A Review of Crystallization Fouling in Heat Exchangers
Abstract
:1. Introduction
2. Crystallization Fouling
- Evaporation of the solvent;
- Cooling a solution of normal solubility salts (their solubility increases with increasing temperature, e.g., NaCl, CaCl2) below the solubility temperature;
- Heating a solution of inverse solubility salts (their solubility decreases with increasing temperature, e.g., CaSO4, CaCO3, Na2So4, MgSiO3) above the solubility temperature;
- Mixing of solutions with different compositions;
- Variation of pH, which can affect solubility.
2.1. Solution Concentration and Composition
2.2. Temperature
2.3. Flow Velocity
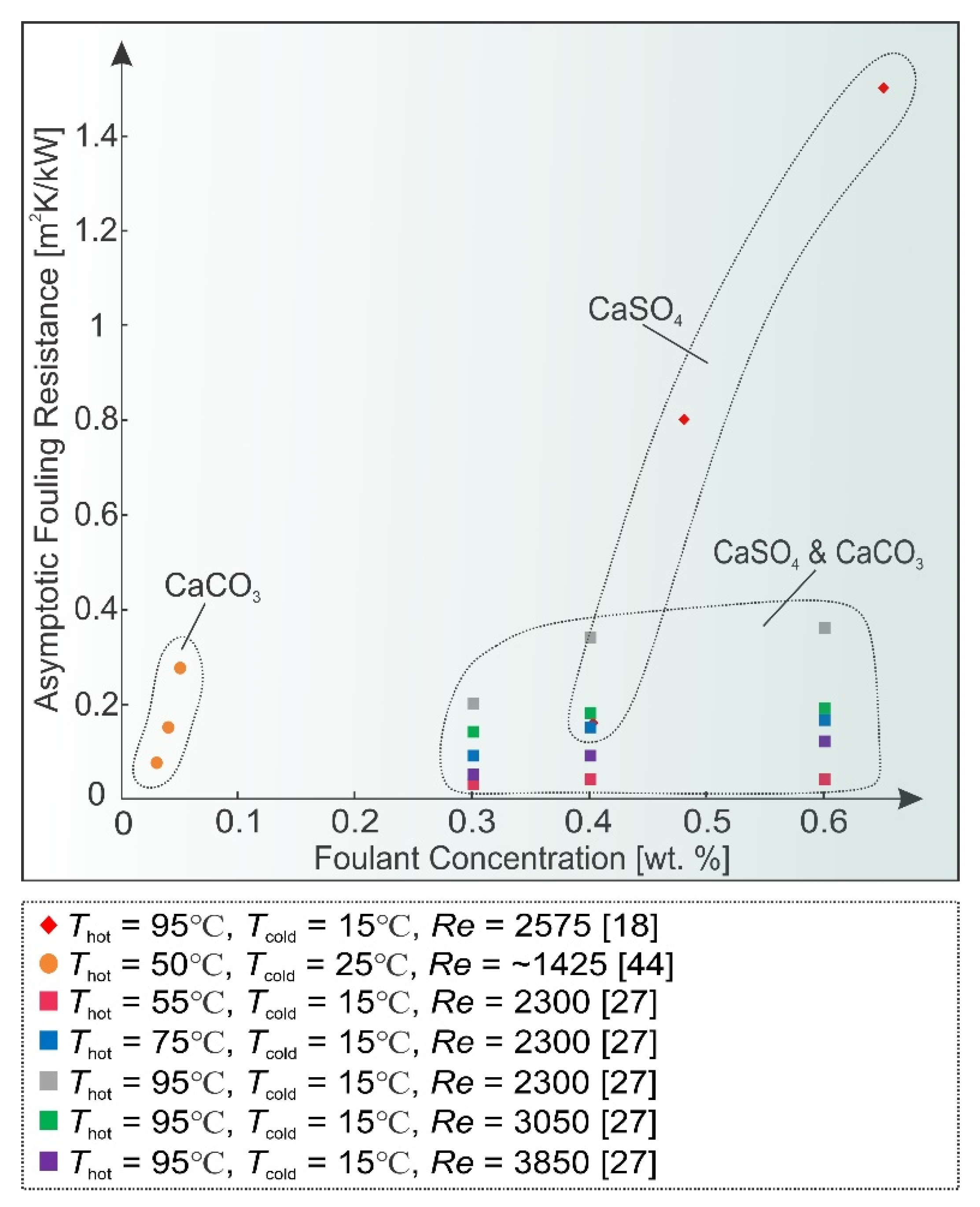
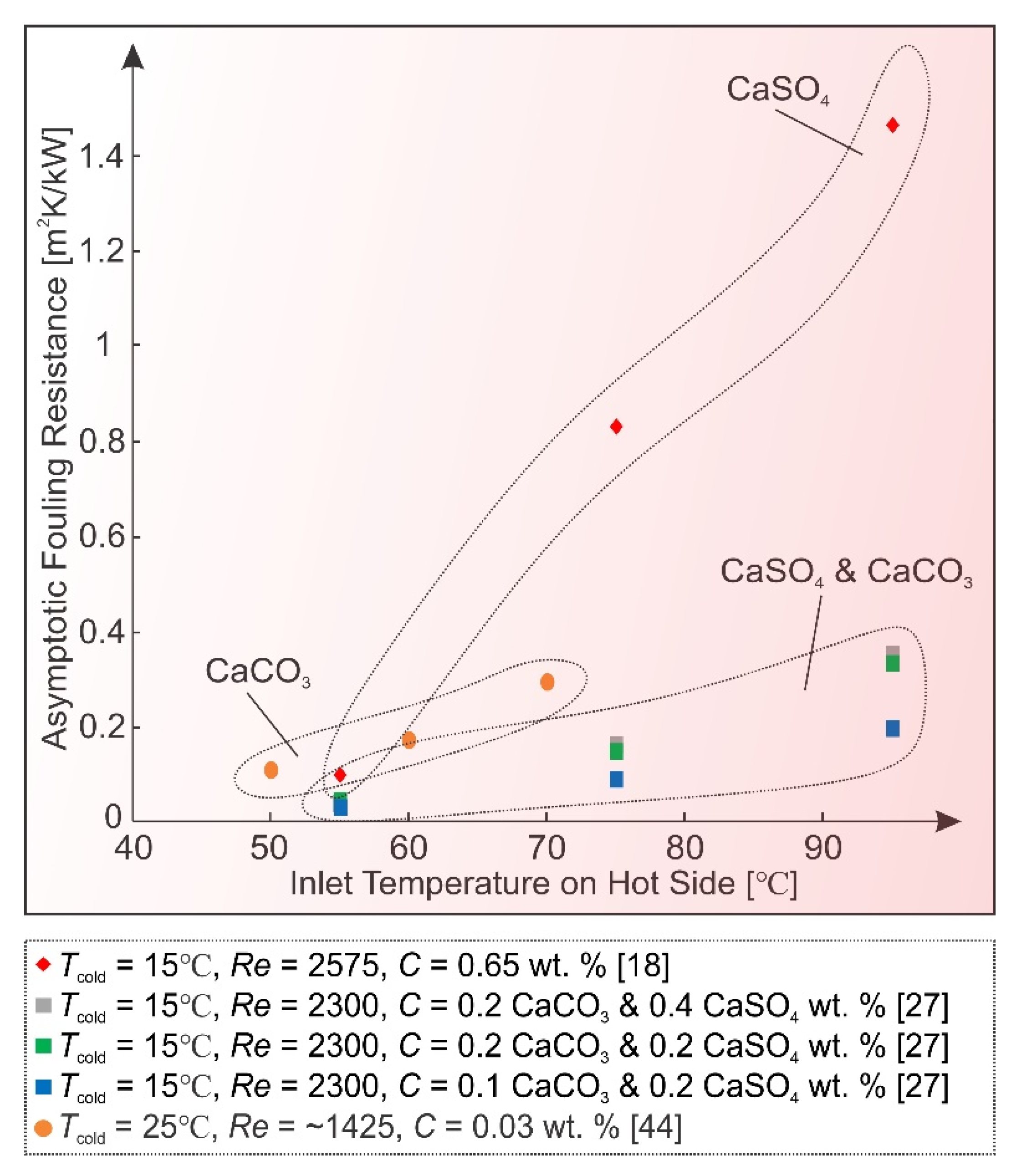
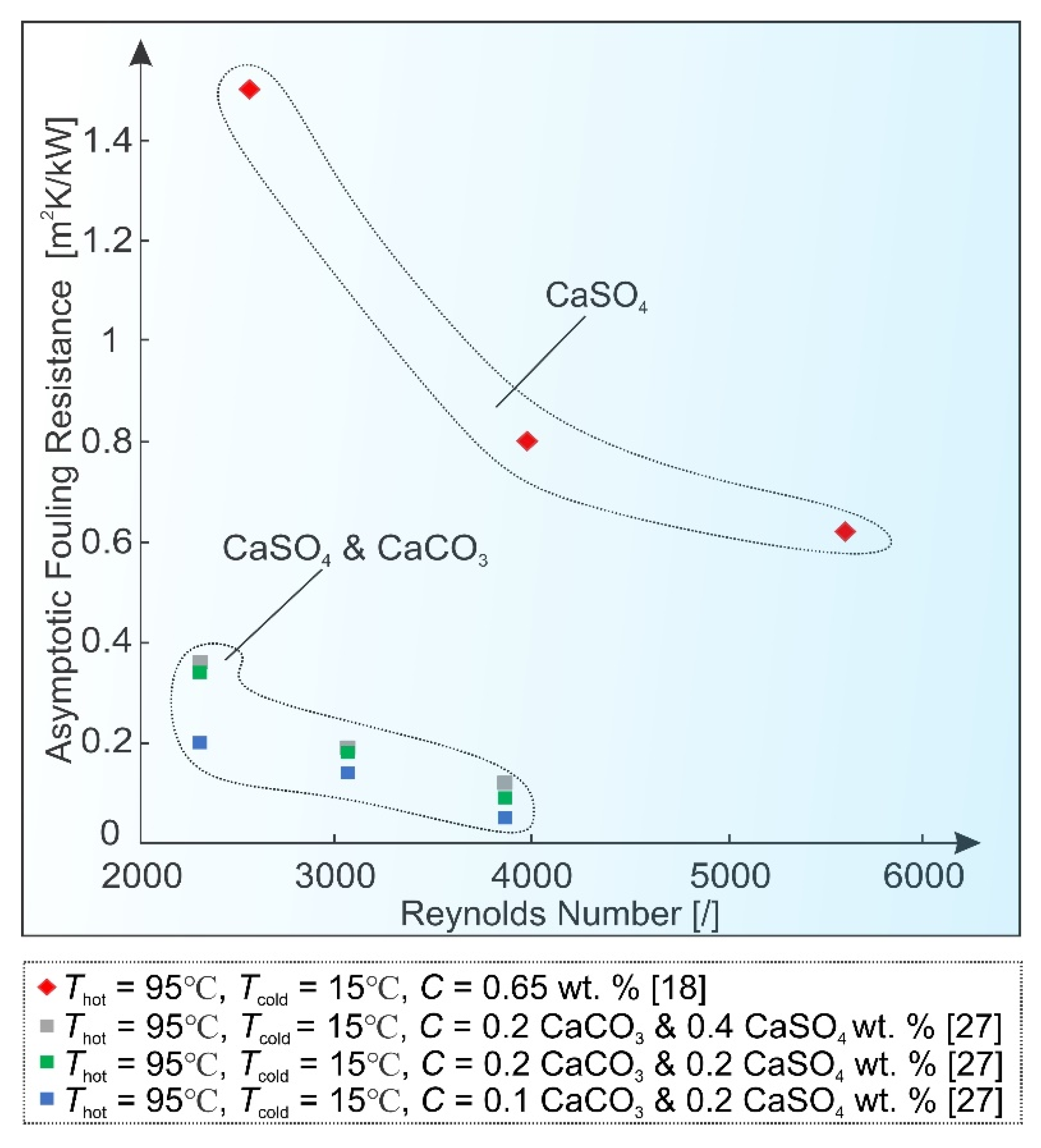
2.4. Surface Condition
2.5. Boiling
2.6. Composite Fouling
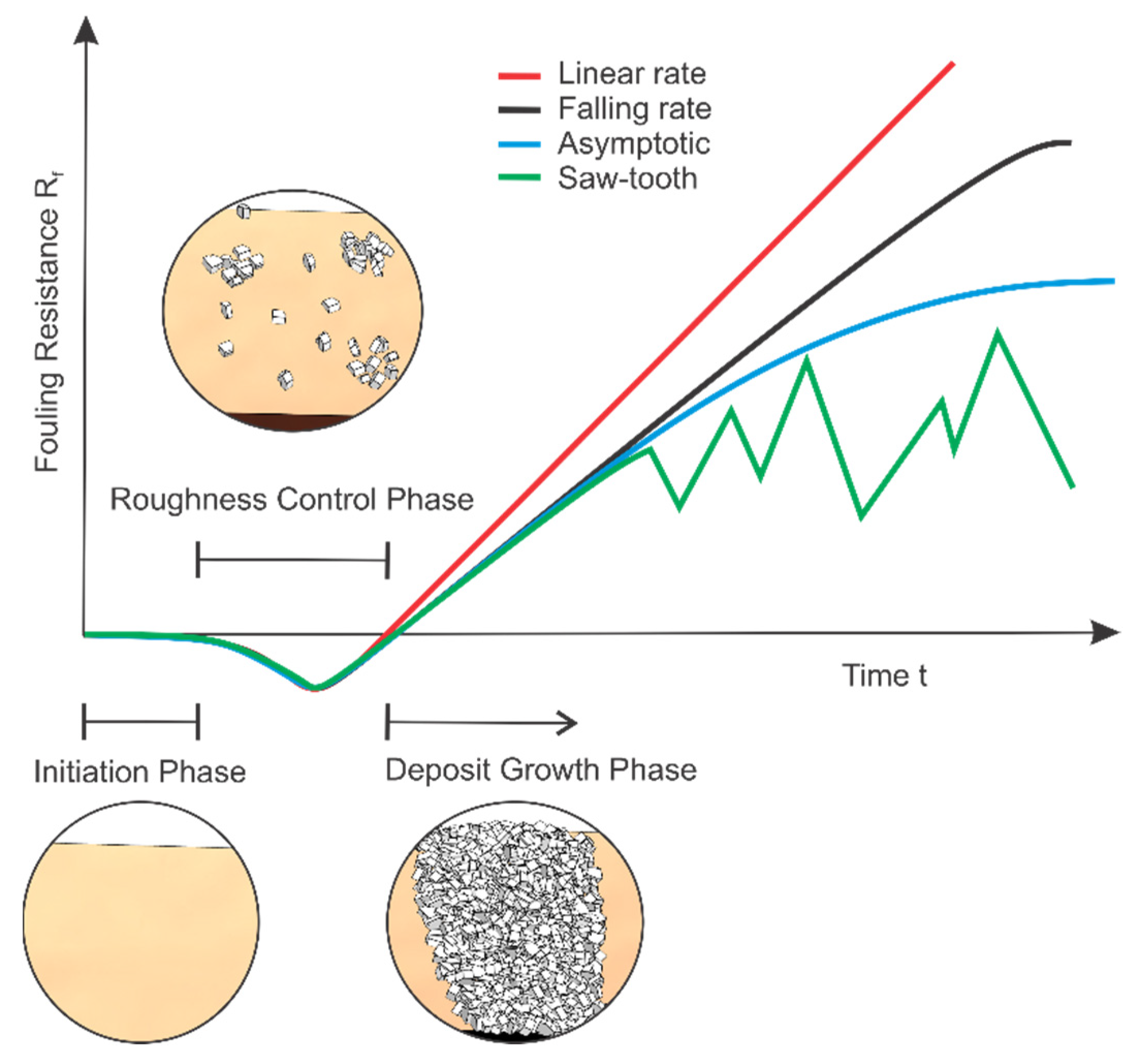
3. Modelling Crystallization Fouling
- The effect of surface roughness and or changing surface area is neglected;
- Only a single mechanism of fouling is assumed to be present;
- Fluid properties are assumed to be constant;
- Fouling layer is assumed to be homogenous;
- Effect of changing flow cross-section is neglected;
- The shape of deposits is ignored;
- The induction and roughness delay periods are ignored;
- Equipment design and material is not incorporated;
- Steady state operation is assumed.
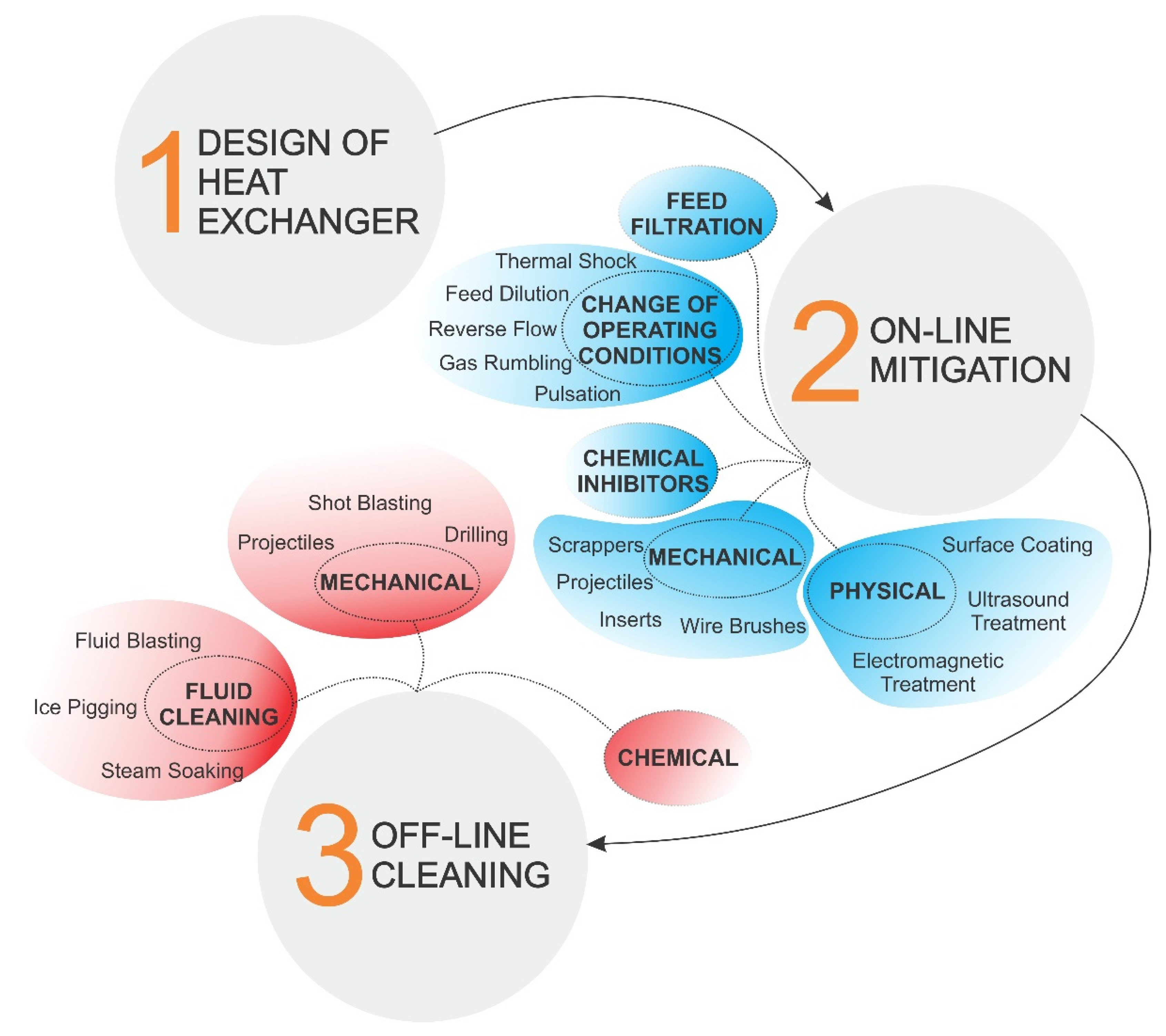
4. Fouling Inhibition and Cleaning
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| Nomenclature | Subscripts | ||
| A | Surface area, m2 | c | clean |
| C | Concentration, g/L | d | deposit |
| D | Diffusion coefficient, m2/s | f | fouling |
| Da | Damköhler number | fl | fluid |
| Total interaction energy, J | r | removal | |
| h | Convective heat transfer coefficient, W/m2 K | sat | saturation |
| Mass transfer coefficient, m/s | Abbreviations | ||
| Reaction rate, m4/kg s | HE | heat exchanger | |
| Deposit solubility product | TOC | total organic carbon | |
| m | Mass, kg | SWRO | seawater reverse osmosis |
| Mass flow rate, kg/s | SEM | scanning electron microscopy | |
| n | Order of reaction | XRD | X-ray diffraction |
| Q | heat, J | CFD | computational fluid dynamics |
| R | Thermal resistance, m2 K/W | VG | vortex generator |
| Rf | Fouling resistance, m2 K/W | MEG | monoethylene glycol |
| Re | Reynolds number | ANN | artificial neural network |
| Sherwood number | DTPA | diethylene triamine pentaacetate | |
| T | Temperature, K | EDTA | ethylenediaminetetraacetic acid |
| t | Time, s | MWCNT | multi-walled carbon nanotubes |
| U | Overall heat transfer coefficient, W/m2 K | SCMC | sodium carboxymethyl cellulose |
| v | Velocity, m/s | HMNS | hierarchical micro/nano structures |
| Work of adhesion, J | EDM | electrical discharge machining | |
| x | Thickness, m | EAF | electronic anti-fouling |
| Greek symbols | AEMF | alternating electromagnetic field | |
| λ | Thermal conductivity, W/m K | US | ultrasonic |
| Density, kg/m3 | |||
| τ | Shear stress, Pa | ||
| ω | Mass fraction | ||
References
- Bott, T.R. Fouling of Heat Exchangers; Elsevier Science & Technology Books: Amsterdam, The Netherlands, 1995; ISBN 0444821864. [Google Scholar]
- Steinhagen, R.; Muller-Steinhagen, H.; Maani, K. Problems and Costs due to Heat Exchanger Fouling in New Zealand Industries. Heat Transf. Eng. 1993, 14, 19–30. [Google Scholar] [CrossRef]
- Smith, S.A.; Watts, R.L.; Garrett-Price, B.A. Fouling of Heat Exchangers: Characteristics, Costs, Prevention, Control, and Removal; Noyes Publications: Park Ridge, IL, USA, 1985. [Google Scholar]
- Stephan, P.; Kabelac, S.; Kind, M.; Mewes, D.; Schaber, K.; Wetzel, T. VDI-Wärmeatlas; Springer Vieweg: Berlin, Germany, 2018; ISBN 9783662529881. [Google Scholar]
- Epstein, N. Thinking about heat transfer fouling: A 5 × 5 matrix. Heat Transf. Eng. 1983, 4, 43–56. [Google Scholar] [CrossRef]
- Lee, S.; Kim, J.; Lee, C.H. Analysis of CaSO4 scale formation mechanism in various nanofiltration modules. J. Memb. Sci. 1999, 163, 63–74. [Google Scholar] [CrossRef]
- Tai, C.Y.; Chen, F.B. Polymorphism of CaCO3 precipitated in a constant-composition environment. AIChE J. 1998, 44, 1790–1798. [Google Scholar] [CrossRef]
- Plummer, L.N.; Busenberg, E. The solubilities of calcite, aragonite and vaterite in CO2-H2O solutions between 0 and 90°C, and an evaluation of the aqueous model for the system CaCO3-CO2-H2O. Geochim. Cosmochim. Acta 1982, 46, 1011–1040. [Google Scholar] [CrossRef]
- Carteret, C.; Dandeu, A.; Moussaoui, S.; Muhr, H.; Humbert, B.; Plasari, E. Polymorphism studied by lattice phonon Raman spectroscopy and statistical mixture analysis method. Application to calcium carbonate polymorphs during batch crystallization. Cryst. Growth Des. 2009, 9, 807–812. [Google Scholar] [CrossRef]
- Hasson, D.; Zahavi, J. Mechanism of Calcium Sulfate Scale Deposition on Heat-Transfer Surfaces. Ind. Eng. Chem. Fundam. 1970, 9, 1–10. [Google Scholar] [CrossRef]
- Sheikholeslami, R.; Ng, M. Calcium sulfate precipitation in the presence of nondominant calcium carbonate: Thermodynamics and kinetics. Ind. Eng. Chem. Res. 2001, 40, 3570–3578. [Google Scholar] [CrossRef]
- Won Tae, K.T.; Cho, Y.I. Experimental study of the crystal growth behavior of caco3 fouling using a microscope. Exp. Heat Transf. 2000, 13, 153–161. [Google Scholar] [CrossRef]
- Albert, F.; Augustin, W.; Scholl, S. Roughness and constriction effects on heat transfer in crystallization fouling. Chem. Eng. Sci. 2011, 66, 499–509. [Google Scholar] [CrossRef]
- Epstein, N. Fouling in Heat Exchanger. In Proceedings of the Sixth International Heat Transfer Conference; Hemisphere Publishing Corporation: New York, NY, USA, 1978; pp. 235–253. [Google Scholar]
- Bansal, B.; Mu¨ller-Steinhagen, H. Crystallization Fouling in Plate Heat Exchangers. J. Heat Transfer 1993, 115, 584–591. [Google Scholar] [CrossRef]
- Kern, D.Q.; Seaton, R.E. A theoretical analysis of thermal surface fouling. Br. Chem. Eng. 1959, 4, 258–262. [Google Scholar]
- Mwaba, M.G.; Golriz, M.R.; Gu, J. A semi-empirical correlation for crystallization fouling on heat exchange surfaces. Appl. Therm. Eng. 2006, 26, 440–447. [Google Scholar] [CrossRef]
- Lee, E.; Jeon, J.; Kang, H.; Kim, Y. Thermal resistance in corrugated plate heat exchangers under crystallization fouling of calcium sulfate (CaSO4). Int. J. Heat Mass Transf. 2014, 78, 908–916. [Google Scholar] [CrossRef]
- Müller-Steinhagen, H. Cooling-Water Fouling in Heat Exchangers. Adv. Heat Transf. 1999, 33, 415–496. [Google Scholar] [CrossRef]
- Bansal, B.; Chen, X.D.; Müller-Steinhagen, H. Analysis of “classical” deposition rate law for crystallisation fouling. Chem. Eng. Process. Process Intensif. 2008, 47, 1201–1210. [Google Scholar] [CrossRef]
- Pääkkönen, T.M.; Riihimäki, M.; Simonson, C.J.; Muurinen, E.; Keiski, R.L. Crystallization fouling of CaCO3–Analysis of experimental thermal resistance and its uncertainty. Int. J. Heat Mass Transf. 2012, 55, 6927–6937. [Google Scholar] [CrossRef]
- Malayeri, M.R.; Müller-Steinhagen, H. Initiation of CASO4 scale formation on heat transfer surfaces under pool boiling conditions. Heat Transf. Eng. 2007, 28, 240–247. [Google Scholar] [CrossRef] [Green Version]
- Al-Gailani, A.; Sanni, O.; Charpentier, T.V.J.; Crisp, R.; Bruins, J.H.; Neville, A. Examining the effect of ionic constituents on crystallization fouling on heat transfer surfaces. Int. J. Heat Mass Transf. 2020, 160, 120180. [Google Scholar] [CrossRef]
- Lv, Y.; Lu, K.; Ren, Y. Composite crystallization fouling characteristics of normal solubility salt in double-pipe heat exchanger. Int. J. Heat Mass Transf. 2020, 156. [Google Scholar] [CrossRef]
- Choi, Y.; Naidu, G.; Jeong, S.; Lee, S.; Vigneswaran, S. Effect of chemical and physical factors on the crystallization of calcium sulfate in seawater reverse osmosis brine. Desalination 2018, 426, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Chong, T.H.; Sheikholeslami, R. Thermodynamics and kinetics for mixed calcium carbonate and calcium sulfate precipitation. Chem. Eng. Sci. 2001, 56, 5391–5400. [Google Scholar] [CrossRef]
- Song, K.S.; Lim, J.; Yun, S.; Kim, D.; Kim, Y. Composite fouling characteristics of CaCO3 and CaSO4 in plate heat exchangers at various operating and geometric conditions. Int. J. Heat Mass Transf. 2019, 136, 555–562. [Google Scholar] [CrossRef]
- Helalizadeh, A.; Müller-Steinhagen, H.; Jamialahmadi, M. Mixed salt crystallisation fouling. Chem. Eng. Process. Process Intensif. 2000, 39, 29–43. [Google Scholar] [CrossRef] [Green Version]
- Helalizadeh, A.; Müller-Steinhagen, H.; Jamialahmadi, M. Mathematical modelling of mixed salt precipitation during convective heat transfer and sub-cooled flow boiling. Chem. Eng. Sci. 2005, 60, 5078–5088. [Google Scholar] [CrossRef]
- Helalizadeh, A.; Müller-Steinhagen, H.; Jamialahmadi, M. Application of fractal theory for characterisation of crystalline deposits. Chem. Eng. Sci. 2006, 61, 2069–2078. [Google Scholar] [CrossRef]
- Augustin, W.; Bohnet, M. Influence of the ratio of free hydrogen ions on crystallization fouling. Chem. Eng. Process. Process Intensif. 1995, 34, 79–85. [Google Scholar] [CrossRef]
- Al-Otaibi, D.A.; Hashmi, M.S.J.; Yilbas, B.S. Fouling resistance of brackish water: Comparision of fouling characteristics of coated carbon steel and titanium tubes. Exp. Therm. Fluid Sci. 2014, 55, 158–165. [Google Scholar] [CrossRef]
- Hasan, B.O.; Nathan, G.J.; Ashman, P.J.; Craig, R.A.; Kelso, R.M. The effects of temperature and hydrodynamics on the crystallization fouling under cross flow conditions. Appl. Therm. Eng. 2012, 36, 210–218. [Google Scholar] [CrossRef]
- Stephan, P.; Kabelac, S.; Kind, M.; Mewes, D.; Schaber, K.; Wetzel, T. VDI Heat Atlas; Springer Verlag: Berlin, Germany, 2010; ISBN 9788578110796. [Google Scholar]
- Bott, T.R. Aspects of crystallization fouling. Exp. Therm. Fluid Sci. 1997, 14, 356–360. [Google Scholar] [CrossRef]
- Genić, S.B.; Jaćimović, B.M.; Mandić, D.; Petrović, D. Experimental Determination of Fouling Factor on Plate Heat Exchangers in District Heating System; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar] [CrossRef]
- Pääkkönen, T.M.; Riihimäki, M.; Simonson, C.J.; Muurinen, E.; Keiski, R.L. Modeling CaCO3 crystallization fouling on a heat exchanger surface–Definition of fouling layer properties and model parameters. Int. J. Heat Mass Transf. 2015, 83, 84–98. [Google Scholar] [CrossRef]
- Watkinson, A.P.; Martinez, O. Scaling of Heat Exchanger Tubes By Calcium Carbonate. Am. Soc. Mech. Eng. 1976, 504–508. [Google Scholar] [CrossRef]
- Al-Gailani, A.; Sanni, O.; Charpentier, T.V.J.; Crisp, R.; Bruins, J.H.; Neville, A. Inorganic fouling of heat transfer surface from potable water during convective heat transfer. Appl. Therm. Eng. 2020, 116271. [Google Scholar] [CrossRef]
- Peyghambarzadeh, S.M.; Vatani, A.; Jamialahmadi, M. Influences of bubble formation on different types of heat exchanger fouling. Appl. Therm. Eng. 2013, 50, 848–856. [Google Scholar] [CrossRef]
- Wang, L.C.; Li, S.F.; Wang, L.B.; Cui, K.; Zhang, Q.L.; Liu, H.B.; Li, G. Relationships between the characteristics of CaCO3 fouling and the flow velocity in smooth tube. Exp. Therm. Fluid Sci. 2016, 74, 143–159. [Google Scholar] [CrossRef]
- Vosough, A.; Assari, M.R.; Peyghambarzadeh, S.M.; Azizi, S. Influence of fluid flow rate on the fouling resistance of calcium sulfate aqueous solution in subcooled flow boiling condition. Int. J. Therm. Sci. 2020, 154, 106397. [Google Scholar] [CrossRef]
- Pääkkönen, T.M.; Ojaniemi, U.; Pättikangas, T.; Manninen, M.; Muurinen, E.; Keiski, R.L.; Simonson, C.J. CFD modelling of CaCO3 crystallization fouling on heat transfer surfaces. Int. J. Heat Mass Transf. 2016, 97, 618–630. [Google Scholar] [CrossRef]
- Teng, K.H.; Kazi, S.N.; Amiri, A.; Habali, A.F.; Bakar, M.A.; Chew, B.T.; Al-Shamma’a, A.; Shaw, A.; Solangi, K.H.; Khan, G. Calcium carbonate fouling on double-pipe heat exchanger with different heat exchanging surfaces. Powder Technol. 2017, 315, 216–226. [Google Scholar] [CrossRef]
- Kazi, S.N.; Duffy, G.G.; Chen, X.D. Fouling and fouling mitigation on heated metal surfaces. Desalination 2012, 288, 126–134. [Google Scholar] [CrossRef]
- Al-Janabi, A.; Malayeri, M.R. Innovative non-metal heat transfer surfaces to mitigate crystallization fouling. Int. J. Therm. Sci. 2019, 138, 384–392. [Google Scholar] [CrossRef]
- Ren, L.; Cheng, Y.; Wang, Q.; Tian, X.; Yang, J.; Zhang, D. Relationship between corrosion product and fouling growth on mild steel, copper and brass surface. Colloids Surfaces A Physicochem. Eng. Asp. 2020, 591, 124502. [Google Scholar] [CrossRef]
- Ren, L.; Cheng, Y.; Wang, Q.; Yang, J. Simulation of the relationship between calcium carbonate fouling and corrosion of iron surface. Colloids Surfaces A Physicochem. Eng. Asp. 2019, 582, 123882. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Miao, R.; Lv, Y.; Wang, X.; Meng, X.; Yang, R.; Zhang, X. Enhanced gypsum scaling by organic fouling layer on nanofiltration membrane: Characteristics and mechanisms. Water Res. 2016, 91, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Großerichter, D.; Stichlmair, J. Crystallization fouling in packed columns. Chem. Eng. Res. Des. 2003, 81, 68–73. [Google Scholar] [CrossRef]
- Zhao, L.; Tang, W.; Wang, L.; Li, W.; Minkowycz, W.J. Heat transfer and fouling characteristics during falling film evaporation in a vertical sintered tube. Int. Commun. Heat Mass Transf. 2019, 109, 104388. [Google Scholar] [CrossRef]
- Herz, A.; Malayeri, M.R.; Müller-Steinhagen, H. Fouling of roughened stainless steel surfaces during convective heat transfer to aqueous solutions. Energy Convers. Manag. 2008, 49, 3381–3386. [Google Scholar] [CrossRef]
- Lei, C.; Peng, Z.; Day, T.; Yan, X.; Bai, X.; Yuan, C. Experimental observation of surface morphology effect on crystallization fouling in plate heat exchangers. Int. Commun. Heat Mass Transf. 2011, 38, 25–30. [Google Scholar] [CrossRef]
- Al-Janabi, A.; Malayeri, M.R.; Badran, O.O. Performance of shot peened surfaces subject to crystallization fouling. Int. J. Therm. Sci. 2017, 111, 379–389. [Google Scholar] [CrossRef]
- Cooper, A.; Suitor, J.W.; Usher, J.D. Cooling water fouling in plate heat exchangers. Heat Transf. Eng. 1980, 1, 50–55. [Google Scholar] [CrossRef]
- Kho, T.; Müller-Steinhagen, H. An experimental and numerical investigation of heat transfer fouling and fluid flow in flat plate heat exchangers. Chem. Eng. Res. Des. 1999, 77, 124–130. [Google Scholar] [CrossRef]
- Bansal, B.; Müller-Steinhagen, H.; Chen, X.D. Performance of plate heat exchangers during calcium sulphate fouling—investigation with an in-line filter. Chem. Eng. Process. Process Intensif. 2000, 39, 507–519. [Google Scholar] [CrossRef]
- Mayer, M.; Bucko, J.; Benzinger, W.; Dittmeyer, R.; Augustin, W.; Scholl, S. The impact of crystallization fouling on a microscale heat exchanger. Exp. Therm. Fluid Sci. 2012, 40, 126–131. [Google Scholar] [CrossRef]
- Han, Z.; Xu, Z.; Wang, J. CaSO4 fouling characteristics on the rectangular channel with half-cylinder vortex generators. Appl. Therm. Eng. 2018, 128, 1456–1463. [Google Scholar] [CrossRef]
- Xu, Z.; Han, Z.; Wang, J.; Li, Y. Numerical simulation of CaSO4 crystallization fouling in a rectangular channel with vortex generators. Int. Commun. Heat Mass Transf. 2019, 101, 42–50. [Google Scholar] [CrossRef]
- Hasan, B.O.; Nathan, G.J.; Ashman, P.J.; Craig, R.A.; Kelso, R.M. The use of turbulence generators to mitigate crystallization fouling under cross flow conditions. Desalination 2012, 288, 108–117. [Google Scholar] [CrossRef]
- Hasan, B.O.; Jwair, E.A.; Craig, R.A. The effect of heat transfer enhancement on the crystallization fouling in a double pipe heat exchanger. Exp. Therm. Fluid Sci. 2017, 86, 272–280. [Google Scholar] [CrossRef]
- Abd-Elhady, M.S.; Malayeri, M.R. Transition of convective heat transfer to subcooled flow boiling due to crystallization fouling. Appl. Therm. Eng. 2016, 92, 122–129. [Google Scholar] [CrossRef]
- Malayeri, M.R.; Müller-Steinhagen, H.; Bartlett, T.H. Fouling of tube bundles under pool boiling conditions. Chem. Eng. Sci. 2005, 60, 1503–1513. [Google Scholar] [CrossRef]
- Peyghambarzadeh, S.M.; Vatani, A.; Jamialahmadi, M. Application of asymptotic model for the prediction of fouling rate of calcium sulfate under subcooled flow boiling. Appl. Therm. Eng. 2012, 39, 105–113. [Google Scholar] [CrossRef]
- Dash, S.; Rapoport, L.; Varanasi, K.K. Crystallization-Induced Fouling during Boiling: Formation Mechanisms to Mitigation Approaches. Langmuir 2018, 34, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Wang, Y.; Tang, Z.; Yao, Y.; Huang, Y.; Wang, X. Experimental study on the interaction between particulate fouling and precipitation fouling in the fouling process on heat transfer tubes. Int. J. Heat Mass Transf. 2019, 138, 1238–1250. [Google Scholar] [CrossRef]
- Zhang, G.M.; Li, G.Q.; Li, W.; Zhang, Z.; Leng, X.L.; Tian, M.C. Particulate fouling and composite fouling assessment in corrugated plate heat exchangers. Int. J. Heat Mass Transf. 2013, 60, 263–273. [Google Scholar] [CrossRef]
- Bansal, B.; Chen, X.D.; Müller-Steinhagen, H. Use of non-crystallising particles to mitigate crystallisation fouling. Int. Commun. Heat Mass Transf. 2003, 30, 695–706. [Google Scholar] [CrossRef]
- Bansal, B.; Chen, X.D.; Muller-Steinhagen, H. Effect of suspended particles on crystallization fouling in plate heat exchangers. J. Heat Transfer 1997, 119, 568–574. [Google Scholar] [CrossRef]
- Broby, M.; Neteland, M.; Ma, X.; Andreassen, J.P.; Seiersten, M. Scaling of calcium carbonate on heated surfaces-Crystallization or particulate fouling? In Proceedings of the SPE International Oilfield Scale Conference and Exhibition, Aberdeen, Scotland, UK, 11– May 2016. [Google Scholar] [CrossRef]
- Somerscales, E.F.C. Fouling of heat transfer surfaces: An historical review. Heat Transf. Eng. 1990, 11, 19–36. [Google Scholar] [CrossRef]
- Müller-Steinhagen, H. Heat transfer fouling: 50 years after the Kern and Seaton model. Heat Transf. Eng. 2011, 32, 1–13. [Google Scholar] [CrossRef]
- Hasson, D. Rate of decrease of heat transfer due to scale deposition. Dechema-Monogr. 1962, 47, 233–252. [Google Scholar]
- Hasson, D.; Avriel, M.; Resnick, W.; Rozenman, T.; Windreich, S. Mechanism of calcium carbonate scale deposition on heat-transfer surfaces. Ind. Eng. Chem. Fundam. 1968, 7, 59–65. [Google Scholar] [CrossRef]
- Nikoo, A.H.; Malayeri, M.R. Incorporation of surface energy properties into general crystallization fouling model for heat transfer surfaces. Chem. Eng. Sci. 2020, 215, 115461. [Google Scholar] [CrossRef]
- Bohnet, M. Fouling of Heat Transfer Surfaces. Chem. Eng. Technol. 1987, 10, 113–125. [Google Scholar] [CrossRef]
- Segev, R.; Hasson, D.; Semiat, R. Rigorous Modeling of the Kinetics of Calcium Carbonate Deposit Formation. AIChE J. 2012, 58, 1222–1229. [Google Scholar] [CrossRef]
- Yang, M.; Young, A.; Niyetkaliyev, A.; Crittenden, B. Modelling fouling induction periods. Int. J. Therm. Sci. 2012, 51, 175–183. [Google Scholar] [CrossRef] [Green Version]
- Kazi, S.N. Fouling and Fouling Mitigation on Heat Exchanger Surfaces. In Heat Exchangers—Basics Design Applications; Books on Demand: Norderstedt, Germany, 2012. [Google Scholar]
- Briançon, S.; Colson, D.; Klein, J.P. Modelling of crystalline layer growth using kinetic data obtained from suspension crystallization. Chem. Eng. J. 1998, 70, 55–64. [Google Scholar] [CrossRef]
- Arsenyeva, O.P.; Crittenden, B.; Yang, M.; Kapustenko, P.O. Accounting for the thermal resistance of cooling water fouling in plate heat exchangers. ATE 2013, 61, 53–59. [Google Scholar] [CrossRef] [Green Version]
- Babuška, I.; Silva, R.S.; Actor, J. Break-off model for CaCO3 fouling in heat exchangers. Int. J. Heat Mass Transf. 2018, 116, 104–114. [Google Scholar] [CrossRef]
- Esawy, M.; Malayeri, M.R. Modeling of CaSO4crystallization fouling of finned tubes during nucleate pool boiling. Chem. Eng. Res. Des. 2017, 118, 51–60. [Google Scholar] [CrossRef]
- Kapustenko, P.O.; Klemeš, J.J.; Matsegora, O.I.; Arsenyev, P.Y.; Arsenyeva, O.P. Accounting for local thermal and hydraulic parameters of water fouling development in plate heat exchanger. Energy 2019, 174, 1049–1059. [Google Scholar] [CrossRef]
- Jamialahmadi, M.; Müller-Steinhagen, H. Heat exchanger fouling and cleaning in the dihydrate process for the production of phosphoric acid. Chem. Eng. Res. Des. 2007, 85, 245–255. [Google Scholar] [CrossRef]
- Souza, A.R.C.; Costa, A.L.H. Modeling and simulation of cooling water systems subjected to fouling. Chem. Eng. Res. Des. 2019, 141, 15–31. [Google Scholar] [CrossRef]
- Bobič, M.; Gjerek, B.; Golobič, I.; Bajsić, I. Dynamic behaviour of a plate heat exchanger: Influence of temperature disturbances and flow configurations. Int. J. Heat Mass Transf. 2020, 163. [Google Scholar] [CrossRef]
- Guelpa, E.; Verda, V. Automatic fouling detection in district heating substations: Methodology and tests. Appl. Energy 2020, 258, 114059. [Google Scholar] [CrossRef]
- Aguel, S.; Meddeb, Z.; Jeday, M.R. Parametric study and modeling of cross-flow heat exchanger fouling in phosphoric acid concentration plant using artificial neural network. J. Process. Control. 2019, 84, 133–145. [Google Scholar] [CrossRef]
- Alsadaie, S.M.; Mujtaba, I.M. Dynamic modelling of Heat Exchanger fouling in multistage flash (MSF) desalination. Desalination 2017, 409, 47–65. [Google Scholar] [CrossRef] [Green Version]
- Sundar, S.; Rajagopal, M.C.; Zhao, H.; Kuntumalla, G.; Meng, Y.; Chang, H.C.; Shao, C.; Ferreira, P.; Miljkovic, N.; Sinha, S.; et al. Fouling modeling and prediction approach for heat exchangers using deep learning. Int. J. Heat Mass Transf. 2020, 159. [Google Scholar] [CrossRef]
- Brahim, F.; Augustin, W.; Bohnet, M. Numerical simulation of the fouling process. Int. J. Therm. Sci. 2003, 42, 323–334. [Google Scholar] [CrossRef]
- Brahim, F.; Augustin, W.; Bohnet, M. Numerical Simulation of the Fouling on Structured Heat Transfer Surfaces (Fouling). In Proceedings of the Heat Exchanger Fouling and Cleaning: Fundamentals and Applications; Institute for Chemical and Thermal Process Engineering: Braunschweig, Germany, 2003. [Google Scholar]
- Walker, P.; Sheikholeslami, R. Assessment of the effect of velocity and residence time in CaSO4 precipitating flow reaction. Chem. Eng. Sci. 2003, 58, 3807–3816. [Google Scholar] [CrossRef]
- Xiao, J.; Li, Z.; Han, J.; Pan, F.; Woo, M.W.; Chen, X.D. A systematic investigation of the fouling induction phenomena with artificial crystal structures and distributions. Chem. Eng. Sci. 2017, 168, 137–155. [Google Scholar] [CrossRef]
- Yang, J. Computational fluid dynamics studies on the induction period of crude oil fouling in a heat exchanger tube. Int. J. Heat Mass Transf. 2020, 159. [Google Scholar] [CrossRef]
- Zhang, F.; Xiao, J.; Chen, X.D. Towards predictive modeling of crystallization fouling: A pseudo-dynamic approach. Food Bioprod. Process. 2015, 93, 188–196. [Google Scholar] [CrossRef]
- Haghshenasfard, M.; Yeoh, G.H.; Dahari, M.; Hooman, K. On numerical study of calcium sulphate fouling under sub-cooled flow boiling conditions. Appl. Therm. Eng. 2015, 81, 18–27. [Google Scholar] [CrossRef]
- Müller-Steinhagen, H.; Malayeri, M.R.; Watkinson, A.P. Heat exchanger fouling: Mitigation and cleaning strategies. Heat Transf. Eng. 2011, 32, 189–196. [Google Scholar] [CrossRef]
- Pugh, S.J.; Hewitt, G.F.; Müller-Steinhagen, H. Fouling during the use of seawater as coolant—The development of a user guide. Heat Transf. Eng. 2005, 26, 35–43. [Google Scholar] [CrossRef]
- Pugh, S.J.; Hewitt, G.F.; Müller-Steinhagen, H. Fouling during the use of fresh water as coolant the development of a user guide. Heat Transf. Eng. 2009, 30, 851–858. [Google Scholar] [CrossRef]
- Maddahi, M.H.; Hatamipour, M.S.; Jamialahmadi, M. Experimental study of calcium sulfate fouling in a heat exchanger during liquid-solid fluidized bed with cylindrical particles. Int. J. Therm. Sci. 2018, 125, 11–22. [Google Scholar] [CrossRef]
- Maddahi, M.H.; Hatamipour, M.S.; Jamialahmadi, M. A model for the prediction of thermal resistance of calcium sulfate crystallization fouling in a liquid–solid fluidized bed heat exchanger with cylindrical particles. Int. J. Therm. Sci. 2019, 145, 106017. [Google Scholar] [CrossRef]
- Pronk, P.; Infante Ferreira, C.A.; Witkamp, G.J. Prevention of crystallization fouling during eutectic freeze crystallization in fluidized bed heat exchangers. Chem. Eng. Process. Process Intensif. 2008, 47, 2140–2149. [Google Scholar] [CrossRef]
- Pronk, P.; Infante Ferreira, C.A.; Witkamp, G.J. Mitigation of ice crystallization fouling in stationary and circulating liquid-solid fluidized bed heat exchangers. Int. J. Heat Mass Transf. 2010, 53, 403–411. [Google Scholar] [CrossRef]
- Shih, W.-Y.; Albrecht, K.; Glater, J.; Cohen, Y. A dual-probe approach for evaluation of gypsum crystallization in response to antiscalant treatment. Desalination 2004, 169, 213–221. [Google Scholar] [CrossRef]
- Sousa, M.F.B.; Bertran, C.A. New methodology based on static light scattering measurements for evaluation of inhibitors for in bulk CaCO3 crystallization. J. Colloid Interface Sci. 2014, 420, 57–64. [Google Scholar] [CrossRef]
- Müller-Steinhagen, H.; Malayeri, M.R.; Watkinson, A.P. Heat exchanger fouling: Environmental impacts. Heat Transf. Eng. 2009, 30, 773–776. [Google Scholar] [CrossRef]
- Kazi, S.N.; Teng, K.H.; Zakaria, M.S.; Sadeghinezhad, E.; Bakar, M.A. Study of mineral fouling mitigation on heat exchanger surface. Desalination 2015, 367, 248–254. [Google Scholar] [CrossRef]
- Teng, K.H.; Amiri, A.; Kazi, S.N.; Bakar, M.A.; Chew, B.T.; Al-Shamma’a, A.; Shaw, A. Retardation of heat exchanger surfaces mineral fouling by water-based diethylenetriamine pentaacetate-treated CNT nanofluids. Appl. Therm. Eng. 2017, 110, 495–503. [Google Scholar] [CrossRef]
- Teng, K.H.; Amiri, A.; Kazi, S.N.; Bakar, M.A.; Chew, B.T. Fouling mitigation on heat exchanger surfaces by EDTA-treated MWCNT-based water nanofluids. J. Taiwan Inst. Chem. Eng. 2016, 60, 445–452. [Google Scholar] [CrossRef]
- Xu, Z.; Zhao, Y.; Wang, J.; Chang, H. Inhibition of calcium carbonate fouling on heat transfer surface using sodium carboxymethyl cellulose. Appl. Therm. Eng. 2019, 148, 1074–1080. [Google Scholar] [CrossRef]
- Qian, J.; Wang, J.; Yue, Z.; Wu, W. Surface crystallization behavior of calcium carbonate in the presence of SMPs secreted by SRB. J. Cryst. Growth 2019, 525, 125208. [Google Scholar] [CrossRef]
- Benecke, J.; Rozova, J.; Ernst, M. Anti-scale effects of select organic macromolecules on gypsum bulk and surface crystallization during reverse osmosis desalination. Sep. Purif. Technol. 2018, 198, 68–78. [Google Scholar] [CrossRef]
- Al-Janabi, A.; Malayeri, M.R. A criterion for the characterization of modified surfaces during crystallization fouling based on electron donor component of surface energy. Chem. Eng. Res. Des. 2015, 100, 212–227. [Google Scholar] [CrossRef]
- Al-Janabi, A.; Malayeri, M.R.; Müller-Steinhagen, H. Experimental fouling investigation with electroless Ni-P coatings. Int. J. Therm. Sci. 2010, 49, 1063–1071. [Google Scholar] [CrossRef]
- Yang, Q.; Ding, J.; Shen, Z. Investigation on fouling behaviors of low-energy surface and fouling fractal characteristics. Chem. Eng. Sci. 2000, 55, 797–805. [Google Scholar] [CrossRef]
- Yang, Q.; Liu, Y.; Gu, A.; Ding, J.; Shen, Z. Investigation of induction period and morphology of CaCO3 fouling on heated surface. Chem. Eng. Sci. 2002, 57, 921–931. [Google Scholar] [CrossRef]
- Cheng, Y.H.; Zou, Y.; Cheng, L.; Liu, W. Effect of the microstructure on the anti-fouling property of the electroless Ni-P coating. Mater. Lett. 2008, 62, 4283–4285. [Google Scholar] [CrossRef]
- Cheng, Y.H.; Chen, H.Y.; Zhu, Z.C.; Jen, T.C.; Peng, Y.X. Experimental study on the anti-fouling effects of Ni-Cu-P-PTFE deposit surface of heat exchangers. Appl. Therm. Eng. 2014, 68, 20–25. [Google Scholar] [CrossRef]
- He, Z.R.; Liu, C.S.; Gao, H.Y.; Jie, X.H.; Lian, W.Q. Experimental study on the anti-fouling effects of EDM machined hierarchical micro/nano structure for heat transfer surface. Appl. Therm. Eng. 2019, 162. [Google Scholar] [CrossRef]
- Oon, C.S.; Kazi, S.N.; Hakimin, M.A.; Abdelrazek, A.H.; Mallah, A.R.; Low, F.W.; Tiong, S.K.; Badruddin, I.A.; Kamanger, S. Heat transfer and fouling deposition investigation on the titanium coated heat exchanger surface. Powder Technol. 2020, 373, 671–680. [Google Scholar] [CrossRef]
- Mayer, M.; Augustin, W.; Scholl, S. Adhesion of single crystals on modified surfaces in crystallization fouling. J. Cryst. Growth 2012, 361, 152–158. [Google Scholar] [CrossRef]
- Reed, J.H.; Gonsalves, A.E.; Román, J.K.; Oh, J.; Cha, H.; Dana, C.E.; Toc, M.; Hong, S.; Hoffman, J.B.; Andrade, J.E.; et al. Ultrascalable Multifunctional Nanoengineered Copper and Aluminum for Antiadhesion and Bactericidal Applications. ACS Appl. Bio Mater. 2019, 2, 2726–2737. [Google Scholar] [CrossRef]
- Razavi, S.M.R.; Oh, J.; Haasch, R.T.; Kim, K.; Masoomi, M.; Bagheri, R.; Slauch, J.M.; Miljkovic, N. Environment-Friendly Antibiofouling Superhydrophobic Coatings. ACS Sustain. Chem. Eng. 2019, 7, 14509–14520. [Google Scholar] [CrossRef]
- Vosough, A.; Peyghambarzadeh, S.M.; Assari, M.R. Influence of thermal shock on the mitigation of calcium sulfate crystallization fouling under subcooled flow boiling condition. Appl. Therm. Eng. 2020, 164, 114434. [Google Scholar] [CrossRef]
- Wang, J.; Liang, Y. Anti-fouling effect of axial alternating electromagnetic field on calcium carbonate fouling in U-shaped circulating cooling water heat exchange tube. Int. J. Heat Mass Transf. 2017, 115, 774–781. [Google Scholar] [CrossRef]
- Fan, C.; Cho, Y.I. Microscopic observation of calcium carbonate particles: Validation of an electronic anti-fouling technology. Int. Commun. Heat Mass Transf. 1997, 24, 747–756. [Google Scholar] [CrossRef]
- Han, Y.; Zhang, C.; Wu, L.; Zhang, Q.; Zhu, L.; Zhao, R. Influence of alternating electromagnetic field and ultrasonic on calcium carbonate crystallization in the presence of magnesium ions. J. Cryst. Growth 2018, 499, 67–76. [Google Scholar] [CrossRef]
- R S T, M.; Epstein, N. Optimum cycles for falling rate processes. Can. J. Chem. Eng. 1981, 59, 631–633. [Google Scholar]
- Pogiatzis, T.; Ishiyama, E.M.; Paterson, W.R.; Vassiliadis, V.S.; Wilson, D.I. Identifying optimal cleaning cycles for heat exchangers subject to fouling and ageing. Appl. Energy 2012, 89, 60–66. [Google Scholar] [CrossRef]
- Diaby, A.L.; Miklavcic, S.J.; Addai-Mensah, J. Optimization of scheduled cleaning of fouled heat exchanger network under ageing using genetic algorithm. Chem. Eng. Res. Des. 2016, 113, 223–240. [Google Scholar] [CrossRef]
- Di Pretoro, A.; D’Iglio, F.; Manenti, F. Optimal cleaning cycle scheduling under uncertain conditions: A flexibility analysis on heat exchanger fouling. Processes 2021, 9, 93. [Google Scholar] [CrossRef]
- Al Ismaili, R.; Lee, M.W.; Wilson, D.I.; Vassiliadis, V.S. Optimisation of heat exchanger network cleaning schedules: Incorporating uncertainty in fouling and cleaning model parameters. Comput. Chem. Eng. 2019, 121, 409–421. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berce, J.; Zupančič, M.; Može, M.; Golobič, I. A Review of Crystallization Fouling in Heat Exchangers. Processes 2021, 9, 1356. https://0-doi-org.brum.beds.ac.uk/10.3390/pr9081356
Berce J, Zupančič M, Može M, Golobič I. A Review of Crystallization Fouling in Heat Exchangers. Processes. 2021; 9(8):1356. https://0-doi-org.brum.beds.ac.uk/10.3390/pr9081356
Chicago/Turabian StyleBerce, Jure, Matevž Zupančič, Matic Može, and Iztok Golobič. 2021. "A Review of Crystallization Fouling in Heat Exchangers" Processes 9, no. 8: 1356. https://0-doi-org.brum.beds.ac.uk/10.3390/pr9081356







