1. Introduction
Roselle (
Hibiscus sabdariffa L.) is an annual shrub belongs to the Malvaceae family and mostly grows in tropical and subtropical areas like African and Asian countries [
1]. The calyces of Roselle have been used in traditional medicine to help to treat many diseases like hypertension, diabetes and liver disorders [
2,
3]. Studies on the phytochemical compositions of Roselle have found that the calyces of Roselle contain a significant number of bioactive compounds, which may contribute to the beneficial health effects of this material such as phenolic acids, anthocyanins, flavonoids and polysaccharides [
4]. The major anthocyanins are delphinidin-3-glucoside and cyanidin-3-sambubioside while quercetin and its glucosides are the principal flavonols and chlorogenic acid is the main phenolic acid present in Roselle [
4,
5]. Tsai et al. (2002) [
6] found that anthocyanins were the principal component contributing to the antioxidant capacity of the extract from Roselle calyxes. Roselle extracts also showed antibacterial activity against various species like
Escherichia coli,
Staphylococcus aureus,
Bacillus subtilis, and
Clostridium sporogenes [
7] and antifungal activity against
Aspergillus,
Trichophyton and
Cryptococcus [
8]. In a study by Liu et al. (2011) Roselle extract was reported to have anticancer activity via protective effects against oxidative damage induced by tert-butyl hydroperoxide (t-BHP) in rat primary hepatocytes [
5].
In addition to the use of folk medicine, Roselle is also commonly used to provide natural purple color for many kinds of food like jellies, beverages, and desserts. The fruit and calyxes of Roselle are also processed to jams, marmalade and, especially, dried to use as herbal tea [
6,
9]. Previous studies have demonstrated that drying, an essential thermal process in producing herbal teas, may cause significant losses of bioactive ingredients and thereby decrease the health benefits and quality of the products [
10,
11]. The brewing conditions such as quality, volume, and temperature of water and brewing time have also been reported to significantly affect the content of the desired components in tea infusion [
12,
13]. The release level of the tasty ingredients and volatile compounds into the tea will decide the sensory quality of the herbal tea. Although there have been some studies on the extraction of Roselle using different types of solvent [
14,
15,
16] and investigations into the drying processes for this material [
17,
18,
19], comprehensive studies to establish a convenient procedure of drying and brewing to achieve high-quality Roselle tea infusion are limited.
In this study, the use of a common hot-air dryer for drying of Roselle and the classical brewing method were investigated to find out the most suitable drying and brewing conditions in order to produce Roselle tea with a high content of bioactive, antioxidant activity and sensory quality. The results help to suggest a convenient procedure of drying, along with brewing, which can be easily used at household or small scale to produce high-quality Roselle tea.
2. Material and Methods
2.1. Material
Roselle fruits were harvested at Daklak province in the Central Highland of Vietnam. After being harvested, the edible parts were separated to be used as the material for the drying process.
Gallic acid, Folin-Ciocalteu reagent and 2,2-diphenyl-1-picrylhydrazyl (DPPH) were purchased from Sigma-Aldrich Pty Ltd. (Singapore City, Singapore). Analytical grade methanol was purchased from Merck Ltd. (Ho Chi Minh City, Vietnam).
2.2. Drying of Roselle
The material was dried at 60, 80, 100, and 120 °C using a hot-air drying oven (UM500, Memmert, Schwabach, Germany) until reaching a moisture content under 8%. During the drying processes, the moisture content of the samples was measured to establish the drying curves at each drying temperature. The dried Roselle samples obtained from different drying temperatures were then analyzed for total phenolic content (TPC), antioxidant capacity and sensory quality.
2.3. Brewing of Dried Roselle
To investigate the effect of brewing temperature on the quality of the Roselle tea, the dried Roselle was brewed using hot water at the temperature of 80, 90, and 100 °C with the ratio of water to dried Roselle of 10:1 (mL/g) for 20 min. The influence of the liquid-solid ratio on the tea quality was investigated by brewing the Roselle at 100 °C for 20 min using different water-Roselle ratios (8:1, 10:1, 12:1 and 14:1). Finally, different brewing periods (20, 30, and 40 min) were used to study the effect of brewing time on tea quality. The Roselle tea samples brewed under different conditions were evaluated for total soluble solid (TSS) content, TPC and antioxidant capacity.
2.4. Measurement of Total Phenolic Content (TPC)
Total phenolic content of the extracts was determined according to the method described by Vuong et al. (2014) [
20] with some modifications. An extract of 0.5 mL from Roselle was mixed with 2.5 mL of Folin-Ciocalteu reagent in a test tube and left at room temperature. After 5 min, 2 mL of 7.5% (w/v) Na
2CO
3 and the mixture was left for reacting at room temperature in a dark room for 1 h. The absorbance of the reacted mixture was then measured at 765 nm using a spectrophotometer (V-630, Jasco, Tokyo, Japan). The total phenolic content of the extracts was expressed as mg gallic acid equivalent (mg GAE) based on the standard curve of gallic acid solutions.
2.5. Determination of Antioxidant Capacity
Antioxidant activity of the extracts from Roselle was evaluated by the free radical scavenging activity of the extracts against 2,2-diphenyl-2-picrylhydrazyl (DPPH) radical. The DPPH antioxidant assay was carried out according to a method described by Nguyen and Eun (2011) [
21] with some modifications.
The working DPPH solution was prepared by diluting stock DPPH solution (0.24 g/L) with methanol to obtain an absorbance of 1.1 ± 0.02 at 515 nm. A volume of 0.15 mL of Roselle extract was then mixed with 2.85 mL of the DPPH working solution in a test tube and the solution was left for 15 min at room temperature in a dark room. The absorbance of the reacted solution was then measured at 515 nm using a spectrophotometer (V-630, Jasco, Japan). The absorbance of a blank sample that contained the same amount of methanol and DPPH solution was also measured.
Radical scavenging activity was calculated by the following formula:
where
Ab is the absorbance of the test with a blank sample and
Ae is the absorbance of the test with Roselle extract solution. The IC
50 of the DPPH radical scavenging activity was calculated based on a plot of scavenging ability versus concentration of extracts.
2.6. Sensory Evaluation
To investigate the effect of drying temperature on the dried Roselle samples, Roselle tea was prepared by brewing 1 g of the dried Roselle with 200 mL of hot water at 90 °C for 15 min. The tea was then decanted into a white ceramic cup, added with 0.5 g of stevia sweetener and left to cool down to 50 °C before being evaluated by 20 panelists (10 men and 10 women) with different ages ranging from 22 to 40 years old at Tay Nguyen University. The sensory characteristics including aroma, taste, aftertaste, color, transparency and the overall acceptance were evaluated by the 9-point hedonic scale [
22].
2.7. Determination of Total Soluble Solid (TSS) Content
The TSS content of the extracts were determined using a digital refractometer (PAL-α, ATAGO, Tokyo, Japan) according to the method described by AOAC (2000) [
23].
2.8. Determination of Moisture Content
Moisture content of the samples were analyzed by drying to a constant weight at 105 °C using a drying oven (UM500, Memmert, Germany) according to the method described by AOAC (2000) [
23].
2.9. Statistical Analysis
Experiments were repeated in triplicates and the results were expressed as the mean values ± standard deviations. The statistical significance for each experiment was determined using the analysis of variance test (ANOVA) and the LSD (Least Significant Difference) post-hoc test was used for the comparison amongst the mean values. Differences were considered to be significant at p < 0.05.
3. Results and Discussion
3.1. Effects of Drying Temperature on Drying Time, Total Phenolic Content, Antioxidant Activity and Sensory Quality of Roselle
3.1.1. Drying Time
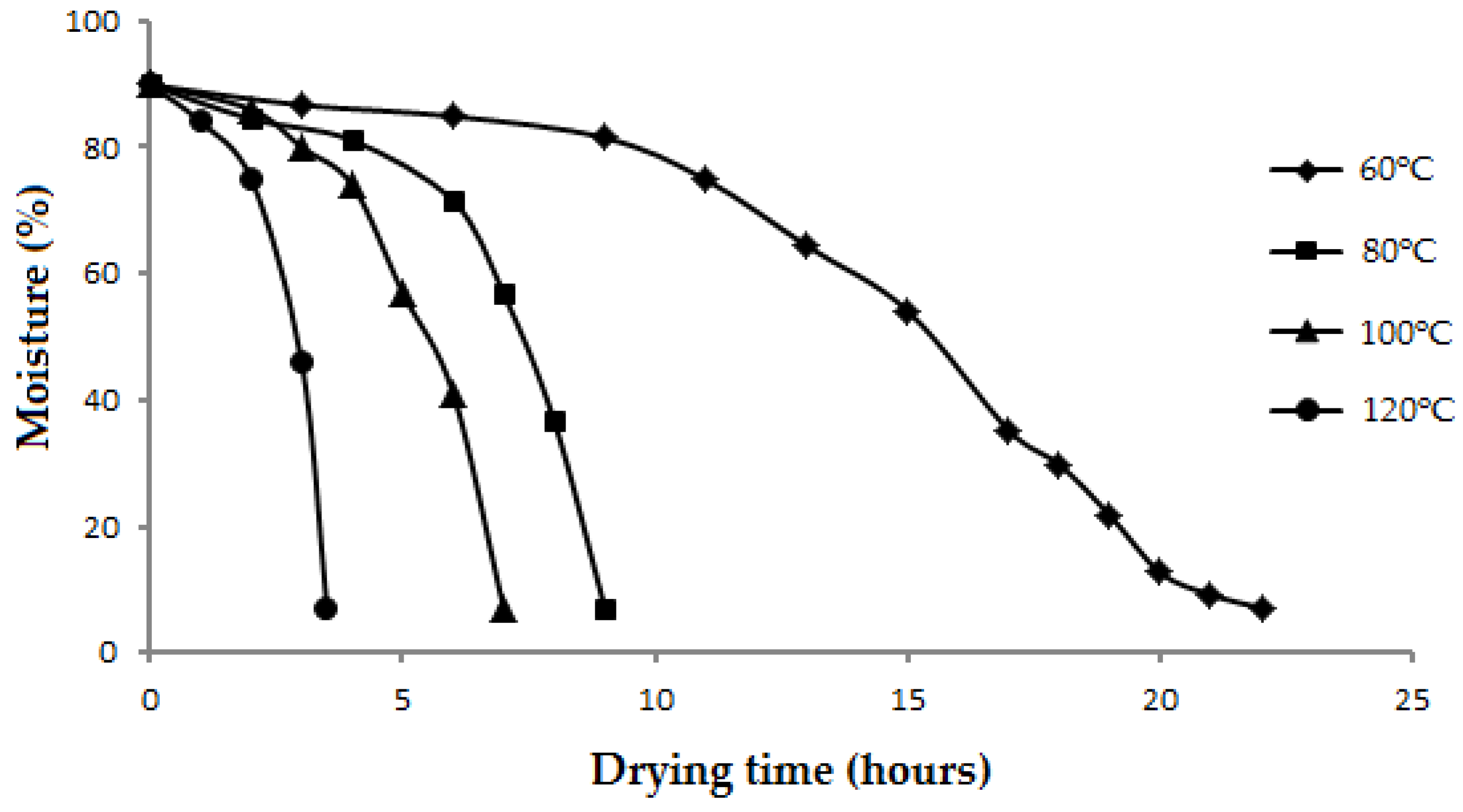
The drying curves of Roselle at 60, 80, 100, and 120 °C are presented in
Figure 1. The drying at 60 °C required 22 h to bring the moisture content of Roselle from 89.9% to 7.1% while the use of drying temperatures at 80, 100, and 120 °C could shorten the time to achieve moisture content of the materials less than 8% to 9, 7, and 3.5 h, respectively.
As expected, the higher the drying temperatures used, the higher the rate of moisture loss was achieved. At the beginning of the drying process, the decrease of moisture content in the material was slow but the drying rate rose rapidly until the end of the processes. This phenomenon is different from the change in drying rate for many other herbs or medicinal plants, which usually reaches a high rate at the beginning and becomes slow at the end of the process when the materials reach a low moisture content [
24,
25,
26]. This difference may be due to the high moisture content in the fresh juicy Roselle (approximately 90%) which requires more heat to reach the evaporation temperature of the water. The reduction in drying rate did not occur at the end of the drying process of the Roselle may be attributed to the soft structure and the thin skin of the material, which allowed moisture to easily escape from the matrix.
3.1.2. Total Phenolic Content in Dried Roselle
Concentrations of total phenolic compounds were significantly different among dried Roselle obtained from different temperatures. The TPC of the sample dried at 80 °C was the highest, followed by the Roselle dried at 60 °C, 100 °C, and 120 °C (
Figure 2). As reported in previous studies [
4,
6], Roselle contains a high level of phenolic compounds which considerably contribute to biological activities of the material but are also very susceptible to processing conditions like high temperature and oxygen.
The thermal drying processes have been reported to cause significant degradation of phenolic compounds in medicinal plants and herbs such as green tea, ginseng and wax apple [
27,
28,
29]. In this study, a remarkable decrease in TPC of dried Roselle samples was observed for the drying at 100 and 120 °C. Although the drying at 60 °C used the lowest temperature, the long drying time led to the longer exposure to heat, light, and oxygen of the bioactive ingredients in the materials which resulted in greater degradation of those compounds compared to the short time drying at higher temperatures. The highest retention of TPC in Roselle dried at 80 °C may be caused by the use of middle drying temperature combined with a relatively short time drying (9 h) which was reduced by 60% compared to that of the drying at 60 °C.
3.1.3. DPPH Radical Scavenging Activity of Roselle Dried at Different Temperatures
The variation in DPPH radical scavenging activity of the dried Roselle samples was in agreement with that of the total phenolic content.
Table 1 shows that extracts from Roselle dried at 80 °C possess higher antioxidant activities than those produced at other temperatures at all tested soluble solid concentrations. Although at low concentration, the antioxidant activity of Roselle extract dried at 60 °C was lower than those at 100 °C and comparable to the extracts at 120 °C, this increased rapidly and became significantly higher than the others at high concentrations (
Table 1). This may be due to the difference in the composition of antioxidants in the Roselle dried at 60 °C compared to that of dried samples at the higher temperatures. This may contain some antioxidants which only show antioxidant activity at high concentration but not at low concentration and those were not present in the samples dried at 100 °C and 120 °C.
As a result, the IC
50 for the DPPH radical scavenging ability of the Roselle dried at 80 °C followed by that dried at 60 °C while the IC
50 values of samples dried at 100 and 120 °C were the lowest (
Figure 3). The same trend in the variation of TPC and antioxidant capacity among the dried Roselle from different drying temperatures is predictable because phenolic acids, anthocyanins, and flavonoids, which all belong to the phenolic group, are the predominant bioactive ingredients in Roselle and mainly contribute to the antioxidant capacity of this material [
4,
6]. Thus it is reasonable when the products having higher TPC show stronger DPPH radical scavenging activity.
3.1.4. Sensory Characteristics of Roselle Dried at Different Temperatures
The images of dried Roselle products from different drying temperatures are shown in
Figure 4. The samples dried at higher temperatures present a brighter appearance while those obtained from lower temperatures were darker. This may be due to the higher concentration of anthocyanins in the samples dried at 60 and 80 °C which were claimed as the principal components contributing to the purple-red color of the Roselle [
6].
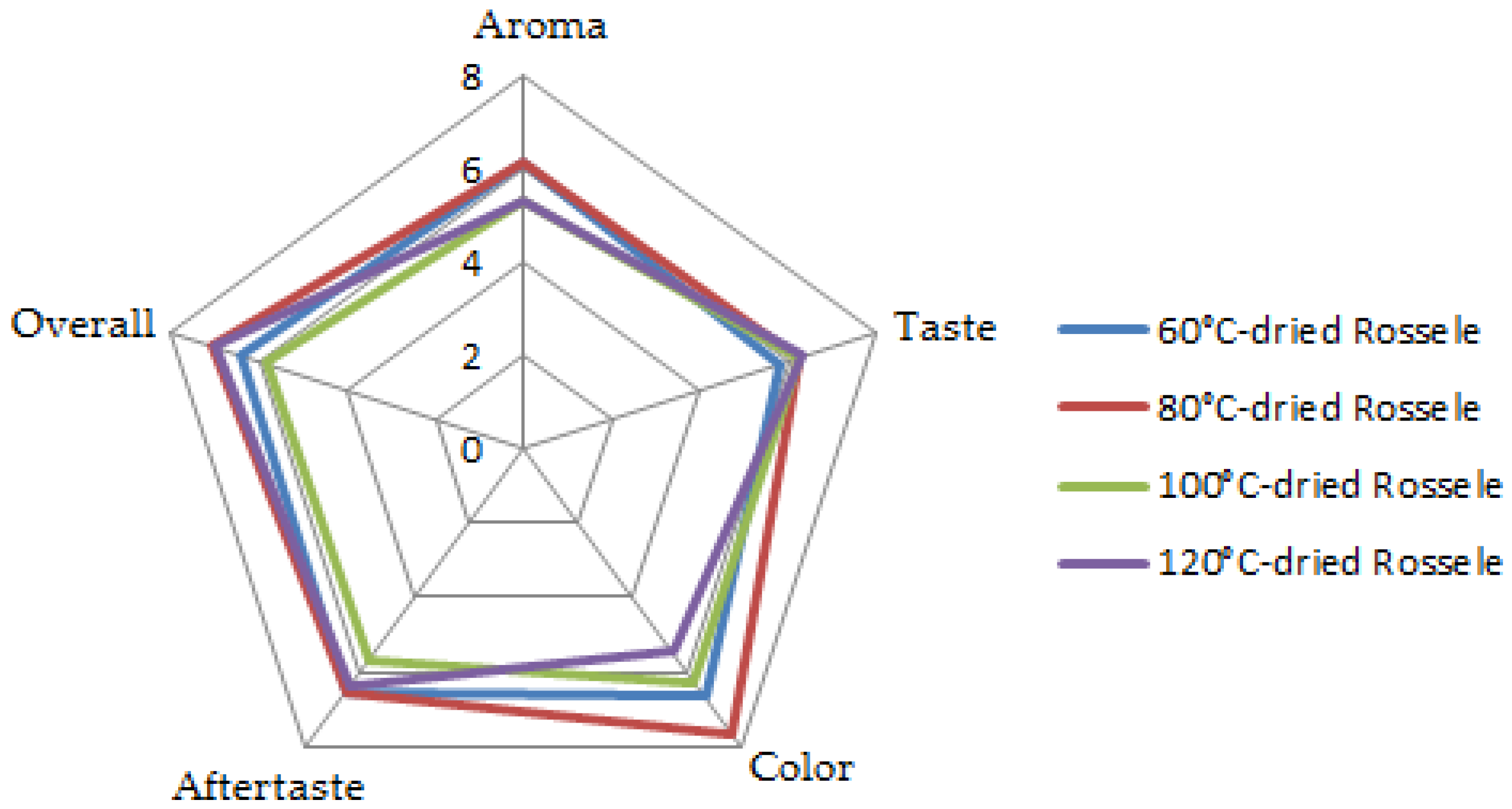
The sensory evaluation using the 9-point hedonic scale with 20 panelists showed great variations in the preference of the panelists with the individual characteristics of the dried Roselle samples (
Table 2). The samples dried at lower temperatures had higher points for aroma, color, and aftertaste while the dried Roselle from higher drying temperatures achieved higher grades for taste (
Figure 5).
Since the phenolic compounds significantly contribute to the bitter taste and astringent mount feel of foods and drinks [
30], the higher concentration of those compounds in Roselle dried at 60 and 80 °C may be the reason of the lower sensory results for the taste. However, the higher TPC resulted in the better aftertaste for those samples compared to the others. The higher retention in the principal color components, anthocyanins, in those samples also contributed to the higher intensity of the Roselle tea infusion which is shown by a very high sensory point for color (7.6) of the Roselle dried at 80 °C. As a result, the Roselle dried at 80 °C also got the highest overall acceptance of the sensory evaluation panel.
3.2. Effects of Brewing Conditions on Total Soluble Solid Content, Total Phenolic Content and Antioxidant Activity of Roselle Tea
3.2.1. Effect of Water Temperature
Table 3 shows that the use of water at 90 °C produces the Roselle infusion with the highest TPC and antioxidant capacity. The increase of water temperature from 80 to 90 °C resulted in an improvement in total soluble solid of the tea but this parameter did not significantly improve when the temperature increased to 100 °C. The use of water at 100 °C for brewing Roselle actually caused a decline in TPC and antioxidant capacity of the tea (
Table 3).
Literature has shown that most of the bioactive compounds, and the phenolic group in particular, from plant sources are very sensitive and very easy to be decomposed under high temperatures [
31]. This characteristic of phenolic compounds explained for the reduction of TPC in Roselle tea brewed at 100 °C. DPPH radical scavenging activity of this sample was also lower than that brewed at 90 °C. The literature has shown that phenolic acids, anthocyanins and flavonoids are the major compounds contributing to the antioxidant capacity of Roselle so the reduction of TPC in Roselle tea brewed at 100 °C may be responsible for the lower DPPH radical scavenging activity compared to that brewed at 90 °C [
4,
6].
3.2.2. Effect of Liquid-Solid Ratio
As reported by many studies related to the recovery of bioactive compounds and phenolics in particular from plant sources, the increase in the volume of solvent would improve the extraction yield of the compounds [
20,
30]. In the brewing of Roselle, the same trend was observed for the increase in TPC and antioxidant capacity of the Roselle tea. However, the significant improvement of those parameters was only obtained when the liquid-solid ratio increased from 8:1 to 10:1 but not at higher ratios (
Table 4).
The improvement in the extraction yield of soluble ingredients from materials with a higher liquid-solid ratio has been well-known to be caused by the higher gap of the concentration of solutes in the material and their concentration in the solvent. This leads to higher concentration gradients and allows the ingredients to diffuse into the solvent faster [
32]. However, the diffusion rate could not proportionally increase with the increase of the solvent amount because the amount of the solutes is limited and the rate depends on the distance from the surface of solid material to the diluted areas of solvent. Thus the increasing rate of the extraction yield declines slowly when an excessive amount of solvent is used [
32]. This trend can be clearly observed in this study when the liquid-solid ratios of 12:1 and 14:1 were used for brewing Roselle tea. Based on this result, Roselle is suggested to be brewed using water at 90 °C instead of boiling water as commonly recommended for the brewing of other herbal teas.
3.2.3. Effect of Brewing Time
The influence of brewing time on the TSS content, TPC, and antioxidant capacity of the Roselle drinks is shown in
Table 5. The TSS content and TPC increased along with the increase in the brewing time from 10 to 30 min but the increase was not significant when the brewing time excess 30 min. The change in the antioxidant capacity of the tea was similar to the increasing trend of TSS content and TPC but then declined when the drying time extended to 40 min.
The literature on the extraction of phenolic compounds reported that the phenolic extraction yield reached the maximum value after a certain extraction time depending on materials and the solvents, then fluctuated at this level or started declining if the extraction time is extended [
33,
34]. The decrease in antioxidant capacity of the Roselle drink after 40 min might be due to the degradation of other bioactive compounds in the solution such as carotenoids which also contribute to the antioxidant capacity of the solutions but are susceptible and degraded by high temperature combined with a long exposure time of the brewing process [
34].
4. Conclusions
In this study, drying temperatures (60, 80, 100, and 120 °C) were used for drying Roselle calyxes to investigate their effects on the retention of total phenolic content, antioxidant capacity and sensory quality of the dried Roselle. The drying at 80 °C produced the dried Roselle with the highest retention of TPC (25.196 mg GAE/g), strongest AC (IC50 of 2.2 mg/mL), and the highest sensory quality compared to the Roselle dried at other temperatures. Effects of brewing conditions including water temperature, water-to-Roselle ratio and brewing time on the quality of Roselle infusion were also evaluated. The brewing process using hot water at 90 °C with the solid-liquid ratio of 1:10 (g/mL) for 30 min resulted in the highest TSS content (5.4 oBx), TPC (12.7 mg GAE/g Roselle), and strongest AC (73.3% DPPH inhibition) of the Roselle tea. Thus, the drying at 80 °C combined with the brewing of dried Roselle for 30 min using 90 °C hot water with the solid-liquid ratio of 1:10 (g/mL) are recommended to produce Roselle tea with the highest content of beneficial ingredients.
Author Contributions
Conceptualization, Q.V.N.; methodology, Q.V.N.; software, Q.V.N. and H.V.C.; validation, Q.V.N. and H.V.C.; formal analysis, Q.V.N. and H.V.C.; investigation, Q.V.N. and H.V.C.; resources, Q.V.N.; data curation, Q.V.N. and H.V.C.; writing—original draft preparation, Q.V.N. and H.V.C.; writing—review and editing, H.V.C.; supervision, Q.V.N. and H.V.C.; project administration, Q.V.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Domínguez-López, A.; Edgardo, R.G.; Navarro-Galindo, S. Thermal Kinetic Degradation of Anthocyanins in a Roselle (Hibiscus sabdariffa L. Cv. ‘Criollo’) Infusion. Int. J. Food Sci. Technol. 2008, 43, 322–325. [Google Scholar]
- Patel, S. Hibiscus Sabdariffa: An Ideal yet under-Exploited Candidate for Nutraceutical Applications. Biomed. Prev. Nutr. 2014, 4, 23–27. [Google Scholar] [CrossRef]
- Hopkins, A.L.; Lamm, M.G.; Funk, J.L.; Ritenbaugh, C. Hibiscus Sabdariffa L. In the Treatment of Hypertension and Hyperlipidemia: A Comprehensive Review of Animal and Human Studies. Fitoterapia 2013, 85, 84–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borrás-Linares, I.; Fernández-Arroyo, S.; Arráez-Roman, D.; Palmeros-Suárez, P.A.; del Val-Díaz, R.; Andrade-Gonzáles, I.; Fernández-Gutiérrez, A.; Gómez-Leyva, J.F.; Segura-Carretero, A. Characterization of Phenolic Compounds, Anthocyanidin, Antioxidant and Antimicrobial Activity of 25 Varieties of Mexican Roselle (Hibiscus sabdariffa). Ind. Crops Prod. 2015, 69, 385–394. [Google Scholar] [CrossRef]
- Lin, H.H.; Chen, J.H.; Wang, C.J. Chemopreventive Properties and Molecular Mechanisms of the Bioactive Compounds in Hibiscus sabdariffa Linne. Curr. Med. Chem. 2011, 18, 1245–1254. [Google Scholar] [CrossRef]
- Pi-Jen, T.; McIntosh, J.; Pearce, P.; Camden, B.; Jordan, B.R. Anthocyanin and Antioxidant Capacity in Roselle (Hibiscus sabdariffa L.) Extract. Food Res. Int. 2002, 35, 351–356. [Google Scholar]
- Liu, K.S.; Tsao, S.M.; Yin, M.C. In Vitro Antibacterial Activity of Roselle Calyx and Protocatechuic Acid. Phytother Res. 2005, 19, 942–945. [Google Scholar] [CrossRef]
- Olaleye, M.T. Cytotoxicity and Antibacterial Activity of Methanolic Extract of Hibiscus Sabdariffa. J. Med. Plants Res. 2007, 1, 9–13. [Google Scholar]
- Herrera-Arellano, A.; Flores-Romero, S.; Chavez-Soto, M.A.; Tortoriello, J. Effectiveness and Tolerability of a Standardized Extract from Hibiscus sabdariffa in Patients with Mild to Moderate Hypertension: A Controlled and Randomized Clinical Trial. Phytomedicine 2004, 11, 375–382. [Google Scholar] [CrossRef]
- Mbondo Naomi, N.; Owino, W.O.; Ambuko, J.; Sila, D.N. Effect of Drying Methods on the Retention of Bioactive Compounds in African Eggplant. Food Sci. Nutr. 2018, 6, 814–823. [Google Scholar] [CrossRef] [Green Version]
- Monika, G.; Siwulski, M.; Magdziak, Z.; Budzyńska, S.; Stuper-Szablewska, K.; Niedzielski, P.; Mleczek, M. The Effect of Drying Temperature on Bioactive Compounds and Antioxidant Activity of Leccinum scabrum (Bull.) Gray and Hericium erinaceus (Bull.) Pers. J. Food Sci. Technol. 2019, 56, 1–13. [Google Scholar]
- Zeinab, N.; Mahdavi, R.; Ghaemmaghami, S.J.; Yagin, N.L.; Nikniaz, L. Effect of Different Brewing Times on Antioxidant Activity and Polyphenol Content of Loosely Packed and Bagged Black Teas (Camellia sinensis L.). Avicenna J. Phytomedicine 2016, 6, 313–321. [Google Scholar]
- Erica, S.; Hua, F.; Schuckers, S.; Andreescu, S.; Bradley, R. Effects of Brewing Conditions on the Antioxidant Capacity of Twenty-Four Commercial Green Tea Varieties. Food Chem. 2016, 192, 380–387. [Google Scholar]
- Peng-Kong, W.; Yusof, S.; Ghazali, H.M.; Man, Y.B.C. Optimization of Hot Water Extraction of Roselle Juice Using Response Surface Methodology: A Comparative Study with Other Extraction Methods. J. Sci. Food Agric. 2003, 83, 1273–1278. [Google Scholar]
- Ben Amor, B.; Allaf, K. Impact of Texturing Using Instant Pressure Drop Treatment Prior to Solvent Extraction of Anthocyanins from Malaysian Roselle (Hibiscus sabdariffa). Food Chem. 2009, 115, 820–825. [Google Scholar] [CrossRef]
- Chumsri, P.; Anchalee, S.; Itharat, A. Studies on the Optimum Conditions for the Extraction and Concentration of Roselle (Hibiscus sabdariffa Linn.) Extract. Songklanakarin J. Sci. Technol. 2008, 30, 133–139. [Google Scholar]
- Gonzalez-Palomares, S.; Estarron-Espinosa, M.; Gomez-Leyva, J.F.; Andrade-Gonzalez, I. Effect of the Temperature on the Spray Drying of Roselle Extracts (Hibiscus sabdariffa L.). Plant Foods Hum. Nutr. 2009, 64, 62–67. [Google Scholar] [CrossRef]
- Leyva, D.D.; Huerta, B.E.B.; Sosa, I.A.; Mendoza, M.G.V. Effect of Fixed Bed Drying on the Retention of Phenolic Compounds, Anthocyanins and Antioxidant Activity of Roselle (Hibiscus sabdariffa L.). Ind. Crops Prod. 2012, 40, 268–276. [Google Scholar]
- Hahn, F.; Hernández, G.; Hernández, J.; Pérez, C.; Vargas, J.M. Optimization of Roselle Drying Time and Drying Quality. Can. Biosyst. Eng. 2011, 53, 31–38. [Google Scholar]
- Quan, V.; Goldsmith, C.; Dang, T.; Nguyen, T.; Bhuyan, D.J.; Sadeqzadeh, E.; Scarlett, C.; Bowyer, M. Optimisation of Ultrasound-Assisted Extraction Conditions for Phenolic Content and Antioxidant Capacity from Euphorbia Tirucalli Using Response Surface Methodology. Antioxidants 2014, 3, 604–617. [Google Scholar]
- Quang, N.; Eun, J. Antioxidant Activity of Solvent Extracts from Vietnamese Medicinal Plants. J. Med. Plants Res. 2011, 5, 2798–2811. [Google Scholar]
- Peryam, D.; Girardot, N. Advanced Taste-Test Method. Food Eng. Rev. 1952, 24, 58–61. [Google Scholar]
- Latimer, G.W.; The Association of Official Agricultural (AOAC). Official Method of Analysis, 17th ed.; AOAC: Washington, DC, USA, 2000. [Google Scholar]
- Chan, E.W.C.; Lim, Y.Y.; Wong, S.K.; Lim, K.K.; Tan, S.P.; Lianto, F.S.; Yong, M.Y. Effects of Different Drying Methods on the Antioxidant Properties of Leaves and Tea of Ginger Species. Food Chem. 2009, 113, 166–172. [Google Scholar] [CrossRef]
- Jung, C.; Dai, Y.; Cheng, H.; Lam, W.; Cheng, Y.; Li, K.; Peng, W.; Pao, L.; Hsieh, M.; Qin, X.; et al. Improving the Concentrations of the Active Components in the Herbal Tea Ingredient, Uraria Crinita: The Effect of Post-Harvest Oven-Drying Processing. Sci. Rep. 2017, 7, 38763. [Google Scholar]
- Chong, K.L.; Lim, Y.Y. Effects of Drying on the Antioxidant Properties of Herbal Tea from Selected Vitex Species. J. Food Qual. 2012, 35, 51–59. [Google Scholar] [CrossRef]
- Minh, N.P.; Anh, N.H.; Pha, P.T.L.; Bach, L.G. Effect of Blanching, Drying and Preservation of Dried Wax Apple (Syzygium samarangense) Leaf Tea. Res. Crops 2018, 19, 730–735. [Google Scholar]
- Xu, L.; Zhang, L.; Lei, H.; Zhang, H.; Cheng, Y.; Zhu, R.; Ruan, R. Effect of Drying Technologies on Quality of Green Tea. Int. Agric. Eng. J. 2010, 19, 30–37. [Google Scholar]
- Guixing, R.; Chen, F. Drying of American Ginseng (Panax Quinquefolium Roots by Microwave-Hot Air Combination. J. Food Eng. 1998, 35, 433–443. [Google Scholar]
- Soares, S.; Kohl, S.; Thalmann, S.; Mateus, N.; Meyerhof, W.; de Freitas, V. Different Phenolic Compounds Activate Distinct Human Bitter Taste Receptors. J. Agric. Food Chem. 2013, 61, 1525–1533. [Google Scholar] [CrossRef]
- Murthy Pushpa, S.; Madhava Naidu, M. Recovery of Phenolic Antioxidants and Functional Compounds from Coffee Industry by-Products. Food Bioprocess Technol. 2012, 5, 897–903. [Google Scholar] [CrossRef]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for Extraction of Bioactive Compounds from Plant Materials: A Review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Gordana, R.; Komes, D.; Likić, S.; Horžić, D.; Kovač, M. Phenolic Content and Antioxidative Capacity of Green and White Tea Extracts Depending on Extraction Conditions and the Solvent Used. Food Chem. 2008, 110, 852–858. [Google Scholar]
- Chuyen Hoang, V.; Tran, X.T.; Roach, P.D.; Golding, J.B.; Parks, S.E.; Nguyen, M.H. Yield of Carotenoids, Phenolic Compounds and Antioxidant Capacity of Extracts from Gac Peel as Affected by Different Solvents and Extraction Conditions. J. Adv. Agric. Technol. 2017, 4, 87–91. [Google Scholar] [CrossRef] [Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).