Angiogenic Effects of Secreted Factors from Periodontal Ligament Stem Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture and CM Collection
2.2. Cell Viability
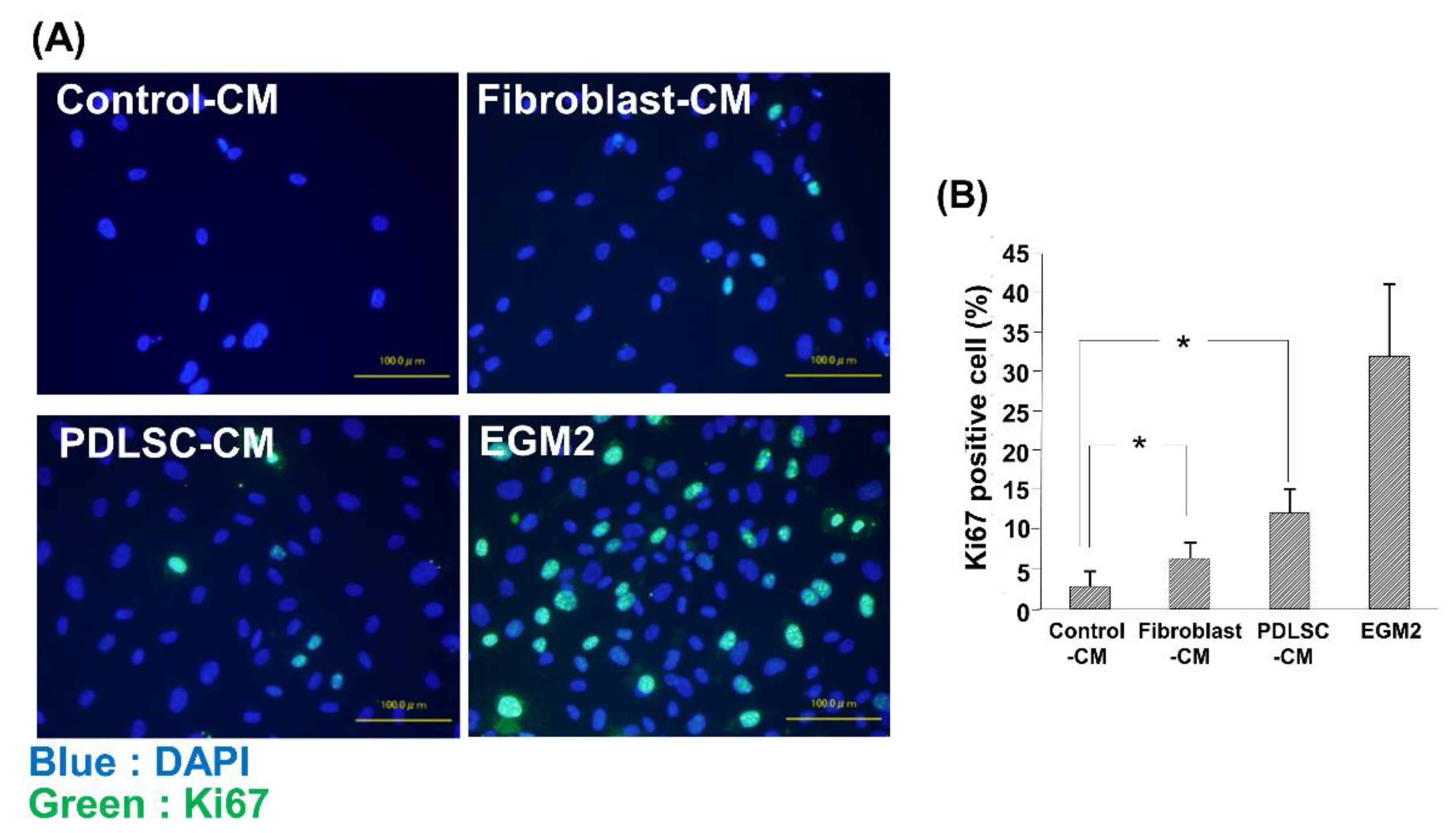
2.3. Ki67 Immunostaining and DAPI Staining
2.4. Network Formation
2.5. Measurement of Growth Factors
2.6. Statistical Analysis
3. Results
3.1. Isolation and Characterization of PDLSCs
3.2. Effect of CMs on the Cell Viability of Periodontal Tissue-Composing Cells
3.3. Effect of CM on the Apoptosis of HUVECs
3.4. Effect of CM on the Cell Proliferation of HUVECs
3.5. Effect of CM on the Network Formation of HUVECs
3.6. Angiogenic Factor in PDLSC-CM
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loos, B.-G.; Van Dyke, T.-E. The role of inflammation and genetics in periodontal disease. Periodontol. 2000 2020, 83, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef] [Green Version]
- Hajishengallis, G.; Chavakis, T.; Lambris, J.-D. Current understanding of periodontal disease pathogenesis and targets for host-modulation therapy. Periodontol. 2000 2020, 81, 14–34. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.D.; Papapanou, P.N.; Philips, K.H.; Offenbacher, S. Periodontal Medicine: 100 Years of Progress. J. Dent. Res. 2019, 98, 1053–1062. [Google Scholar] [CrossRef]
- Williams, R.C.; Offenbacher, S. Periodontal medicine: The emergence of a new branch of periodontology. Periodontol. 2000 2000, 23, 9–12. [Google Scholar] [CrossRef]
- Bosshardt, D.D.; Sculean, A. Does periodontal tissue regeneration really work? Periodontol. 2000 2009, 51, 208–219. [Google Scholar] [CrossRef]
- Reynolds, M.A.; Kao, R.T.; Camargo, P.M.; Caton, J.G.; Clem, D.S.; Fiorellini, J.P.; Geisinger, M.L.; Mills, M.P.; Nares, S.; Nevins, M.L. Periodontal regeneration-intrabony defects: A consensus report from the AAP Regeneration Workshop. J. Periodontol. 2015, 86, S105–S107. [Google Scholar] [CrossRef] [Green Version]
- Esposito, M.; Grusovin, M.G.; Papanikolaou, N.; Coulthard, P.; Worthington, H.V. Enamel matrix derivative (Emdogain(R)) for periodontal tissue regeneration in intrabony defects. Cochrane Database Syst. Rev. 2009, CD003875. [Google Scholar] [CrossRef] [Green Version]
- Needleman, I.G.; Worthington, H.V.; Giedrys-Leeper, E.; Tucker, R.J. Guided tissue regeneration for periodontal infra-bony defects. Cochrane Database Syst. Rev. 2006, CD001724. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Chailakhjan, R.K.; Lalykina, K.S. The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell Tissue Kinet. 1970, 3, 393–403. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prockop, D.J. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 1997, 276, 71–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, B.-M.; Miura, M.; Gronthos, S.; Bartold, P.M.; Batouli, S.; Brahim, J.; Young, M.; Robey, P.G.; Wang, C.-Y.; Shi, S. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004, 364, 149–155. [Google Scholar] [CrossRef]
- Akizuki, T.; Oda, S.; Komaki, M.; Tsuchioka, H.; Kawakatsu, N.; Kikuchi, A.; Yamato, M.; Okano, T.; Ishikawa, I. Application of periodontal ligament cell sheet for periodontal regeneration: A pilot study in beagle dogs. J. Periodontal Res. 2005, 40, 245–251. [Google Scholar] [CrossRef]
- Bassir, S.H.; Wisitrasameewong, W.; Raanan, J.; Ghaffarigarakani, S.; Chung, J.; Freire, M.; Andrada, L.C.; Intini, G. Potential for Stem Cell-Based Periodontal Therapy. J. Cell. Physiol. 2016, 231, 50–61. [Google Scholar] [CrossRef] [Green Version]
- Bright, R.; Hynes, K.; Gronthos, S.; Bartold, P.M. Periodontal ligament-derived cells for periodontal regeneration in animal models: A systematic review. J. Periodontal Res. 2015, 50, 160–172. [Google Scholar] [CrossRef]
- Chen, F.-M.; Gao, L.-N.; Tian, B.-M.; Zhang, X.-Y.; Zhang, Y.-J.; Dong, G.-Y.; Lu, H.; Chu, Q.; Xu, J.; Yu, Y.; et al. Treatment of periodontal intrabony defects using autologous periodontal ligament stem cells: A randomized clinical trial. Stem Cell Res. Ther. 2016, 7, 33. [Google Scholar] [CrossRef] [Green Version]
- Hasegawa, M.; Yamato, M.; Kikuchi, A.; Okano, T.; Ishikawa, I. Human periodontal ligament cell sheets can regenerate periodontal ligament tissue in an athymic rat model. Tissue Eng. 2005, 11, 469–478. [Google Scholar] [CrossRef]
- Iwasaki, K.; Komaki, M.; Yokoyama, N.; Tanaka, Y.; Taki, A.; Honda, I.; Kimura, Y.; Takeda, M.; Akazawa, K.; Oda, S.; et al. Periodontal regeneration using periodontal ligament stem cell-transferred amnion. Tissue Eng. Part A 2014, 20, 693–704. [Google Scholar] [CrossRef] [Green Version]
- Iwasaki, K.; Akazawa, K.; Nagata, M.; Komaki, M.; Honda, I.; Morioka, C.; Yokoyama, N.; Ayame, H.; Yamaki, K.; Tanaka, Y.; et al. The Fate of Transplanted Periodontal Ligament Stem Cells in Surgically Created Periodontal Defects in Rats. Int. J. Mol. Sci. 2019, 20, 192. [Google Scholar] [CrossRef] [Green Version]
- Nagata, M.; Iwasaki, K.; Akazawa, K.; Komaki, M.; Yokoyama, N.; Izumi, Y.; Morita, I. Conditioned Medium from Periodontal Ligament Stem Cells Enhances Periodontal Regeneration. Tissue Eng. Part A 2017, 23, 367–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.S.; Feng, Z.H.; Wu, G.F.; Bai, S.Z.; Dong, Y.; Chen, F.M.; Zhao, Y.M. The use of platelet-rich fibrin combined with periodontal ligament and jaw bone mesenchymal stem cell sheets for periodontal tissue engineering. Sci. Rep. 2016, 6, 28126. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Li, Z.; Tian, H.; Wang, W.; Cui, D.; Zhou, Z.; Chen, X.; Cheung, H.S.; Xu, G.T.; Chen, Y. Adult human periodontal ligament-derived stem cells delay retinal degeneration and maintain retinal function in RCS rats. Stem Cell Res. Ther. 2017, 8, 290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albuquerque-Souza, E.; Schulte, F.; Chen, T.; Hardt, M.; Hasturk, H.; Van Dyke, T.E.; Holzhausen, M.; Kantarci, A. Maresin-1 and Resolvin E1 Promote Regenerative Properties of Periodontal Ligament Stem Cells Under Inflammatory Conditions. Front. Immunol. 2020, 11, 585530. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Iwasaki, K.; Feghali, K.; Komaki, M.; Ishikawa, I.; Izumi, Y. Comparison of characteristics of periodontal ligament cells obtained from outgrowth and enzyme-digested culture methods. Arch. Oral Biol. 2011, 56, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa, J.; Komaki, M.; Yoshida, T.; Nakahama, K.; Amagasa, T.; Morita, I. Cell-printing and transfer technology applications for bone defects in mice. J. Tissue Eng. Regen. Med. 2011, 5, 695–703. [Google Scholar] [CrossRef]
- Mandelkow, R.; Gümbel, D.; Ahrend, H.; Kaul, A.; Zimmermann, U.; Burchardt, M.; Stope, M.B. Detection and Quantification of Nuclear Morphology Changes in Apoptotic Cells by Fluorescence Microscopy and Subsequent Analysis of Visualized Fluorescent Signals. Anticancer Res. 2017, 37, 2239–2244. [Google Scholar] [CrossRef] [Green Version]
- Mezentsev, A.; Merks, R.M.H.; O’Riordan, E.; Chen, J.; Mendelev, N.; Goligorsky, M.S.; Brodsky, S.V. Endothelial microparticles affect angiogenesis in vitro: Role of oxidative stress. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H1106–H1114. [Google Scholar] [CrossRef]
- Humphreys, B.D.; Cantaluppi, V.; Portilla, D.; Singbartl, K.; Yang, L.; Rosner, M.H.; Kellum, J.A.; Ronco, C. Acute Dialysis Quality Initiative (ADQI) XIII Work Group Targeting Endogenous Repair Pathways after AKI. J. Am. Soc. Nephrol. 2016, 27, 990–998. [Google Scholar] [CrossRef] [Green Version]
- Rowart, P.; Erpicum, P.; Detry, O.; Weekers, L.; Grégoire, C.; Lechanteur, C.; Briquet, A.; Beguin, Y.; Krzesinski, J.-M.; Jouret, F. Mesenchymal Stromal Cell Therapy in Ischemia/Reperfusion Injury. J. Immunol. Res. 2015, 2015, 602597. [Google Scholar] [CrossRef]
- Iwasaki, K.; Komaki, M.; Yokoyama, N.; Tanaka, Y.; Taki, A.; Kimura, Y.; Takeda, M.; Oda, S.; Izumi, Y.; Morita, I. Periodontal ligament stem cells possess the characteristics of pericytes. J. Periodontol. 2013, 84, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Liang, M. Periodontal ligament stem cells: Current status, concerns, and future prospects. Stem Cells Int. 2015, 2015, 972313. [Google Scholar] [CrossRef] [Green Version]
- Apte, R.S.; Chen, D.S.; Ferrara, N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell 2019, 176, 1248–1264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karaman, S.; Leppänen, V.-M.; Alitalo, K. Vascular endothelial growth factor signaling in development and disease. Development 2018, 145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bae, Y.-K.; Kim, G.-H.; Lee, J.C.; Seo, B.-M.; Joo, K.-M.; Lee, G.; Nam, H. The Significance of SDF-1α-CXCR4 Axis in in vivo Angiogenic Ability of Human Periodontal Ligament Stem Cells. Mol. Cells 2017, 40, 386–392. [Google Scholar] [CrossRef] [Green Version]
- Yeasmin, S.; Ceccarelli, J.; Vigen, M.; Carrion, B.; Putnam, A.J.; Tarle, S.A.; Kaigler, D. Stem cells derived from tooth periodontal ligament enhance functional angiogenesis by endothelial cells. Tissue Eng. Part A 2014, 20, 1188–1196. [Google Scholar] [CrossRef] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iwasaki, K.; Akazawa, K.; Nagata, M.; Komaki, M.; Peng, Y.; Umeda, M.; Watabe, T.; Morita, I. Angiogenic Effects of Secreted Factors from Periodontal Ligament Stem Cells. Dent. J. 2021, 9, 9. https://0-doi-org.brum.beds.ac.uk/10.3390/dj9010009
Iwasaki K, Akazawa K, Nagata M, Komaki M, Peng Y, Umeda M, Watabe T, Morita I. Angiogenic Effects of Secreted Factors from Periodontal Ligament Stem Cells. Dentistry Journal. 2021; 9(1):9. https://0-doi-org.brum.beds.ac.uk/10.3390/dj9010009
Chicago/Turabian StyleIwasaki, Kengo, Keiko Akazawa, Mizuki Nagata, Motohiro Komaki, Yihao Peng, Makoto Umeda, Tetsuro Watabe, and Ikuo Morita. 2021. "Angiogenic Effects of Secreted Factors from Periodontal Ligament Stem Cells" Dentistry Journal 9, no. 1: 9. https://0-doi-org.brum.beds.ac.uk/10.3390/dj9010009




