Risk Factors, Clinical Characteristics, and Prognosis of Acute Kidney Injury in Hospitalized COVID-19 Patients: A Retrospective Cohort Study
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Design
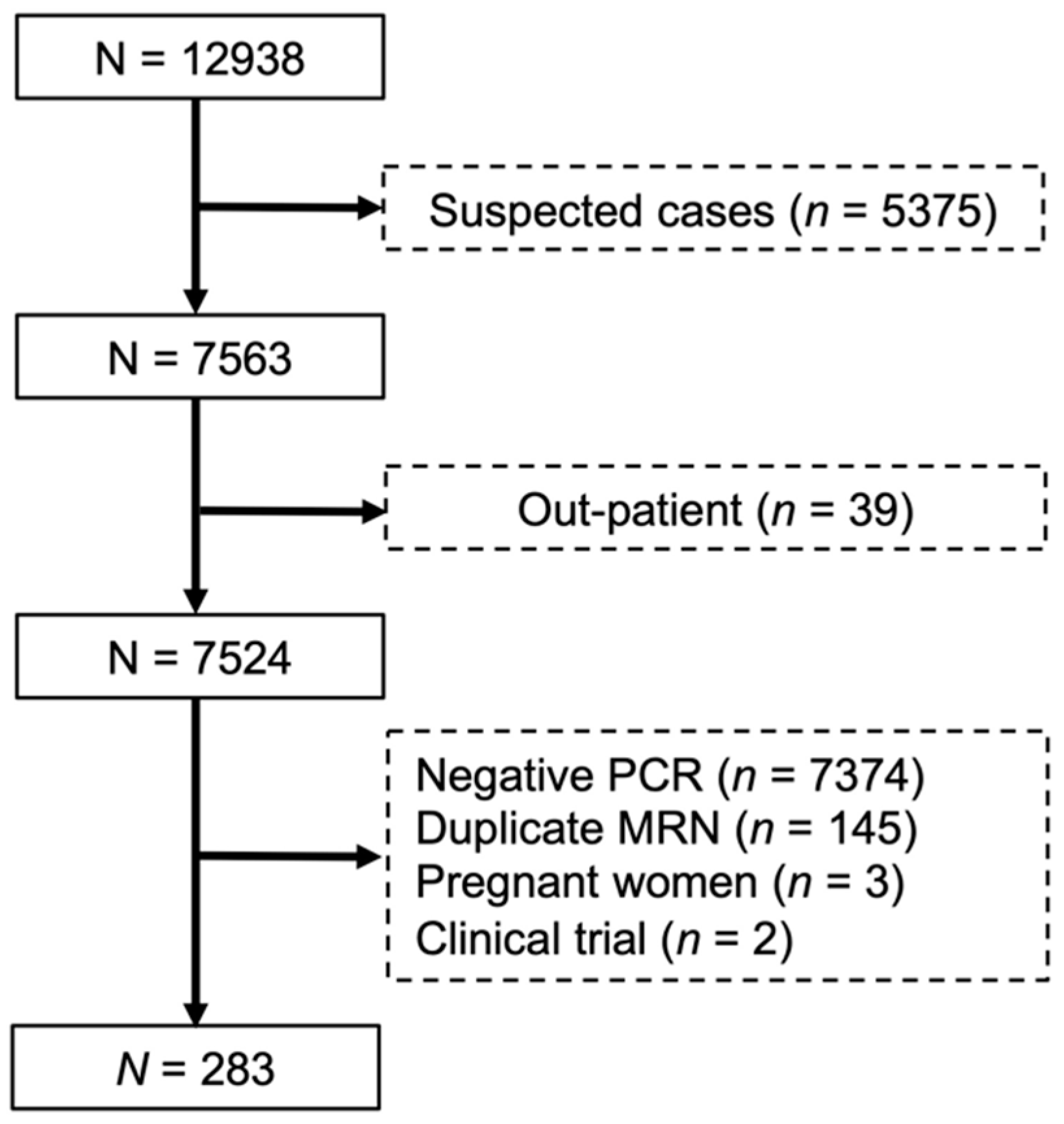
2.2. Data Source and Patient Population
2.3. Data Collection
2.4. Study Outcomes
2.5. Logistic Regression Analysis
2.6. Sensitivity Analysis
2.7. Mortality Analysis
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics, Interventions and Patient Outcomes
3.2. Univariate Analysis
3.3. Multivariate Analysis
3.4. Characteristics of Patients with AKI Classified by Its Origin
3.5. Characteristics of Patients with Intrinsic AKI
3.6. Mortality Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sawhney, S.; Marks, A.; Fluck, N.; Levin, A.; Prescott, G.; Black, C. Intermediate and Long-term Outcomes of Survivors of Acute Kidney Injury Episodes: A Large Population-Based Cohort Study. Am. J. Kidney Dis. 2017, 69, 18–28. [Google Scholar] [CrossRef]
- Lombardi, R.; Nin, N.; Peñuelas, Ó.; Ferreiro, A.; Rios, F.; Marin, M.C.; Raymondos, K.; Lorente, J.A.; Koh, Y.; Hurtado, J.; et al. Acute Kidney Injury in Mechanically Ventilated Patients: The Risk Factor Profile Depends on the Timing of Aki Onset. Shock Inj. Inflamm. Sepsis Lab. Clin. Approaches 2017, 48, 411–417. [Google Scholar] [CrossRef]
- Wang, H.E.; Muntner, P.; Chertow, G.M.; Warnock, D.G. Acute Kidney Injury and Mortality in Hospitalized Patients. Am. J. Nephrol. 2012, 35, 349–355. [Google Scholar] [CrossRef]
- World Health Organization. Coronavirus Disease (COVID-19) Pandemic. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 29 August 2020).
- Hansrivijit, P.; Qian, C.; Boonpheng, B.; Thongprayoon, C.; Vallabhajosyula, S.; Cheungpasitporn, W.; Ghahramani, N. Incidence of acute kidney injury and its association with mortality in patients with COVID-19: A meta-analysis. J. Investig. Med. 2020, 1–10. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W. The Northwell COVID-19 Research Consortium. Presenting Characteristics, Comorbidities, and Outcomes among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Khwaja, A. KDIGO Clinical Practice Guidelines for Acute Kidney Injury. Nephron Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., 3rd; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Kidney Disease Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2009, 113, S1–130. [Google Scholar] [CrossRef]
- Ronco, C. Cardiorenal Syndromes: Definition and Classification. Contrib. Nephrol. 2010, 164, 33–38. [Google Scholar] [CrossRef]
- Arroyo, V.; Ginès, P.; Gerbes, A.L.; Dudley, F.J.; Gentilini, P.; Laffi, G.; Reynolds, T.B.; Ring-Larsen, H.; Schölmerich, J. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. Hepatology 1996, 23, 164–176. [Google Scholar] [CrossRef]
- Heller, F.; Frischmann, S.; Gr\ufcnbaum, M.; Zidek, W.; Westhoff, T.H. Urinary Calprotectin and the Distinction between Prerenal and Intrinsic Acute Kidney Injury. Clin. J. Am. Soc. Nephrol. 2011, 6, 2347–2355. [Google Scholar] [CrossRef]
- Hansrivijit, P.; Yarlagadda, K.; Cheungpasitporn, W.; Thongprayoon, C.; Ghahramani, N. Hypoalbuminemia is associated with increased risk of acute kidney injury in hospitalized patients: A meta-analysis. J. Crit. Care 2021, 61, 96–102. [Google Scholar] [CrossRef]
- Cheng, Y.; Luo, R.; Wang, K.; Zhang, M.; Wang, Z.; Dong, L.; Li, J.; Yao, Y.; Ge, S.; Xu, G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020, 97, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.V.; Meghani, N.J.; Powell, B.M.; Newell, I.; Craven, R.; Skilton, G.; Bagg, L.J.; Yaqoob, I.; Dixon, M.J.; Evans, E.J.; et al. Patient characteristics and predictors of mortality in 470 adults admitted to a district general hospital in England with Covid-19. Epidemiol. Infect. 2020, 148, 1–28. [Google Scholar] [CrossRef]
- Lim, J.H.; Park, S.H.; Jeon, Y.; Cho, J.-H.; Jung, H.-Y.; Choi, J.-Y.; Kim, C.-D.; Lee, Y.-H.; Seo, H.; Lee, J.; et al. Fatal Outcomes of COVID-19 in Patients with Severe Acute Kidney Injury. J. Clin. Med. 2020, 9, 1718. [Google Scholar] [CrossRef]
- Hirsch, J.S.; Ng, J.H.; Ross, D.W.; Sharma, P.; Shah, H.H.; Barnett, R.L.; Hazzan, A.D.; Fishbane, S.; Jhaveri, K.D.; Abate, M.; et al. Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020, 98, 209–218. [Google Scholar] [CrossRef]
- Finlay, S.; Bray, B.; Lewington, A.J.; Hunter-Rowe, C.T.; Banerjee, A.; Atkinson, J.M.; Jones, M.C. Identification of risk factors associated with acute kidney injury in patients admitted to acute medical units. Clin. Med. 2013, 13, 233–238. [Google Scholar] [CrossRef]
- Ye, Q.; Wang, B.; Mao, J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J. Infect. 2020, 80, 607–613. [Google Scholar] [CrossRef]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. New Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Drury, D.R.; Henry, J.P.; Goodman, J. The effects of continuous pressure breathing on kidney function. J. Clin. Investig. 1947, 26, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Koyner, J.L.; Murray, P.T. Mechanical Ventilation and the Kidney. Blood Purif. 2010, 29, 52–68. [Google Scholar] [CrossRef]
- Santoriello, D.; Khairallah, P.; Bomback, A.S.; Xu, K.; Kudose, S.; Batal, I.; Barasch, J.; Radhakrishnan, J.; D’Agati, V.; Markowitz, G. Postmortem Kidney Pathology Findings in Patients with COVID-19. J. Am. Soc. Nephrol. 2020, 31, 2158–2167. [Google Scholar] [CrossRef] [PubMed]
- Golmai, P.; Larsen, C.P.; DeVita, M.V.; Wahl, S.J.; Weins, A.; Rennke, H.G.; Bijol, V.; Rosenstock, J.L. Histopathologic and Ultrastructural Findings in Postmortem Kidney Biopsy Material in 12 Patients with AKI and COVID-19. J. Am. Soc. Nephrol. 2020, 31, 1944–1947. [Google Scholar] [CrossRef]
- Su, H.; Yang, M.; Wan, C.; Yi, L.-X.; Tang, F.; Zhu, H.-Y.; Yi, F.; Yang, H.-C.; Fogo, A.B.; Nie, X.; et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020, 98, 219–227. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | All Patients (n = 283) | No AKI (n = 168) | AKI (n = 115) | p-Value |
|---|---|---|---|---|
| Male | 159 (56.2) | 86 (51.2) | 73 (63.5) | 0.041 * |
| Age (year) | 64.1 (15.9) † | 60.8 (16.4) † | 68.8 (14.0) † | <0.001 * |
| Ethnicity | ||||
| Caucasian | 143 (50.5) | 85 (50.6) | 58 (50.4) | |
| African-American | 88 (31.1) | 48 (28.6) | 40 (34.8) | |
| Hispanic/Latino | 32 (11.3) | 20 (11.9) | 12 (10.4) | 0.540 |
| Asian | 17 (6.0) | 13 (7.7) | 4 (3.5) | |
| Others | 3 (1.1) | 2 (1.2) | 1 (0.9) | |
| Co-morbidities | ||||
| Obesity (BMI ≥ 30 kg/m2) | 132 (46.6) | 78 (46.4) | 54 (47.0) | 0.930 |
| Current/former smokers | 109 (38.5) | 64 (38.1) | 45 (39.1) | 0.860 |
| Hypertension | 189 (66.8) | 104 (61.9) | 85 (73.9) | 0.035 * |
| Diabetes mellitus | 108 (38.2) | 52 (31.0) | 56 (48.7) | 0.003 * |
| Hyperlipidemia | 121 (42.8) | 65 (38.7) | 56 (48.7) | 0.095 |
| Coronary artery disease | 50 (17.7) | 25 (14.9) | 25 (21.7) | 0.137 |
| Heart failure/cardiomyopathy | 53 (18.7) | 27 (16.1) | 26 (22.6) | 0.166 |
| Arrhythmia/conduction disorders | 45 (15.9) | 19 (11.3) | 26 (22.6) | 0.011 * |
| Chronic kidney disease | 66 (23.3) | 22 (13.1) | 44 (38.3) | <0.001 * |
| Asthma/COPD | 73 (25.8) | 40 (23.8) | 33 (28.7) | 0.357 |
| Cerebrovascular disease | 40 (14.1) | 20 (11.9) | 20 (17.4) | 0.193 |
| Signs and symptoms | ||||
| Cough | 185 (65.4) | 121 (72.0) | 64 (55.7) | 0.004 * |
| Dyspnea | 203 (71.7) | 127 (75.6) | 76 (66.1) | 0.081 |
| Hypoxia (SpO2 < 95%) | 178 (62.9) | 101 (60.1) | 77 (67.0) | 0.242 |
| Rhinorrhea | 29 (10.2) | 18 (10.7) | 11 (9.6) | 0.754 |
| Fever/chills | 179 (63.3) | 108 (64.3) | 71 (61.7) | 0.663 |
| Chest pain | 35 (12.4) | 25 (14.9) | 10 (8.7) | 0.121 |
| Headache | 28 (9.9) | 20 (11.9) | 8 (7.0) | 0.224 |
| Gastrointestinal symptoms | 79 (27.9) | 49 (29.2) | 30 (26.1) | 0.571 |
| Asymptomatic | 8 (2.8) | 4 (2.4) | 4 (3.5) | 0.719 |
| Rales/crackles | 57 (20.1) | 32 (19.0) | 25 (21.7) | 0.579 |
| Rhonchi | 57 (20.1) | 27 (16.1) | 30 (26.1) | 0.039 * |
| Reduced breath sound | 63 (22.3) | 40 (23.8) | 23 (20.0) | 0.449 |
| Laboratory findings | ||||
| Leukopenia (WBC < 4000/uL) | 56 (19.8) | 37 (22.0) | 19 (16.5) | 0.254 |
| Leukocytosis (WBC > 10,000/uL) | 80 (28.3) | 26 (15.5) | 54 (47.0) | <0.001 * |
| Lymphocytopenia (ALC < 1000/uL) | 109 (38.7) | 31 (18.5) | 26 (22.6) | 0.392 |
| Thrombocytopenia (<140,000/uL) | 57 (20.1) | 14 (8.3) | 17 (14.8) | 0.088 |
| Thrombocytosis (>400,000/uL) | 31 (11.0) | 54 (32.1) | 55 (48.2) | 0.006 * |
| Respiratory acidosis | 43 (21.3) | 8 (7.3) | 35 (38.0) | < 0.001 * |
| Transaminitis (ALT > 3X UNL) | 33 (12.4) | 9 (5.8) | 24 (21.6) | < 0.001 * |
| Serum creatinine (mg/dL) on admission | 1.06 (0.72) ‡ | 1.37 (1.95) † | 1.92 (1.38)† | 0.005 * |
| eGFR (mL/min/1.73 m2) on admission | 64.2 (51.0) † | 76.5 (35.2) † | 46.2 (22.7)† | <0.001 * |
| Troponin I (>0.03 ng/mL) | 91 (39.1) | 31 (22.8) | 60 (61.9) | <0.001 * |
| Inflammatory markers | ||||
| D-dimer (>500 ng/mL) | 135 (80.4) | 64 (72.7) | 71 (88.8) | 0.009 * |
| Ferritin (>336 ng/mL) | 109 (65.3) | 47 (52.8) | 62 (79.5) | <0.001 * |
| Lactate dehydrogenase (>200 U/L) | 108 (73.0) | 50 (62.5) | 58 (85.3) | 0.002 * |
| C-reactive protein (>1 mg/dL) | 152 (87.4) | 82 (86.3) | 70 (88.6) | 0.651 |
| Procalcitonin (>0.25 ng/mL) | 73 (47.1) | 27 (32.5) | 46 (63.9) | <0.001 * |
| ST-T change on EKG | 84 (31.8) | 39 (25.0) | 45 (41.7) | 0.004 * |
| Radiographic findings | ||||
| Opacity/infiltrate | 209 (73.9) | 122 (72.6) | 87 (75.7) | 0.568 |
| Ground glass appearance | 79 (27.9) | 42 (25.0) | 37 (32.2) | 0.186 |
| Pleural effusion | 24 (8.5) | 11 (6.5) | 13 (11.3) | 0.158 |
| Pulmonary congestion | 30 (10.6) | 15 (8.9) | 15 (13.0) | 0.269 |
| Oxygen therapy/delivery | ||||
| Nasal cannula | 207 (73.1) | 121 (72.0) | 86 (74.8) | 0.607 |
| High-flow nasal cannula | 36 (12.7) | 18 (10.7) | 18 (15.7) | 0.221 |
| NIPPV | 28 (9.9) | 5 (3.0) | 23 (20.0) | <0.001 * |
| Mechanical ventilation | 58 (20.5) | 11 (6.5) | 47 (40.9) | <0.001 * |
| Intervention | ||||
| Intensive care unit | 89 (31.4) | 33 (19.6) | 56 (48.7) | <0.001 * |
| ECMO | 2 (0.7) | 1 (0.6) | 1 (0.9) | 1.000 |
| RRT | 16 (5.7) | 0 (0) | 16 (13.9) | <0.001 * |
| Vasopressor | 53 (18.7) | 9 (5.4) | 44 (38.5) | <0.001 * |
| Antibiotics | 220 (77.7) | 131 (78.0) | 89 (77.4) | 0.908 |
| Treatment | ||||
| Azithromycin | 182 (64.3) | 109 (64.9) | 73 (63.5) | 0.809 |
| Hydroxychloroquine | 67 (23.7) | 31 (18.5) | 36 (31.3) | 0.012 * |
| Steroids | 46 (16.3) | 16 (9.5) | 30 (26.1) | <0.001 * |
| Ascorbic acid | 57 (20.1) | 32 (19.0) | 25 (21.7) | 0.579 |
| Zinc | 54 (19.1) | 29 (17.3) | 25 (21.7) | 0.346 |
| Tocilizumab | 12 (4.2) | 3 (1.8) | 9 (7.8) | 0.017 * |
| Convalescent plasma | 36 (12.7) | 16 (9.5) | 20 (17.4) | 0.051 |
| Remdesivir | 25 (8.8) | 17 (10.1) | 8 (7.0) | 0.401 |
| Complications | ||||
| ARDS | 53 (18.7) | 13 (7.7) | 40 (34.8) | <0.001 * |
| Arrhythmias | 31 (11.0) | 11 (6.5) | 20 (17.4) | 0.004 * |
| QT prolongation | 25 (8.8) | 9 (5.4) | 16 (13.9) | 0.018 * |
| Venous thromboembolism | 10 (3.5) | 4 (2.4) | 6 (5.2) | 0.326 |
| Arterial thrombosis | 1 (0.4) | 0 (0) | 1 (0.9) | 0.406 |
| Cerebrovascular event | 3 (1.1) | 1 (0.6) | 2 (1.7) | 0.568 |
| Myocardial infection | 5 (1.8) | 1 (0.6) | 4 (3.5) | 0.162 |
| Heart failure | 16 (5.7) | 9 (5.4) | 7 (6.1) | 0.799 |
| Superimposed bacteremia | 19 (6.7) | 7 (4.2) | 12 (10.4) | 0.052 |
| Hospital stay (day) | 6.0 (7.0) ‡ | 6.1 (6.2) † | 10.1 (8.8) † | <0.001 * |
| Outcome | ||||
| Recovery/discharge | 223 (78.8) | 150 (89.3) | 73 (63.5) | <0.001 * |
| Remained hospitalized | 2 (0.7) | 1 (0.6) | 1 (0.9) | 1.000 |
| Death | 55 (19.4) | 15 (8.9) | 40 (34.8) | <0.001 * |
| Characteristics | Odds Ratio | 95% CI | p-Value |
|---|---|---|---|
| Male | 1.657 | 1.020–2.693 | 0.041 * |
| Age (1-year increment) | 1.034 | 1.017–1.051 | <0.001 * |
| Hypertension | 1.742 | 1.036–2.933 | 0.036 * |
| Diabetes mellitus | 2.119 | 1.297–3.460 | 0.003 * |
| Arrhythmia/conduction disorders | 2.294 | 1.199–4.357 | 0.012 * |
| Chronic kidney disease | 4.115 | 2.294–7.407 | <0.001 * |
| Cough | 0.487 | 0.296–0.803 | 0.005 * |
| Rhonchi | 1.842 | 1.017–3.311 | 0.041 * |
| Leukocytosis (WBC > 10,000/uL) | 4.831 | 2.770–8.403 | <0.001 * |
| Thrombocytosis (>400,000/uL) | 1.908 | 0.900–4.049 | 0.092 |
| Respiratory acidosis | 7.813 | 3.401–18.182 | <0.001 * |
| Transaminitis (ALT > 3X UNL) | 4.505 | 2.004–10.101 | <0.001 * |
| Serum creatinine (1.0 mg/dL increment) | 1.210 | 1.036–1.413 | 0.016 * |
| eGFR (1.0 mL/min/1.73 m2 increment) | 0.970 | 0.961–0.978 | <0.001 * |
| Troponin I (>0.03 ng/mL) | 5.495 | 3.096–9.709 | <0.001 * |
| D-dimer (>500 ng/mL) | 2.959 | 1.280–6.849 | 0.011 * |
| Ferritin (>336 ng/mL) | 3.460 | 1.739–6.897 | <0.001 * |
| Lactate dehydrogenase (>200 U/L) | 3.484 | 1.548–7.813 | 0.003 * |
| Procalcitonin (>0.25 ng/mL) | 3.663 | 1.887–7.143 | <0.001 * |
| ST-T change on EKG | 2.141 | 1.266–3.623 | 0.005 * |
| NIPPV | 8.130 | 2.994–22.222 | <0.001 * |
| Mechanical ventilation | 9.901 | 4.831–20.000 | <0.001 * |
| Intensive care unit admission | 3.876 | 2.288–6.579 | <0.001 * |
| Vasopressor | 10.989 | 5.076–23.810 | <0.001 * |
| Hydroxychloroquine | 2.012 | 1.157–3.509 | 0.013 * |
| Steroids | 3.356 | 1.730–6.494 | <0.001 * |
| Tocilizumab | 4.673 | 1.236–17.544 | 0.023 * |
| ARDS | 6.369 | 3.205–12.658 | <0.001 * |
| Arrhythmias as complication | 3.003 | 1.379–6.536 | 0.006 * |
| QT prolongation | 2.857 | 1.215–6.711 | 0.016 * |
| Characteristics | Statistics | ||
|---|---|---|---|
| Odds Ratio | 95% CI | p-Value | |
| Model 1 | |||
| Male | 2.285 | 1.338–3.902 | 0.002 * |
| Age (1-year increment) | 1.050 | 1.029–1.071 | <0.001 * |
| Hypertension | 1.087 | 0.604–1.957 | 0.780 |
| Diabetes mellitus | 1.855 | 1.094–3.145 | 0.022 * |
| Model 2 | |||
| Chronic kidney disease | 2.488 | 1.302–4.762 | 0.006 * |
| Cough | 0.578 | 0.328–1.019 | 0.058 |
| Rhonchi | 1.667 | 0.880–3.155 | 0.117 |
| Leukocytosis (WBC > 10,000/uL) | 4.525 | 2.494–8.197 | <0.001 * |
| Thrombocytosis (>400,000/uL) | 1.961 | 0.868–4.425 | 0.105 |
| Transaminitis (ALT > 3X UNL) | 4.405 | 1.770–10.870 | 0.001 * |
| Troponin I (>0.03 ng/mL) | 4.032 | 2.179–7.519 | <0.001 * |
| Model 3 | |||
| Serum creatinine (1.0 mg/dL increment) | 1.081 | 0.933–1.252 | 0.300 |
| eGFR (1.0 mL/min/1.73 m2 decrement) | 1.027 | 1.016–1.037 | <0.001 * |
| NIPPV | 6.329 | 2.179–18.519 | 0.001 * |
| Mechanical ventilation | 9.434 | 4.310–20.408 | <0.001 * |
| Intensive care unit admission | 3.597 | 2.000–6.494 | <0.001 * |
| Vasopressor | 10.309 | 4.505–23.810 | <0.001 * |
| ARDS | 4.926 | 2.342–10.309 | <0.001 * |
| Model 4 | |||
| Respiratory acidosis | 7.042 | 2.710–18.182 | <0.001 * |
| D-dimer (>500 ng/mL) | 1.992 | 0.766–5.181 | 0.157 |
| Ferritin (>336 ng/mL) | 2.710 | 1.093–6.711 | 0.031 * |
| Lactate dehydrogenase (>200 U/L) | 3.663 | 1.376–9.804 | 0.009 * |
| Procalcitonin (>0.25 ng/mL) | 3.448 | 1.595–7.463 | 0.002 * |
| Hydroxychloroquine | 1.689 | 0.897–3.185 | 0.104 |
| Steroids | 2.591 | 1.205–5.556 | 0.015 * |
| Tocilizumab | 4.032 | 0.986–16.393 | 0.052 |
| Model 5 | |||
| Arrhythmia/conduction disorders | 1.721 | 0.779–3.802 | 0.179 |
| ST-T change on EKG | 1.852 | 1.020–3.356 | 0.043 * |
| Arrhythmias as complication | 2.217 | 0.927–5.319 | 0.074 |
| QT prolongation | 2.114 | 0.821–5.435 | 0.121 |
| Characteristics | Community-Acquired AKI (n = 77) | Hospital-Acquired AKI (n = 21) | p-Value |
|---|---|---|---|
| Male | 48 (62.3) | 11 (52.4) | 0.456 |
| Age (year) † | 67.6 (15.1) | 71.3 (10.8) | 0.214 |
| Blood chemistry | |||
| Baseline serum creatinine (mg/dL) † | 0.95 (0.34) | 0.95 (0.27) | 0.894 |
| Peak serum creatinine (mg/dL) † | 2.35 (1.69) | 3.45 (1.80) | 0.018 * |
| Lowest eGFR (mL/kg/1.73 m2) † | 38.6 (21.1) | 24.2 (18.2) | 0.004 * |
| Blood urea nitrogen (mg/dL) † | 49.0 (33.8) | 80.8 (42.4) | 0.004 * |
| Serum sodium (mEq/L) §,† | 139 (10) | 142 (7) | 0.213 |
| Serum potassium (mEq/L) §,† | 5.1 (5.0) | 5.1 (0.9) | 0.929 |
| AKI staging | |||
| Stage 1 | 36 (46.8) | 5 (23.8) | |
| Stage 2 | 24 (31.2) | 5 (23.8) | 0.021 * |
| Stage 3 | 17 (22.1) | 11 (52.4) | |
| Urine studies § | |||
| Urine WBC > 3 or positive leukocyte esterase | 23 (40.4) | 5 (26.3) | 0.272 |
| Urine RBC > 3 or positive blood | 37 (64.9) | 12 (63.2) | 1.000 |
| Urine protein | |||
| Trace | 1 (1.8) | 0 (0) | |
| 1+ | 21 (36.8) | 4 (21.1) | |
| 2+ | 16 (28.1) | 6 (31.6) | 0.661 |
| 3+ | 5 (8.8) | 3 (15.8) | |
| Urine chemistry § | |||
| Fractional excretion of sodium † (%, n = 19) | 0.6 (0.5) | 0.2 (0.1) | 0.015 * |
| Fractional excretion of urea † (%, n = 14) | 18.4 (9.7) | 23.4 (21.1) | 0.727 |
| Etiology | |||
| Pre-renal | 64 (83.1) | 4 (19.0) | <0.001 * |
| Cardiorenal | 1 (1.3) | 1 (4.8) | 0.320 |
| Hepatorenal | 0 (0) | 0 (0) | - |
| Intrinsic AKI | 9 (11.7) | 16 (76.2) | <0.001 * |
| Post-renal | 3 (3.9) | 0 (0) | 0.358 |
| Co-morbidities | |||
| Obesity (BMI ≥ 30 kg/m2) | 30 (39.0) | 13 (61.9) | 0.083 |
| Current/former smokers | 30 (39.0) | 10 (47.6) | 0.617 |
| Hypertension | 56 (72.7) | 15 (71.4) | 1.000 |
| Diabetes mellitus | 33 (42.9) | 13 (61.9) | 0.144 |
| Hyperlipidemia | 35 (45.5) | 11 (52.4) | 0.627 |
| Coronary artery disease | 16 (20.8) | 5 (23.8) | 0.764 |
| Heart failure/cardiomyopathy | 17 (22.1) | 5 (23.8) | 0.866 |
| Arrhythmia/conduction disorders | 16 (20.8) | 3 (14.3) | 0.505 |
| Chronic kidney disease | 25 (32.5) | 7 (33.3) | 0.941 |
| Asthma/COPD | 25 (32.5) | 5 (23.8) | 0.445 |
| Cerebrovascular disease | 15 (19.5) | 4 (19.0) | 0965 |
| Outcome | |||
| Need for RRT | 4 (5.2) | 7 (33.3) | <0.001 * |
| Hospital stay † (day) | 8.4 (7.5) | 12.4 (9.9) | 0.098 |
| Death | 16 (20.8) | 14 (66.7) | <0.001 * |
| Characteristics | Intrinsic AKI (n = 40) | Other Causes of AKI (n = 75) | p-Value |
|---|---|---|---|
| Male | 31 (77.5) | 42 (56.0) | 0.026 * |
| Age (year) † | 69.8 (11.1) | 68.2 (15.4) | 0.551 |
| Hospital-acquired | 16 (40.0) | 5 (6.7) | <0.001 * |
| Blood chemistry | |||
| Baseline serum creatinine (mg/dL) † | 1.20 (0.40) | 0.90 (0.32) | <0.001 * |
| Peak serum creatinine (mg/dL) † | 4.35 (1.95) | 1.93 (1.13) | <0.001 * |
| Lowest eGFR (mL/kg/1.73 m2) † | 17.8 (13.1) | 41.8 (19.7) | <0.001 * |
| Blood urea nitrogen (mg/dL) † | 85 (36) | 45 (30) | <0.001 * |
| Serum sodium (mEq/L) §,† | 142 (8) | 140 (10) | 0.172 |
| Serum potassium (mEq/L) §,† | 5.2 (0.8) | 5.0 (5.1) | 0.742 |
| AKI staging | |||
| Stage 1 | 5 (12.5) | 41 (54.7) | |
| Stage 2 | 12 (30.0) | 22 (29.3) | <0.001 * |
| Stage 3 | 23 (57.5) | 12 (16.0) | |
| Urine studies § | |||
| Urine WBC or leukocyte esterase | 15 (40.5) | 18 (32.7) | 0.444 |
| Urine RBC or blood | 27 (73.0) | 32 (58.2) | 0.147 |
| Urine protein | |||
| Trace | 2 (5.4) | 2 (3.6) | |
| 1+ | 9 (24.3) | 22 (40.0) | |
| 2+ | 10 (27.0) | 14 (25.5) | 0.149 |
| 3+ | 8 (21.6) | 3 (5.5) | |
| Urine chemistry § | |||
| Fractional excretion of sodium † (%, n = 19) | 0.7 (0.6) | 0.4 (0.2) | 0.084 |
| Fractional excretion of urea † (%, n = 14) | 23.8 (13.2) | 20.1 (13.4) | 0.616 |
| Outcome | |||
| Need for RRT | 16 (40.0) | 0 (0) | <0.001 * |
| Hospital stay † (day) | 12.6 (9.4) | 8.7 (8.1) | 0.030 * |
| Death | 27 (67.5) | 13 (17.3) | <0.001 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hansrivijit, P.; Gadhiya, K.P.; Gangireddy, M.; Goldman, J.D. Risk Factors, Clinical Characteristics, and Prognosis of Acute Kidney Injury in Hospitalized COVID-19 Patients: A Retrospective Cohort Study. Medicines 2021, 8, 4. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines8010004
Hansrivijit P, Gadhiya KP, Gangireddy M, Goldman JD. Risk Factors, Clinical Characteristics, and Prognosis of Acute Kidney Injury in Hospitalized COVID-19 Patients: A Retrospective Cohort Study. Medicines. 2021; 8(1):4. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines8010004
Chicago/Turabian StyleHansrivijit, Panupong, Kinjal P. Gadhiya, Mounika Gangireddy, and John D. Goldman. 2021. "Risk Factors, Clinical Characteristics, and Prognosis of Acute Kidney Injury in Hospitalized COVID-19 Patients: A Retrospective Cohort Study" Medicines 8, no. 1: 4. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines8010004





