β-Thujaplicin Enhances TRAIL-Induced Apoptosis via the Dual Effects of XIAP Inhibition and Degradation in NCI-H460 Human Lung Cancer Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Trypan Blue Staining
2.4. Measurements of Cell Viability
2.5. Evaluation of the Combinatorial Effects of Drugs
2.6. Caspase-3/7 Activity Assay
2.7. Western Blotting
2.8. Expression and Purification of Recombinant GST-Tagged XIAP-BIR3 Domain in an Escherichia coli Expression System
2.9. Fluorescence Polarization Binding Assay
3. Results
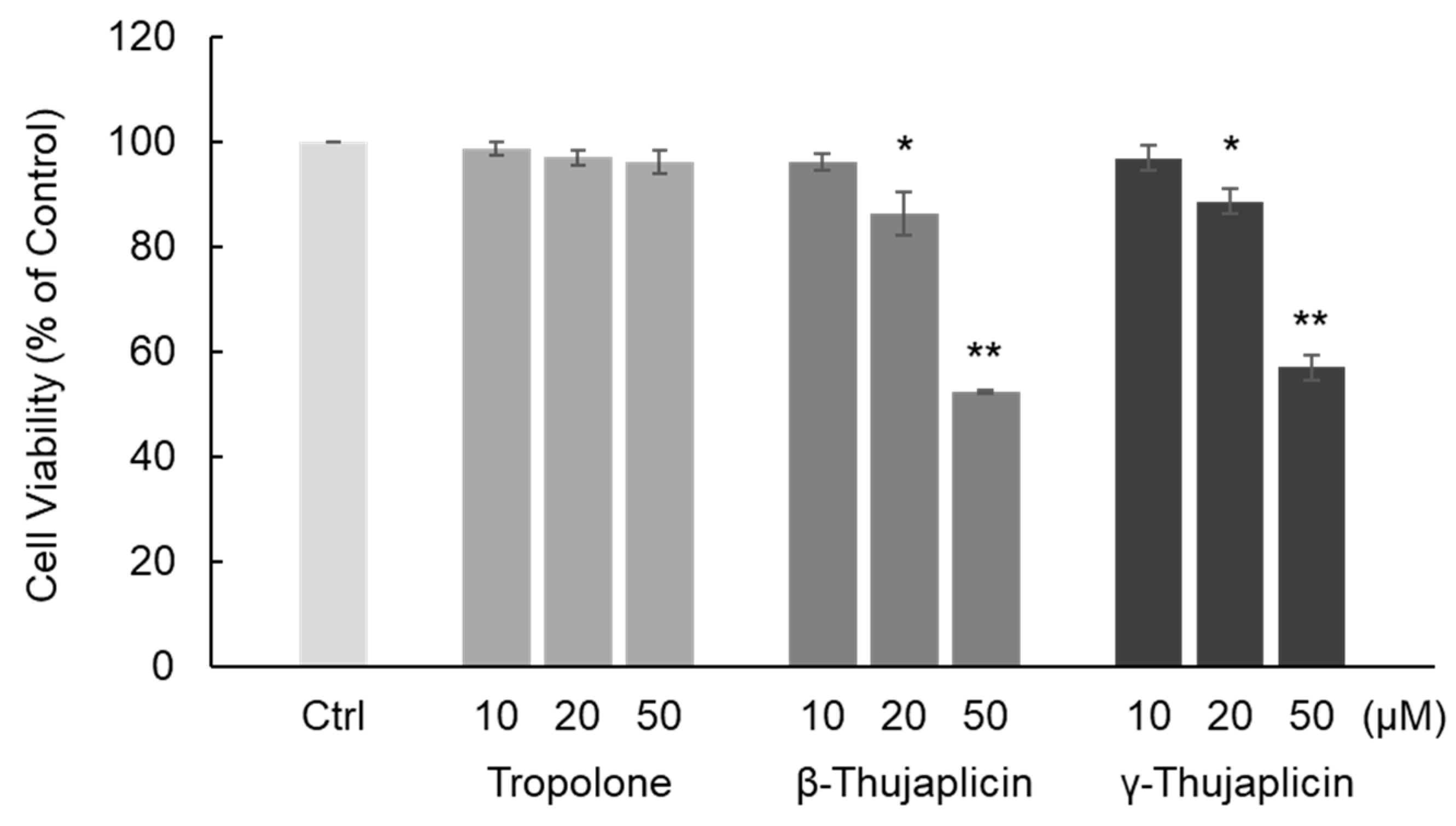
3.1. β-. and γ-Thujaplicins Decrease the Viability of NCI-H460 Cells
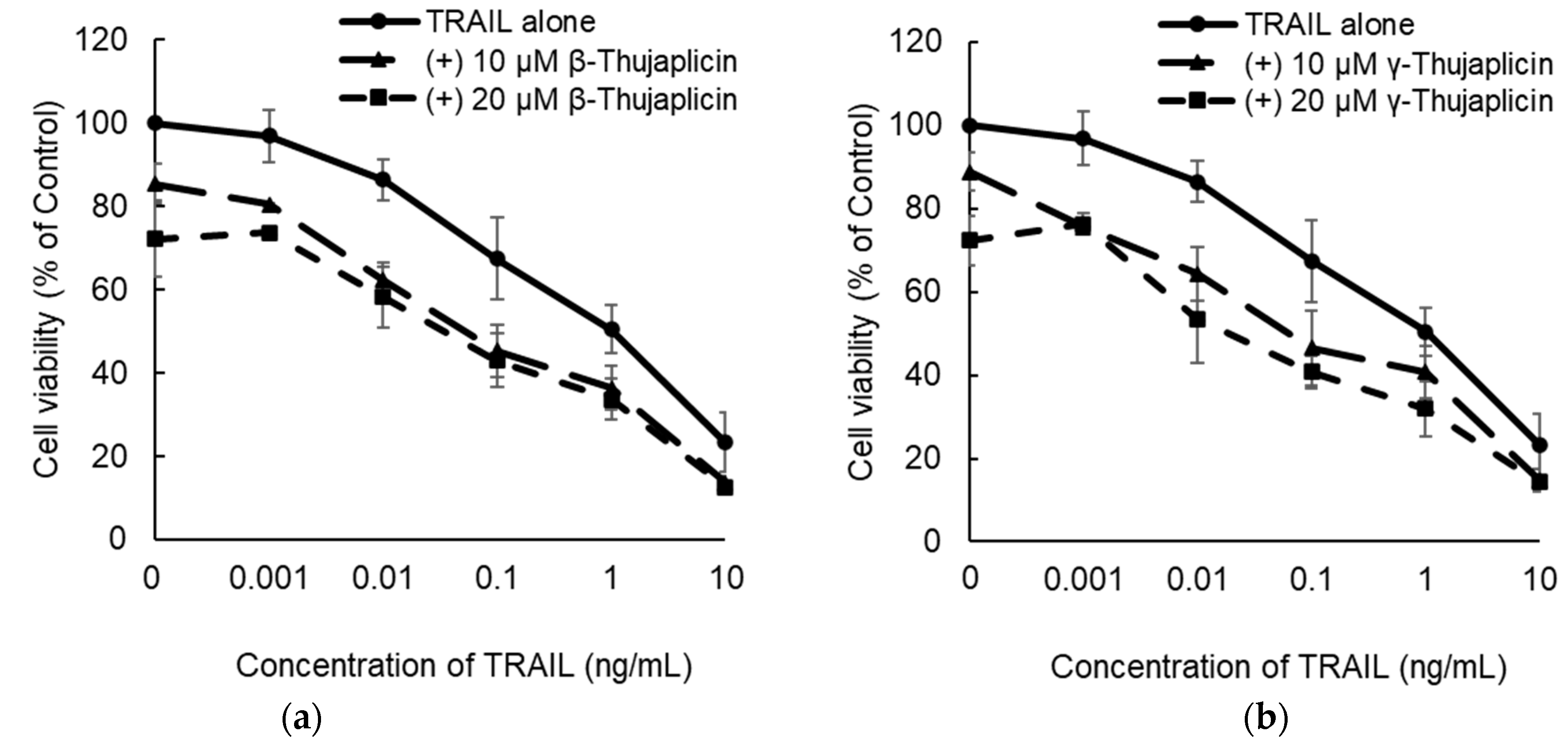
3.2. β-. and γ-Thujaplicins Enhance the Antiproliferative Effect of TRAIL on NCI-H460 Cells
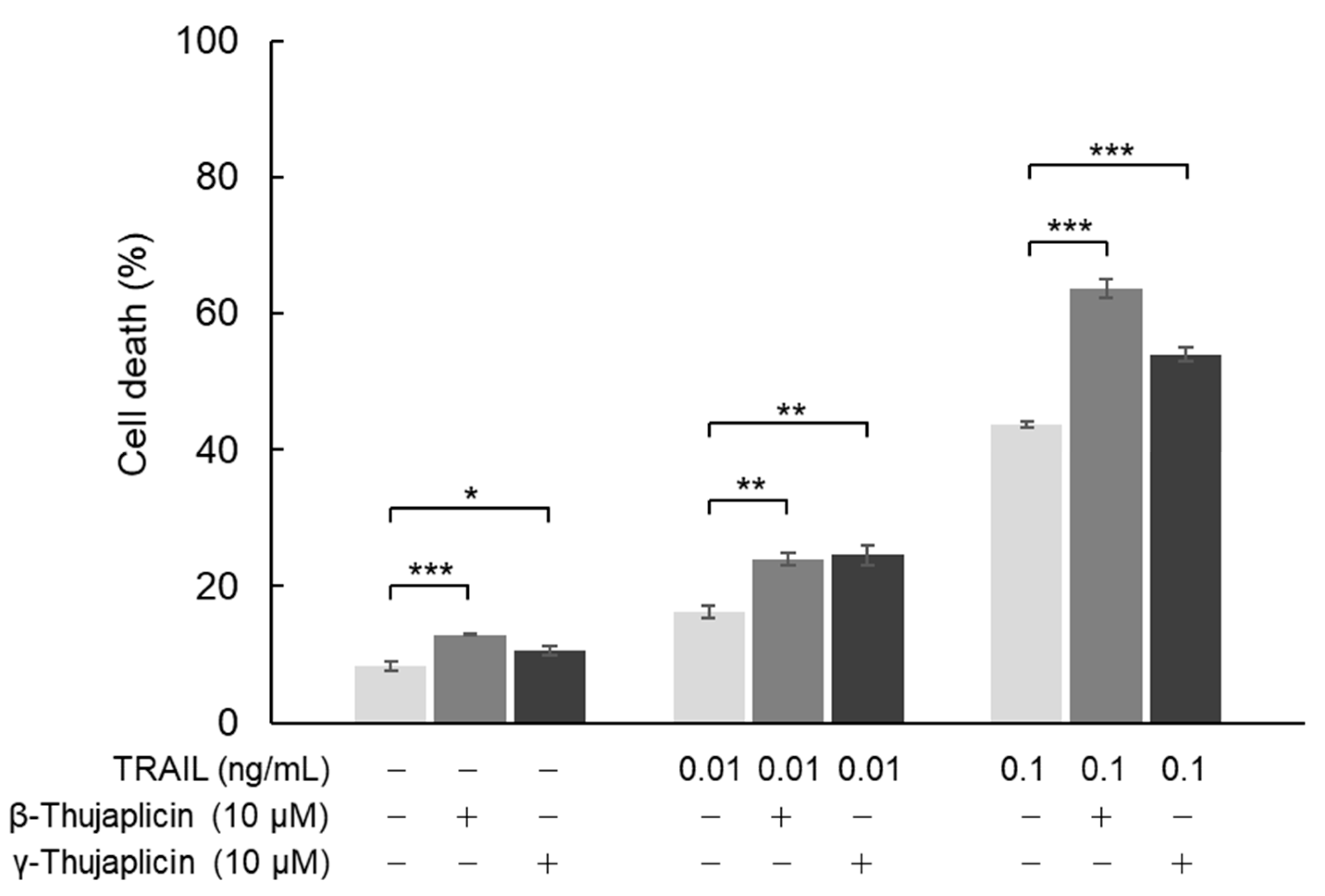
3.3. β-. Thujaplicin Enhances the Cell Death Induction Effect of TRAIL on NCI-H460 Cells
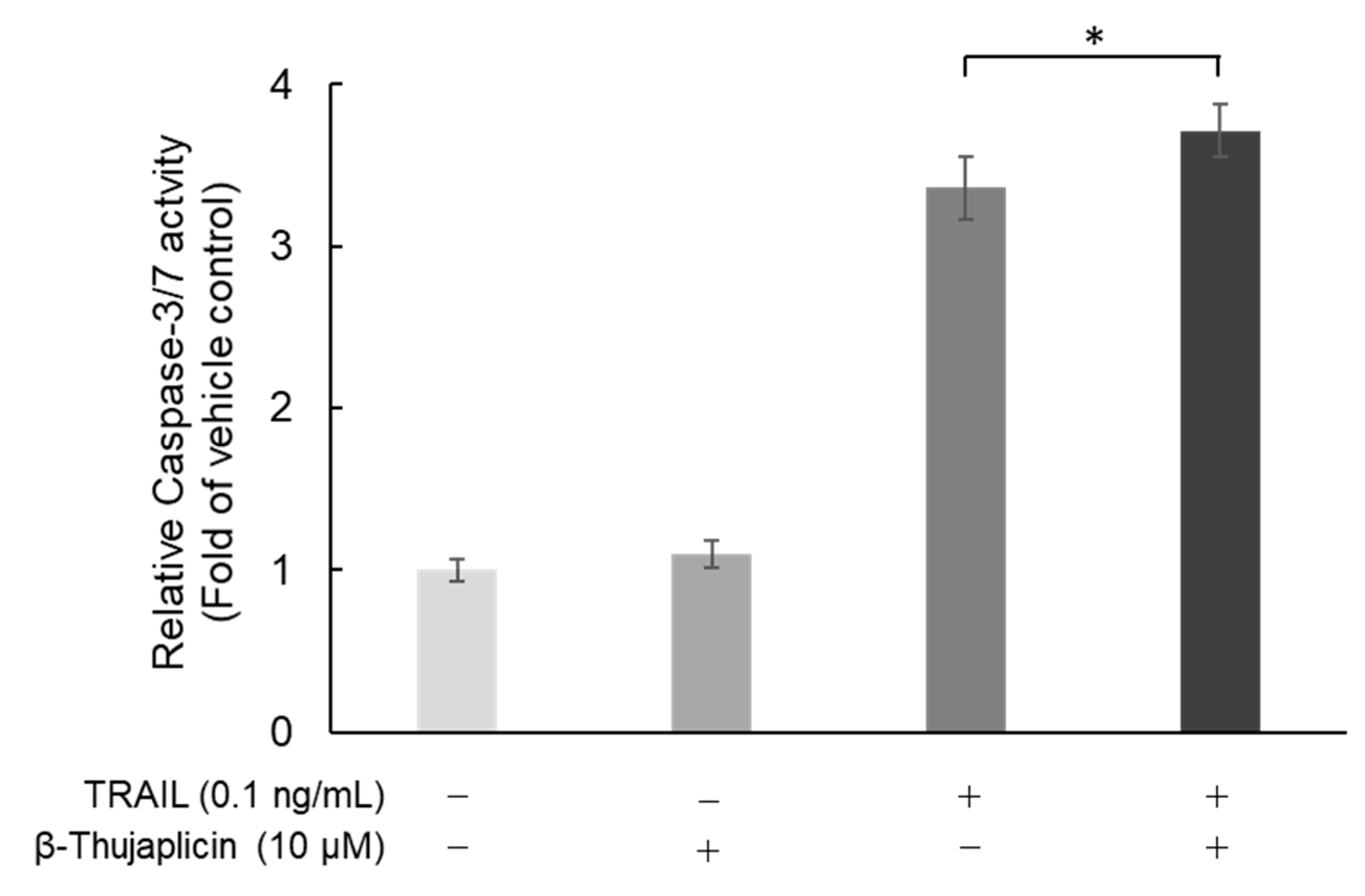
3.4. Potentiating Effect of β-Thujaplicin on TRAIL-Induced Cell Death in NCI-H460 Cells Is Accompanied by Enhanced Caspase-3/7 Activity
3.5. Tropolone, β-Thujaplicin, and γ-Thujaplicin Treatments Decrease the Amount of XIAP Protein in NCI-H460 Cells
3.6. Tropolone, β-Thujaplicin, and γ-Thujaplicin Inhibit the Binding of the Smac N-Terminal Peptide to the XIAP-BIR3 Domain
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Miyamoto, D.; Endo, N.; Oku, N.; Arima, Y.; Suzuki, T.; Suzuki, Y. β-thujaplicin zinc chelate induces apoptosis in mouse high metastatic melanoma B16BL6 cells. Biol. Pharm. Bull. 1998, 21, 1258–1262. [Google Scholar] [CrossRef]
- Inamori, Y.; Shinohara, S.; Tsujibo, H.; Okabe, T.; Morita, Y.; Sakagami, Y.; Kumeda, Y.; Ishida, N. Antimicrobial activity and metalloprotease inhibition of hinokitiol-related compounds, the constituents of Thujopsis dolabrata S. and Z. hondai MAK. Biol. Pharm. Bull. 1999, 22, 990–993. [Google Scholar] [CrossRef]
- Fotopoulou, T.; Ćirić, A.; Kritsi, E.; Calhelha, R.C.; Ferreira, I.C.; Soković, M.; Zoumpoulakis, P.; Koufaki, M. Antimicrobial/antibiofilm activity and cytotoxic studies of β-Thujaplicin derivatives. Arch. Pharm. 2016, 349, 698–709. [Google Scholar] [CrossRef]
- Baya, M.; Soulounganga, P.; Gelhaye, E.; Gérardin, P. Fungicidal activity of β-thujaplicin analogues. Pest. Manag. Sci. 2001, 57, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, D.; Kusagaya, Y.; Endo, N.; Sometani, A.; Takeo, S.; Suzuki, T.; Arima, Y.; Nakajima, K.; Suzuki, Y. Thujaplicin-copper chelates inhibit replication of human influenza viruses. Antivir. Res. 1998, 39, 89–100. [Google Scholar] [CrossRef]
- Inamori, Y.; Tsujibo, H.; Ohishi, H.; Ishii, F.; Mizugaki, M.; Aso, H.; Ishida, N. Cytotoxic effect of hinokitiol and tropolone on the growth of mammalian cells and on blastogenesis of mouse splenic T cells. Biol. Pharm. Bull. 1993, 16, 521–523. [Google Scholar] [CrossRef]
- Liu, S.; Yamauchi, H. p27-associated G1 arrest induced by hinokitiol in human malignant melanoma cells is mediated via down-regulation of pRb, Skp2 ubiquitin ligase, and impairment of Cdk2 function. Cancer Lett. 2009, 286, 240–249. [Google Scholar] [CrossRef]
- Shih, Y.H.; Chang, K.W.; Hsia, S.M.; Yu, C.C.; Fuh, L.J.; Chi, T.Y.; Shieh, T.M. In vitro antimicrobial and anticancer potential of hinokitiol against oral pathogens and oral cancer cell lines. Microbiol. Res. 2013, 168, 254–262. [Google Scholar] [CrossRef]
- Lee, Y.S.; Choi, K.M.; Kim, W.; Jeon, Y.S.; Lee, Y.M.; Hong, J.T.; Yun, Y.P.; Yoo, H.S. Hinokitiol inhibits cell growth through induction of S-phase arrest and apoptosis in human colon cancer cells and suppresses tumor growth in a mouse xenograft experiment. J. Nat. Prod. 2013, 76, 2195–2202. [Google Scholar] [CrossRef] [PubMed]
- Li, L.H.; Wu, P.; Lee, J.Y.; Li, P.R.; Hsieh, W.Y.; Ho, C.C.; Ho, C.L.; Chen, W.J.; Wang, C.C.; Yen, M.Y.; et al. Hinokitiol induces DNA damage and autophagy followed by cell cycle arrest and senescence in gefitinib-resistant lung adenocarcinoma cells. PLoS ONE 2014, 9, e104203. [Google Scholar] [CrossRef]
- Chen, S.M.; Wang, B.Y.; Lee, C.H.; Lee, H.T.; Li, J.J.; Hong, G.C.; Hung, Y.C.; Chien, P.J.; Chang, C.Y.; Hsu, L.S.; et al. Hinokitiol up-regulates miR-494-3p to suppress BMI1 expression and inhibits self-renewal of breast cancer stem/progenitor cells. Oncotarget 2017, 8, 76057–76068. [Google Scholar] [CrossRef]
- Jayakumar, T.; Liu, C.H.; Wu, G.Y.; Lee, T.Y.; Manubolu, M.; Hsieh, C.Y.; Yang, C.H.; Sheu, J.R. Hinokitiol inhibits migration of A549 lung cancer cells via suppression of MMPs and induction of antioxidant enzymes and apoptosis. Int. J. Mol. Sci. 2018, 19, 939. [Google Scholar] [CrossRef]
- Zhang, G.; He, J.; Ye, X.; Zhu, J.; Hu, X.; Shen, M.; Ma, Y.; Mao, Z.; Song, H.; Chen, F. β-Thujaplicin induces autophagic cell death, apoptosis, and cell cycle arrest through ROS-mediated Akt and p38/ERK MAPK signaling in human hepatocellular carcinoma. Cell Death Dis. 2019, 10, 255. [Google Scholar] [CrossRef] [PubMed]
- Eckelman, B.P.; Salvesen, G.S.; Scott, F.L. Human inhibitor of apoptosis proteins: Why XIAP is the black sheep of the family. EMBO Rep. 2006, 7, 988–994. [Google Scholar] [CrossRef] [PubMed]
- Schimmer, A.D.; Dalili, S.; Batey, R.A.; Riedl, S.J. Targeting XIAP for the treatment of malignancy. Cell Death Differ. 2006, 13, 179–188. [Google Scholar] [CrossRef]
- Kluger, H.M.; McCarthy, M.M.; Alvero, A.B.; Sznol, M.; Ariyan, S.; Camp, R.L.; Rimm, D.L.; Mor, G. The X-linked inhibitor of apoptosis protein (XIAP) is up-regulated in metastatic melanoma, and XIAP cleavage by phenoxodiol is associated with carboplatin sensitization. J. Transl. Med. 2007, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, F.; Luo, Q.; Wu, X.; Liu, Z.; Chen, H.; Huang, Y. Inhibition of XIAP increases carboplatin sensitivity in ovarian cancer. Onco Targets Ther. 2018, 11, 8751–8759. [Google Scholar] [CrossRef]
- Makhov, P.; Golovine, K.; Uzzo, R.G.; Rothman, J.; Crispen, P.L.; Shaw, T.; Scoll, B.J.; Kolenko, V.M. Zinc chelation induces rapid depletion of the X-linked inhibitor of apoptosis and sensitizes prostate cancer cells to TRAIL-mediated apoptosis. Cell Death Differ. 2008, 15, 1745–1751. [Google Scholar] [CrossRef]
- Nikolovska-Coleska, Z.; Xu, L.; Hu, Z.; Tomita, Y.; Li, P.; Roller, P.P.; Wang, R.; Fang, X.; Guo, R.; Zhang, M.; et al. Discovery of embelin as a cell-permeable, small-molecular weight inhibitor of XIAP through structure-based computational screening of a traditional herbal medicine three-dimensional structure database. J. Med. Chem. 2004, 47, 2430–2440. [Google Scholar] [CrossRef]
- Sun, S.Y.; Yue, P.; Zhou, J.Y.; Wang, Y.; Choi Kim, H.R.; Lotan, R.; Wu, G.S. Overexpression of BCL2 blocks TNF-related apoptosis-inducing ligand (TRAIL)-induced apoptosis in human lung cancer cells. Biochem. Biophys. Res. Commun. 2001, 280, 788–797. [Google Scholar] [CrossRef]
- Yang, L.; Mashima, T.; Sato, S.; Mochizuki, M.; Sakamoto, H.; Yamori, T.; Oh-hara, T.; Tsuruo, T. Predominant suppression of apoptosome by inhibitor of apoptosis protein in non-small cell lung cancer H460 cells: Therapeutic effect of a novel polyarginine-conjugated Smac peptide. Cancer Res. 2003, 63, 831–837. [Google Scholar] [PubMed]
- Borisy, A.A.; Elliott, P.J.; Hurst, N.W.; Lee, M.S.; Lehar, J.; Price, E.R.; Serbedzija, G.; Zimmermann, G.R.; Foley, M.A.; Stockwell, B.R.; et al. Systematic discovery of multicomponent therapeutics. Proc. Natl. Acad. Sci. USA 2003, 100, 7977–7982. [Google Scholar] [CrossRef] [PubMed]
- Rotin, L.E.; Gronda, M.; MacLean, N.; Hurren, R.; Wang, X.; Lin, F.H.; Wrana, J.; Datti, A.; Barber, D.L.; Minden, M.D.; et al. Ibrutinib synergizes with poly(ADP-ribose) glycohydrolase inhibitors to induce cell death in AML cells via a BTK-independent mechanism. Oncotarget 2016, 7, 2765–2779. [Google Scholar] [CrossRef]
- Manzoni, L.; Gornati, D.; Manzotti, M.; Cairati, S.; Bossi, A.; Arosio, D.; Lecis, D.; Seneci, P. Dual action Smac mimetics–zinc chelators as pro-apoptotic antitumoral agents. Bioorg. Med. Chem. Lett. 2016, 26, 4613–4619. [Google Scholar] [CrossRef]



| Compound | Structure | IC50 (μM) a |
|---|---|---|
| Tropolone |  | 83.1 ± 6.9 |
| β-thujaplicin |  | 58.5 ± 2.8 |
| γ-thujaplicin |  | 56.2 ± 2.9 |
| Embelin |  | 3.1 ± 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seno, S.; Kimura, M.; Yashiro, Y.; Kimura, R.; Adachi, K.; Terabayashi, A.; Takahashi, M.; Oyama, T.; Abe, H.; Abe, T.; et al. β-Thujaplicin Enhances TRAIL-Induced Apoptosis via the Dual Effects of XIAP Inhibition and Degradation in NCI-H460 Human Lung Cancer Cells. Medicines 2021, 8, 26. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines8060026
Seno S, Kimura M, Yashiro Y, Kimura R, Adachi K, Terabayashi A, Takahashi M, Oyama T, Abe H, Abe T, et al. β-Thujaplicin Enhances TRAIL-Induced Apoptosis via the Dual Effects of XIAP Inhibition and Degradation in NCI-H460 Human Lung Cancer Cells. Medicines. 2021; 8(6):26. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines8060026
Chicago/Turabian StyleSeno, Saki, Minori Kimura, Yuki Yashiro, Ryutaro Kimura, Kanae Adachi, Aoi Terabayashi, Mio Takahashi, Takahiro Oyama, Hideaki Abe, Takehiko Abe, and et al. 2021. "β-Thujaplicin Enhances TRAIL-Induced Apoptosis via the Dual Effects of XIAP Inhibition and Degradation in NCI-H460 Human Lung Cancer Cells" Medicines 8, no. 6: 26. https://0-doi-org.brum.beds.ac.uk/10.3390/medicines8060026





