1. Introduction
Fresh fruit and vegetables are unique in being the only food group that remain living entities during postharvest handling and marketing. It is readily accepted that produce are living, when attached to the parent plant, but after harvest they still need to continue all the same metabolic systems in order to maintain cellular function and integrity. These systems require energy that is generated through respiration involving the oxidation of sugars and other substrates with the loss of carbon as CO2 and water by transpiration. However, the act of harvesting prevents the replenishment of nutrients from the parent plant or by photosynthesis. Thus, over time, the ability of all produce to sustain normal metabolism declines which leads to increasing cellular dysfunction. From a marketing perspective, this results in loss of quality due to changes in appearance, texture or taste and enhanced susceptibility to microbial infection. Fresh fruit and vegetables are thus classified as perishables.
The need for some innovation to slow the rate of metabolism and thus extend the postharvest life of fruit and vegetables arises when produce are marketed some distance from the production area or where an economic advantage can be gained by extending to marketing period. Indeed, the current situation in many countries is for a wide range of produce to be traded internationally and consumers demanding year-round availability for seasonal commodities, situations that have exacerbated the requirement for postharvest technology.
The primary technological intervention to extend the marketing period of fruit and vegetables is to reduce the temperature of produce. The benefit of storage at reduced temperature has long been known in countries with a cold winter. For example, storage of Chinese cabbages in China and apples in North America and Europe in cellars or caves was practiced at village and farm level and provided a much needed food source during winter. The advent of mechanical refrigeration in the 1850s transformed the use of low-temperature storage into a reliable technology. Low temperature refrigerated storage of food was well established at the beginning of the twentieth century, with apples and pears being the major stored horticultural crops [
1,
2].
Numerous research studies around the world have now identified the optimum low temperature at which individual fruit and vegetables achieve maximum postharvest life and comprehensive lists of recommended storage temperatures for individual produce have been published, for example by the USDA [
3]. These recommendations are based on a reduction in metabolism being optimal just above the freezing temperature of about −1 °C, but the recommended temperature is higher for produce where abnormal metabolism leads to chilling-related injury, physiological disorders, or failure to fully ripen. The gold standard for the postharvest handling of fruit and vegetables is now considered to be cool-chain management where the optimum temperature is consistently maintained around produce at all stages from the farm to wholesale, retail and the consumer, and in the transport systems linking these destinations.
However, cool-chain management is energy-intensive. A review on energy usage in the postharvest horticultural industry by East [
4] identified that it utilised a significant proportion of the total energy consumption. Cited examples included that cooling and storage of fresh horticultural produce in California consumed about 1 billion kWh of energy annually, and marketing of fruit and vegetables accounted for about 2.5% of total UK CO
2 emissions. Refrigerated storage became
de rigeur when energy was cheap and concern about greenhouse gas emissions did not exist. The world has moved to an energy minimization mode and horticulture, in common with many other industries, needs to reduce its carbon footprint from an economic perspective, due to the increasing cost of energy, and to be seen by the community as a good corporate citizen.
In an energy-conscious world, it is appropriate to ask if there is an alternative methodology that can reduce the use of refrigeration across a wide range of produce. The option proposed in this paper is to reduce the impact of ethylene on postharvest metabolism by either reducing exposure to ethylene, or inhibiting ethylene action. It will review data available for a wide range of produce on the effect of ethylene concentration and its interaction with temperature in postharvest life with the aim of quantifying a reduced need for refrigeration. Technologies available to utilise reduced ethylene and barriers to implementation will also be discussed.
2. Function and Presence of Ethylene in Fruit and Vegetable Environments
Ethylene (CH
2=CH
2) is notable as the defining metabolic difference between plant and animal systems. It is synthesized by plants and is universally accepted as having a major regulatory role in germination, growth, development and senescence of all plants at quite low concentrations. Postharvest effects of ethylene on fruit and vegetables have been known for many centuries, even if its identity remained unknown due to inadequate analytical techniques. It was not until the 1930s that ethylene was identified with Gane [
5] reporting ethylene synthesis by plants and Crocker et al. [
6] proposing ethylene as the agent responsible for fruit ripening and senescence of vegetative tissues. The advent of gas chromatography in the 1960s provided the analytical sensitivity to below parts per million (µL L
−1) levels, which enabled expanded research efforts into the biosynthesis of ethylene and its presence in produce and postharvest situations.
Any intervention technology needs to recognize the differing role of ethylene in: (1) initiating ripening in climacteric fruit; and (2) accelerating senescence of non- climacteric fruit and vegetables [
7,
8]. Climacteric fruits have a distinct ripening pattern, with the defining characteristic being a pronounced increase in respiration to a peak value or climacteric. They are often harvested at a mature but unripe state, with the aim of preventing ripening during storage and transport to markets and then ripened close to consumer outlets. By contrast, non-climacteric produce, which can be fruits or non-seed-bearing vegetables, are harvested at or close to desirable eating quality with no dramatic changes occurring after harvest. The response of non-climacteric produce to increased ethylene is an acceleration of the rate of normal senescence. However, for both climacteric and non-climacteric produce, the presence of ethylene during marketing reduces the marketing life.
While all fruits and vegetables synthesise ethylene, albeit at different rates [
9], in a commercial postharvest environment, the source of ethylene affecting a produce can also be from other produce held in the same chamber. In addition, ethylene in the ambient air can be generated from a range of industrial sources, such as motor vehicle exhaust, natural gas leakage, or from a range of industrial effluent sources [
10]. The rapid diffusivity of ethylene into plant tissues means that all exogenous sources of ethylene are as effective as endogenous ethylene in modifying metabolic and physiological behaviour. In order to consider methods of ameliorating the effects of ethylene, it is therefore important to know the actual concentration of ethylene that produce are subject to during storage and marketing.
However, in the relatively few published case studies on ethylene concentrations throughout the postharvest chain have found measureable but widely varying level at all operational phases. Morris et al. [
11] determined the ethylene concentration in cartons of lettuce at nine stages from the field through to domestic refrigerator and the range from 455 measurements was 0.01 to 2.95 µL L
−1 with a mean ethylene level during postharvest chain of 0.20 µL L
−1. Schouten [
12] in a smaller trial surveyed the level of ethylene in a shipment of mixed vegetables at six times during a 58 h transport in a closed truck and a ventilated trailer and found mean ethylene values of 5.9 and 0.68 µL L
−1, respectively. A study by Wills et al. [
13] conducted over a three-year period determined the ethylene concentration in 700 atmospheric samples collected from handling and storage areas in the Sydney fruit and vegetables wholesale markets, produce distribution centres, supermarket retail stores and domestic refrigerators. They found a measureable ethylene concentration, defined as >0.005 µL L
−1 which was the limit of detection of their gas chromatograph, in all samples. A concentration of 0.1 µL L
−1 was found to be common in many areas with levels of 0.2 to 1 µL L
−1 in fruit storage chambers. It is worth noting that an ethylene concentration of 0.1 µL L
−1 has long been cited as the threshold atmospheric level below which no physiological effects are induced [
9,
14] but the concept was developed at a time when this concentration was close to the prevailing analytical sensitivity.
A series of laboratory studies was conducted by Wills and colleagues [
15,
16] to determine the effect on postharvest quality of holding a wide range of produce in ethylene concentrations that ranged from 0.001 to 10 µL L
−1. They found a progressive increase in postharvest life across 30 types of produce as the ethylene concentration was reduced. The relationship for all produce was a linear increase in postharvest life with logarithmic decrease in ethylene. Examples of data from some of their produce are illustrated in
Figure 1.
Pranamornkith et al. [
17] conducted a similar study with kiwifruit stored at 1.5 °C and found fruit ripening was progressively delayed as the ethylene concentration was decreased in a logarithmic manner from 1 to 0.001 µL L
−1. These findings imply there is no “safe” level of ethylene that does not cause a deleterious effect on postharvest life. Hence, for any current postharvest situation, any reduction in ethylene concentration will result in an increase in postharvest life.
3. Interaction of Temperature and Ethylene Concentration on Postharvest Life
If ethylene management is to be a partial or complete substitute for low temperature refrigeration, it is necessary to know the relationship between temperature and ethylene concentration in ability to inhibit ripening of individual climacteric fruit and senescence of non-climacteric fruit and vegetables.
3.1. Ripening of Bananas
Banana is a climacteric fruit that is harvested in most countries when mature but unripe with marketing system geared to transporting the fruit in an unripe condition and ripened with ethylene on arrival at the target market. Bananas are among the most internationally traded fruit crops with 22 million tonnes exported in 2020 with substantial imports into temperate countries in Europe, USA and Japan from a range of tropical countries [
18]. This long-distance transport of bananas is by sea under refrigeration at 13–15 °C in order to prevent ripening in transit [
19]. In addition, in a large country such as Australia, banana production occurs a considerable distance from major urban centres and refrigerated road trucks at 14–16 °C is the standard mode of transport [
20]. The energy used in such transportation is considerable and Lescot [
21] has estimated that the refrigerated shipping of bananas by sea is the largest source of greenhouse gas emissions in the banana production chain. Similarly, with refrigerated trucking of bananas, where hiring of non-refrigerated trucks is 20–25% cheaper [
22]. The extra charge for refrigerated trucks is primarily due to higher consumption of diesel. Allowing for the many fixed costs such as labour and trucking fees it is estimated that the energy savings with non-refrigerated trucks would be at least 50%.
Wills et al. [
23] quantified the relationship between temperature, ethylene concentration and time to ripen of Cavendish bananas that were stored at 15, 20 and 25 °C in an atmosphere containing <0.001, 0.01, 0.1 and 1.0 μL L
−1 ethylene (
Figure 2). The equation they derived from the data showed a linear relationship for time to ripen and temperature but a logarithmic relationship with ethylene. Thus, there was greater benefit by reducing ethylene than by reducing temperature. The equation was applied to the shipping of Cavendish bananas from Costa Rica to Italy, which is at 13°C with a voyage time of up to 16 days. Using international meteorological data, the maximum shipping temperature (July) is 24 °C, and it indicates that fruit could be shipped without ripening if ethylene was maintained at 0.01 µL L
−1. For the minimum shipping temperature (January) of 17 °C, fruit could be shipped if ethylene was maintained at 0.08 µL L
−1. While such ethylene concentrations can be achieved with a range of technologies, Wills et al. [
24] found the ventilation of banana cartons with ambient air, (generally <0.01 µL L
−1) at a flow rate of 14 L hr
−1 maintained ethylene around fruit at a concentration of 0.01 µL L
−1. Thus, it is possible to ship fruit for a 16-day voyage over the year without refrigeration. However, if it was deemed desirable to have a safety buffer of up to 4 days to allow for any delay in shipping or unloading,
Figure 2 indicates a temperature of 20 °C would ensure fruit did not ripen in transit in an atmosphere of 0.01 µL L
−1 ethylene. Such a shipping temperature is 7 °C higher than the current temperature and would give a considerable energy saving for the voyage.
An innovation over the last decade in the transport of bananas from Central America to Europe by Dole and other shippers [
25] is to pack fruit in modified atmosphere (MA) bags but to still use refrigeration. The ability of the low oxygen and high carbon dioxide atmospheres generated in such bags to retard ripening of bananas was first reported as long ago as 1966 [
26]. Scott and Gandanegara [
27] subsequently examined a range of storage temperatures and found that at 24 °C, use of an MA bag inhibited the ripening of bananas for 22 days compared to 14 days for bananas stored in air. They also found inclusion of potassium permanganate impregnated pellets to absorb ethylene into the MA bags further increased the time to ripen to 42 days. Since these times are well in excess of current shipping times and therefore it would seem feasible for sealed MA bag transport of bananas from the Central America to Europe to not require refrigeration.
Wills et al. [
23] also applied the equation of temperature, ethylene concentration and green life of Cavendish bananas to the Australian trucking situation. They modelled a five-day 3000 km road transport route from the tropical production area to the Sydney urban market. Using meteorological data, they predicted that bananas transported in the prevailing mean summer temperature of 25 °C would not require refrigeration if the ethylene level did not exceed 0.58 µL L
−1 while transport at the mean winter temperature of 14 °C fruit could withstand a level of about 0.90 µL L
−1 without ripening
en route. These are quite high ethylene levels and considered unlikely to occur during the five-day transit. This contention is supported by Wills et al. [
28], who in a survey over 12 months of 363 banana cartons on arrival at the Sydney wholesale markets found a mean ethylene concentration of 0.06 µL L
−1 with the highest measurement being 0.28 µL L
−1, albeit that fruit were from refrigerated shipments.
3.2. Senescence of Leafy Vegetables
Li et al. [
29] stored four non-climacteric green vegetables at a temperature range of 0 to 20 °C and ethylene range of 0.001 to 1.0 μL L
−1. They found that the postharvest life, as determined by consumer acceptance criteria of yellowing for pak choy and broccoli, leaf abscission for mint, and pod softening and chilling injury for green bean, increased as the temperature and ethylene concentration decreased.
The recommended storage temperature for pak choy, broccoli and mint is 0 °C ([
30,
31,
32], respectively) but these recommendations are based on laboratory studies to find the maximum possible postharvest life. The study by Li et al., [
29] found the postharvest life at 0 °C of these produce ranged from 40–60 days with the longer times at the lower concentrations of ethylene. However, it is questionable as to the amount of produce that requires a 40–60-day market life with many trading chains only requiring a postharvest life of ≤3 weeks. The regression equations Li et al. [
29] generated for each produce allowed calculation of the temperature and ethylene levels required for any nominated postharvest life. Their data for broccoli to achieve a 7-, 14- or 21-day postharvest life are given in
Table 1. This shows firstly, that for all storage periods, the temperature that broccoli need to be held increases by 4 C as the ethylene is reduced from 1 to 0.001 μL L
−1. They found a similar effect for pak choy and mint. Using a 14-day postharvest life as an example, maintaining ethylene at 0.001 μL L
−1 would allow a storage temperature of about 10 °C. This is well above the current recommendation of 0 °C and would result in substantial energy savings. Indeed, in many temperate regions, 10 °C is close to the mean ambient temperature during cooler months so no refrigeration would be needed.
For green beans the recommended storage temperature is 5.0–7.5 °C due to the development of chilling injury at lower temperatures [
33]. Li et al. [
29] found that beans were more sensitive to ethylene than pak choy, broccoli and mint.
Table 2 shows that for a seven day postharvest life, the temperature that beans need to be held increases by 9 °C as the ethylene is reduced from 1 to 0.001 μL L
−1. For the longer storage periods of 14 and 21 days, it was not possible to maintain produce in an acceptable condition unless the ethylene level was maintained at ≤0.1 and ≤0.01 μL L
−1, respectively. However, if ethylene was maintained at 0.001 μL L
−1 then storage temperatures of 18 and 13 °C, respectively, would allow marketing of beans after 14 and 21 days, respectively. The commercial relevance of maintaining a low ethylene level was shown by Wills and Kim [
34] who reported hessian bags and polystyrene boxes of beans in retail and wholesale markets contained ethylene from 0.17 to 1.17 μL L
−1 with a mean concentration of 0.45 μL L
−1.
An added benefit for all vegetables could be from a reduced respiration that occurs when the ethylene level is lower [
29,
35]. This was demonstrated by Becker and Fricker [
36], who used the equation for rate of heat generated during respiration, and calculated that a decrease in ethylene from 0.1 to 0.001 μL L
−1 around a 10 kg box of green bean stored at 10 °C would require 61 J less energy to be removed per hour, or 1460 J per day. This would thus further reduce the refrigeration needed to maintain the storage temperature.
3.3. Marketing Produce in Tropical Regions
Many countries in tropical regions are less developed but have a substantial horticulture industry with produce grown on small holdings and marketed soon after harvest to local or nearby villages. With the short market chain in terms of time and distance, there was little need for technology to limit postharvest change. However, with economic development there is growing interest in trucking produce to larger urban centres within the country or to neighbouring countries. In order to market to these more distant destinations and cope with unexpected delays there is a need for some technology to maintain produce quality during the longer transport and distribution time.
In developed countries, the technology of choice is to reduce the storage temperature of produce. However, in regional areas of most developing countries, refrigerated storage or transport facilities are either not available or are expensive to utilize by small traders. In addition, if refrigeration becomes more accessible its usage will add to the world energy consumption. The management of ethylene levels around produce is a possible alternate technology to maintain the required produce quality during marketing.
3.3.1. Tomato Ripening
The benefit of reducing the ethylene concentration around unripe tomatoes without temperature control was demonstrated by Ku et al. [
37]. The marketing of tomatoes in Laos involves packing fruit into polystyrene boxes that are transported 750 km over about 24 h to the capital city, Vientiane. Traders report that substantial ripening occurs during the journey, which limits marketing options to withhold fruit from the market if the price is depressed or fruit is in oversupply. Ku et al. [
37] added sachets of an ethylene absorbent into polystyrene boxes of green tomatoes and assessed the proportion of fruit remaining green during nine days at an ambient temperature of 26–32 °C in Vientiane. On the assumption that an acceptable market outcome is achieved if 75% of tomatoes remain green, they found boxes with ethylene absorbent sachets had a 7-day supply chain market life, which was double the 3.5 days for fruit in control containers. Thus, the market life doubled without any temperature management. The study, however, did not have the ability to measure the change in ethylene concentration in the boxes.
3.3.2. Senescence of Vegetables
A more detailed study under controlled conditions of temperature and ethylene concentration was carried out with a range of vegetables by Wills et al. [
38]. Since they found no published data for levels of ethylene around produce during marketing at tropical ambient temperatures, they conducted a simulated commercial trial in the laboratory and found that after one day at 30 °C, ethylene accumulation around different produce ranged from 0.3 to 1.5 μL L
−1 with a mean value across all produce of 0.8 μL L
−1. They then examined the effect of atmospheric ethylene at 0.001, 0.01, 0.1 and 1.0 μL L
−1 on the retention of visual quality of cucumber, golden squash, bitter melon, green bean, pak choy, choy sum, parsley held at a typical tropical ambient temperature of 30 °C [
39] at high humidity. Reducing atmospheric ethylene progressively increased the postharvest life of all produce. The extension in market life in 0.001 μL L
−1 was about 13 days for cucumber, nine days for golden squash and three days for green bean. Market life extension was 1–2 days for bitter melon, coriander, parsley and pak choy but this represents an approximate doubling of the market life that occurred at 1 μL L
−1 and is considered commercially worthwhile in many developing countries. The marked benefit of reduced ethylene for a 2–day increase in postharvest life is illustrated in
Figure 3 where the appearance of bitter melon after three days at 30 °C shows melons held in 1.0 µL L
−1 were unmarketable while those held in 0.001 µL L
−1 were still of high quality. Thus, the higher ethylene levels used in this study can be considered likely around stack of produce held at tropical temperatures and the extension of market life found when ethylene was reduced from 1 to 0.001 µL L
−1 could reflect expectations of actual commercial benefits at 30 °C.
For comparison, Wills et al. [
38] also stored produce at the commonly accepted ambient temperature in temperate regions of 20 °C in the same range of ethylene concentrations. All produce held at 30 °C in 0.001 µL L
−1 were found to have a similar postharvest life to that stored at 20 °C in 0.1 µL L
−1, a common ethylene level around produce in temperate markets [
13]. Thus, maintaining a low ethylene atmosphere at 30 °C was equivalent to reducing the storage temperature by 10 °C.
Cucumber is widely available in temperate and tropical countries with a recommended storage temperature of 10 °C [
41]. Wills et al. [
38] published data for cucumber stored at 30 and 20 °C, they also had unpublished data for cucumber stored at 10 °C [
40] which is now included in
Table 3. This shows firstly, that at 30 °C storage under current commercial conditions where cucumbers are exposed to 1 µL L
−1, they have a short postharvest life but this is increased to about 15 days if the ethylene level is reduced to ≤0.01 µL L
−1. It can also be seen that the postharvest life of about 15 days is similar to that of cucumbers stored at 20 and 10 °C in the common ethylene level of 0.1 µL L
−1 experienced at these temperatures. Thus, reducing ethylene to ≤0.01 µL L
−1 around cucumbers at 30 °C gives an equivalent postharvest life to 20 and 10 °C held under current marketing conditions.
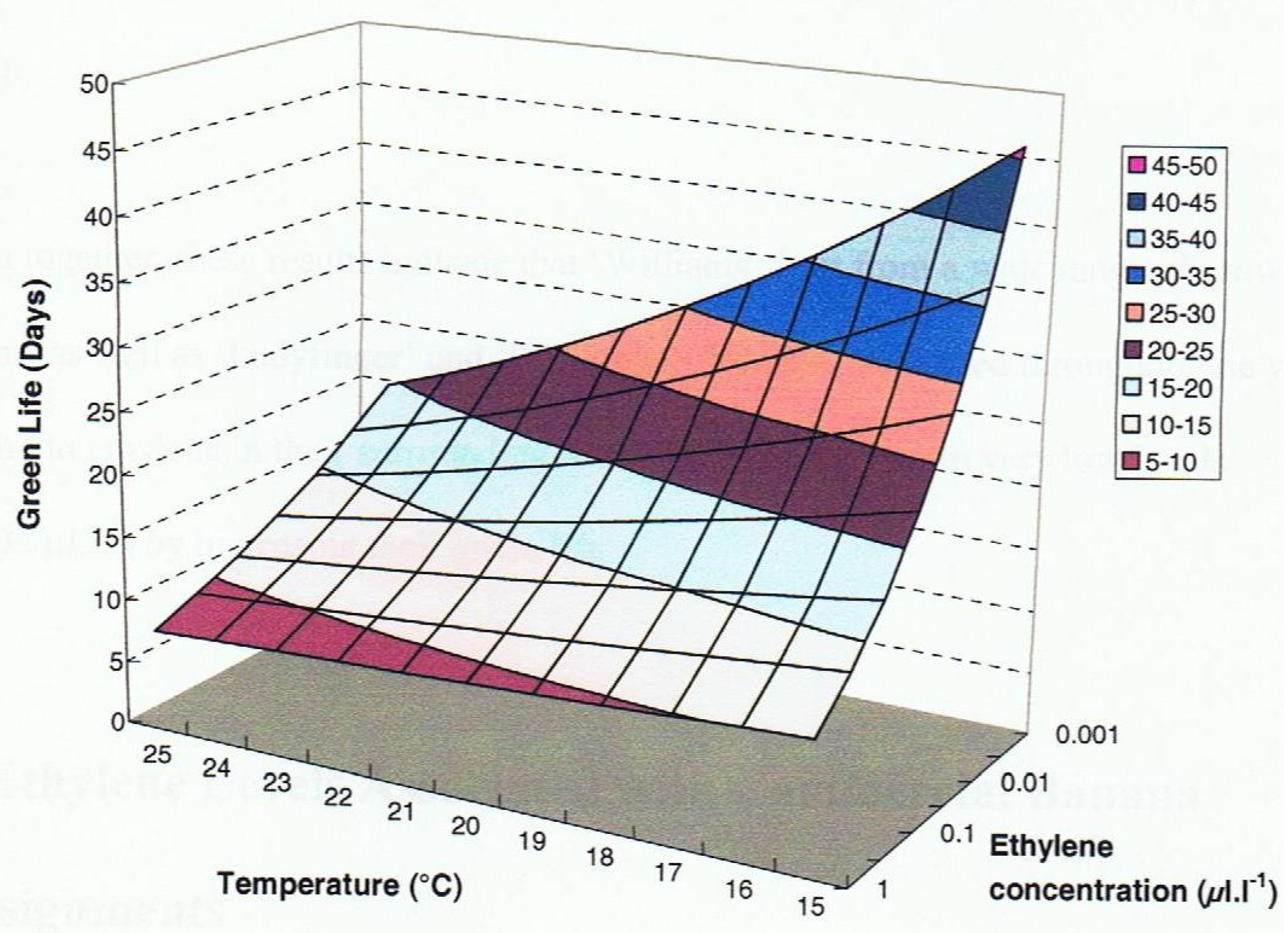
3.3.3. Green Colour Retention of Limes
Persian (Tahitian) limes are grown in tropical and sub-tropical regions and mature fruit are harvested when the skin is still green. The postharvest retention of a green colour is of major importance, as yellow fruit is of considerably lower value to consumers. The recommended storage temperature for limes to retain a green peel colour is 10 °C, which is the threshold temperature below which the fruit is susceptible to chilling injury [
42]. Li et al. [
43] examined the effect on green life of limes held in various ethylene concentrations at the recommended storage temperature (10 °C), and the ambient temperature in temperate regions (20 °C) and tropical regions (30 °C). The findings in
Figure 4 show that the green life at 10 °C increased as the ethylene concentration decreased and was maximal in ≤0.01 µL L
−1 ethylene at about 24 days. Increasing the temperature to 20 °C showed a similar pattern of change in green life with ethylene concentration but at every concentration the green life was lower than at 10 °C with a maximum green life of about 21 days in ≤0.01 µL L
−1 ethylene. However, storing limes at 30 °C resulted in a longer green life at all concentrations than at 10 °C with the maximum green life of about 27 days achieved in ethylene concentrations of ≤0.1 µL L
−1. Thus, holding limes at 30 °C generated a longer green life than at the recommended temperature (10 °C) and it was achieved in the presence of a higher concentration of ethylene. There would therefore be a marketing advantage of not using refrigeration for limes grown and marketed in tropical regions.
3.4. Controlled Atmosphere Storage of Apples
Apples are grown widely around the world with global production of about 85 million tonnes [
44]. Considerable volumes are stored for up to 12 months at low temperature, and distributed through extensive domestic and international marketing chains. The use of storage in a low oxygen-controlled atmosphere (CA) is widespread and is often supplemented by pre-storage treatment with 1-methylcyclopropene (1-MCP). 1-MCP acts by binding irreversibly to ethylene-binding sites, thereby preventing the action of ethylene on produce (see
Section 4.3). For apples, this benefit is in the maintenance of firmness and acidity, and reduction in the number of various physiological disorders [
45].
Kittemann et al. [
46] examined the potential for apples treated with 1-MCP to be stored in CA at a higher temperature than untreated fruit. The aim was to evaluate the energy saving benefits against any potential loss in fruit quality at the higher temperature. Three cultivars of untreated apples were stored for seven months in a commonly used CA in Germany of 1% O
2 and 2.5% CO
2 at 1 °C. 1-MCP-treated fruit were stored in the same CA in a similar size commercial chamber but at 5 °C.
After seven months storage, the energy consumption for each room was calculated from the operating times of refrigeration compressor, ventilation fans, defrosting and CO2 scrubber machinery. Total energy use was reduced by 70% in the 5 °C chamber with the 1-MCP-treated fruit compared to the 1 °C chamber with the untreated fruit.
Contrary to what might be expected, overall fruit quality of the 1-MCP-treated fruit stored at 5 °C was superior to that of the untreated fruit at 1 °C. This was reflected in:
Weight loss of all three cultivars was lower for apples stored at 5 °C.
Firmness of Jonagold apples was greater in the 1-MCP-treated fruit at 5 °C, with no significant difference for Golden Delicious and Pinova apples between the treatments.
Incidence of fungal rots was reduced for Pinova fruit. It was ≤5% for both treatments of Jonagold and Golden Delicious.
Sensory analyses after storage plus seven days at 20 °C was conducted only on Jonagold and Pinova apples. The texture of Jonagold apples was rated higher for the 1-MCP-treated fruit and accordingly, its purchase preference was higher. There was no difference in the texture preferences of Pinova apples for each treatment.
Thus, for all quality attributes, storage of 1-MCP-treated fruit at 5 °C was either superior in quality to untreated fruit at 1 °C or was not significantly different. Hence, the use of an ethylene-inhibiting agent allowed substantial energy savings through storage at a higher temperature and better retention of fruit quality.
The findings are in agreement with a conference paper by McCormick et al. [
47] who similarly compared storage of German Gala apples in CA (1% O
2 and 2% CO
2) with untreated fruit at 1.5 °C and 1-MCP-treated fruit at 4 °C. After 5.5 months storage there was a 35% reduction in energy usage for the room with 1-MCP treated fruit, which were firmer, contained higher acid and preferred by a sensory panel than fruit stored at 1.5 °C.
5. Discussion
It has been shown reducing the concentration of ethylene around horticultural produce increases postharvest life and that reducing ethylene has a similar effect to reducing the storage temperature., There would seem to be general acceptance by the horticultural industry that a reduction in the use of refrigerated storage and transport of fruit and vegetables and the commensurate reduction in energy use leading to reduced greenhouse gas emissions would be of benefit for the industry and the environment. However, it is noteworthy that it in the 10+ years since East [
4] highlighted the need to reduce energy in storage and handling, only a few research groups have published studies on how energy reduction can be achieved. This paper has shown that management of ethylene levels around produce can be at least a partial substitute for refrigeration and in some instances a replacement for refrigeration.
However, industry remains reluctant to change current practices. A major impediment is considered to be overcoming risk aversion in the upending of accepted wisdom over many years that cool chain management is the gold standard for postharvest handling of fresh fruit and vegetables. This risk aversion is illustrated by the example noted in
Section 3.1 of some international banana traders who place a polyethylene bag around bananas in a carton but still maintain the container at a low temperature. Research shows either technology will deliver the desired outcome, so why are both systems used concurrently?
Change in industry practice would be assisted if there was greater interest in ethylene metabolism by postharvest researchers. For this to occur, there firstly, needs to be greater understanding by researchers that 0.1 L L
−1 is not the threshold concentration for activity of ethylene and that ethylene above 0.001 L L
−1 is detrimental to postharvest quality. It is telling that while numerous published papers evaluate the effectiveness of some technology or compound on a wide range of quality factors, the vast majority do not even measure ethylene. Thus, conclusions in these papers on the mode of action of the applied treatment may be erroneous. Indeed, in one of the few papers to include ethylene measurement, a time course study by Al Ubeed et al. [
70] of the beneficial effect of hydrogen sulphide fumigation on the green leafy vegetable, pak choy, found that the first quality factor to be affected by hydrogen sulphide was a reduction in ethylene synthesis. They concluded that reducing ethylene metabolism was the key action of hydrogen sulphide that led to inhibition of other senescence attributes.
The paucity of studies on ethylene is considered to be in large part due to the relative difficulty in measuring ethylene at the low physiologically effective concentrations important for fruit and vegetables. The standard method for measuring ethylene is with flame ionisation gas chromatography (FIDC). With a good instrument held in a controlled temperature environment, it is possible to attain a sensitivity of 0.001 L L
−1 but many FIDC systems struggle to measure 0.01 L L
−1. However, the cost and expertise needed to sustain a FIDC system is beyond the capability of many small research laboratories especially in less developed countries, and it is impractical to locate instrumentation at most cool stores or packing sheds. While many types of ethylene sensor devices are commercially available [
71] our experience has been that most are not able to accurately measure levels of ≤0.01 µL L
−1 ethylene.
The inability to rapidly measure low levels of ethylene is a major hurdle in demonstrating to an industry participant that there is a need to reduce ethylene in their storage situation. In addition, how does someone implementing an ethylene-reducing technology know if the intervention is effective? This is in contrast to measurement of temperature, which is cheap, generates a continuous measurement and no training is needed to take a measurement. Thus, a critical development is the availability of a low-cost, portable, digital readout instrument that gives real time assessment of the ethylene concentration. It would seem that companies marketing ethylene-reduction technologies should be more proactive in supporting development of such an instrument, as they would be a major beneficiary of the technology.
A marketing advantage of reduced energy usage could be through increasing community concerns about climate change with the horticultural industry being seen as a good corporate citizen. In the 1990s, the United Kingdom developed an energy rating for foods based on the concept of food miles that measures the distance the food has travelled from the farm gate to the consumer. Such a measure is flawed as postharvest transport is only a small fraction of the energy used to produce food and is poorly correlated to the total energy usage during production and marketing. However, it is a simple concept that consumers can relate to and there would be marketing value in a trader who can advertise that its produce was less energy intensive than those of competitors.
The greatest benefit accruing to industry participants would be in reducing costs by holding produce at a higher temperature. Of course, this needs to be balanced by any cost to introduce some new technology to reduce ethylene. It can be a daunting process to contemplate how to capture the cost benefit of less refrigeration usage and is best tackled in a systematic manner. The first step should be to know the maximum time required for a particular marketing chain for a specific produce and then determine the temperature required to maintain quality for this period. Where a relatively short market life is required, it may be possible to hold produce at ambient temperature without having any ethylene reduction intervention. For longer marketing periods, an investigation should be made of the level of ethylene in the current storage and transport system and then determine the increase in temperature that accompanies a reduction in ethylene around produce. While it is not commercially practical to modify the storage temperature by small increments, it could be feasible to replace storage at 0 °C with storage at 5 °C or 10 °C, temperatures that are in commercial usage. If the required temperature is 15 °C or some other sub-ambient temperature, especially in tropical regions, use of standard air conditioning may be cost effective.
Selection of an ethylene reduction technology obviously needs to be effective, reliable, manageable and cost-effective. The most universal technology currently available is potassium permanganate-based products as they are commercially available from a wide range of manufacturers as small sachets suitable for single packages up to tubes suitable for storage rooms or containers. However, they do have a limited life and without regular measurement of ethylene levels it is currently it is not easy to decide when to replace the product. 1-MCP is highly effective in inhibiting ethylene action but only in a limited range of produce and the technology currently requires batch fumigation which tends to make it suitable only for higher volume produce. Ventilation with ambient air is worthy of consideration as it is an endless free resource and is chemical-free both in usage and post-usage supply. The issues that need to be managed are to maintain the air stream at a sufficient high humidity to prevent excessive weight loss from produce, and where the incoming air is at a higher temperature than that of the storage chamber a heat exchange unit may be required. The technology is only suitable for storage rooms and transport vehicles and containers. As mentioned in
Section 4.2, if the ethylene-porous membrane proves to be highly selective for ethylene, it could make ventilation of storage chambers a viable option as the membrane should last many seasons and requires no energy input.