Oral Healthcare and Idiopathic Thrombocytopenic Purpura: Early Recognition, Dental Management and Case Report
Abstract
:1. Introduction
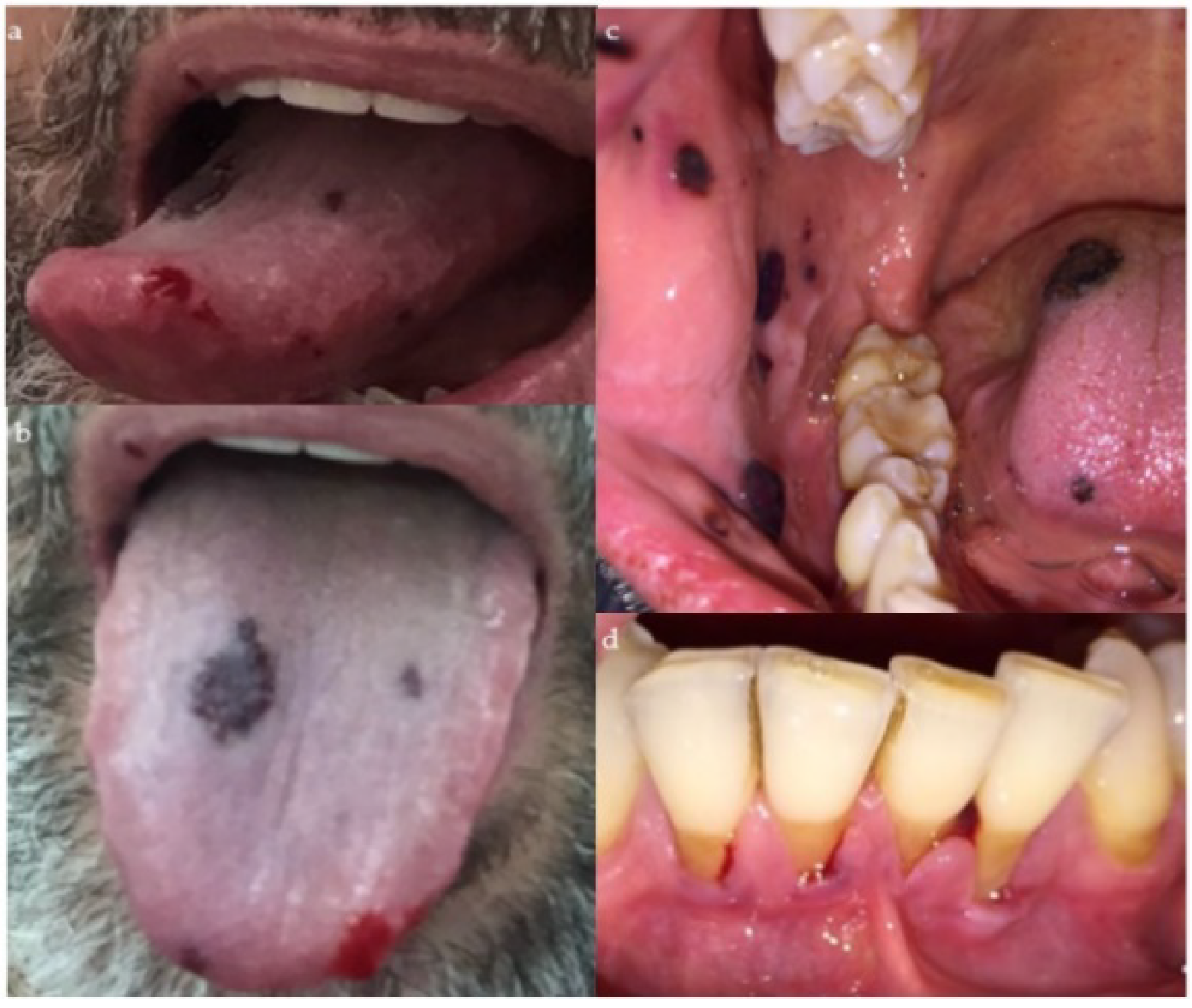
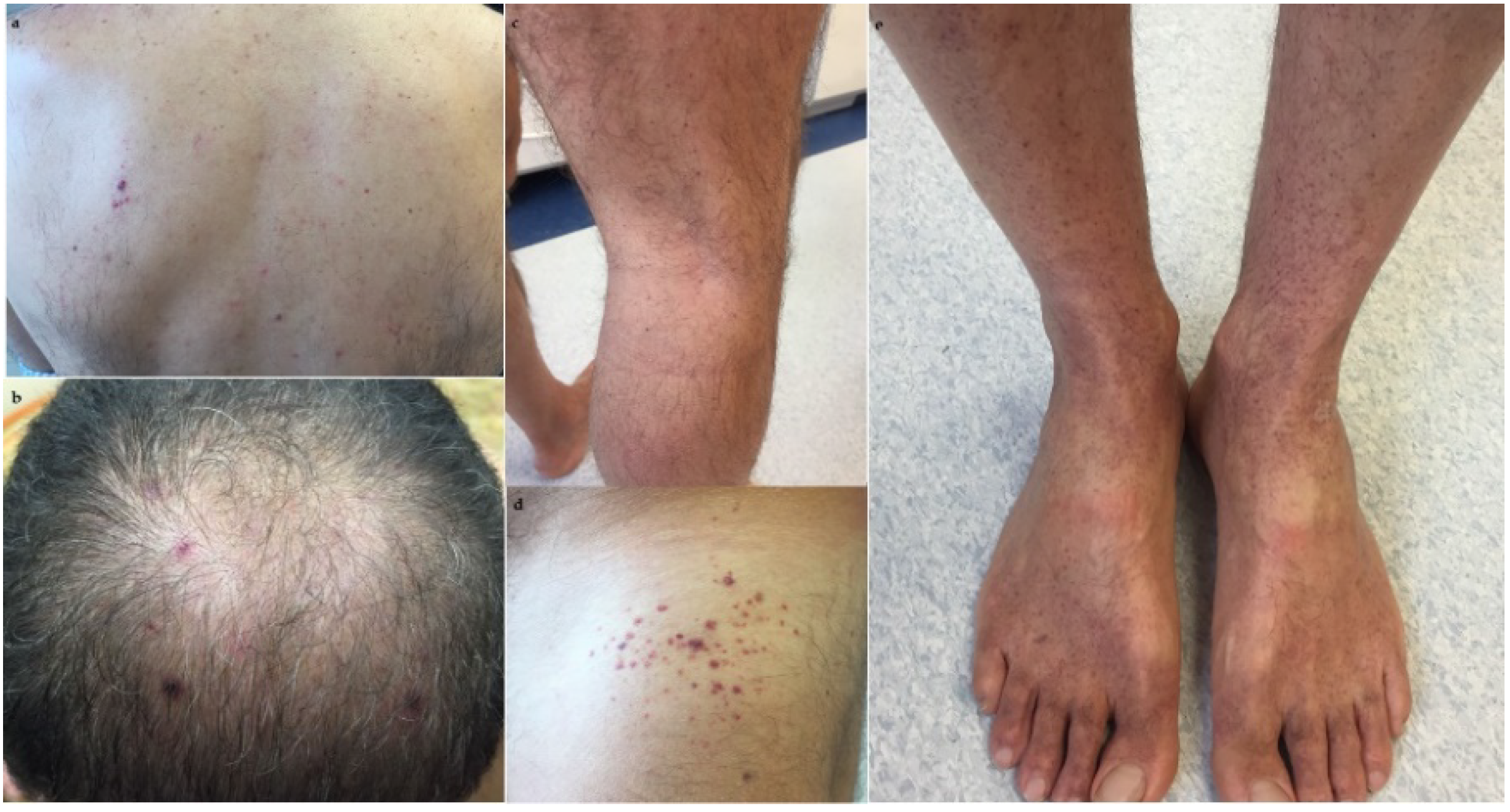
2. Case Presentation
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Uchiyama, M.; Hattori, A.; Tanaka, T.; Miyaji, T.; Matsuki, Y.; Fujii, T.; Ichiwata, T.; Hayashi, T.; Ikeda, T. Acute Idiopathic Thrombocytopenic Purpura Complicated with Diffuse Alveolar Hemorrhage in an Elderly Patient. Intern. Med. 2009, 48, 1449–1452. [Google Scholar] [CrossRef] [Green Version]
- Moulis, G.; Palmaro, A.; Montastruc, J.-L.; Godeau, B.; Lapeyre-Mestre, M.; Sailler, L. Epidemiology of incident immune thrombocytopenia: A nationwide population-based study in France. Blood 2014, 124, 3308–3315. [Google Scholar] [CrossRef] [Green Version]
- Cines, D.B.; Bussel, J.B.; Liebman, H.A.; Prak, E.T.L. The ITP syndrome: Pathogenic and clinical diversity. Blood 2009, 113, 6511–6521. [Google Scholar] [CrossRef] [Green Version]
- Rodeghiero, F.; Stasi, R.; Gernsheimer, T.; Michel, M.; Provan, D.; Arnold, D.M.; Bussel, J.B.; Cines, D.B.; Chong, B.H.; Cooper, N.; et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: Report from an international working group. Blood 2009, 113, 2386–2393. [Google Scholar] [CrossRef] [PubMed]
- Provan, D.; Stasi, R.; Newland, A.C.; Blanchette, V.S.; Bolton-Maggs, P.; Bussel, J.B.; Chong, B.H.; Cines, D.B.; Gernsheimer, T.B.; Godeau, B.; et al. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood 2010, 115, 168–186. [Google Scholar] [CrossRef]
- Neunert, C.; Lim, W.; Crowther, M.A.; Cohen, A.; Solberg, L. The American Society of Hematology 2011 evidence-based practice guideline for immune thrombocytopenia. Blood 2011, 117, 4190–4207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michel, M. Immune Thrombocytopenia Nomenclature, Consensus Reports, and Guidelines: What Are the Consequences for Daily Practice and Clinical Research? Semin. Hematol. 2013, 50, S50–S54. [Google Scholar] [CrossRef] [Green Version]
- Cines, D.B.; Blanchette, V.S. Immune thrombocytopenic purpura. N. Engl. J. Med. 2002, 346, 995–1008. [Google Scholar] [CrossRef] [PubMed]
- McMillan, R. The pathogenesis of chronic immune (idiopathic) thrombocytopenic purpura. Semin. Hematol. 2000, 37, 5–9. [Google Scholar] [CrossRef]
- Portielje, J.; Westendorp, R.G.J.; Kluin-Nelemans, H.C.; Brand, A. Morbidity and mortality in adults with idiopathic thrombocytopenic purpura. Blood 2001, 97, 2549–2554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lloyd, H.; Smith, J.; Wyngaarden, J.B. Cecil Textbook of Medicine; W B Saunders Co.: Amsterdam, The Netherlands, 1991. [Google Scholar]
- Sangwan, A.; Tewari, S.; Narula, S.C.; Sharma, R.K.; Sangwan, P. Significance of Periodontal Health in Primary Immune Thrombocytopenia: A Case Report and Review of Literature. J. Dent. 2013, 10, 197–202. [Google Scholar]
- Thachil, J.; Hall, G.W. Is this immune thrombocytopenic purpura? Arch. Dis. Child. 2008, 93, 76–81. [Google Scholar] [CrossRef]
- Guzeldemir, E. The role of oral hygiene in a patient with idiopathic thrombocytopenic purpura. Int. J. Dent. Hyg. 2009, 7, 289–293. [Google Scholar] [CrossRef]
- Geddis, E.A.; Balduini, C.L. Diagnosis of immune thrombocytopenic purpura in children. Curr. Opin. Hematol. 2007, 14, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Provan, D.; Newland, A.; Norfolk, D.; Bolton-Maggs, P.; Lilleyman, J.; Greer, I.; May, A.; Murphy, M.; Ouwehand, W.; Watson, S.; et al. Guidelines for the investigation and management of idiopathic thrombocytopenic purpura in adults, children and in pregnancy. Br. J. Haematol. 2003, 120, 574–596. [Google Scholar] [CrossRef] [Green Version]
- George, J.N.; Woolf, S.H.; Raskob, G.E.; Wasser, J.S.; Aledort, L.M.; Ballem, P.J.; Blanchette, V.S.; Bus-sel, J.B.; Cines, D.B.; Kelton, J.G.; et al. Idiopathic thrombocytopenic purpura: A practice guideline developed by explicit methods for the American Society of Hematology. Blood 1996, 88, 3–40. [Google Scholar] [CrossRef] [Green Version]
- Isola, G.; Matarese, G.; Cordasco, G.; Rotondo, F.; Crupi, A.; Ramaglia, L. Anticoagulant therapy in pa-tients undergoing dental interventions: A critical review of the literature and current perspectives. Minerva Stomatol. 2015, 64, 21–46. [Google Scholar] [PubMed]
- Matarese, G.; Isola, G.; Anastasi, G.P.; Favaloro, A.; Milardi, D.; Vermiglio, G.; Vita, G.; Cordasco, G.; Cutroneo, G. Immunohistochemical analysis of TGF-β1 and VEGF in gingival and periodontal tissues: A role of these biomarkers in the pathogenesis of scleroderma and periodontal disease. Int. J. Mol. Med. 2012, 30, 502–508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cirasino, L.; Robino, A.M.; Cattaneo, M.; Pioltelli, P.E.; Pogliani, E.M.; Terranova, L.; Morra, E.; Colombo, P.; Palmieri, G.A.; Piscitelli, P. Appropriate hospital management of adult immune thrombocytopenic purpura patients in major Italian institutions in 2000–2002: A retrospective analysis. Blood Coagul. Fibrinolysis 2010, 21, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, H.; Schmidt, K. The Incidence of Idiopathic Thrombocytopenic Purpura in Adults Increases With Age. Blood 1999, 94, 909–913. [Google Scholar] [CrossRef] [PubMed]
- Sbordone, C.; Toti, P.; Brevi, B.; Martuscelli, R.; Sbordone, L.; Di Spirito, F. Computed tomography-aided descriptive analysis of maxillary and mandibular atrophies. J. Stomatol. Oral Maxillofac. Surg. 2019, 120, 99–105. [Google Scholar] [CrossRef]
- James, W.D.; Guiry, C.; Grote, W.R. Acute idiopathic thrombocytopenic purpura. Oral Surg. Oral Med. Oral Pathol. 1984, 57, 149–151. [Google Scholar] [CrossRef]
- Di Spirito, F.; Pelella, S.; Argentino, S.; Sisalli, L.; Sbordone, L. Oral manifestations and the role of the oral healthcare workers in COVID-19. Oral Dis. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; Iacono, V.J.; Alfredo, I.; Alessandra, A.; Sbordone, L.; Lanza, A. Evidence-based Recommendations on Periodontal Practice and the Management of Periodontal Patients During and after the COVID-19 Era: Challenging Infectious Diseases Spread by Airborne Transmission. Open Dent. J. 2021, 15, 325–336. [Google Scholar] [CrossRef]
- Dunphy, L.; Williams, R. Immune thrombocytopenic purpura presenting with spontaneous gingival haemorrhage in pregnancy. BMJ Case Rep. 2019, 12, e228309. [Google Scholar] [CrossRef]
- Owais, Z.; Dane, J.; Cumming, C.G. Unprovoked periodontal hemorrhage, life-threatening anemia and idiopathic thrombocytopenia purpura: An unusual case report. Spéc. Care Dent. 2003, 23, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; Sbordone, L.; Pilone, V.; D’Ambrosio, F. Obesity and Periodontal Disease: A Narrative Review on Current Evidence and Putative Molecular Links. Open Dent. J. 2019, 13, 526–536. [Google Scholar] [CrossRef]
- Di Spirito, F.; Toti, P.; Pilone, V.; Carinci, F.; Lauritano, D.; Sbordone, L. The Association between Periodontitis and Human Colorectal Cancer: Genetic and Pathogenic Linkage. Life 2020, 10, 211. [Google Scholar] [CrossRef]
- Di Spirito, F.; La Rocca, M.; De Bernardo, M.; Rosa, N.; Sbordone, C.; Sbordone, L. Possible Association of Periodontal Disease and Macular Degeneration: A Case-Control Study. Dent. J. 2020, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Kunicki, T.J.; Newman, P.J. The molecular immunology of human platelet proteins. Blood 1992, 80, 1386–1404. [Google Scholar] [CrossRef]
- Panzer, S.; Rieger, M.; Vormittag, R.; Eichelberger, B.; Dunkler, D.; Pabinger, I. Platelet function to estimate the bleeding risk in autoimmune thrombocytopenia. Eur. J. Clin. Investig. 2007, 37, 814–819. [Google Scholar] [CrossRef]
- Lilleyman, J.S. Intracranial haemorrhage in idiopathic thrombocytopenic purpura. PaediatricHaematology Forum of the British Society for Haematology. Arch. Dis. Child. 1994, 71, 251–253. [Google Scholar] [CrossRef] [Green Version]
- Woerner, S.J.; Abilgard, C.F.; French, B.N. Intracranial hemorrhage in children with idiophaticthrom-bocytopenic purport. Pediatrics 1981, 67, 453–460. [Google Scholar] [PubMed]
- Barone, A.; Chatelain, S.; Derchi, G.; Di Spirito, F.; Martuscelli, R.; Porzio, M.; Sbordone, L. Antibiotic’s effectiveness after erupted tooth extractions: A retrospective study. Oral Dis. 2020, 26, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Ramaglia, L.; Di Spirito, F.; Sirignano, M.; La Rocca, M.; Esposito, U.; Sbordone, L. A 5-year longitudinal cohort study on crown to implant ratio effect on marginal bone level in single implants. Clin. Implant. Dent. Relat. Res. 2019, 21, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Cortelazzo, S.; Finazzi, G.; Buelli, M.; Molteni, A.; Viero, P.; Barbui, T. High risk of severe bleeding in aged pa-tients with chronic idiopathic thrombocytopenic purpura. Blood 1991, 77, 31–33. [Google Scholar] [CrossRef] [Green Version]
- Laino, L.; Cicciù, M.; Fiorillo, L.; Crimi, S.; Bianchi, A.; Amoroso, G.; Monte, I.P.; Herford, A.S.; Cervino, G. Surgical Risk on Patients with Coagulopathies: Guidelines on Hemophiliac Patients for Oro-Maxillofacial Surgery. Int. J. Environ. Res. Public Health 2019, 16, 1386. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaisman, B.; Medina, A.C.; Ramírez, G. Dental treatment for children with chronic idiopathic thrombocytopaenicpurpura: A report of two cases. Int. J. Paediatr. Dent. 2004, 14, 355–362. [Google Scholar] [CrossRef]
- Bansal, N.; Jindal, M.; Gupta, N.D.; Shukla, P. Clinical guidelines for periodontal management of idiopathic thrombocytopenic purpura: Current considerations. Int. J. Oral Health Sci. 2017, 7, 30. [Google Scholar] [CrossRef]


| Hematocrit | 48.6% | CK | 167 |
| Hemoglobin | 16.4 g/dL | AMS | 76 |
| White blood cells | 7.740 × 103 µL | AST | 25 |
| Red blood cells | 5.790 × 106 µL | LDH | 192 |
| Lymphocytes | 1.340 × 103 µL | PT (INR)/PTT | 89/29 |
| Neutrophils | 5.760 × 103 µL | D-dimers/INR | Absence/0.97 |
| MCV | 84 fL | ALC | 1.340 µL |
| Platelets | 1000 per mm3 | ANC | 5.760 µL |
| Cutaneous vasculitis | Vessel-related purpura characterized by an inflammatory process that mainly damages the vessels’ walls.In these the involvement of the skin appears prevalent but also that of the visible mucous membranes: polymorphic lesions appear (papules, purpuric or necrotic elements, wheals). The clinical manifestations vary according to the clinical form, whether it is subacute, acute or chronic, and the affected skin sites also vary from limited to diffuse forms. The substantial difference between the cutaneous manifestations of purpura and vasculitis can be seen in the objective clinical examination in which non-palpable purpurin papules and palpable vasculitic papules are observed [1,2,8]. |
| Rendu-Osler’s disease or hereditary hemorrhagic telangiectasia | Inherited autosomal dominant congenital disease. Characterized by epistaxis, multiple telangiectasias (cutaneous-mucous border of the lips, back of the tongue, mucosa of the inside of cheeks, gums, nose, fingers, etc.). Cutaneous and mucosal angiomas discolor or turn pale on digital pressure and this does not occur in purpura that does not discolor [3,9]. |
| Acute meningococcemia | The causative agent, Neisseria Meningitidis, colonizes the mucous membranes causing a wide spectrum of clinical syndromes including acute meningococcemia with early rash (isolated papules ranging in color from pink to purplish and purple), which heralds disseminated intravascular coagulation with relative skin manifestation of fulminant purpura [2]. |
| Schoenlei-Henoch syndrome or rheumatoid purpura | Hypersensitivity cutaneous vasculitis, common in childhood with a maximum peak of incidence between 5 and 6 years, but all age groups can be involved. It has palpable purpura and multiple point like hemorrhages that can be detected in the oral cavity, but no gingivorrhagia [2,8]. |
| Glanzman’sthromboathenia | Thrombopathy characterized by non-aggregation of platelets due to a defect in membrane glycoproteins. Severe mucosal bleeding associated with petechial and ecchymoticpurpura can occur [3,5,10]. |
| Leukemia | Neoplastic of hematopoietic cells, classified into myeloid or lymphoid, acute and chronic. The first symptoms are often represented by oral manifestations in acute leukemic disease, while in chronic they are less frequent. The neoplastic cells proliferate in the bone marrow and then in the tissues, giving pancytopenia and oral manifestations, secondary to thrombocytopenia or thrombocytopathies derived from the leukemic clone, such as: hemorrhagic blisters and bleeding of the oral mucosa as well as gastrointestinal, pulmonary and cerebral [3,9,10,20]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanza, A.; Di Spirito, F.; Petrosino, S.; Sbordone, L. Oral Healthcare and Idiopathic Thrombocytopenic Purpura: Early Recognition, Dental Management and Case Report. Dent. J. 2021, 9, 108. https://0-doi-org.brum.beds.ac.uk/10.3390/dj9090108
Lanza A, Di Spirito F, Petrosino S, Sbordone L. Oral Healthcare and Idiopathic Thrombocytopenic Purpura: Early Recognition, Dental Management and Case Report. Dentistry Journal. 2021; 9(9):108. https://0-doi-org.brum.beds.ac.uk/10.3390/dj9090108
Chicago/Turabian StyleLanza, Antonio, Federica Di Spirito, Serena Petrosino, and Ludovico Sbordone. 2021. "Oral Healthcare and Idiopathic Thrombocytopenic Purpura: Early Recognition, Dental Management and Case Report" Dentistry Journal 9, no. 9: 108. https://0-doi-org.brum.beds.ac.uk/10.3390/dj9090108






