Early Aberrant Angiogenesis Due to Elastic Fiber Fragmentation in Aortic Valve Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Human Valve Tissue
2.2. Histochemistry
2.3. Immunohistochemistry
2.4. Transmission Electron Microscopy
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. Early-Onset AVD Specimens Demonstrate Aberrant Angiogenesis without Inflammation or Atherosclerosis
3.3. A Distinct Pattern of Elastic Fiber Components Is Associated with Early-Onset AVD Neovessels
3.4. WS but Not MFS Aortic Valves Demonstrate Aberrant Angiogenesis
3.5. The Aortic Root Is Composed in Part of Valve Tissue
3.6. Different Elastic Fiber Defects Result in Different Types of EFF
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nkomo, V.T.; Gardin, J.M.; Skelton, T.N.; Gottdiener, J.S.; Scott, C.G.; Enriquez-Sarano, M. Burden of valvular heart diseases: A population-based study. Lancet 2006, 368, 1005–1011. [Google Scholar] [CrossRef]
- Go, A.S.; Mozaffarian, D.; Roger, V.L.; Benjamin, E.J.; Berry, J.D.; Blaha, M.J.; Dai, S.; Ford, E.S.; Fox, C.S.; Franco, S.; et al. Heart Disease and Stroke Statistics—2014 Update: A Report From the American Heart Association. Circulation 2013, 129, e28–e292. [Google Scholar] [CrossRef] [Green Version]
- Bonow, R.O.; Carabello, B.A.; Chatterjee, K.; de Leon, A.C.; Faxon, D.P.; Freed, M.D.; Gaasch, W.H.; Lytle, B.W.; Nishimura, R.A.; O’Gara, P.T.; et al. 2008 Focused Update Incorporated Into the ACC/AHA 2006 Guidelines for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1998 Guidelines for the Management of Patients With Valvular Heart Disease) Endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J. Am. Coll. Cardiol. 2008, 52, e1–e142. [Google Scholar] [CrossRef] [Green Version]
- Otto, C.M. Valvular Aortic Stenosis: Disease Severity and Timing of Intervention. J. Am. Coll. Cardiol. 2006, 47, 2141–2151. [Google Scholar] [CrossRef]
- Rajamannan, N.M.; Evans, F.J.; Aikawa, E.; Grande-Allen, K.J.; Demer, L.L.; Heistad, D.D.; Simmons, C.A.; Masters, K.S.; Mathieu, P.; O’Brien, K.D.; et al. Calcific aortic valve disease: Not simply a degenerative process: A review and agenda for research from the National Heart and Lung and Blood Institute Aortic Stenosis Working Group. Circulation 2011, 124, 1783–1791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schoen, F.J. Evolving concepts of cardiac valve dynamics: The continuum of development, functional structure, pathobiology, and tissue engineering. Circulation 2008, 118, 1864–1880. [Google Scholar] [CrossRef] [PubMed]
- Hinton, R.B.; Yutzey, K.E. Heart Valve Structure and Function in Development and Disease. Annu. Rev. Physiol. 2011, 73, 29–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Folkman, J. Angiogenesis: An organizing principle for drug discovery? Nat. Rev. Drug Discov. 2007, 6, 273–286. [Google Scholar] [CrossRef]
- Risau, W. Mechanisms of angiogenesis. Nat. Cell Biol. 1997, 386, 671–674. [Google Scholar] [CrossRef]
- Hakuno, D.; Kimura, N.; Yoshioka, M.; Mukai, M.; Kimura, T.; Okada, Y.; Yozu, R.; Shukunami, C.; Hiraki, Y.; Kudo, A.; et al. Periostin advances atherosclerotic and rheumatic cardiac valve degeneration by inducing angiogenesis and MMP production in humans and rodents. J. Clin. Investig. 2010, 120, 2292–2306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajamannan, N.M.; Nealis, T.B.; Subramaniam, M.; Pandya, S.; Stock, S.R.; Ignatiev, C.I.; Sebo, T.J.; Rosengart, T.K.; Edwards, W.D.; McCarthy, P.M.; et al. Calcified rheumatic valve neoangiogenesis is associated with vascular endothelial growth factor expression and osteoblast-like bone formation. Circulation 2005, 111, 3296–3301. [Google Scholar] [CrossRef] [Green Version]
- Soini, Y.; Salo, T.; Satta, J. Angiogenesis is involved in the pathogenesis of nonrheumatic aortic valve stenosis. Hum. Pathol. 2003, 34, 756–763. [Google Scholar] [CrossRef]
- Syvaranta, S.; Helske, S.; Laine, M.; Lappalainen, J.; Kupari, M.; Mayranpaa, M.I.; Lindstedt, K.A.; Kovanen, P.T. Vascular endothelial growth factor-secreting mast cells and myofibroblasts: A novel self-perpetuating angiogenic pathway in aortic valve stenosis. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 1220–1227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caira, F.C.; Stock, S.R.; Gleason, T.G.; McGee, E.C.; Huang, J.; Bonow, R.O.; Spelsberg, T.C.; McCarthy, P.M.; Rahimtoola, S.H.; Rajamannan, N.M. Human degenerative valve disease is associated with up-regulation of low-density lipoprotein receptor-related protein 5 receptor-mediated bone formation. J. Am. Coll. Cardiol. 2006, 47, 1707–1712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohler, E.R.; Gannon, F.; Reynolds, C.; Zimmerman, R.; Keane, M.G.; Kaplan, F.S. Bone Formation and Inflammation in Cardiac Valves. Circulation 2001, 103, 1522–1528. [Google Scholar] [CrossRef] [PubMed]
- Rossebø, A.B.; Pedersen, T.R.; Boman, K.; Brudi, P.; Chambers, J.B.; Egstrup, K.; Gerdts, E.; Gohlke-Bärwolf, C.; Holme, I.; Kesäniemi, Y.A.; et al. Intensive Lipid Lowering with Simvastatin and Ezetimibe in Aortic Stenosis. N. Engl. J. Med. 2008, 359, 1343–1356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schoen, F.J. Aortic valve structure-function correlations: Role of elastic fibers no longer a stretch of the imagination. J. Hear. Valve Dis. 1997, 6, 1–6. [Google Scholar]
- Vesely, I. The role of elastin in aortic valve mechanics. J. Biomech. 1997, 31, 115–123. [Google Scholar] [CrossRef]
- Fondard, O.; Detaint, D.; Iung, B.; Choqueux, C.; Adle-Biassette, H.; Jarraya, M.; Hvass, U.; Couetil, J.-P.; Henin, D.; Michel, J.-B.; et al. Extracellular matrix remodelling in human aortic valve disease: The role of matrix metalloproteinases and their tissue inhibitors. Eur. Heart J. 2005, 26, 1333–1341. [Google Scholar] [CrossRef]
- Hinton, R.B.; Lincoln, J.; Deutsch, G.H.; Osinska, H.; Manning, P.B.; Benson, D.W.; Yutzey, K. Extracellular Matrix Remodeling and Organization in Developing and Diseased Aortic Valves. Circ. Res. 2006, 98, 1431–1438. [Google Scholar] [CrossRef] [Green Version]
- Wirrig, E.E.; Hinton, R.B.; Yutzey, K.E. Differential expression of cartilage and bone-related proteins in pediatric and adult diseased aortic valves. J. Mol. Cell. Cardiol. 2011, 50, 561–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eronen, M.; Peippo, M.; Hiippala, A.; Raatikka, M.; Arvio, M.; Johansson, R.; Kähkönen, M. Cardiovascular manifestations in 75 patients with Williams syndrome. J. Med. Genet. 2002, 39, 554–558. [Google Scholar] [CrossRef] [PubMed]
- Roberts, W.C.; Honig, H.S. The spectrum of cardiovascular disease in the Marfan syndrome: A clinico-morphologic study of 18 necropsy patients and comparison to 151 previously reported necropsy patients. Am. Heart J. 1982, 104, 115–135. [Google Scholar] [CrossRef]
- O’Connor, W.N.; Davis, J.B.; Geissler, R.; Cottrill, C.M.; Noonan, J.A.; Todd, E.P. Supravalvular aortic stenosis. Clinical and pathologic observations in six patients. Arch. Pathol. Lab. Med. 1985, 109, 179–185. [Google Scholar] [PubMed]
- Senior, R.M.; Griffin, G.L.; Mecham, R.P. Chemotactic activity of elastin-derived peptides. J. Clin. Investig. 1980, 66, 859–862. [Google Scholar] [CrossRef] [PubMed]
- Robinet, A.; Fahem, A.; Cauchard, J.-H.; Huet, E.; Vincent, L.; Lorimier, S.; Antonicelli, F.; Soria, C.; Crepin, M.; Hornebeck, W.; et al. Elastin-derived peptides enhance angiogenesis by promoting endothelial cell migration and tubulogenesis through upregulation of MT1-MMP. J. Cell Sci. 2005, 118, 343–356. [Google Scholar] [CrossRef] [Green Version]
- Yacoub, M.H.; Kilner, P.J.; Birks, E.J.; Misfeld, M. The aortic outflow and root: A tale of dynamism and crosstalk. Ann. Thorac. Surg. 1999, 68, S37–S43. [Google Scholar] [CrossRef]
- Anderson, R.H. Clinical anatomy of the aortic root. Heart 2000, 84, 670–673. [Google Scholar] [CrossRef] [Green Version]
- Gross, L.; Kugel, M.A. Topographic Anatomy and Histology of the Valves in the Human Heart*. Am. J. Pathol. 1931, 7, 445–474.7. [Google Scholar]
- Wagenseil, J.E.; Mecham, R.P. New insights into elastic fiber assembly. Birth Defects Res. Part C Embryo Today Rev. 2007, 81, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Kielty, C.M. Elastic fibres in health and disease. Expert Rev. Mol. Med. 2006, 8, 1–23. [Google Scholar] [CrossRef]
- Tseng, H.; Grande-Allen, K. Elastic fibers in the aortic valve spongiosa: A fresh perspective on its structure and role in overall tissue function. Acta Biomater. 2011, 7, 2101–2108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cleaver, O.; Krieg, P.A. Vascular Development. In Heart Development and Regeneration; Elsevier BV: Amsterdam, The Netherlands, 2010; pp. 487–528. [Google Scholar]
- Alexopoulos, A.; Bravou, V.; Peroukides, S.; Kaklamanis, L.; Varakis, J.; Alexopoulos, D.; Papadaki, H. Bone regulatory factors NFATc1 and Osterix in human calcific aortic valves. Int. J. Cardiol. 2010, 139, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Roberts, W.C.; Ko, J.M. Frequency by Decades of Unicuspid, Bicuspid, and Tricuspid Aortic Valves in Adults Having Isolated Aortic Valve Replacement for Aortic Stenosis, With or Without Associated Aortic Regurgitation. Circulation 2005, 111, 920–925. [Google Scholar] [CrossRef] [PubMed]
- Stewart, B.; Siscovick, D.; Lind, B.K.; Gardin, J.M.; Gottdiener, J.S.; Smith, V.E.; Kitzman, D.W.; Otto, C.M. Clinical Factors Associated With Calcific Aortic Valve Disease. J. Am. Coll. Cardiol. 1997, 29, 630–634. [Google Scholar] [CrossRef] [Green Version]
- Perrotta, I.; Russo, E.; Camastra, C.; Filice, G.; Di Mizio, G.; Colosimo, F.; Ricci, P.; Tripepi, S.; Amorosi, A.; Triumbari, F.; et al. New evidence for a critical role of elastin in calcification of native heart valves: Immunohistochemical and ultrastructural study with literature review. Histopathology 2011, 59, 504–513. [Google Scholar] [CrossRef]
- Aikawa, E.; Aikawa, M.; Libby, P.; Figueiredo, J.-L.; Rusanescu, G.; Iwamoto, Y.; Fukuda, D.; Kohler, R.H.; Shi, G.-P.; Jaffer, F.A.; et al. Arterial and Aortic Valve Calcification Abolished by Elastolytic Cathepsin S Deficiency in Chronic Renal Disease. Circulation 2009, 119, 1785–1794. [Google Scholar] [CrossRef] [Green Version]
- Jain, D.; Dietz, H.C.; Oswald, G.L.; Maleszewski, J.J.; Halushka, M.K. Causes and histopathology of ascending aortic disease in children and young adults. Cardiovasc. Pathol. 2011, 20, 15–25. [Google Scholar] [CrossRef] [Green Version]
- Holbrook, K.A.; Byers, P.H. Structural abnormalities in the dermal collagen and elastic matrix from the skin of patients with inherited connective tissue disorders. J. Investig. Dermatol. 1982, 79, 7s–16s. [Google Scholar] [CrossRef]
- Balguid, A.; Driessen, N.J.; Mol, A.; Schmitz, J.P.; Verheyen, F.; Bouten, C.V.; Baaijens, F.P. Stress related collagen ultrastructure in human aortic valves—implications for tissue engineering. J. Biomech. 2008, 41, 2612–2617. [Google Scholar] [CrossRef]
- Wynn, T.A. Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J. Clin. Investig. 2007, 117, 524–529. [Google Scholar] [CrossRef] [Green Version]
- Otto, C.; Kuusisto, J.; Reichenbach, D.D.; Gown, A.M.; O’Brien, K. Characterization of the early lesion of ’degenerative’ valvular aortic stenosis. Histological and immunohistochemical studies. Circulation 1994, 90, 844–853. [Google Scholar] [CrossRef] [Green Version]
- Mohler, E.R.; Nichols, R.; Harvey, W.P.; Sheridan, M.J.; Waller, B.F. Development and progression of aortic valve stenosis: Atherosclerosis risk factors-a causal relationship? a clinical morphologic study. Clin. Cardiol. 1991, 14, 995–999. [Google Scholar] [CrossRef]
- Doetschman, T.; Barnett, J.V.; Runyan, R.; Camenisch, T.D.; Heimark, R.L.; Granzier, H.L.; Conway, S.J.; Azhar, M. Transforming growth factor beta signaling in adult cardiovascular diseases and repair. Cell Tissue Res. 2011, 347, 203–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markwald, R.R.; Norris, R.A.; Moreno-Rodriguez, R.; Levine, R.A. Developmental basis of adult cardiovascular diseases: Valvular heart diseases. Ann. N. Y. Acad. Sci. 2010, 1188, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Gendron, N.; Rosa, M.; Blandinieres, A.; Sottejeau, Y.; Rossi, E.; Van Belle, E.; Idelcadi, S.; Lecourt, S.; Vincentelli, A.; Cras, A.; et al. Human Aortic Valve Interstitial Cells Display Proangiogenic Properties During Calcific Aortic Valve Disease. Arter. Thromb. Vasc. Biol. 2020, 41, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Arevalos, C.A.; Berg, J.M.; Nguyen, J.M.V.; Godfrey, E.L.; Iriondo, C.; Grande-Allen, K.J.; Nguyen, J.M.V. Valve Interstitial Cells Act in a Pericyte Manner Promoting Angiogensis and Invasion by Valve Endothelial Cells. Ann. Biomed. Eng. 2016, 44, 2707–2723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katsi, V.; Magkas, N.; Antonopoulos, A.; Trantalis, G.; Toutouzas, K.; Tousoulis, D. Aortic valve: Anatomy and structure and the role of vasculature in the degenerative process. Acta Cardiol. 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Antonicelli, F.; Bellon, G.; Debelle, L.; Hornebeck, W. Elastin-elastases and inflamm-aging. Curr. Top Dev. Biol. 2007, 79, 99–155. [Google Scholar] [PubMed]
- Pezet, M.; Jacob, M.-P.; Escoubet, B.; Gheduzzi, D.; Tillet, E.; Perret, P.; Huber, P.; Quaglino, D.; Vranckx, R.; Li, D.Y.; et al. Elastin Haploinsufficiency Induces Alternative Aging Processes in the Aorta. Rejuvenation Res. 2008, 11, 97–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jacob, M.P. Extracellular matrix remodeling and matrix metalloproteinases in the vascular wall during aging and in pathological conditions. Biomed. Pharmacother. 2003, 57, 195–202. [Google Scholar] [CrossRef]
- Mariko, B.; Pezet, M.; Escoubet, B.; Bouillot, S.; Andrieu, J.-P.; Starcher, B.; Quaglino, D.; Jacob, M.-P.; Huber, P.; Ramirez, F.; et al. Fibrillin-1 genetic deficiency leads to pathological ageing of arteries in mice. J. Pathol. 2011, 224, 33–44. [Google Scholar] [CrossRef] [Green Version]
- Karnik, S.K.; Brooke, B.S.; Bayes-Genis, A.; Sorensen, L.; Wythe, J.D.; Schwartz, R.S.; Keating, M.T.; Li, D.Y. A critical role for elastin signaling in vascular morphogenesis and disease. Development 2003, 130, 411–423. [Google Scholar] [CrossRef] [Green Version]
- Sullivan, K.M.; Bissonnette, R.; Yanagisawa, H.; Hussain, S.N.; Davis, E.C. Fibulin-5 functions as an endogenous angiogenesis inhibitor. Lab. Investig. 2007, 87, 818–827. [Google Scholar] [CrossRef]
- Long, M.M.; King, V.J.; Prasad, K.U.; Freeman, B.A.; Urry, D.W. Elastin repeat peptides as chemoattractants for bovine aortic endothelial cells. J. Cell. Physiol. 1989, 140, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Hollinger, J.O.; Schmitz, J.P.; Yaskovich, R.; Long, M.M.; Prasad, K.U.; Urry, D.W. A synthetic polypentapeptide of elastin for initiating calcification. Calcif. Tissue Int. 1988, 42, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Cocciolone, A.J.; Johnson, E.O.; Shao, J.-Y.; Wagenseil, J.E. Elastic Fiber Fragmentation Increases Transmural Hydraulic Conductance and Solute Transport in Mouse Arteries. J. Biomech. Eng. 2018, 141, 021013. [Google Scholar] [CrossRef] [PubMed]
- Thubrikar, M.J.; Aouad, J.; Nolan, S.P. Patterns of calcific deposits in operatively excised stenotic or purely regurgitant aortic valves and their relation to mechanical stress. Am. J. Cardiol. 1986, 58, 304–308. [Google Scholar] [CrossRef]
- Balachandran, K.; Sucosky, P.; Jo, H.; Yoganathan, A.P. Elevated cyclic stretch alters matrix remodeling in aortic valve cusps: Implications for degenerative aortic valve disease. Am. J. Physiol. Circ. Physiol. 2009, 296, H756–H764. [Google Scholar] [CrossRef] [PubMed]
- Grande, K.J.; Cochran, R.P.; Reinhall, P.G.; Kunzelman, K.S. Stress Variations in the Human Aortic Root and Valve: The Role of Anatomic Asymmetry. Ann. Biomed. Eng. 1998, 26, 534–545. [Google Scholar] [CrossRef]
- Peacock, J.A. An in vitro study of the onset of turbulence in the sinus of Valsalva. Circ. Res. 1990, 67, 448–460. [Google Scholar] [CrossRef] [Green Version]
- Woo, K.V.; Qu, X.; Babaev, V.R.; Linton, M.F.; Guzman, R.J.; Fazio, S.; Baldwin, H.S. Tie1 attenuation reduces murine atherosclerosis in a dose-dependent and shear stress–specific manner. J. Clin. Investig. 2011, 121, 1624–1635. [Google Scholar] [CrossRef]
- Hinton, R.B.; Adelman-Brown, J.; Witt, S.; Krishnamurthy, V.K.; Osinska, H.; Sakthivel, B.; James, J.F.; Li, D.Y.; Narmoneva, D.A.; Mecham, R.P.; et al. Elastin Haploinsufficiency Results in Progressive Aortic Valve Malformation and Latent Valve Disease in a Mouse Model. Circ. Res. 2010, 107, 549–557. [Google Scholar] [CrossRef] [Green Version]
- Krishnamurthy, V.K.; Opoka, A.M.; Kern, C.B.; Guilak, F.; Narmoneva, D.A.; Hinton, R.B. Maladaptive matrix remodeling and regional biomechanical dysfunction in a mouse model of aortic valve disease. Matrix Biol. 2012, 31, 197–205. [Google Scholar] [CrossRef] [Green Version]
- Votteler, M.; Berrio, D.A.; Horke, A.; Sabatier, L.; Reinhardt, D.P.; Nsair, A.; Aikawa, E.; Schenke-Layland, K. Elastogenesis at the onset of human cardiac valve development. Development 2013, 140, 2345–2353. [Google Scholar] [CrossRef] [Green Version]
- Munjal, C.; Opoka, A.; Osinska, H.; James, J.F.; Bressan, G.M.; Hinton, R.B. TGF-Beta Mediates Early Angiogenesis and Latent Fibrosis in an Emilin1-Deficient Model of Aortic Valve Disease. Dis. Models Mech. 2014, 7, 987–996. [Google Scholar] [CrossRef] [Green Version]
- Hanada, K.; Vermeij, M.; Garinis, G.A.; De Waard, M.C.; Kunen, M.G.; Myers, L.; Maas, A.; Duncker, D.J.; Meijers, C.; Dietz, H.C.; et al. Perturbations of Vascular Homeostasis and Aortic Valve Abnormalities in Fibulin-4 Deficient Mice. Circ. Res. 2007, 100, 738–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ng, C.M.; Cheng, A.; Myers, L.A.; Martinez-Murillo, F.; Jie, C.; Bedja, D.; Gabrielson, K.L.; Hausladen, J.M.W.; Mecham, R.P.; Judge, D.P.; et al. TGF-beta-dependent pathogenesis of mitral valve prolapse in a mouse model of Marfan syndrome. J. Clin. Investig. 2004, 114, 1586–1592. [Google Scholar] [CrossRef] [Green Version]
- Dor, Y.; Camenisch, T.D.; Itin, A.; Fishman, G.I.; McDonald, J.A.; Carmeliet, P.; Keshet, E. A novel role for VEGF in endocardial cushion formation and its potential contribution to congenital heart defects. Development 2001, 128, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Snider, P.; Hinton, R.B.; Moreno-Rodriguez, R.A.; Wang, J.; Rogers, R.; Lindsley, A.; Li, F.; Ingram, D.A.; Menick, D.; Field, L.; et al. Periostin Is Required for Maturation and Extracellular Matrix Stabilization of Noncardiomyocyte Lineages of the Heart. Circ. Res. 2008, 102, 752–760. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, M.; Yuasa, S.; Matsumura, K.; Kimura, K.; Shiomi, T.; Kimura, N.; Shukunami, C.; Okada, Y.; Mukai, M.; Shin, H.; et al. Chondromodulin-I maintains cardiac valvular function by preventing angiogenesis. Nat. Med. 2006, 12, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Barolet, A.W.; Nili, N.; Cheema, A.; Robinson, R.; Natarajan, M.K.; O’Blenes, S.; Li, J.; Eskandarian, M.R.; Sparkes, J.; Rabinovitch, M.; et al. Arterial Elastase Activity After Balloon Angioplasty and Effects of Elafin, an Elastase Inhibitor. Arter. Thromb. Vasc. Biol. 2001, 21, 1269–1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otto, C.; Lind, B.K.; Kitzman, D.W.; Gersh, B.J.; Siscovick, D.S. Association of Aortic-Valve Sclerosis with Cardiovascular Mortality and Morbidity in the Elderly. N. Engl. J. Med. 1999, 341, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Schoen, F.J. Mechanisms of Function and Disease of Natural and Replacement Heart Valves. Annu. Rev. Pathol. Mech. Dis. 2012, 7, 161–183. [Google Scholar] [CrossRef]
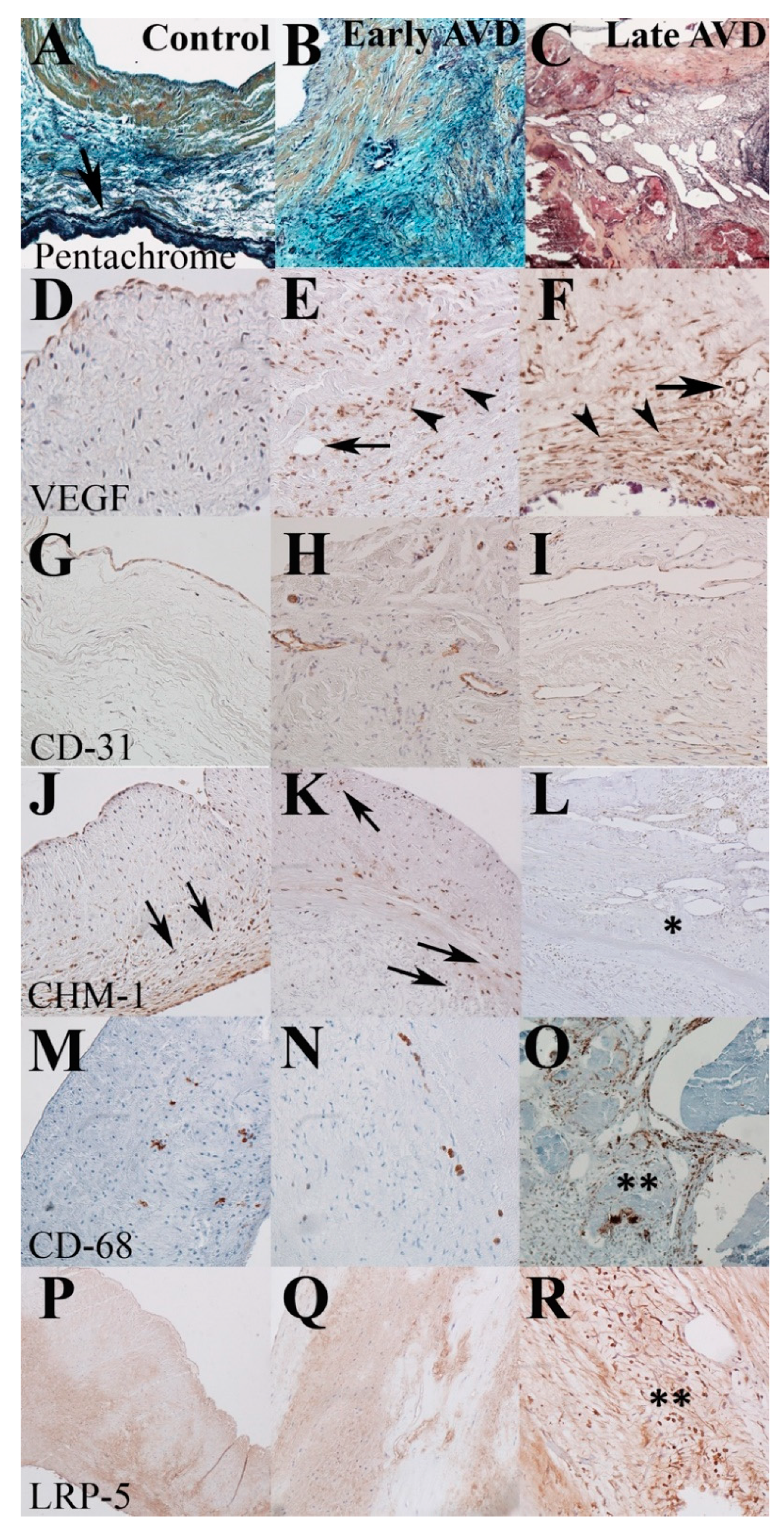
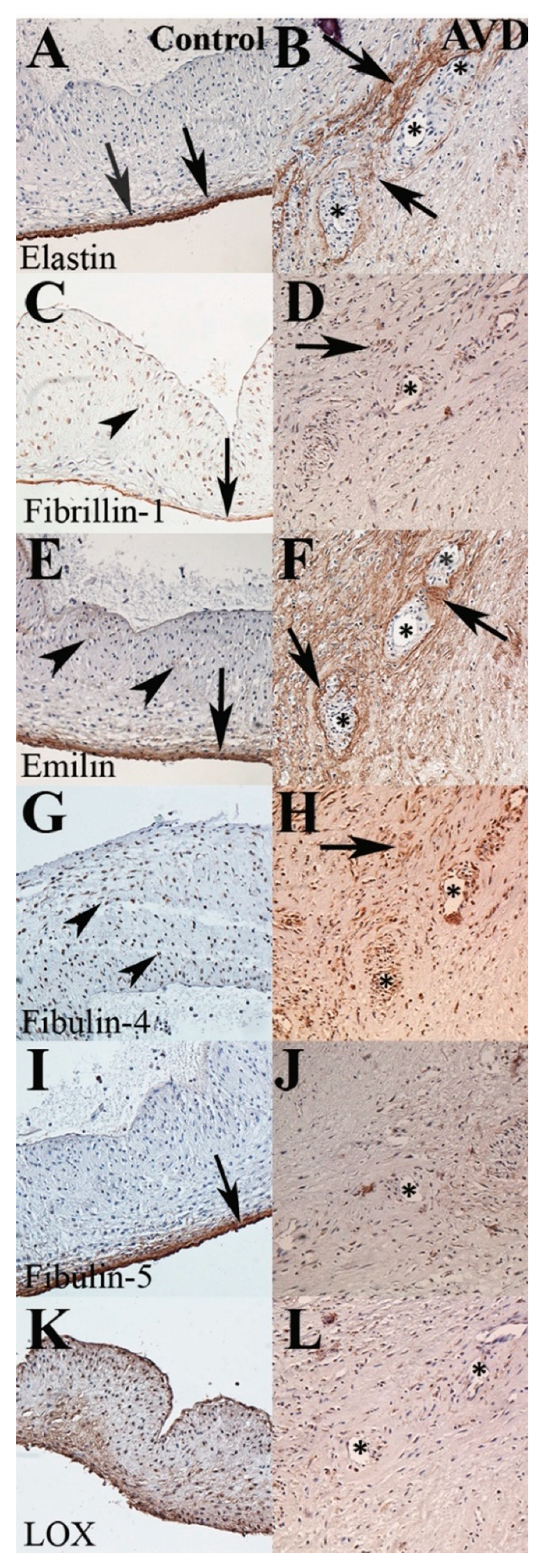


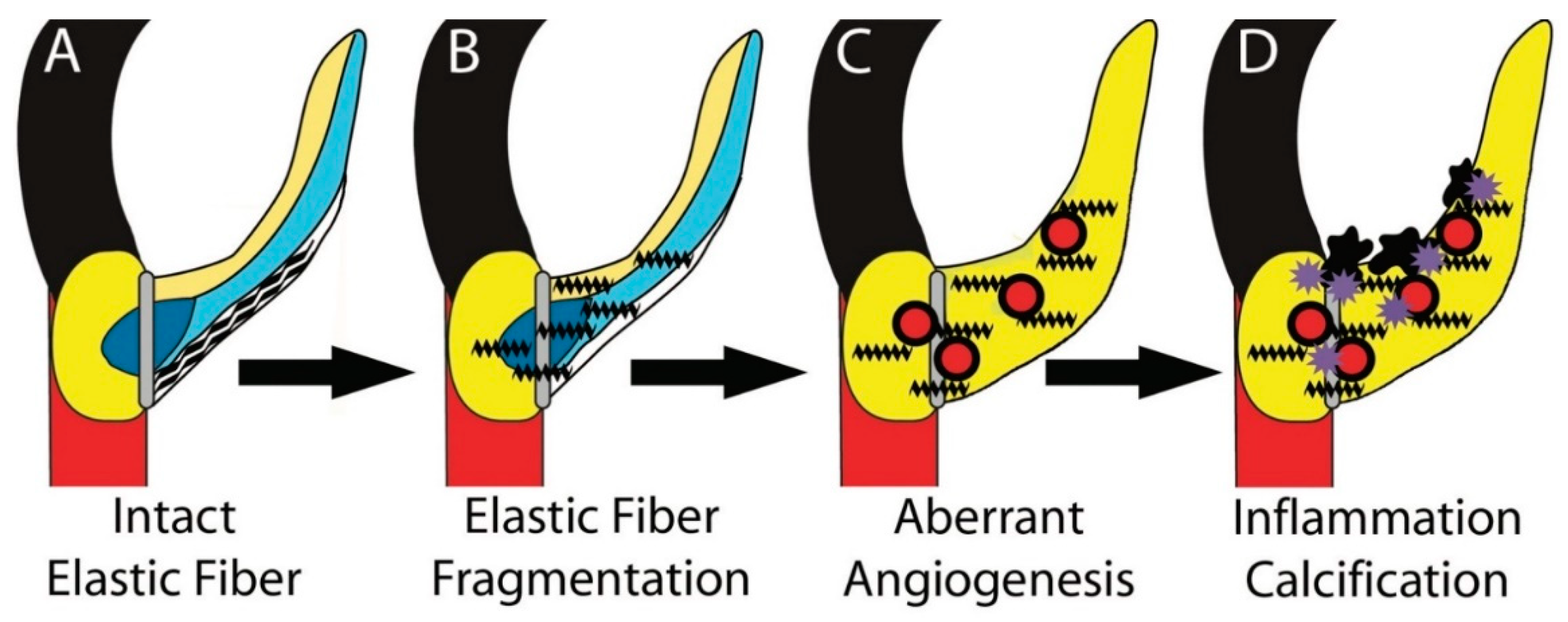
| Early-Onset AVD | Late-Onset AVD | p | |
|---|---|---|---|
| N | 21 | 11 | - |
| Mean Age (Range) | 15 (1, 32) | 67 (44, 85) | - |
| Male (%) | 81 | 73 | NS |
| BAV (%) | 48 | 27 | 0.08 |
| CAD (%) | 0 | 45 | <0.003 |
| HT (%) | 10 | 55 | <0.002 |
| TAA (%) | 10 | 18 | NS |
| DM (%) | 5 | 36 | 0.08 |
| CKD (%) | 0 | 9 | <0.001 |
| Microvessels | Arterioles | Total (Vessels/10×) | |
|---|---|---|---|
| Control | 0 | 0 | 0 |
| Early AVD | 0.7 | 0.2 | 0.9 |
| Late AVD | 3.7 | 1.9 | 5.6 |
| VEGF-A | CD-68 | |||||
|---|---|---|---|---|---|---|
| Score | Control | Early AVD | Late AVD | Control | Early AVD | Late AVD |
| 0 (0–10% + cells) | 100% | 0% | 18% | 100% | 73% | 20% |
| 1 (10–35% + cells) | 0% | 21% | 27% | 0% | 18% | 40% |
| 2 (35–70% + cells) | 0% | 57% | 36% | 0% | 9% | 40% |
| 3 (>70% + cells) | 0% | 21% | 18% | 0% | 0% | 0% |
| Mean (SD) | 0 ± 0 | 2.0 ± 0.7 * | 1.6 ± 1.0 * | 0 ± 0 | 0.4 ± 0.7 | 1.3 ± 0.8 *,# |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hinton, R.B.; Juraszek, A.L.; Opoka, A.M.; Landis, B.J.; Smith, J.M.; Mecham, R.P.; Bove, K.E. Early Aberrant Angiogenesis Due to Elastic Fiber Fragmentation in Aortic Valve Disease. J. Cardiovasc. Dev. Dis. 2021, 8, 75. https://0-doi-org.brum.beds.ac.uk/10.3390/jcdd8070075
Hinton RB, Juraszek AL, Opoka AM, Landis BJ, Smith JM, Mecham RP, Bove KE. Early Aberrant Angiogenesis Due to Elastic Fiber Fragmentation in Aortic Valve Disease. Journal of Cardiovascular Development and Disease. 2021; 8(7):75. https://0-doi-org.brum.beds.ac.uk/10.3390/jcdd8070075
Chicago/Turabian StyleHinton, Robert B., Amy L. Juraszek, Amy M. Opoka, Benjamin J. Landis, J. Michael Smith, Robert P. Mecham, and Kevin E. Bove. 2021. "Early Aberrant Angiogenesis Due to Elastic Fiber Fragmentation in Aortic Valve Disease" Journal of Cardiovascular Development and Disease 8, no. 7: 75. https://0-doi-org.brum.beds.ac.uk/10.3390/jcdd8070075





