Screening for Melanoma and Other Skin Cancer Shows a Higher Early Melanoma Incidence: Social Educational Program “Life Fear-Free”
Abstract
:1. Introduction
2. Materials and Methods
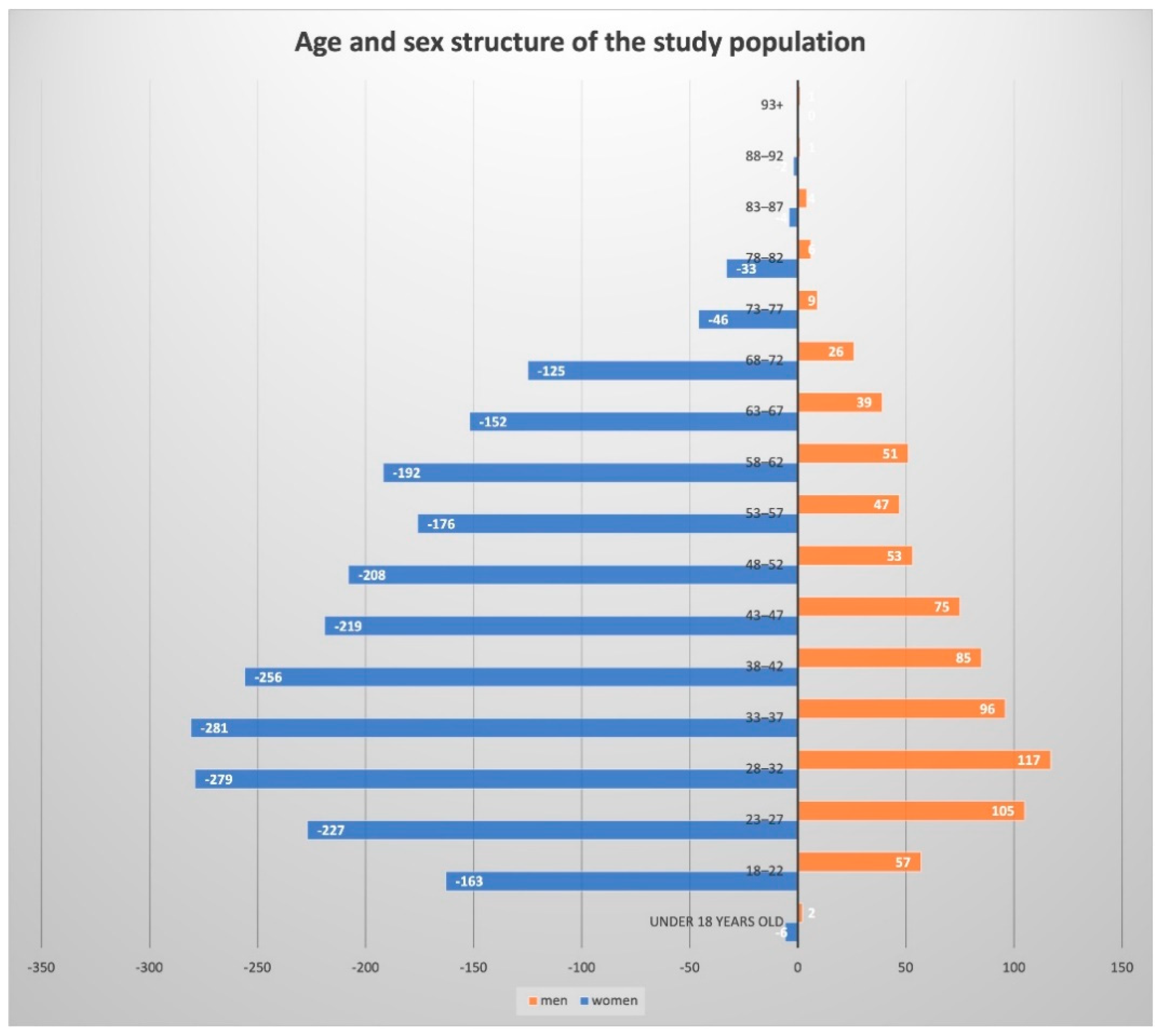
2.1. Program Participants
2.2. Analysis of Melanoma Awareness
- design and launch of information and educational “long-read” websites about melanoma and the importance of its early diagnosis;
- launch of advertising and informational videos on TV;
- launch of advertising and information programs on the radio;
- presentations of leading oncologists and/or dermatologists of the region on TV;
- announcement of the LFF program on the regional news channels and information websites;
- announcement of the LFF program in social networks;
- contextual advertising; and
- information in bloggers’ posts.
2.3. Examinations
2.4. Statistics
3. Results
3.1. Awareness of the Population about Early Detection of Skin Cancer
3.2. Organization of Skin Examination
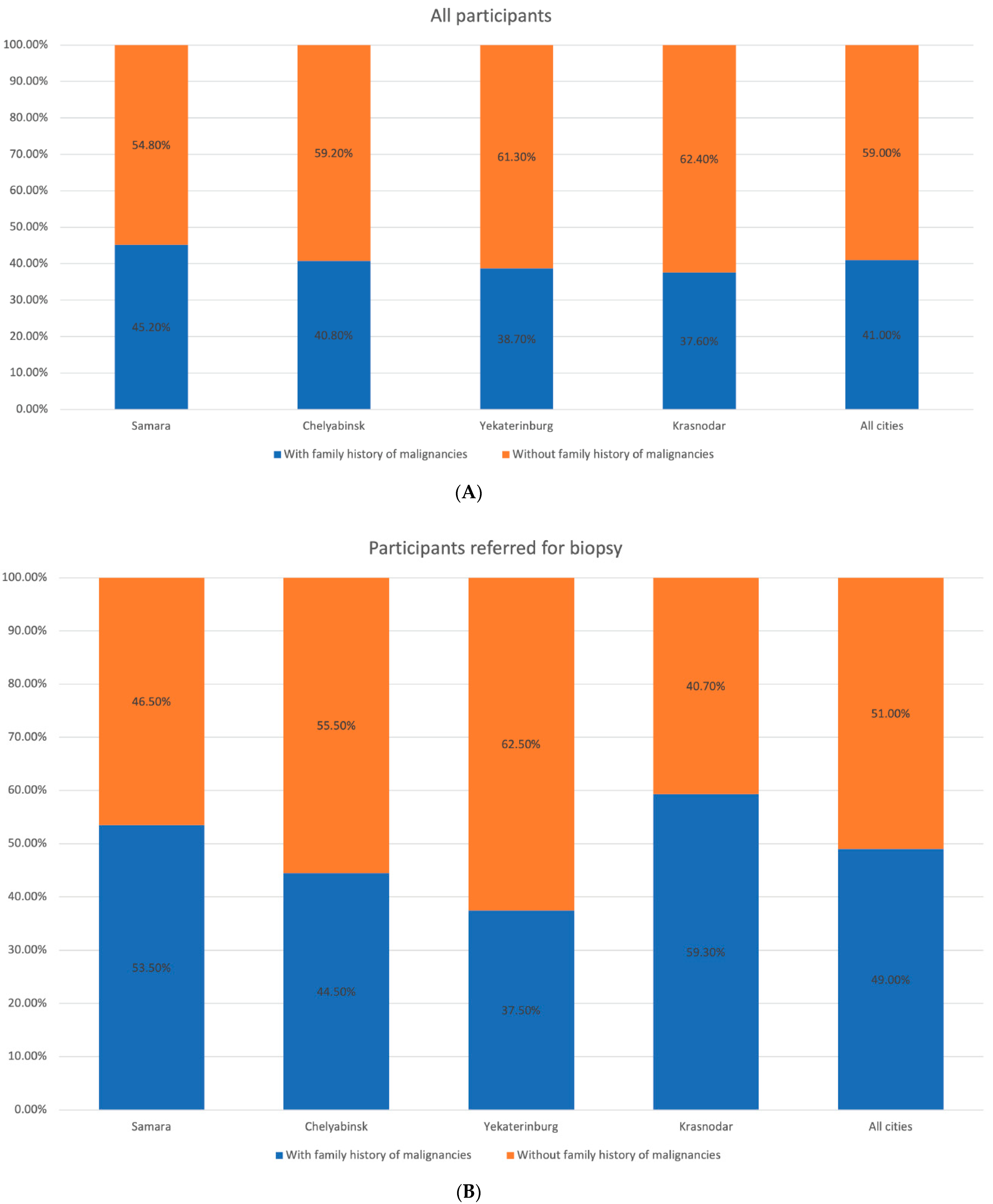
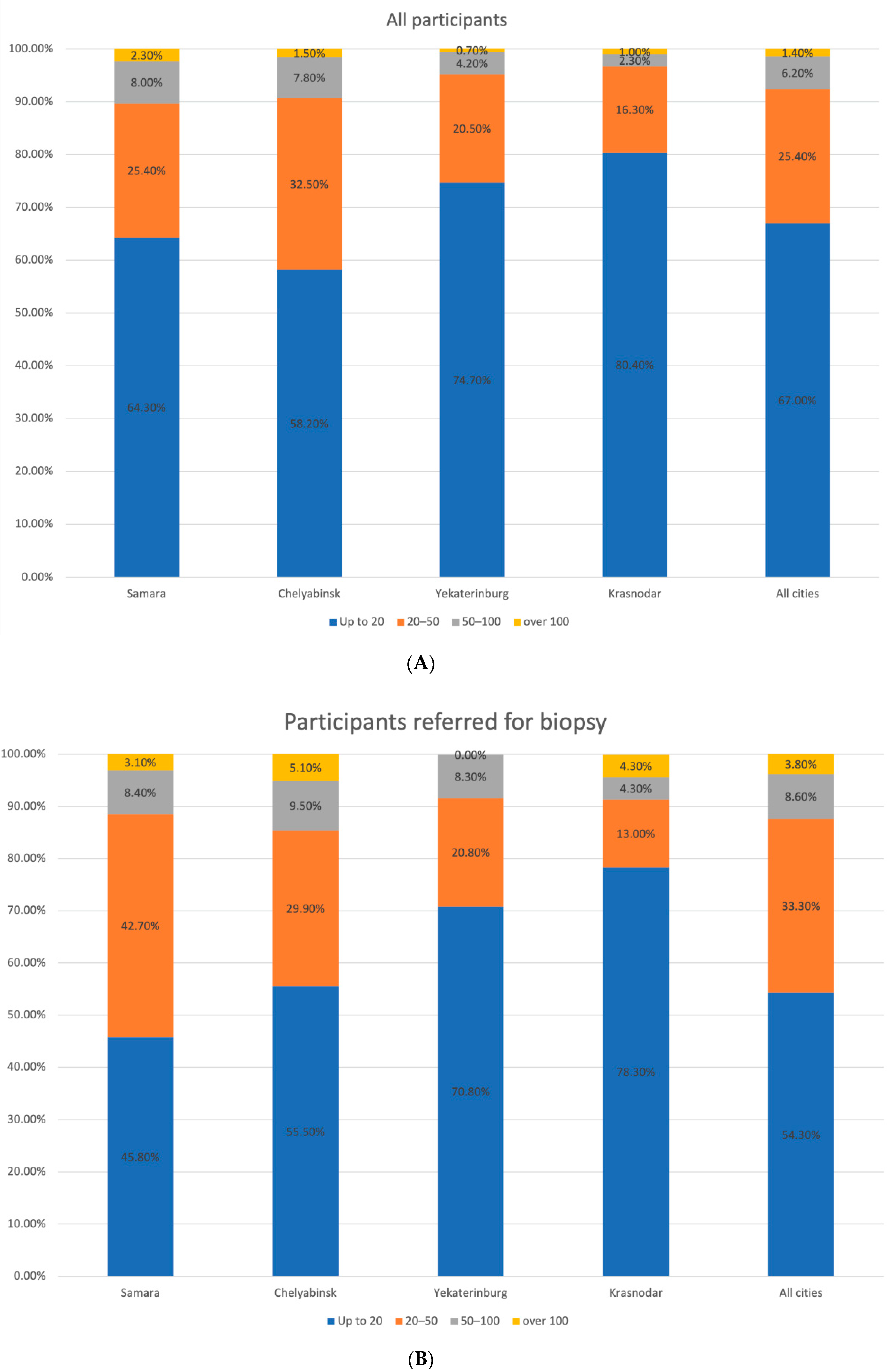
3.3. Evaluation of Risk Factors for CM
3.4. CM and NMSC Incidence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Kaprin, A.; Starinsky, V.; Shakhzadova, O. Status of Oncological Healthcare for the Population of Russia in 2019; P.A. Herzen Moscow Oncology Research Center affiliated to FSBI NMRRC of the Ministry of Health of Russia: Moscow, Russia, 2020; 239p. [Google Scholar]
- Van Akkooi, A.C.; de Wilt, J.H.; Verhoef, C.; Schmitz, P.I.; van Geel, A.N.; Eggermont, A.M.; Kliffen, M. Clinical relevance of melanoma micrometastases (<0.1 mm) in sentinel nodes: Are these nodes to be considered negative? Ann. Oncol. 2006, 17, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Samoylenko, I.; Kharkevich, G.; Utyashev, I.A.; Vishnevskaya, Y.V.; Orlova, K.V.; Demidov, L.V. Do we have early detection of cutaneous melanoma in Russia? Single-institution observational study. J. Clin. Oncol. 2014, 32 (Suppl. 15), e20027. [Google Scholar]
- Elmore, J.G.; Barnhill, R.L.; Elder, D.E.; Longton, G.M.; Pepe, M.S.; Reisch, L.M.; Carney, P.A.; Titus, L.J.; Nelson, H.D.; Onega, T. Pathologists’ diagnosis of invasive melanoma and melanocytic proliferations: Observer accuracy and reproducibility study. BMJ 2017, 28, 357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Analytical Report on the Level of Awareness of Russians about Melanoma and Willingness to Monitor the Condition of Their moles on Their Own or by a Doctor. 2019. Available online: https://wciom.ru/index.php?id=236&uid=9941 (accessed on 5 June 2020).
- Open Information and Educational Materials on the Prevention and Diagnosis of Cancer. 2019. Available online: https://xn--80aabdqdkeb7fkm5b.xn--p1ai (accessed on 5 June 2020).
- Krol, S.; Keijser, L.M.; van der Rhee, H.J.; Welvaart, K. Screening for skin cancer in The Netherlands. Acta Derm Venereol. 1991, 71, 317–321. [Google Scholar] [PubMed]
- Berwick, M.; Begg, C.B.; Fine, J.A.; Roush, G.C.; Barnhill, R.L. Screening for cutaneous melanoma by skin self-examination. J. Natl. Cancer Inst. 1996, 88, 17–23. [Google Scholar] [CrossRef] [PubMed]
- McPherson, M.; Elwood, M.; English, D.R.; Baade, P.D.; Youl, P.H.; Aitken, J.F. Presentation and detection of invasive melanoma in a high-risk population. J. Am. Acad. Dermatol. 2006, 54, 783–792. [Google Scholar] [CrossRef] [PubMed]
- Paoli, J.; Danielsson, M.; Wennberg, A.M. Results of the ‘Euromelanoma Day’ screening campaign in Sweden 2008. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 1304–1310. [Google Scholar] [CrossRef] [PubMed]
- Seidenari, S.; Benati, E.; Ponti, G.; Borsari, S.; Ferrari, C.; Albertini, G.; Arcangeli, F.; Aste, N.; Bernengo, M.G.; Bongiorno, M.R. Italian Euromelanoma Day Screening Campaign (2005–2007) and the planning of melanoma screening strategies. Eur J. Cancer Prev. 2012, 21, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Cristofolini, M.; Boi, S.; Cattoni, D.; Sicher, M.C.; Decarli, A.; Micciolo, R. A 10-Year Follow-Up Study of Subjects Recruited in a Health Campaign for the Early Diagnosis of Cutaneous Melanoma: Suggestions for the Screening Timetable. Dermatology 2015, 231, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Wernli, K.J.; Henrikson, N.B.; Morrison, C.C.; Nguyen, M.; Pocobelli, G.; Whitlock, E.P. Screening for Skin Cancer in Adults: An. Updated Systematic Evidence Review for the US Preventive Services Task Force; U.S. Preventive Services Task Force Evidence Syntheses, formerly Systematic Evidence Reviews: Rockville, MD, USA, 2016. [Google Scholar]
- Ferris, L.K.; Saul, M.I.; Lin, Y.; Ding, F.; Weinstock, M.A.; Geller, A.C.; Yuan, J.M.; Neuren, E.; Maddukuri, S.; Solano, F.X.; et al. A Large Skin Cancer Screening Quality Initiative: Description and First-Year Outcomes. JAMA Oncol. 2017, 3, 1112–1115. [Google Scholar] [CrossRef] [PubMed]
- Hubner, J.; Waldmann, A.; Eisemann, N.; Noftz, M.; Geller, A.C.; Weinstock, M.A.; Volkmer, B.; Greinert, R.; Breitbart, E.W.; Katalinic, A. Association between risk factors and detection of cutaneous melanoma in the setting of a population-based skin cancer screening. Eur. J. Cancer Prev. 2018, 27, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Palkina, N.; Sergeeva, O.; Ruksha, T.; Artyukhov, I. Melanoma Screening Day in Krasnoyarsk Krai of the Russian Federation: Results from 2015–2016. Asian Pac. J. Cancer Prev. 2018, 19, 503–507. [Google Scholar] [PubMed]
- Breitbart, E.W.; Waldmann, A.; Nolte, S.; Capellaro, M.; Greinert, R.; Volkmer, B.; Katalinic, A. Systematic skin cancer screening in Northern Germany. J. Am. Acad. Dermatol. 2012, 66, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Stang, A.; Jockel, K.H. Does skin cancer screening save lives? A detailed analysis of mortality time trends in Schleswig-Holstein and Germany. Cancer 2016, 122, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.S.; Moore, D.H.; Mendelsohn, M.L. Screening program reduced melanoma mortality at the Lawrence Livermore National Laboratory, 1984 to 1996. J. Am. Acad. Dermatol. 2008, 58, 741–749. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aitken, J.F.; Elwood, J.M.; Lowe, J.B.; Firman, D.W.; Balanda, K.P.; Ring, I.T. A randomized trial of population screening for melanoma. J. Med. Screen. 2002, 9, 33–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van der Leest, R.J.; de Vries, E.; Bulliard, J.L.; Paoli, J.; Peris, K.; Stratigos, A.J.; Trakatelli, M.; Maselis, T.J.E.M.L.; Šitum, M.; Pallouras, A.C.; et al. The Euromelanoma skin cancer prevention campaign in Europe: Characteristics and results of 2009 and 2010. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
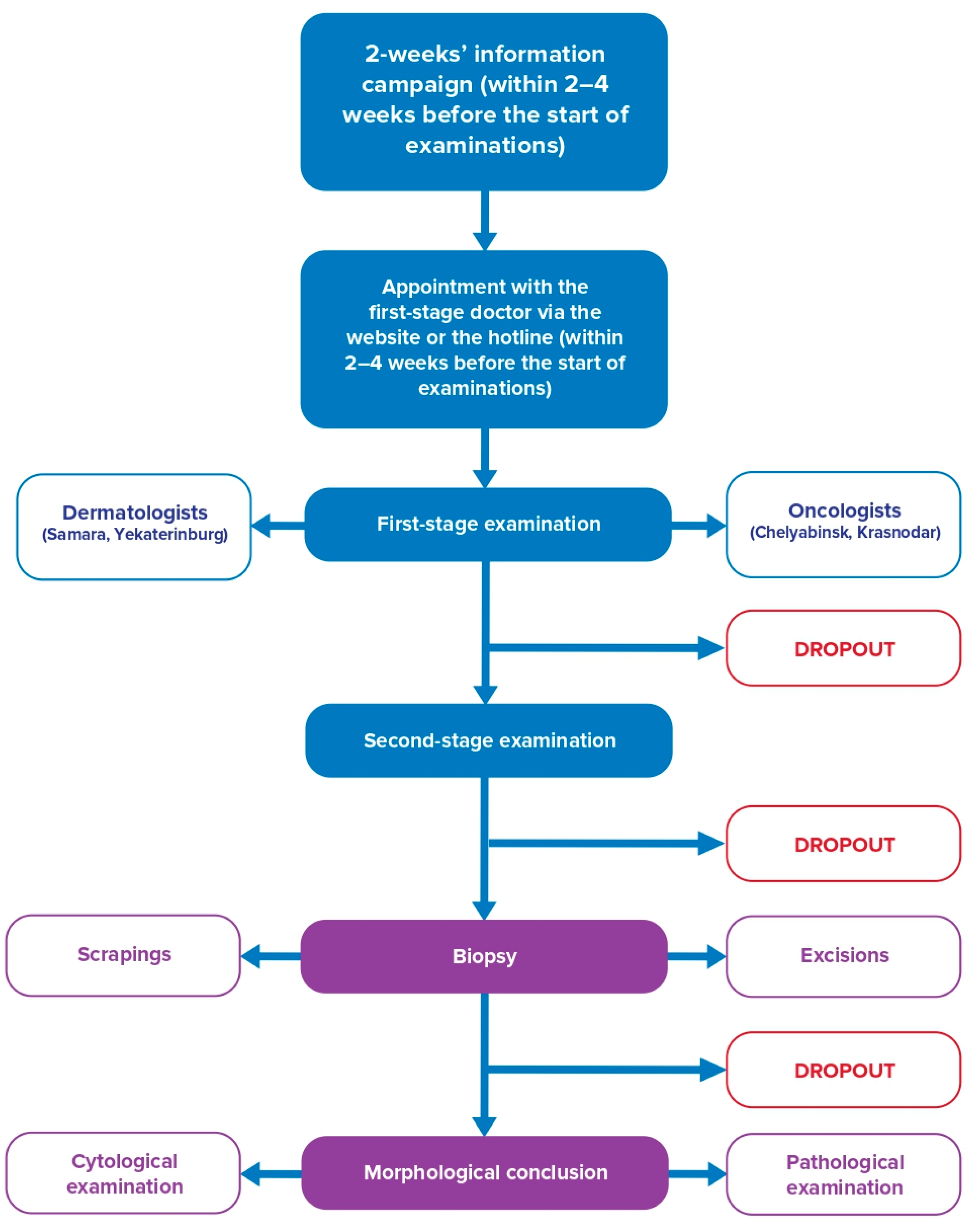
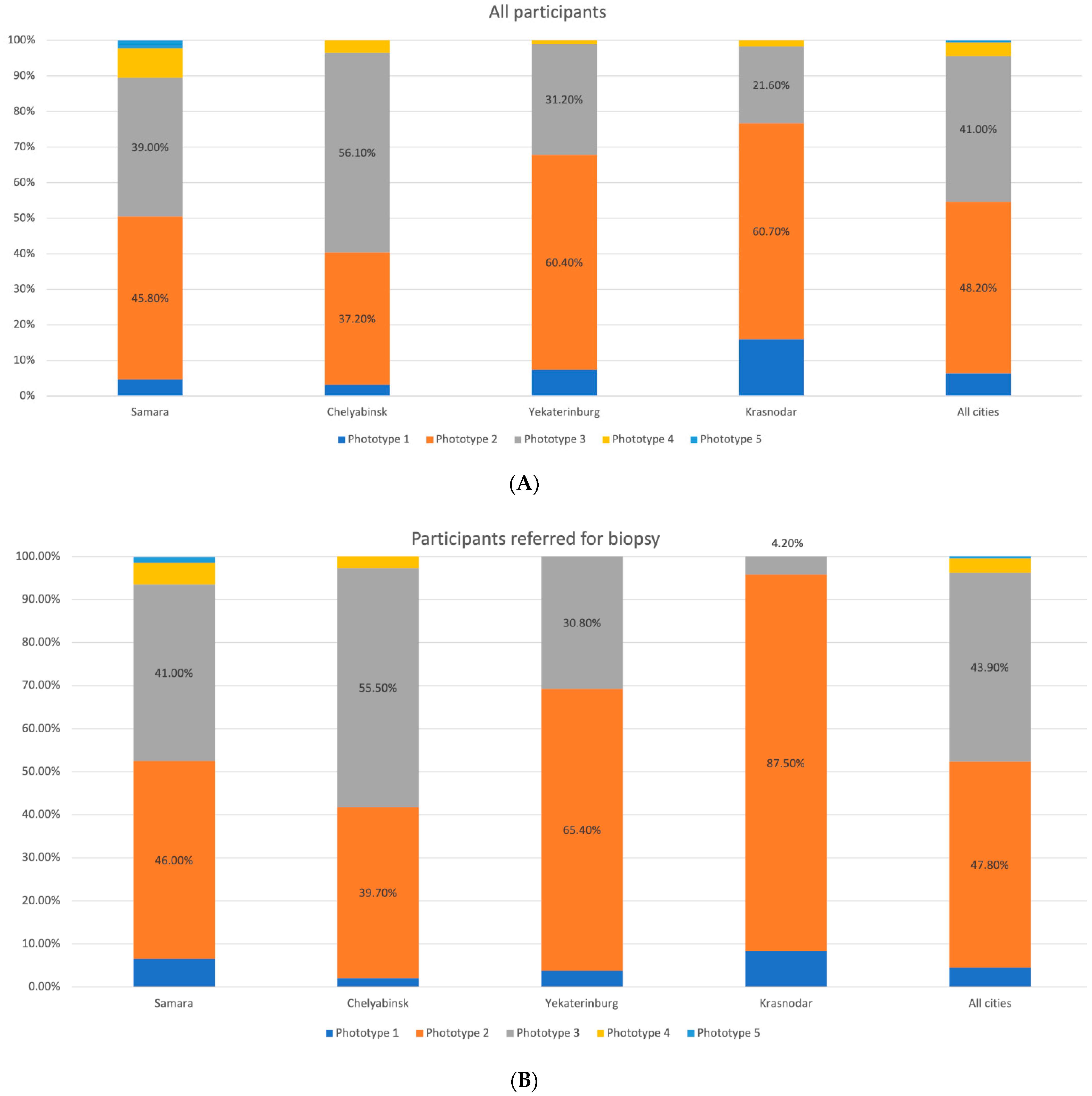
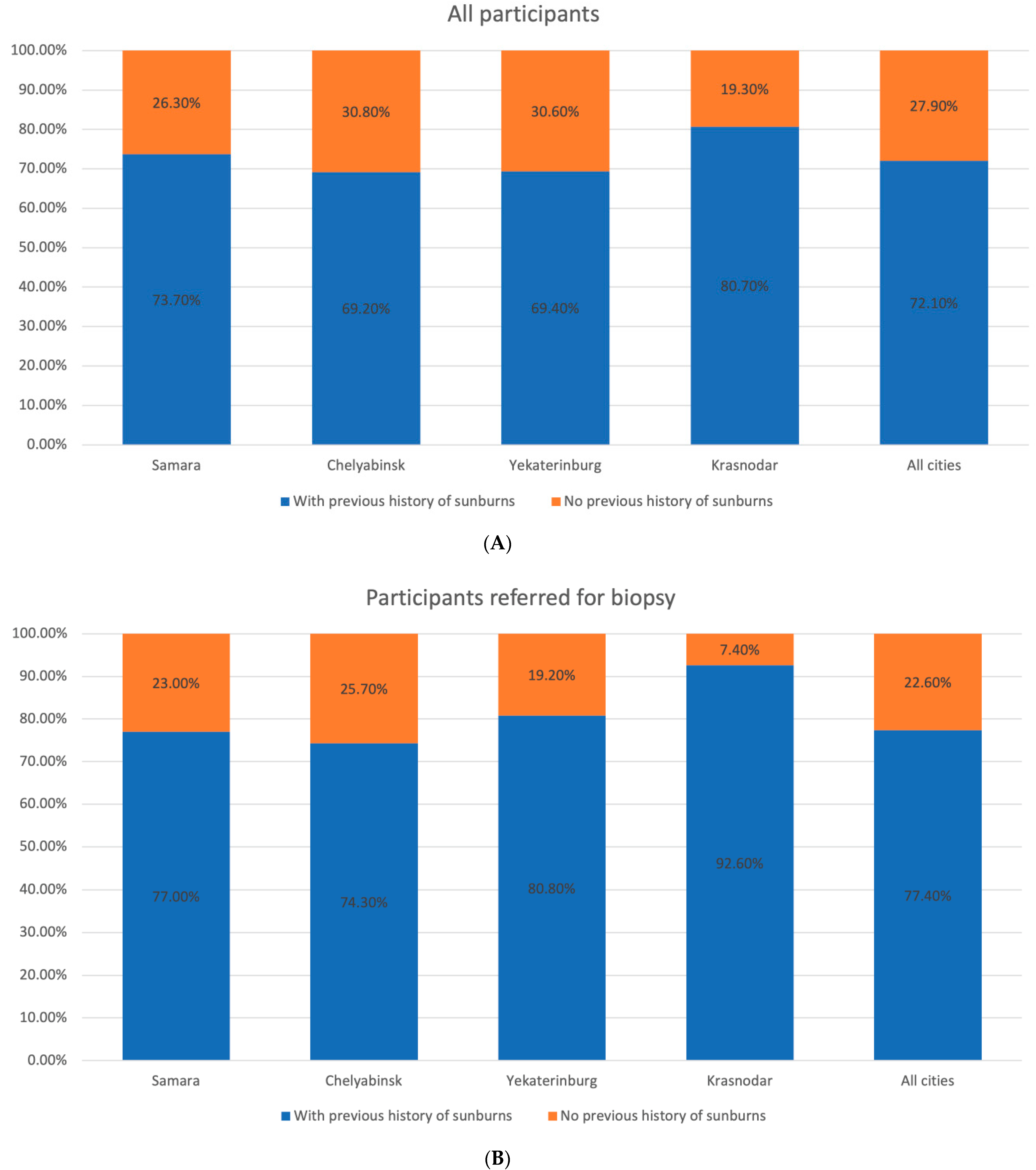

| Samara | Chelyabinsk | Yekaterinburg | Krasnodar | Total | |
|---|---|---|---|---|---|
| Information and communication campaign start date | 5 October 2019 | 8 October 2019 | 12 November 2019 | 26 November 2019 | - |
| Duration of the information and communication campaign before the start of the examinations (days) | 11 days | 18 days | 18 days | 18 days | - |
| Start and end dates for examination appointment | 5–14 October 2019 | 2 October 1–22 October 2019 | 8 October 1–2 December 2019 | 23 November 1–7 December 2019 | - |
| Start date for first-stage examinations | 15 October 2019 | 26 October 2019 | 3 December 2019 | 14 December 2019 | - |
| Duration of first-stage examinations (days) | 4 days (15–18 October) | 2 days (26–27 October) | 5 days (3–7 December) | 1 day (14 December) | 12 |
| Start date for second-stage examinations | 16 October 2019 | 26 October 2019 | 7 December 2019 | 14 December 2019 | - |
| Duration of second-stage examinations (days) | 4 days (16–19 October) | 2 days (26–27 October) | 6 days (7 December, 9–13 December) | 1 day (14 December) | 13 |
| City | Sex (M/F) | Age (Year of Birth) | Number of Nevi | Skin Photo Type | Sunburns | Previous Cancer in Personal History | Previous Cancer in Family History | Diagnosis | Depth of the Lesion |
|---|---|---|---|---|---|---|---|---|---|
| Yekaterinburg | M | 72 (1947) | Up to 20 | II | No | No | No | Melanoma | Up to 1 mm |
| Chelyabinsk | M | 40 (1979) | Up to 20 | III | Yes | No | Yes (father, gastric cancer) | Melanoma | 1 mm |
| Samara | F | 68 (1951) | Up to 20 | I | Yes | No | No | Melanoma | 1 mm |
| Krasnodar | F | 73 (1946) | Up to 20 | II | Yes | No | Yes (father, lung cancer) | Basal cell carcinoma | |
| Krasnodar | F | 72 (1947) | Up to 20 | II | Yes | No | Yes (mother, skin cancer) | Basal cell carcinoma | |
| Krasnodar | M | 71 (1947) | Up to 20 | II | Yes | Yes (prostate cancer) | No | Bowen’s disease | |
| Yekaterinburg | F | 68 (1951) | Up to 20 | II | Yes | No | No | Basal cell carcinoma | |
| Yekaterinburg | F | 66 (1953) | Up to 20 | II | Yes | No | No | Basal cell carcinoma | |
| Yekaterinburg | M | 68 (1951) | Up to 20 | III | Yes | No | Yes (father, mother) | Basal cell carcinoma | |
| Yekaterinburg | F | 81 (1938) | Up to 20 | II | Yes | No | Yes (mother, uterine cancer) | Basal cell carcinoma | |
| Yekaterinburg | F | 83 (1936) | Up to 20 | II | Yes | No | Yes (mother: lung cancer, sister: ovarian cancer) | Basal cell carcinoma | |
| Yekaterinburg | F | 56 (1963) | 20–50 | II | Yes | No | Yes (mother) | Basal cell carcinoma | |
| Chelyabinsk | M | 51 (1968) | 50–100 | III | Yes | No | Yes (father, gastric cancer) | Basal cell carcinoma | |
| Chelyabinsk | M | 48 (1970) | 50–100 | II | Yes | No | Yes | Basal cell carcinoma | |
| Chelyabinsk | M | 80 (1939) | Up to 20 | II | No | No | No | Basal cell carcinoma | |
| Chelyabinsk | F | 71 (1948) | Up to 20 | III | Yes | No | No | Basal cell carcinoma | |
| Samara | F | 70 (1949) | Up to 20 | III | No | No | No | Basal cell carcinoma | |
| Samara | F | 61 (1958) | Over 100 | III | Yes | No | No | Basal cell carcinoma | |
| Samara | F | 59 (1960) | 20–50 | II | Yes | Yes (basal cell carcinoma) | No data | Basal cell carcinoma |
| Biopsy Results | ||||
|---|---|---|---|---|
| Absolute Number | Percentage of All Participants (n = 3143) | Percentage of Participants Who were Referred for Biopsy/Underwent Biopsy without Referral (n = 100) 1 | ||
| Participants referred for biopsy: | Non-informative | 11 | 0.3 | 11.0 |
| Melanocytic nevus | 23 | 0.7 | 23.0 | |
| Other benign skin lesions | 3 | 0.1 | 3.0 | |
| Hemangioma | 1 | 0.0 | 1.0 | |
| Seborrheic keratosis | 5 | 0.2 | 5.0 | |
| Inflammatory skin diseases | 3 | 0.1 | 3.0 | |
| Sebaceous hyperplasia | 2 | 0.1 | 2.0 | |
| Ulcer | 1 | 0.0 | 1.0 | |
| Skin melanoma | 3 | 0.1 | 3.0 | |
| Skin cancer | 16 | 0.5 | 16.0 | |
| Did not have biopsy performed | 5 | 0.2 | 5.0 | |
| Did not attend a biopsy appointment | 27 | 0.9 | 27.0 | |
| Total | 100 | 3.2 | 100.0 | |
| Participants not referred for biopsy: | 3043 | 96.8 | ||
| Total | 3143 | 100.0 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demidov, L.; Samoylenko, I.; Vand, N.; Utyashev, I.; Shubina, I.; Sinelnikov, I. Screening for Melanoma and Other Skin Cancer Shows a Higher Early Melanoma Incidence: Social Educational Program “Life Fear-Free”. Dermatopathology 2021, 8, 54-68. https://0-doi-org.brum.beds.ac.uk/10.3390/dermatopathology8010011
Demidov L, Samoylenko I, Vand N, Utyashev I, Shubina I, Sinelnikov I. Screening for Melanoma and Other Skin Cancer Shows a Higher Early Melanoma Incidence: Social Educational Program “Life Fear-Free”. Dermatopathology. 2021; 8(1):54-68. https://0-doi-org.brum.beds.ac.uk/10.3390/dermatopathology8010011
Chicago/Turabian StyleDemidov, Lev, Igor Samoylenko, Nina Vand, Igor Utyashev, Irina Shubina, and Igor Sinelnikov. 2021. "Screening for Melanoma and Other Skin Cancer Shows a Higher Early Melanoma Incidence: Social Educational Program “Life Fear-Free”" Dermatopathology 8, no. 1: 54-68. https://0-doi-org.brum.beds.ac.uk/10.3390/dermatopathology8010011