Microfluidic Adaptation of Density-Gradient Centrifugation for Isolation of Particles and Cells
Abstract
:1. Introduction
2. Materials and Methods
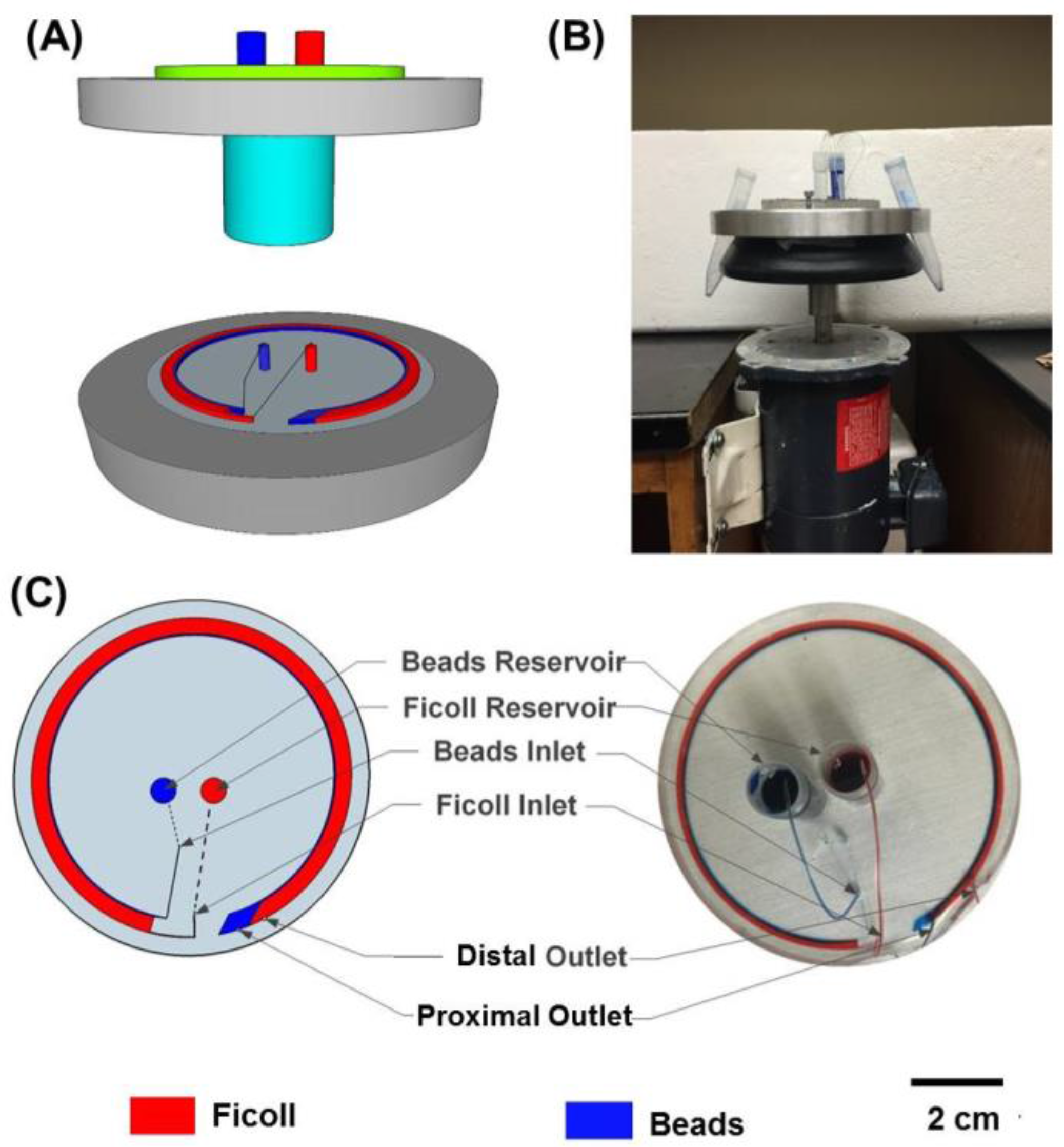
2.1. Materials Microfluidic Device Fabrication
2.2. Centrifugation System
2.3. Particles and Fluids
2.4. Flow Characterization
2.5. Evaluation of Samples
3. Results
3.1. Device Dimensions for Optimal Laminar Flow and Layering
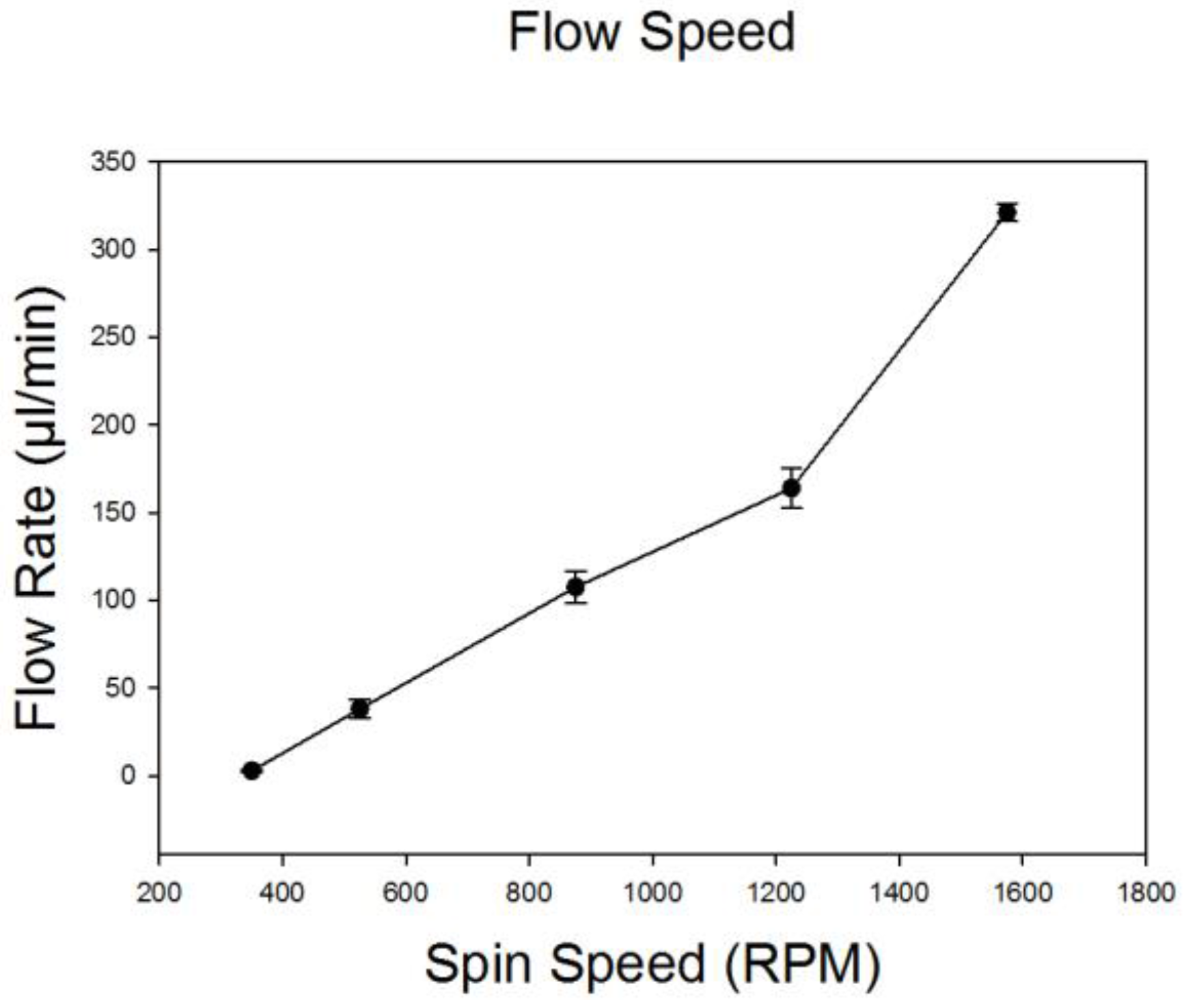
3.2. Characterization of Total and Relative Flow Rates
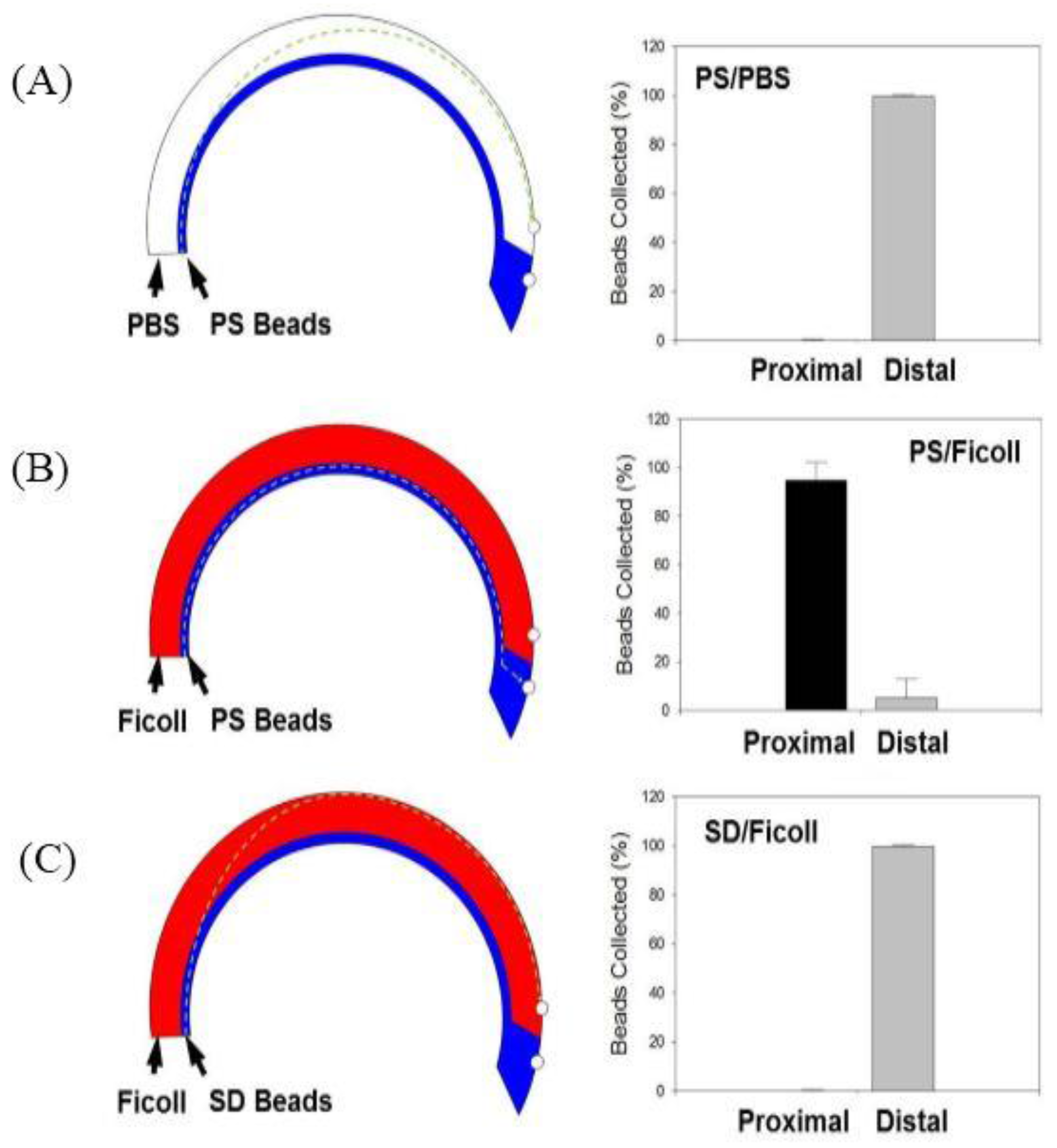
3.3. PS Bead Separation
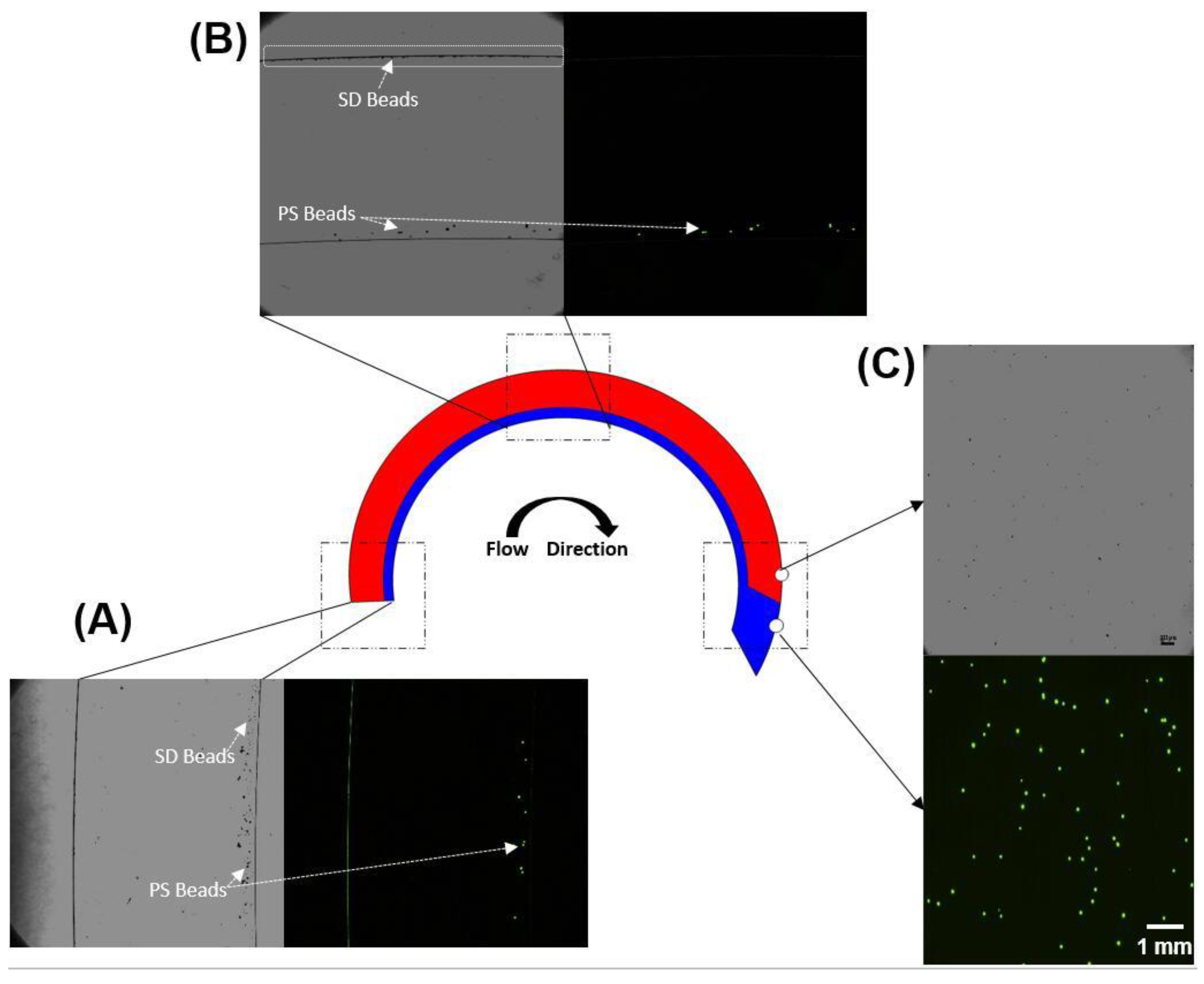
3.5. Separation of PS–SD Bead Mixture
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Byeon, Y.; Ki, C.S.; Han, K.H. Isolation of nucleated red blood cells in maternal blood for Non-invasive prenatal diagnosis. Biomed. Microdevices 2015, 17, 118. [Google Scholar] [CrossRef] [PubMed]
- Aghazarian, A.; Stancik, I.; Huf, W.; Pfluger, H. Evaluation of Leukocyte Threshold Values in Semen to Detect Inflammation Involving Seminal Interleukin-6 and Interleukin-8. Urology 2015, 86, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Schafer, D.; Dressen, P.; Brettner, S.; Rath, N.F.; Molderings, G.J.; Jensen, K.; Ziemann, C. Prostaglandin D2-supplemented ”functional eicosanoid testing and typing” assay with peripheral blood leukocytes as a new tool in the diagnosis of systemic mast cell activation disease: An explorative diagnostic study. J. Transl. Med. 2014, 12, 213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wojcik, M.; Zieleniak, A.; Zurawska-Klis, M.; Cypryk, K.; Wozniak, L.A. Increased expression of immune-related genes in leukocytes of patients with diagnosed gestational diabetes mellitus (GDM). Exp. Biol. Med. (Maywood N.J.) 2016, 241, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Hashemian, A.M.; Ahmadi, K.; Zamani Moghaddam, H.; Zakeri, H.; Davoodi Navakh, S.A.; Sharifi, M.D.; Bahrami, A. Diagnostic Value of Leukocyte Esterase Test Strip Reagents for Rapid Clinical Diagnosis of Spontaneous Bacterial Peritonitis in Patients Admitted to Hospital Emergency Departments in Iran. Iran. Red Crescent Med. J. 2015, 17, e21341. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, L.; Lu, L. Detection of cyprinid herpesvirus 2 in peripheral blood cells of silver crucian carp, Carassius auratus gibelio (Bloch), suggests its potential in viral diagnosis. J. Fish Dis. 2016, 39, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Mariucci, S.; Rovati, B.; Manzoni, M.; Della Porta, M.G.; Comolli, G.; Delfanti, S.; Danova, M. Lymphocyte subpopulation and dendritic cell phenotyping during antineoplastic therapy in human solid tumors. Clin. Exp. Med. 2011, 11, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Gupta, A.; Chen, C.; Tompkins, R.G.; Rodriguez, W.; Toner, M. Enhancing the performance of a point-of-care CD4+ T-cell counting microchip through monocyte depletion for HIV/AIDS diagnostics. Lab Chip 2009, 9, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Mitroulis, I.; Alexaki, V.I.; Kourtzelis, I.; Ziogas, A.; Hajishengallis, G.; Chavakis, T. Leukocyte integrins: Role in leukocyte recruitment and as therapeutic targets in inflammatory disease. Pharmacol. Ther. 2015, 147, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Chernyshev, A.V.; Tarasov, P.A.; Semianov, K.A.; Nekrasov, V.M.; Hoekstra, A.G.; Maltsev, V.P. Erythrocyte lysis in isotonic solution of ammonium chloride: Theoretical modeling and experimental verification. J. Theor. Biol. 2008, 251, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Brosseron, F.; Marcus, K.; May, C. Isolating peripheral lymphocytes by density gradient centrifugation and magnetic cell sorting. Methods Mol. Biol. (Clifton, N.J.) 2015, 1295, 33–42. [Google Scholar] [CrossRef]
- Pelak, O.; Kuzilkova, D.; Thurner, D.; Kiene, M.L.; Stanar, K.; Stuchly, J.; Vaskova, M.; Stary, J.; Hrusak, O.; Stadler, H.; et al. Lymphocyte enrichment using CD81-targeted immunoaffinity matrix. Cytom. Part A J. Int. Soc. Anal. Cytol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.A.; Thiel, M.; Hogg, N. Signaling mechanisms and the activation of leukocyte integrins. J. Leukoc. Biol. 1997, 61, 422–426. [Google Scholar] [PubMed]
- Zhou, L.; Somasundaram, R.; Nederhof, R.F.; Dijkstra, G.; Faber, K.N.; Peppelenbosch, M.P.; Fuhler, G.M. Impact of human granulocyte and monocyte isolation procedures on functional studies. Clin. Vaccine Immunol. CVI 2012, 19, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Bhuvanendran Nair Gourikutty, S.; Chang, C.P.; Puiu, P.D. Microfluidic immunomagnetic cell separation from whole blood. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1011, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.D.; Fachin, F.; Colombini, E.; Wardle, B.L.; Toner, M. Nanoporous micro-element arrays for particle interception in microfluidic cell separation. Lab Chip 2012, 12, 3159–3167. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Huang, N.T.; Oh, B.; Lam, R.H.; Fan, R.; Cornell, T.T.; Shanley, T.P.; Kurabayashi, K.; Fu, J. Surface-micromachined microfiltration membranes for efficient isolation and functional immunophenotyping of subpopulations of immune cells. Adv. Healthc. Mater. 2013, 2, 965–975. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, W.; Liu, G.; Lu, W.; Fu, J. Continuous-flow microfluidic blood cell sorting for unprocessed whole blood using surface-micromachined microfiltration membranes. Lab Chip 2014, 14, 2565–2575. [Google Scholar] [CrossRef] [PubMed]
- Xiang, N.; Ni, Z. High-throughput blood cell focusing and plasma isolation using spiral inertial microfluidic devices. Biomed. Microdevices 2015, 17, 110. [Google Scholar] [CrossRef] [PubMed]
- Darabi, J.; Guo, C. Continuous isolation of monocytes using a magnetophoretic-based microfluidic Chip. Biomed. Microdevices 2016, 18, 77. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Peng, Z.; Lin, S.C.; Geri, M.; Li, S.; Li, P.; Chen, Y.; Dao, M.; Suresh, S.; Huang, T.J. Cell separation using tilted-angle standing surface acoustic waves. Proc. Natl. Acad. Sci. USA 2014, 111, 12992–12997. [Google Scholar] [CrossRef] [PubMed]
- Grenvall, C.; Magnusson, C.; Lilja, H.; Laurell, T. Concurrent isolation of lymphocytes and granulocytes using prefocused free flow acoustophoresis. Anal. Chem. 2015, 87, 5596–5604. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Lin, X.; Su, Y.; Dong, H.; Wu, J. Screen-printed microfluidic dielectrophoresis chip for cell separation. Biosens. Bioelectron. 2015, 63, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Al-Faqheri, W.; Thio, T.H.G.; Qasaimeh, M.A.; Dietzel, A.; Madou, M. Particle/cell separation on microfluidic platforms based on centrifugation effect: A review. Microfluid. Nanofluid. 2017, 21, 102. [Google Scholar] [CrossRef]
- Balter, M.L.; Chen, A.I.; Colinco, C.A.; Gorshkov, A.; Bixon, B.; Martin, V.; Fromholtz, A.; Maguire, T.J.; Yarmush, M.L. Differential Leukocyte Counting via Fluorescent Detection and Image Processing on a Centrifugal Microfluidic Platform. Anal. Methods Adv. Methods Appl. 2016, 8, 8272–8279. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.T.F.; Joseph, J.G.; Liu, S.X.; Cheung, M.K.; Haffey, P.J.; Kurabayashi, K.; Fu, J. Centrifugal microfluidics for sorting immune cells from whole blood. Sens. Actuators B Chem. 2017, 245, 1050–1061. [Google Scholar] [CrossRef]
- Ramachandraiah, H.; Amasia, M.; Cole, J.; Sheard, P.; Pickhaver, S.; Walker, C.; Wirta, V.; Lexow, P.; Lione, R.; Russom, A. Lab-on-DVD: Standard DVD drives as a novel laser scanning microscope for image based point of care diagnostics. Lab Chip 2013, 13, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.I., III; Sommer, G.J.; Schaff, U.Y.; Hahn, P.S.; Jaffe, G.J.; Murthy, S.K. A centrifugal fluidic immunoassay for ocular diagnostics with an enzymatically hydrolyzed fluorogenic substrate. Lab Chip 2014, 14, 2673–2680. [Google Scholar] [CrossRef] [PubMed]
- Schaff, U.Y.; Sommer, G.J. Whole blood immunoassay based on centrifugal bead sedimentation. Clin. Chem. 2011, 57, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, Q.; Liu, M.; Yang, J. A lab-on-CD prototype for high-speed blood separation. J. Micromech. Microeng. 2008, 18, 125025. [Google Scholar] [CrossRef]
- Kinahan, D.J.; Kearney, S.M.; Glynn, M.T.; Ducrée, J. Spira mirabilis enhanced whole blood processing in a lab-on-a-disk. Sens. Actuators A Phys. 2014, 215, 71–76. [Google Scholar] [CrossRef]
- Kinahan, D.J.; Kearney, S.M.; Kilcawley, N.A.; Early, P.L.; Glynn, M.T.; Ducrée, J. Density-gradient mediated band extraction of leukocytes from whole blood using centrifugo-pneumatic siphon valving on centrifugal microfluidic discs. PLoS ONE 2016, 11, e0155545. [Google Scholar] [CrossRef] [PubMed]
- Moen, S.T.; Hatcher, C.L.; Singh, A.K. A centrifugal microfluidic platform that separates whole blood samples into multiple removable fractions due to several discrete but continuous density gradient sections. PLoS ONE 2016, 11, e0153137. [Google Scholar] [CrossRef] [PubMed]
- Ukita, Y.; Oguro, T.; Takamura, Y. Density-gradient-assisted centrifugal microfluidics: An approach to continuous-mode particle separation. Biomed. Microdevices 2017, 19, 24. [Google Scholar] [CrossRef] [PubMed]
- Patibandla, P.K.; Rogers, A.J.; Giridharan, G.A.; Pallero, M.A.; Murphy-Ullrich, J.E.; Sethu, P. Hyperglycemic arterial disturbed flow niche as an in vitro model of atherosclerosis. Anal. Chem. 2014, 86, 10948–10954. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Sethu, P. Microfluidic Adaptation of Density-Gradient Centrifugation for Isolation of Particles and Cells. Bioengineering 2017, 4, 67. https://0-doi-org.brum.beds.ac.uk/10.3390/bioengineering4030067
Sun Y, Sethu P. Microfluidic Adaptation of Density-Gradient Centrifugation for Isolation of Particles and Cells. Bioengineering. 2017; 4(3):67. https://0-doi-org.brum.beds.ac.uk/10.3390/bioengineering4030067
Chicago/Turabian StyleSun, Yuxi, and Palaniappan Sethu. 2017. "Microfluidic Adaptation of Density-Gradient Centrifugation for Isolation of Particles and Cells" Bioengineering 4, no. 3: 67. https://0-doi-org.brum.beds.ac.uk/10.3390/bioengineering4030067




