regulators. Comp. Funct. Genom. 2012, 2012, 141386. [Google Scholar] [CrossRef] [Green Version]
- Trudel, M.; Lanoix, J.; Barisoni, L.; Blouin, M.J.; Desforges, M.; L’Italien, C.; D’Agati, V. C-myc–induced apoptosis in polycystic kidney disease is Bcl-2 and p53 independent. J. Exp. Med. 1997, 186, 1873–1884. [Google Scholar] [CrossRef] [Green Version]
- Fan, L.X.; Zhou, X.; Sweeney, W.E.; Wallace, D.P.; Avner, E.D.; Grantham, J.J.; Li, X. Smac-mimetic-induced epithelial cell death reduces the growth of renal cysts. J. Am. Soc. Nephrol. 2013, 24, 2010–2022. [Google Scholar] [CrossRef] [Green Version]
- Millet-Boureima, C.; Chingle, R.; Lubell, W.D.; Gamberi, C. Cyst reduction in a polycystic kidney disease Drosophila model using Smac mimics. Biomedicines 2019, 7, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Kean, L.; Yang, J.; Allan, A.K.; Davies, S.A.; Herzyk, P.; Dow, J.A.T. Function-informed transcriptome analysis of Drosophila renal tubule. Genome Biol. 2004, 5, R69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Millet-Boureima, C.; Porras, J.M.; Gamberi, C. Modeling renal disease “on the fly”. BioMed Res. Int. 2018, 2018, 5697436. [Google Scholar] [CrossRef] [Green Version]
- Sozen, M.A.; Armstrong, J.D.; Yang, M.; Kaiser, K.; Dow, J.A.T. Functional domains are specified to single-cell resolution in a Drosophila epithelium. Proc. Natl. Acad. Sci. USA 1997, 94, 5207–5212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saberi, K.; Pasbakhsh, P.; Omidi, A.; Borhani-Haghighi, M.; Nekoonam, S.; Omidi, N.; Ghasemi, S.; Kashani, I.R. Melatonin preconditioning of bone-marrow derived mesenchymal stem cells promotes their engraftment and improves renal regeneration in a rat model of chronic kidney disease. J. Mol. Hist. 2019, 50, 129–140. [Google Scholar] [CrossRef]
- Zhao, L.; Hu, C.; Zhang, P.; Jiang, H.; Chen, J. Melatonin preconditioning is an effective strategy for mesenchymal stem cell-based therapy for kidney disease. J. Cell. Mol. Med. 2019, 24, 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Gong, Y.; Xiong, K.; Ye, Y.; Xiong, Y.; Zhuang, Z.; Luo, Y.; Jiang, Q.; He, F. Melatonin mediates protective effects on inflammatory response induced by interleukin-1 beta in human mesenchymal stem cells. J. Pineal Res. 2013, 55, 14–25. [Google Scholar] [CrossRef]
- Klawitter, J.; Reed-Gitomer, B.Y.; McFann, K.; Pennington, A.; Klawitter, J.; Abebe, K.Z.; Klepacki, J.; Cadnapaphornchai, M.A.; Brosnahan, G.; Chonchoi, M.; et al. Endothelial dysfunction and oxidative stress in polycystic kidney disease. Am. J. Physiol. Renal Physiol. 2014, 307, F1198–F1206. [Google Scholar] [CrossRef] [Green Version]
- Andries, A.; Daenen, K.; Jouret, F.; Bammens, B.; Mekahli, D.; Van Schepdael, A. Oxidative stress in autosomal dominant polycystic kidney disease: Player and/or early predictor for disease progression? Pediatr. Nephrol. 2019, 34, 993–1008. [Google Scholar] [CrossRef] [Green Version]
- Ishimoto, Y.; Inagi, R.; Yoshihara, D.; Kugita, M.; Nagao, S.; Shimizu, A.; Takeda, N.; Wake, M.; Honda, K.; Zhou, J.; et al. Mitochondrial abnormality facilitates cyst formation in autosomal dominant polycystic kidney disease. Mol. Cell. Biol. 2017, 37, e00337-17. [Google Scholar] [CrossRef] [Green Version]
- Padovano, V.; Podrini, C.; Boletta, A.; Caplan, M.J. Metabolism and mitochondria in polycystic kidney disease research and therapy. Nat. Rev. Nephrol. 2018, 14, 678–687. [Google Scholar] [CrossRef]
- Peterson, K.M.; Franchi, F.; Loeffler, D.L.; Psaltis, P.J.; Harris, P.C.; Lerman, L.O.; Lerman, A.; Rodriguez-Porcel, M. Endothelial dysfunction occurs prior to clinical evidence of polycystic kidney disease. Am. J. Nephrol. 2013, 38, 233–240. [Google Scholar] [CrossRef] [Green Version]
- Mehrzadi, M.H.; Hosseinzadeh, A.; Juybari, K.B.; Mehrzadi, S. Melatonin and urological cancers: A new therapeutic approach. Cancer Cell Int. 2020, 20, 444. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.L.; Zhang, B.L.; Reiter, R.J.; Liu, Y.X. Melatonin ameliorates inflammation and oxidative stress by suppressing the p38MAPK signaling pathway in LPS-induced sheep orchitis. Antioxidants 2020, 9, 1277. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, Z.; Sun, H.; Song, J.; Chen, X.; Huang, J.; Lin, X.; Zhou, R. Effect of melatonin on T/B cell activation and immune regulation in pinealectomy mice. Life Sci. 2020, 242, 117191. [Google Scholar] [CrossRef] [PubMed]
- Zhen, G.; Liang, W.; Jia, H.; Zheng, X. Melatonin relieves sepsis-induced myocardial injury via regulating JAK2/STAT3 signaling pathway. Minerva Med. 2020. [Google Scholar] [CrossRef]
- Ma, S.; Zhu, L.; Fan, X.; Luo, T.; Liu, D.; Liang, Z.; Hu, X.; Shi, T.; Tan, W.; Wang, Z. Melatonin derivatives combat with inflammation-related cancer by targeting the Main Culprit STAT3. Eur. J. Med. Chem. 2021, 211, 113027. [Google Scholar] [CrossRef]
- Lu, K.H.; Su, S.C.; Lin, C.W.; Hsieh, Y.H.; Lin, Y.C.; Chien, M.H.; Reiter, R.J.; Yang, S.F. Melatonin attenuates osteosarcoma cell invasion by suppression of C-C motif chemokine ligand 24 through inhibition of the c-Jun N-terminal kinase pathway. J. Pineal Res. 2018, 65, e12507. [Google Scholar] [CrossRef]
- Wei, J.; Wang, Y.; Qi, X.; Fan, Z.; Wu, Y. Melatonin ameliorates hyperglycaemia-induced renal inflammation by inhibiting the activation of TLR4 and TGF-b1/Smad3 signalling pathway. Am. J. Transl. Res. 2020, 12, 1584–1599. [Google Scholar]
- Seeger-Nukpezah, T.; Geynisman, D.M.; Nikonova, A.S.; Benzing, T.; Golemis, E.A. The hallmarks of cancer: Relevance to the pathogenesis of polycystic kidney disease. Nat. Rev. Nephrol. 2015, 11, 515–534. [Google Scholar] [CrossRef] [Green Version]
- Rowe, I.; Chiaravalli, M.; Mannella, V.; Ulisse, V.; Quilici, G.; Pema, M.; Song, X.W.; Xu, H.; Mari, S.; Qian, F.; et al. Defective glucose metabolism in polycystic kidney disease identifies a new therapeutic strategy. Nat. Med. 2013, 19, 488–493. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, M.; Li, L.; Chen, L. Involvement of the Warburg effect in non-tumor diseases processes. J. Cell Physiol. 2018, 233, 2839–2849. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Chandel, N.S. We need to talk about the Warburg effect. Nat. Metab. 2020, 2, 127–129. [Google Scholar] [CrossRef]
- Reiter, R.J.; Sharma, R.; Ma, Q. Switching diseased cells from cytosolic aerobic glycolysis to mitochondrial oxidative phosphorylation: A metabolic rhythm regulated by melatonin? J. Pineal Res. 2021, 70, e12677. [Google Scholar] [CrossRef]
- de Lima Mota, A.; Jardim-Perassi, B.V.; de Castro, T.B.; Colombo, J.; Sonehara, N.M.; Nishiyama, V.K.G.; Pierri, V.A.G.; de Campos Zuccari, D.A.P. Melatonin modifies tumor hypoxia and metabolism by inhibiting HIF-1a and energy metabolic pathway in the in vitro and in vivo models of breast cancer. Melatonin Res. 2019, 2, 83–98. [Google Scholar] [CrossRef]
- Millet-Boureima, C.; Selber-Hnatiw, S.; Gamberi, C. Drug discovery and chemical probing in Drosophila. Genome 2021, 64, 147–159. [Google Scholar] [CrossRef]
- Wilson, S.T.; Blask, D.E.; Lemus-Wilson, A.M. Melatonin augments the sensitivity of MCF-7 human breast cancer cells to tamoxifen in vitro. J. Clin. Endocrinol. Metab. 1992, 75, 669–670. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Sainz, R.M.; Mayo, J.C.; Lopez-Burillo, S. Melatonin: Reducing the toxicity and increasing the efficacy of drugs. J. Pharm. Pharmacol. 2002, 54, 1299–1321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lissoni, P.; Brivio, F.; Fumagalli, L.; Messina, G.; Vigore, L.; Parolini, D.; Colciago, M.; Rovelli, F. Neuroimmunomodulation in medical oncology: Application of psychoneuroimmunology with subcutaneous low-dose IL-2 and the pineal hormone melatonin in patients with untreatable metastatic solid tumors. Anticancer Res. 2008, 28, 1377–1381. [Google Scholar] [PubMed]
- Ruiz-Rabelo, J.; Vazquez, R.; Arjona, A.; Perea, D.; Montilla, P.; Tunez, I.; Muntane, J.; Padillo, J. Improvement of capecitabine antitumoral activity by melatonin in pancreatic cancer. Pancreas 2011, 40, 410–414. [Google Scholar] [CrossRef]
- Kim, J.H.; Jeong, S.J.; Kim, B.; Yun, S.M.; Choi, D.Y.; Kim, S.H. Melatonin synergistically enhances cisplatin-induced apoptosis via the dephosphorylation of ERK/p90 ribosomal S6 kinase/heat shock protein 27 in SK-OV-3 cells. J. Pineal Res. 2012, 52, 244–252. [Google Scholar] [CrossRef]
- Margheri, M.; Pacini, N.; Tani, A.; Nosi, D.; Squecco, R.; Dama, A.; Masala, E.; Francini, F.; Zecchi-Orlandini, S.; Formigli, L. Combined effects of melatonin and all-trans retinoic acid and somatostatin on breast cancer cell proliferation and death: Molecular basis for the anticancer effect of these molecules. Eur. J. Pharmacol. 2012, 681, 34–43. [Google Scholar] [CrossRef]
- Uguz, A.C.; Cig, B.; Espino, J.; Bejarano, I.; Naziroglu, M.; Rodriguez, A.B.; Pariente, J.A. Melatonin potentiates chemotherapy-induced cytotoxicity and apoptosis in rat pancreatic tumor cells. J. Pineal Res. 2012, 53, 91–98. [Google Scholar] [CrossRef]
- Jung, J.H.; Sohn, E.J.; Shin, E.A.; Lee, D.; Kim, B.; Jung, D.B.; Kim, J.H.; Yun, M.; Lee, H.J.; Park, Y.K.; et al. Melatonin suppresses the expression of 45S preribosomal RNA and upstream binding factor and enhances the antitumor activity of puromycin in MDA-MB-231 breast cancer cells. Evid.-Based Complement. Altern. Med. 2013, 2013, 879746. [Google Scholar] [CrossRef] [PubMed]
- Kosar, P.A.; Naziroglu, M.; Ovey, I.S.; Cig, B. Synergic e_ects of doxorubicin and melatonin on apoptosis and mitochondrial oxidative stress in MCF-7 breast cancer cells: Involvement of TRPV1 channels. J. Membr. Biol. 2016, 249, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.M.; Woo, S.H.; Oh, S.T.; Hong, S.E.; Choe, T.B.; Ye, S.K.; Kim, E.K.; Seong, M.K.; Kim, H.A.; Noh, W.C.; et al. Melatonin enhances arsenic trioxide-induced cell death via sustained upregulation of Redd1 expression in breast cancer cells. Mol. Cell. Endocrinol. 2016, 422, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Ho_man, K.; Gao, C.; Petrulionis, M.; Herr, I.; Schemmer, P. Melatonin promotes sorafenib-induced apoptosis through synergistic activation of JNK/c-jun pathway in human hepatocellular carcinoma. J. Pineal Res. 2017, 62, e12398. [Google Scholar] [CrossRef]
- Torres, V.E.; Chapman, A.B.; Devuyst, O.; Gansevoort, R.T.; Grantham, J.J.; Higashihara, E.; Perrone, R.D.; Krasa, H.B.; Ouyang, J.; Czerwiec, F.S. Tolvaptan in patients with autosomal dominant polycystic kidney disease. N. Engl. J. Med. 2012, 367, 2407–2418. [Google Scholar] [CrossRef] [Green Version]
- Torres, V.E.; Chapman, A.B.; Devuyst, O.; Gansevoort, R.T.; Perrone, R.D.; Koch, G.; Ouyang, J.; McQuade, R.D.; Blais, J.D.; Czerwiec, F.S.; et al. Tolvaptan in later-stage autosomal dominant polycystic kidney disease. N. Engl. J. Med. 2017, 377, 1930–1942. [Google Scholar] [CrossRef]
- Torres, V.E.; Chapman, A.B.; Devuyst, O.; Gansevoort, R.T.; Perrone, R.D.; Dandurand, A.; Ouyang, J.; Czerwiec, F.S.; Blais, J.D. Multicenter, open-label, extension trial to evaluate the long-term efficacy and safety of early versus delayed treatment with tolvaptan in autosomal dominant polycystic kidney disease: The TEMPO 4:4 Trial. Nephrol. Dial. Transplant 2018, 33, 477–489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, S.; Luoma, S.E.; St Armour, G.E.; Thakkar, E.; Mackay, T.F.C.; Anholt, R.R.H. A Drosophila model for toxicogenomics: Genetic variation in susceptibility to heavy metal exposure. PLoS Genet. 2017, 13, e1006907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Affleck, J.G.; Walker, V.K. Drosophila as a model for developmental toxicology: Using and extending the Drosophotoxicology model. Methods Mol. Biol. 2019, 1965, 139–153. [Google Scholar] [CrossRef]
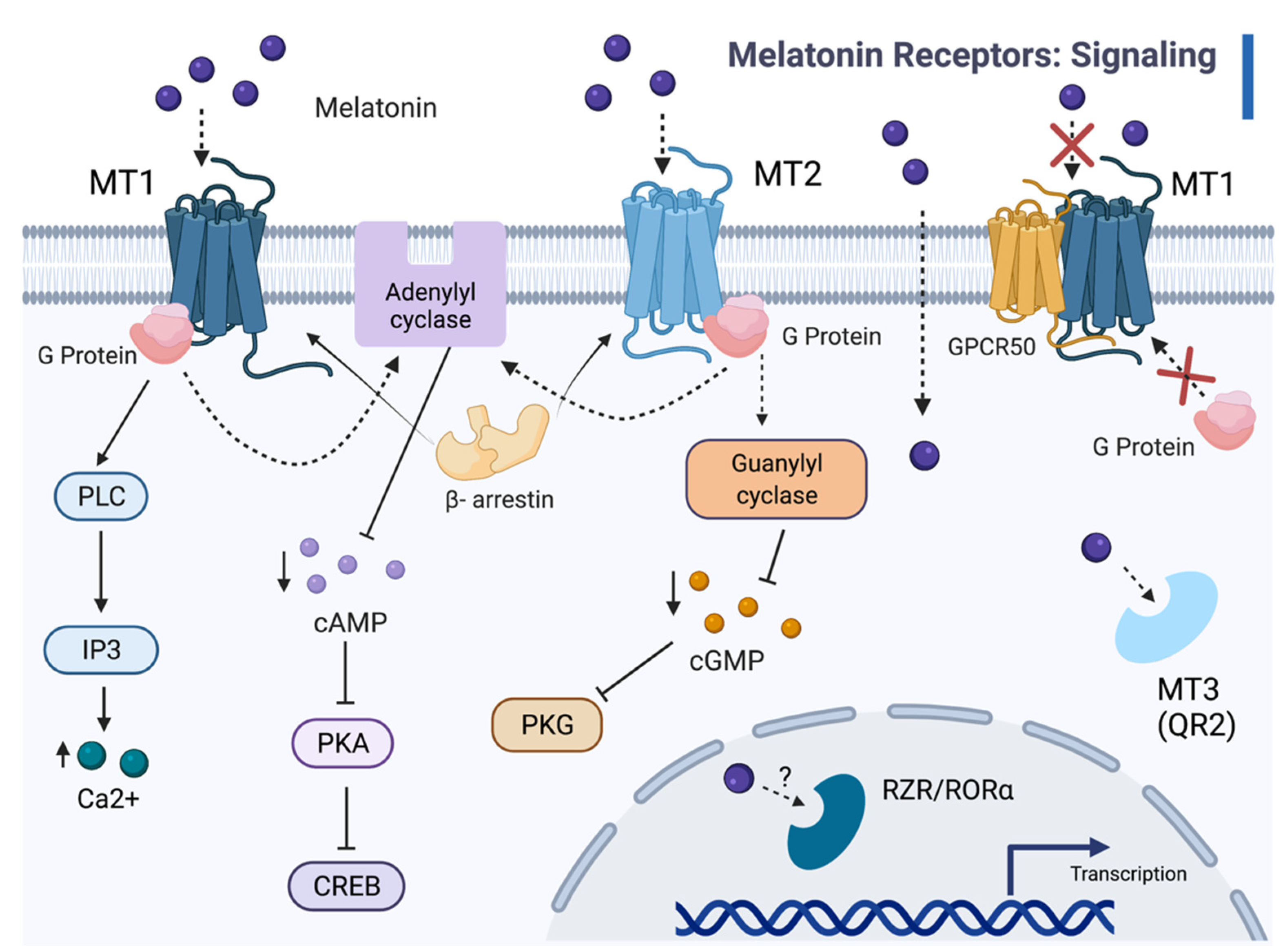
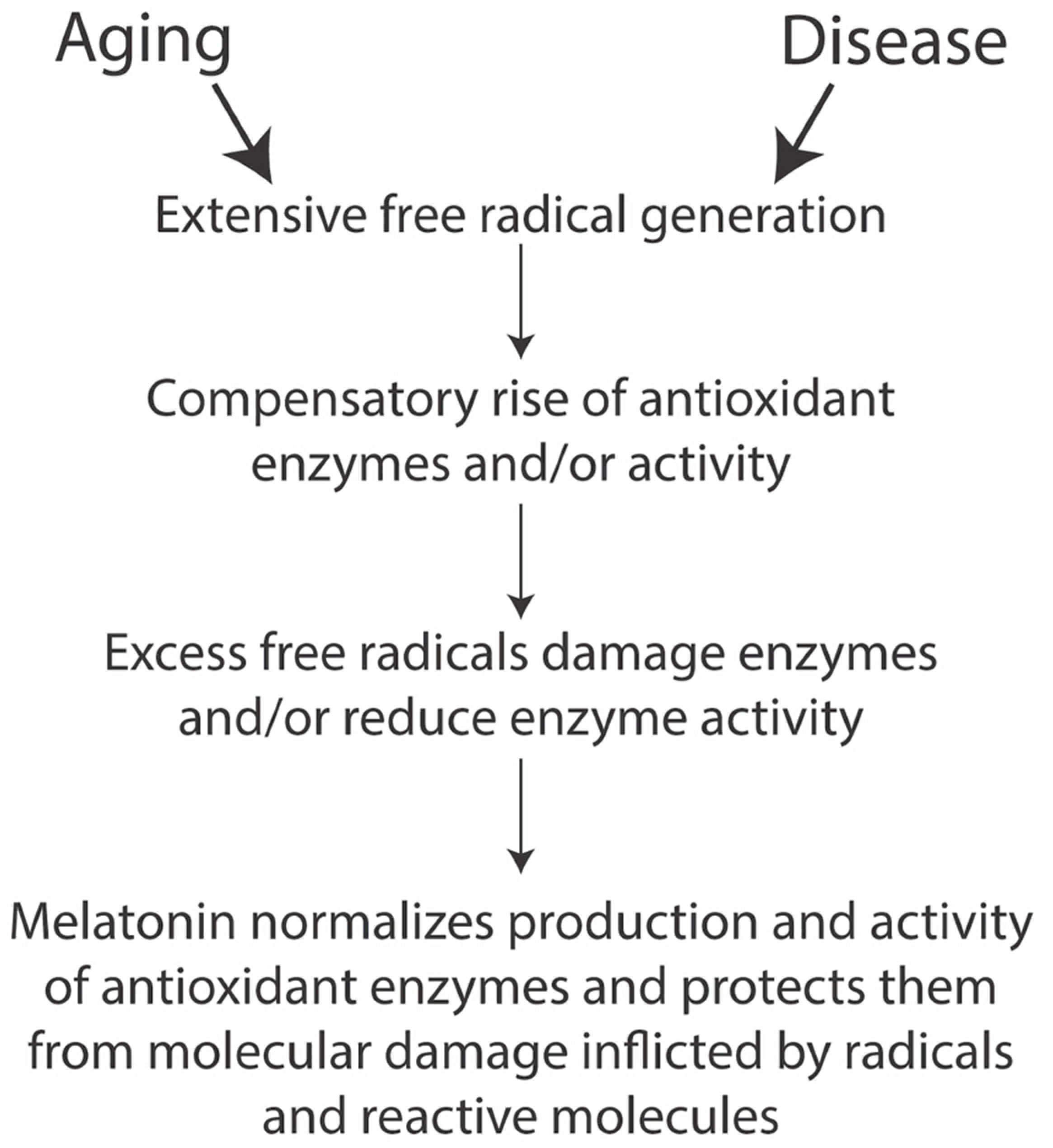
| Receptor | Target | Function | Reference(s) |
|---|---|---|---|
| MT1 | Pituitary | Decreases functions of luteinizing hormone (LH), follicle-stimulating hormone (FSH), and prolactin (PRL) | [65,66,67,68] |
| MT1 | Testes | Decreases testosterone | [69] |
| MT1 | Adrenal cortex | Reduces cortisol secretion | [70,71] |
| MT1 | Metabolism | Limits insulin secretion and increases leptin production | [72,73] |
| MT1/MT2 | Vasculature system | MT1 inhibits vasoconstriction; MT2 inhibits vasodilation | [74,75,76,77,78] |
| MT1/MT2 | Cancer cells | MT1 reduces proliferation of cancer cells; MT2 reduces the proliferation of JAr cells | [79,80] |
| MT1/MT2/MT3 | Immune system | MT1 counteracts prostaglandin E (PGE) inhibition of interleukin (IL-2) production; MT2 increases B splenocyte proliferation, anti-keyhole limpet hemocyanin (KLH) IgG levels and decreases leukocyte rolling; MT3 is responsible for leukocyte adhesion | [81,82,83,84,85] |
| MT1 | Mammalian pituitary and SCN | Inhibition of cAMP accumulation by pertussis toxin (PTX)-sensitive G proteins | [26,41,48,86,87] |
| MT1 | Melatonin binding activates Gi proteins which inhibits adneylyl cyclase activity and decreases cAMP | [38,46] | |
| MT1 | Inner Rectifier Potassium (Kir) channels, phosphorylation of mitogen-activated protein kinase (MAPK), and extracellular signal-regulated kinase 1 and 2 | Increases potassium conductance | [38] |
| MT2 | SCN | Inhibits forskolin-stimulated cAMP production in addition to cGMP formation and activation of PKC | [38] |
| MT1 | Activation of multiple types of G proteins | [34] | |
| MT2 | Inhibition of adenylyl cyclase and guanylyl cyclase, as well as phosphoinositide production | [48,88] |
| Enzymes | Drosophila Homolog | HD | PD | ADPKD | References |
|---|---|---|---|---|---|
| Superoxide dismutase (SOD) | Sod | No effect on neurodegeneration | Reduced activity as PD progresses | Reduced levels | [295,296,297] |
| Glutathione peroxidase (GPx) | PHGPx | Neuroprotective in different models of HD | Reduced activity possibly leading to dopamine neuron loss | Reduced activity | [298,299,300,301] |
| Catalase (CAT) | Cat | Reduced activity | Low activity possibly due to CAT inhibition by α-synuclein | Reduced activity | [302,303,304] |
| Glutathione reductase (GR) | - | Reduced activity causing redox imbalance | Increased levels suggesting attempt to maintain glutathione levels | Activity inhibited by acid pH in proximal tubules | [305,306,307,308] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Millet-Boureima, C.; Ennis, C.C.; Jamison, J.; McSweeney, S.; Park, A.; Gamberi, C. Empowering Melatonin Therapeutics with Drosophila Models. Diseases 2021, 9, 67. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases9040067
Millet-Boureima C, Ennis CC, Jamison J, McSweeney S, Park A, Gamberi C. Empowering Melatonin Therapeutics with Drosophila Models. Diseases. 2021; 9(4):67. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases9040067
Chicago/Turabian StyleMillet-Boureima, Cassandra, Caroline C. Ennis, Jurnee Jamison, Shana McSweeney, Anna Park, and Chiara Gamberi. 2021. "Empowering Melatonin Therapeutics with Drosophila Models" Diseases 9, no. 4: 67. https://0-doi-org.brum.beds.ac.uk/10.3390/diseases9040067







