Investigating the Potential of an In-Situ Method for Monitoring the Malting of Barley Using Solid Phase Microextraction with a Portable Gas Chromatography Mass Spectrometry Instrument
Abstract
:1. Introduction
2. Material and Methods
2.1. Analytical Methods
2.2. Malting Process
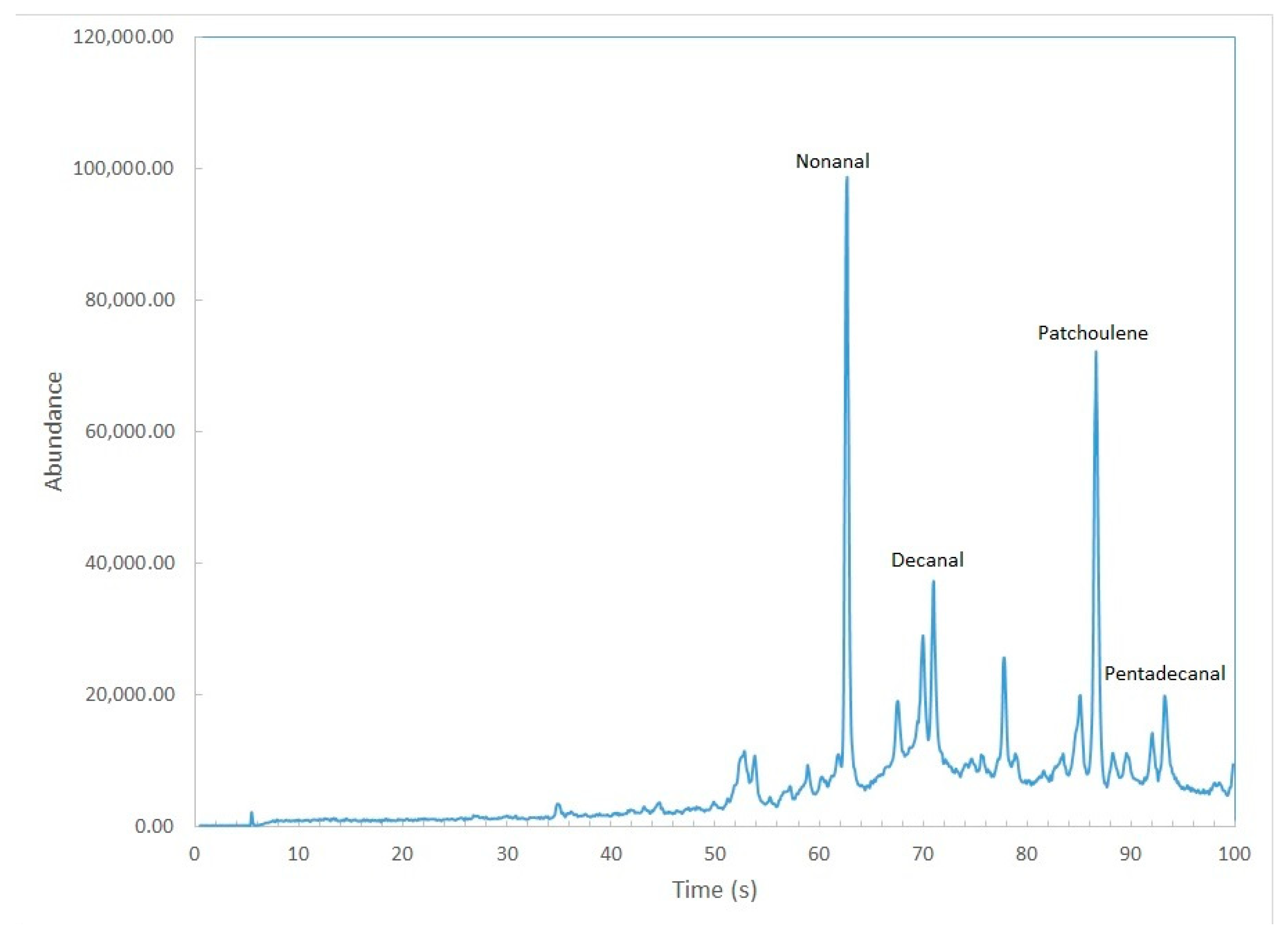
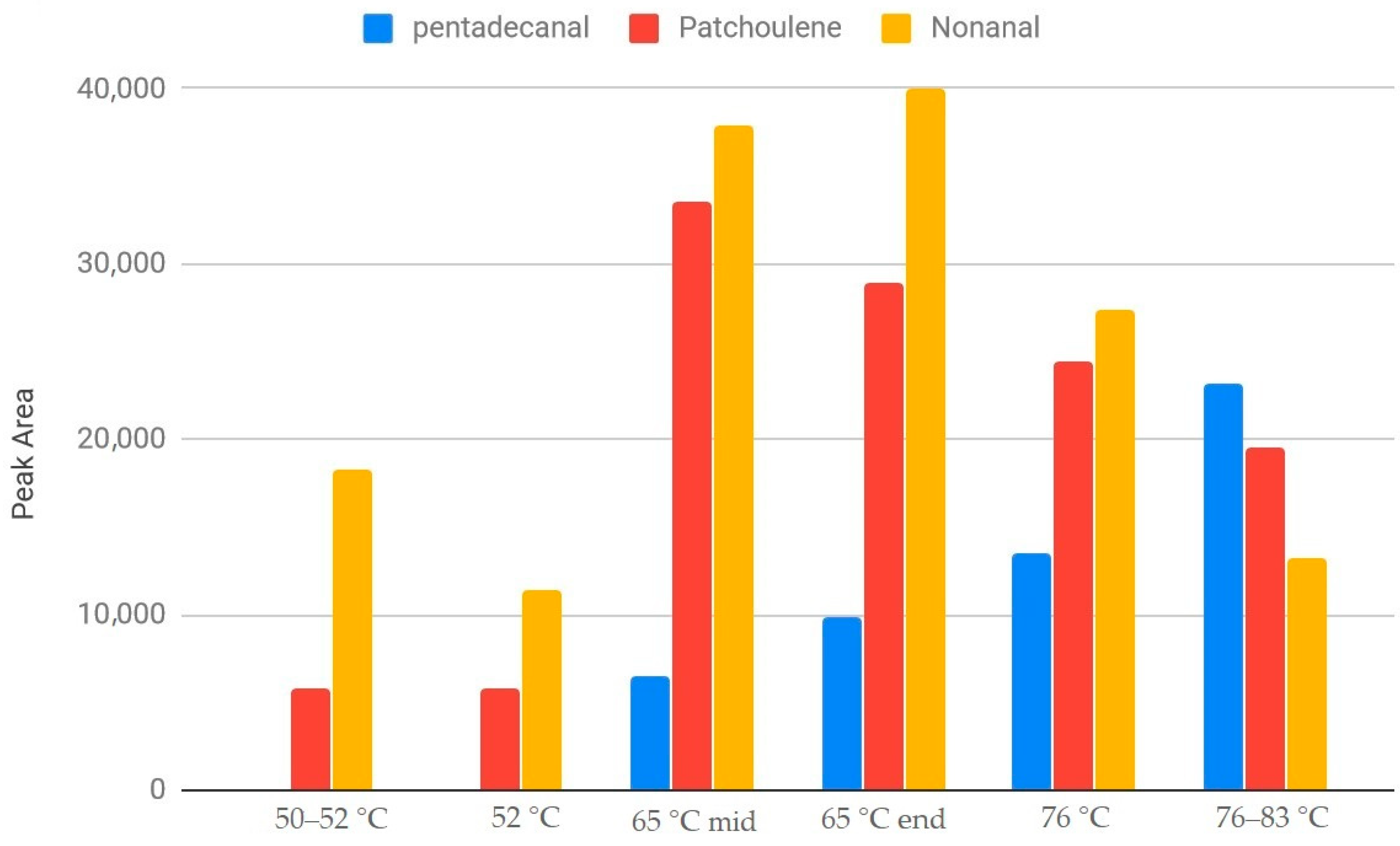
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beer Serves America: A Study of the U.S. Beer Industry’s Economic Contribution in 2018, A Report Prepared for The Beer Institute. Available online: http://beerservesamerica.org/wp-content/uploads/2019/05/BEER-SERVES-AMERICA-A-Study-of-the-U.S.-Beer-Industry%E2%80%99s-Economic-Contribution-in-2018-.pdf (accessed on 17 July 2019).
- De Schutter, D.P.; Saison, D.; Delvaux, F.; Derdelinckx, G.; Rock, J.-M.; Neven, H.; Delvaux, F.R. Characterization of volatiles in unhopped wort. J. Agric. Food Chem. 2008, 56, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.R.; Soprano, S.E.; Wickham, L.M.; Fitzgerald, N.; Edwards, J.C. Nuclear magnetic resonance and headspace solid-phase microextraction gas chromatography as complementary methods for the analysis of beer samples. Beverages 2017, 3, 21. [Google Scholar] [CrossRef] [Green Version]
- Kleinová, J.; Klejdus, B. Determination of volatiles in beer using solid-phase microextraction in combination with gas chromatography/mass spectrometry. Czech J. Food Sci. 2014, 32, 241–248. [Google Scholar] [CrossRef] [Green Version]
- Beal, A.D.; Mottram, S. Compounds contributing to the characteristic aroma of malted barley. J. Agric. Food Chem. 1994, 42, 2880–2884. [Google Scholar] [CrossRef]
- Vandecan, S.M.G.; Saison, D.; Schouppe, N.; Delvaux, F.; Delvaux, F.R. Optimization of speciality malt volatile analysis by headspace solid-phase microextraction in combination with gas chromatography and mass spectrometry. Anal. Chim. Acta 2010, 671, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Piao, Y.; Zhang, X.; Zhao, C.; Hou, Y.; Hou, Y.; Shi, Z. Analysis of volatile compounds from a malting process using headspace solid-phase micro-extraction and GC-MS. Food Res. Int. 2013, 51, 783–789. [Google Scholar] [CrossRef]
- Eckenrode, B.A. Environmental and forensic applications of field-portable GC-MS: An overview. J. Am. Soc. Mass Spectrom. 2001, 12, 683–693. [Google Scholar] [CrossRef] [Green Version]
- Cockburn, P. Method Development for Identification of Adulterated Spirits Using Field Portable GC/MS, Application Note, Perkin Elmer Inc., Waltham, MA. 2016. Available online: https://www.perkinelmer.com/lab-solutions/resources/docs/APP_Method-Dev-ID-Adulterated-Spirits-using-Field-Portable-GC-MS_012711_01.pdf (accessed on 1 September 2019).
- Reddy, S. Rapid Fingerprinting of Volatile Components in Coffee Varieties Using the Portable Torion T-9 Portable GC/MS System, Application Note, Perkin Elmer Inc., Shelton, CT. 2016. Available online: https://www.perkinelmer.com/lab-solutions/resources/docs/APP_Volatile-Components-in-Coffee_013202_01.pdf (accessed on 1 September 2019).


| Steeping Cycles | |
| Time (h) | Grain Environment |
| 8 | Wet |
| 14 | Dry |
| 6 | Wet |
| 12 | Dry |
| 1 | Wet |
| Kilning Conditions | |
| Time (h) | Temperature (°C) |
| 5.5 | 50 |
| 41 * | 52 |
| 3.25 | 65 |
| 4.5 | 76 |
| 2.25 | 85 |
| Alkanes | 3-methyl hexane, 4-methyl octane, 3 ethyl-5-(2-ethylbutyl) octadecane |
| Aldehydes | Hexanal, heptanal, nonanal, 2-nonenal, decanal, pentadecanal |
| Ketones | 2-decanone |
| Alcohols | 2-decen-1-ol |
| Esters | eicosanoic acid, phenylmethyl ester |
| Terpenes | patchoulene |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodges, M.D.; Fitzgerald, N. Investigating the Potential of an In-Situ Method for Monitoring the Malting of Barley Using Solid Phase Microextraction with a Portable Gas Chromatography Mass Spectrometry Instrument. Beverages 2020, 6, 72. https://0-doi-org.brum.beds.ac.uk/10.3390/beverages6040072
Hodges MD, Fitzgerald N. Investigating the Potential of an In-Situ Method for Monitoring the Malting of Barley Using Solid Phase Microextraction with a Portable Gas Chromatography Mass Spectrometry Instrument. Beverages. 2020; 6(4):72. https://0-doi-org.brum.beds.ac.uk/10.3390/beverages6040072
Chicago/Turabian StyleHodges, Mark D., and Neil Fitzgerald. 2020. "Investigating the Potential of an In-Situ Method for Monitoring the Malting of Barley Using Solid Phase Microextraction with a Portable Gas Chromatography Mass Spectrometry Instrument" Beverages 6, no. 4: 72. https://0-doi-org.brum.beds.ac.uk/10.3390/beverages6040072






