1. Introduction
Guava (
Psidium guajava) is tropical fruit cultivated in tropical and subtropical regions. It contains high levels of antioxidants such as beta-carotene, lycopene which played an important role in combating free radicals. It is mostly consumed in fresh or processed form (puree, concentrated, and juice); guava juice is favored in tropical regions due to its sweet and refreshing flavor. Moreover, it is an excellent source of several important vitamins and minerals [
1].
Nowadays, fruit juice consumption is increased due to its relief of thirst, nutrition, and easy digestibility [
2]. Moreover, a large amount of refined sugar is used to sweeten fruit juice for preferable flavor, taste, and color [
3]. They also play a crucial role in the appearance, physicochemical, and rheology of fruit juices [
4]. However, it affected health problems, including obesity, diabetes, and hyperlipidemia [
5]. Therefore, sweeteners are alternative compounds used to partly or completely replace sugar in fruit juice [
6].
A sweetener is a substance used to replace sucrose which can provide the same sweet taste as sucrose. The type of sweetener used can influence the physicochemical properties which may limit its addition to a product [
6] using each sweetener in situations for which it is best suited [
7]. In addition, six sweeteners, including saccharin, aspartame, sucralose, neotame, acesulfame-K, and stevia have been classified under the generally recognized as safe (GRAS) category [
8]. Several sweeteners are allowed for use in food and beverages [
9].
Neotame is an artificial sweetener that has the same characteristics as aspartame. The sweetness is 5000 times that of sucrose, and is sweeter than aspartame. It also has a desirable flavor more than aspartame [
10]. Derta and Bolini [
11] reported that there was no significant difference in appearance and aroma of acerola nectar sweetened with sucrose, neotame, and stevia. In terms of vitamin C content and total titratable acidity, there was also no significant difference (
p > 0.05).
Stevia is produced from the leaf of Stevia rebaudiana (Bertoni, The Netherlands) which has a relative sweetness of 200 times of sucrose [
12]. Rebaudioside A, one of the major sweet components of the plant Stevia rebaudiana [
13] is sweeter and stable, and less bitter than stevioside [
14]. Additionally, stevia is used to replace sugar in many beverage products such as juice and nectar [
15]. Mango nectar mixed with stevia had significant effects on the total soluble solids (°Brix), viscosity and pH which were affected by concentration. In addition flow behavior was not different from mango containing sucrose [
15]. Moreover, the physicochemical properties of mango nectar were evaluated, including color, pH, titratable acidity, and soluble solids in mango nectar sweetened during storage time. The sample nectar blended with stevia showed similar physicochemical properties to sucrose in 0 days. However, sweeteners such as acesulfame-K, sucralose, neotame, and stevia in nectar displayed good stability in mango nectar [
16]. Furthermore, the rheological properties of juice are also affected by sugar content. Sweetened products with sugar substitutes should have similar rheology to the original product. According to Miele et al. [
17] yogurt with different sweeteners replacing sugar did not affect the yogurt’s rheological properties.
However, sucrose replacement by different sweeteners is a challenging alternative that can affect the physicochemical property and rheology of guava nectar. In this study, guava nectar was sweetened with sucrose and two different sweeteners, including neotame and stevia. While stevia is a natural sweetener from a plant that has been used as a sweetener in beverages for decades, neotame is an artificial sweetener with a taste profile similar to sucrose. It promoted desirable flavors, including sweet taste and mouthfeel [
18]. Therefore, the objective of this study was to determine physicochemical property, antioxidant activity, and rheological properties in guava nectar with different sweeteners during refrigerated storage. This will be a positive approach to the reduction of sugar consumption in a low calories drink.
2. Materials and Methods
2.1. Raw Materials
Fresh guava (Psidium guajava) of the Kimju variety was purchased from the local market in Bangkok, Thailand. The sweetener used for the sweetened guava juice was from Chemipan Corporation Co., Ltd., Bangkok, Thailand. The other ingredients such as salt (Prungthip) and sugar (Mitr-Phol) were from a supermarket.
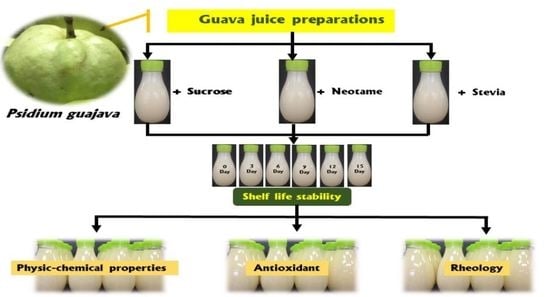
2.2. Preparation Guava Nectar Formulation
The raw material was washed with running water and then cut into pieces. The pulp was mixed with distilled water in a ratio of 1:1 (w/v) by a blender. The mixture was transferred to a nylon bag which was applied pressure to separate the fruit juice from the pulp. Citric acid (0.15%, w/v) and NaCl (0.25%, w/v) were added to juice. The soluble solid was adjusted to 8 °Brix. After mixing, the beverage was homogenized in a rotor-stator type homogenizer (Walita®, São Paulo, Brazil) for 5 min.
2.3. Preparation of Sweetened Guava Nectar
The guava nectar was mixed with neotame and stevia (0.01% and 0.05%) with 0.1% guar gum. Briefly, intense sweetener and guar gum were gradually mixed with guava nectar. For guava nectar without sweetener, sucrose was added to nectar for 4% (w/v), 0.15% (w/v) citric acid and 0.25% (w/v) NaCl as control. The mixture was homogenized and heated to 80 °C for 30 s, then cooled in the ice bath to 40 °C. Finally, guava nectar and sweetened guava nectar were filled in a pasteurized clear bottle (80 °C for 5 s), sealed, and stored refrigerator (4 °C) for 15 days. All juice samples were analyzed immediately after preparation. The shelf life of guava nectar and sweetened guava nectar were determined every 3 days for physicochemical properties, antioxidant activity, and rheology.
2.4. Analytical Methods for Shelf Life Evaluation
The pasteurized guava nectar shelf life was evaluated at 5 °C ± 2 °C to determine physicochemical properties and antioxidant activity which were performed on days 0, 3, 6, 9, 12, and 15.
2.5. Physico-Chemical Property
The cloud value was evaluated using a UV-1601 spectrophotometer (Shimadzu, Japan) that was calibrated with distilled water according to Abid et al. [
19]. The samples were centrifuged at 618×
g for 20 min, then absorbance at 660 nm of the supernatant was recorded. The transmittance rate was calculated as the following equation:
where
The color of guava nectar samples was measured according to the method described by Wang et al. [
20]. The colorimeter (ColorFlex EZ spectrophotometer, Horsham, PA, USA) was used to evaluate the color during storage.
The parameters including L*, a*, and b* of each sample were recorded. The results were expressed as total color difference (ΔE) values which were calculated as follows
where ΔL, Δa* and Δb* are a difference of lightness, redness, and yellowness, respectively, for 0, 3, 6, 9, 12 and 15 days of storage.
The pH of guava nectar and sweetened guava nectar was measured at 25 °C by using a digital pH meter (Denver Instrument, Denver, CO, USA) with a glass electrode.
The acidity of nectar was evaluated by using an auto titrator (Titriline 700, Columbus, OH, USA). Fruit juice (10 mL) mixed with 20 mL of distilled water that was titrated with 0.1 NaOH to a pH value of 8.3 as previously described by Xiang et al., (2018). The results were expressed as g citric acid/100 mL of guava nectar.
2.6. Total Carotenoid Content
Total carotenoid was determined by spectrophotometer (UV-1601 spectrophotometer, Shimadzu, Japan) as proposed by Kotíková et al. [
21]. The nectar samples (2 mL) were added to 15 mL of acetone and incubated for 2 h. The excess acetone was removed by centrifuge at 4000×
g for 5 min, then adjusted volume to 25 with acetone. The samples were measured absorbance at 662 nm, 645 nm, and 470 nm and calculated with the equation.
2.7. Antioxidant Activity
2.7.1. ABTS•+ Radical Scavenging Activity Assay
The ABTS
•+ scavenging activity was performed according to Re et al. [
22] as follows: The ABTS
•+ solution was prepared by mixing two solutions of ABTS
•+ (7 mM) and potassium persulfate (2.45 mM). The mixture was allowed to stand in the dark condition at room temperature for 16 h before use. The ABTS
•+ solution was diluted to the absorbance of 0.700 ± 0.02 at 734 nm. Then, guava juice (1 mL) was added with 3 mL of ABTS
•+ solution, mixed, and kept in the dark for 6 min. The absorbance was measured at 734 nm. The scavenging activity was calculated as:
where
2.7.2. DPPH• Radical Scavenging Activity Assay
Antioxidant activity was assessed as DPPH
• radical scavenging activity according to Bhat and Goh. [
23]. Briefly,
100 µL of the nectar was added to 2 mL of 0.1 mM DPPH
• in 95% ethanol. The mixture was incubated at room temperature (dark condition) for 30 min. The absorbance was recorded at 517 nm with a spectrophotometer (UV-1601 spectrophotometer, Shimadzu, Japan). The antioxidant activity was expressed as the scavenging activity according to the equation
where
2.8. Flow Behavior and Apparent Viscosity of Guava Juice
The apparent viscosity of nectar samples was determined by using a rheometer (Haake Mars 40 model, Thermo Fisher Scientific, Waltham, MA, USA) equipped with cup and coaxial cylinder geometry. The shear rate was varied from 0.01 s
−1 to 10 s
−1 at 540 s
−1 at 5 °C and 37 °C with a gap of 0.01 mm. The nectar samples (17 mL) were placed into a cup and allowed to rest at 5 °C and 37 °C for 120 s
−1 [
24]. A total of 30 data points were recorded at 15 s
−1 during shearing.
2.9. Volatile Compound Gas Chromatography
The volatile compound of all guava nectar samples was performed by chromatography-mass spectrometry (Agilent DB-WAX column, 60 m × 0.25 mm × 0.25 μm film thickness dimensions, Beijing, China) with flame ionization detector (FID). The sample (3 µL) was extracted by using headspace mode at 30 °C for 30 min. After that, volatile compounds were subjected to solid-phase microextraction (SPME fiber 50/30/µm Divinylbenzene/Carbon wide range/Polydimethylsiloxane stableflex 2 cm, Suppleco) and injection was performed in a splitless mode at 250 °C that helium was the carrier gas. The gas chromatography (GC) temperature was 50 °C for 2.5 min up to 200 °C in 4 min. The volatile compound was identified by comparing their GC retention time (RT) as proposed in NIST 2005 v 2.0 (Natl. Inst. of Standards and Technology, Gaithersburg, MD, USA) according to Guo et al. [
25] with slight modification.
2.10. Statistical Analysis
The physicochemical and antioxidant properties were performed in triplicate in a Randomized Complete Block Design. The result was subjected to a one-way analysis of variance (ANOVA) with Statistical Analysis System (SAS, version 6.0) (SAS Institute, Cary, NC, USA). Duncan’s Multiple Range Test was used to determine significant differences between means (p < 0.05). All data were presented as average values with the standard deviation.