Histological Changes Related to Symptomatic Improvement of Spontaneous Keloids Treated with a Low-Dosage Regimen of UVA-1 Phototherapy
Abstract
:1. Introduction
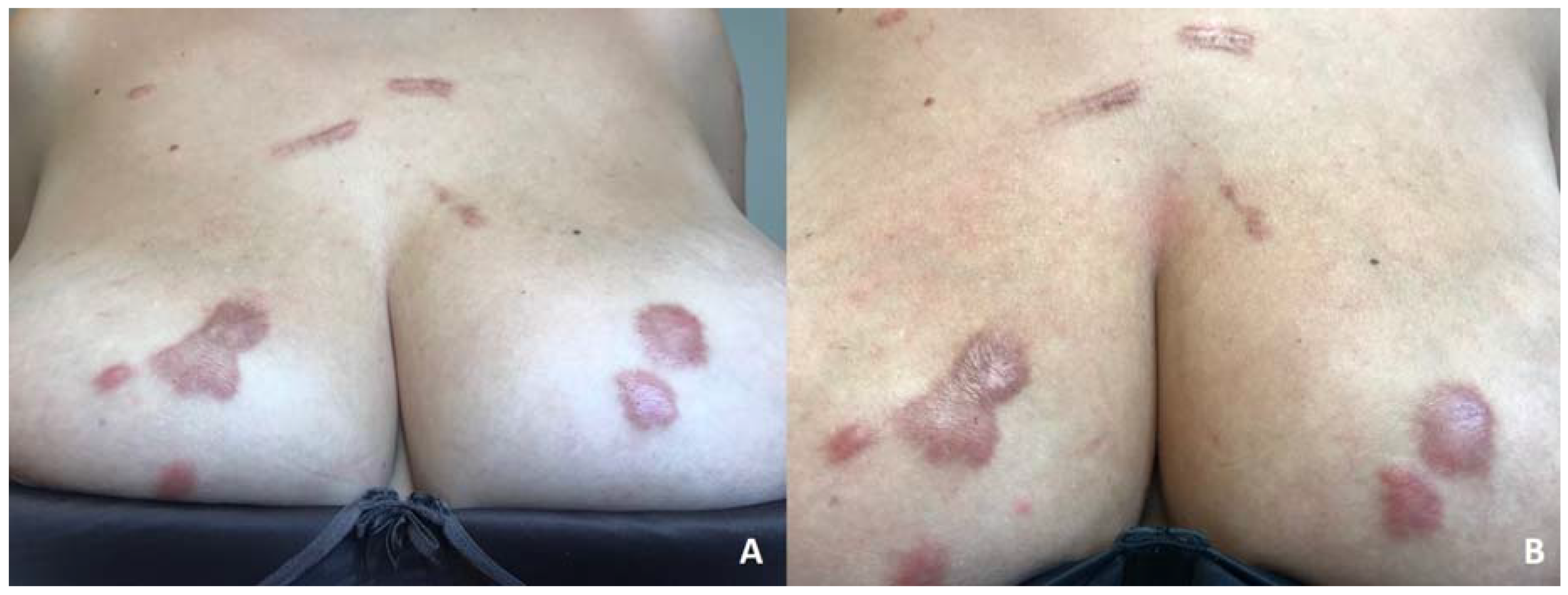
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Viera, M.H.; Vivas, A.C.; Berman, B. Update on Keloid Management: Clinical and Basic Science Advances. Adv. Wound Care 2012, 1, 200–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawe, R.S. Ultraviolet A1 phototherapy. Br. J. Dermatol. 2003, 148, 626–637. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.J.; Hansen, C.; Craig, S. Ultraviolet A irradiation stimulates collagenase production in cultured human fibroblasts. J. Investig. Dermatol. 1992, 99, 440–444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Breuckmann, F.; Stuecker, M.; Altmeyer, P.; Kreuter, A. Modulation of endothelial dysfunction and apoptosis: UVA1-mediated skin improvement in systemic sclerosis. Arch. Dermatol. Res. 2004, 296, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, R.; Akaishi, S. Endothelial dysfunction may play a key role in keloid and hypertrophic scar pathogenesis-Keloids and hypertrophic scars may be vascular disorders. Med. Hypotheses 2016, 96, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.P. Keloids. Dermatol. Clin. 1988, 6, 413–424. [Google Scholar] [CrossRef]
- Kerr, A.C.; Ferguson, J.; Attili, S.K.; Beattie, P.E.; Coleman, A.J.; Dawe, R.S.; Eberlein, B.; Goulden, V.; Ibbotson, S.H.; Menage Hdu, P.; et al. Ultraviolet A1 phototherapy: A British Photodermatology Group workshop report. Clin. Exp. Dermatol. 2012, 37, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Polat, M.; Kaya, H.; Sahin, A. A New Approach in the Treatment of Keloids: UVA-1 Laser. Photomed. Laser Surg. 2016, 34, 130–133. [Google Scholar] [CrossRef]
- Asawanonda, P.; Khoo, L.S.; Fitzpatrick, T.B.; Taylor, C.R. UV-A1 for keloid. Arch. Dermatol. 1999, 135, 348–349. [Google Scholar] [CrossRef]
- Hannuksela-Svahn, A.; Grandal, O.J.; Thorstensen, T.; Christensen, O.B. UVA1 for treatment of keloids. Acta Derm. Venereol. 1999, 79, 490. [Google Scholar] [CrossRef] [PubMed]
- Tuchinda, C.; Kerr, H.A.; Taylor, C.R.; Jacobe, H.; Bergamo, B.M.; Elmets, C.; Rivard, J.; Lim, H.W. UVA1 phototherapy for cutaneous diseases: An experience of 92 cases in the United States. Photodermatol. Photoimmunol. Photomed. 2006, 22, 247–253. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuenca-Barrales, C.; Aneiros-Fernández, J.; Pérez-López, I.; Rodríguez-Pérez, J.; Ruiz-Villaverde, R.; Espelt-Otero, J.L. Histological Changes Related to Symptomatic Improvement of Spontaneous Keloids Treated with a Low-Dosage Regimen of UVA-1 Phototherapy. Dermatopathology 2020, 7, 53-56. https://0-doi-org.brum.beds.ac.uk/10.3390/dermatopathology7030009
Cuenca-Barrales C, Aneiros-Fernández J, Pérez-López I, Rodríguez-Pérez J, Ruiz-Villaverde R, Espelt-Otero JL. Histological Changes Related to Symptomatic Improvement of Spontaneous Keloids Treated with a Low-Dosage Regimen of UVA-1 Phototherapy. Dermatopathology. 2020; 7(3):53-56. https://0-doi-org.brum.beds.ac.uk/10.3390/dermatopathology7030009
Chicago/Turabian StyleCuenca-Barrales, Carlos, José Aneiros-Fernández, Israel Pérez-López, Julia Rodríguez-Pérez, Ricardo Ruiz-Villaverde, and Jorge Luis Espelt-Otero. 2020. "Histological Changes Related to Symptomatic Improvement of Spontaneous Keloids Treated with a Low-Dosage Regimen of UVA-1 Phototherapy" Dermatopathology 7, no. 3: 53-56. https://0-doi-org.brum.beds.ac.uk/10.3390/dermatopathology7030009