Non-Coding RNAs in Retinoic Acid as Differentiation and Disease Drivers
Abstract
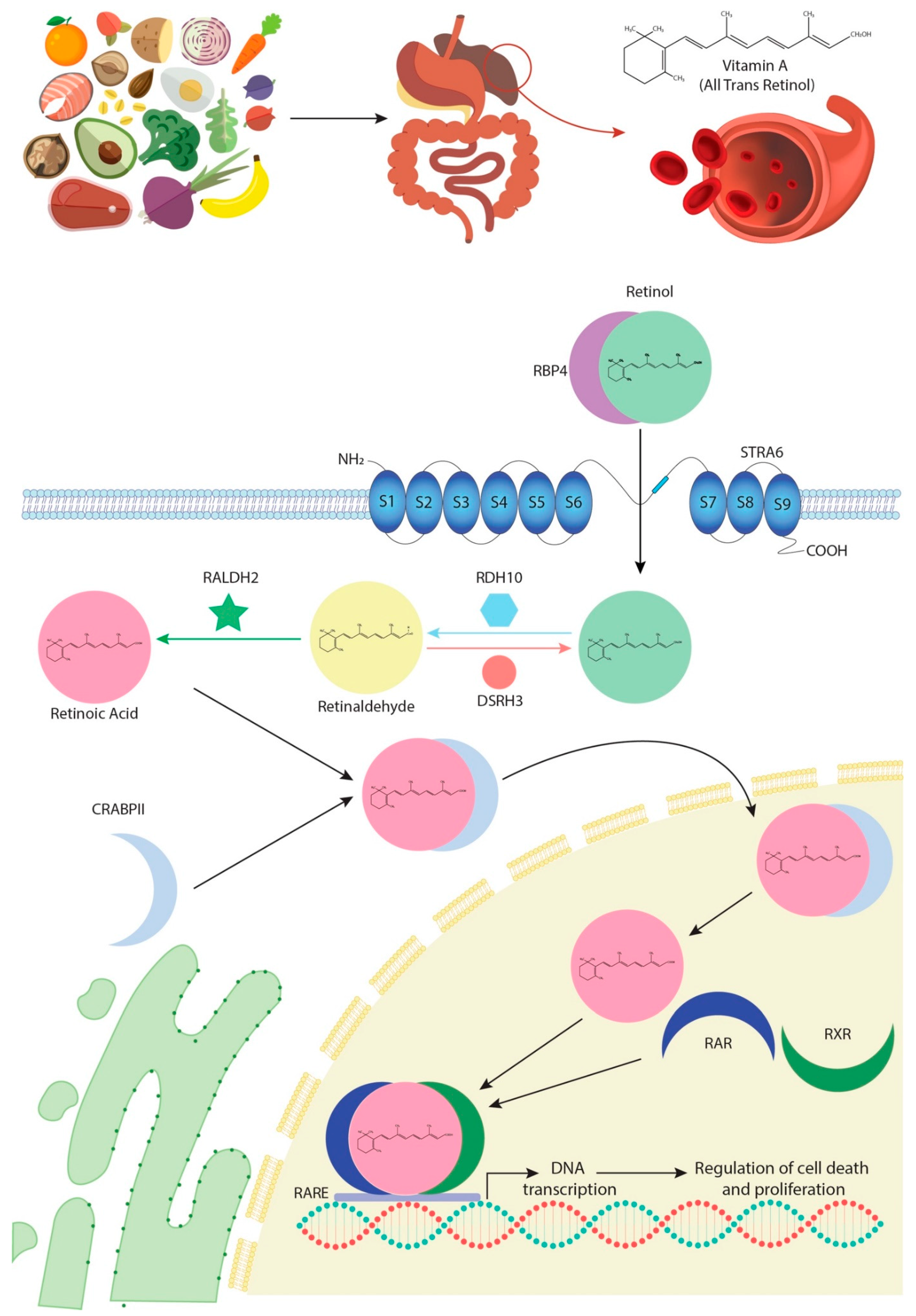
:1. Retinoic Acid Biogenesis
2. Classification and Function of ncRNAs
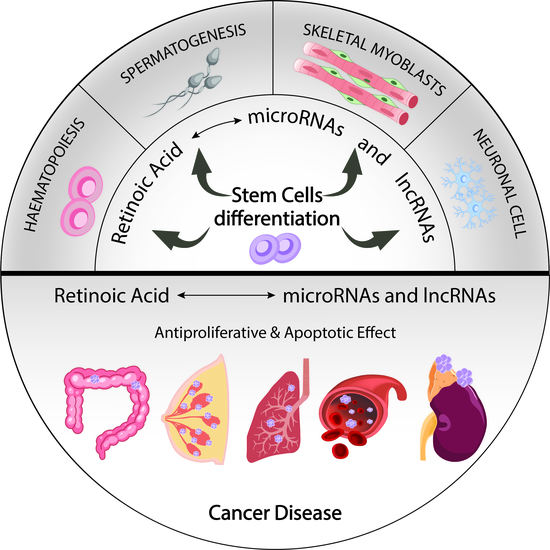
3. Role of MicroRNAs as Differentiation Drivers Mediated by RA
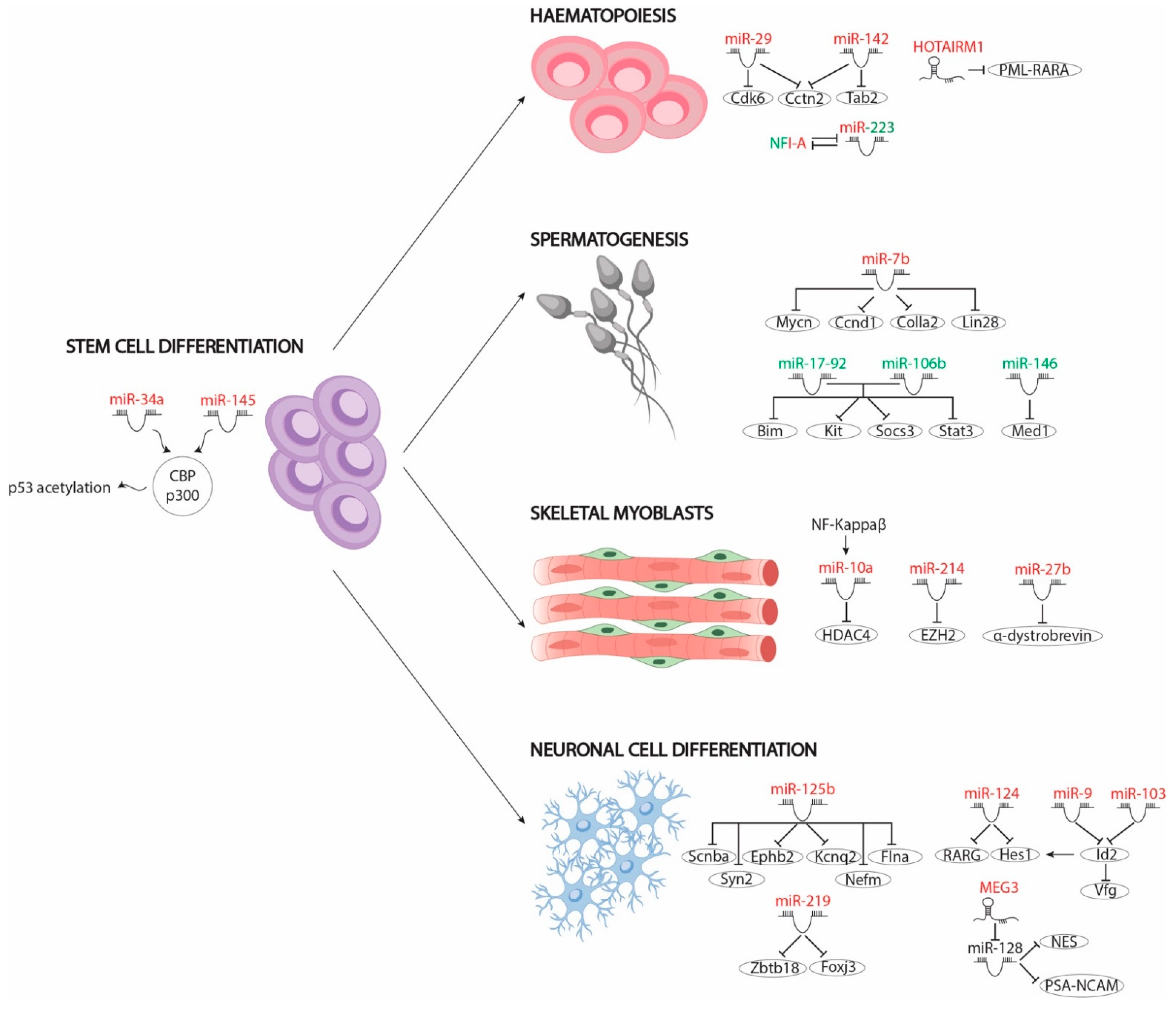
3.1. MicroRNAs Involved in RA Signalling That Mediate Cell Differentiation of Pluripotent Stem Cells
3.2. MiRNAs in RA Signalling-Mediated Neuronal Cell Differentiation
3.3. MiRNAs Involved in RA Signalling-Mediated Haematopoietic Differentiation
3.4. MiRNAs Involved in RA Signalling-Mediated Skeletal Myoblast Differentiation
3.5. MiRNAs Involved in RA Signalling-Mediated Spermatogonia Differentiation
4. The Role of RA-Modulated MicroRNAs in Cancer
4.1. Protective Roles of RA-Mediated MiRNAs in Breast Cancer
4.2. Protective Roles of RNA-Mediated MiRNAs in Colon Cancer
4.3. Protective Roles of RA-Mediated MiRNAs in Neuroblastoma
4.4. Protective Roles of RA-Mediated MiRNAs in Lung Cancer
5. Role of lncRNAs as RA Mediated Differentiation Driver
6. Role of lncRNAs in an RA-Mediated Diseases
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ulven, S.M.; Gundersen, T.E.; Weedon, M.S.; Landaas, V.Ø.; Sakhi, A.K.; Fromm, S.H.; Geronimo, B.A.; Moskaug, J.O.; Blomhoff, R. Identification of Endogenous Retinoids, Enzymes, Binding Proteins, and Receptors during Early Postimplantation Development in Mouse: Important Role of Retinal Dehydrogenase Type 2 in Synthesis of All-trans-Retinoic Acid. Dev. Biol. 2000, 220, 379–391. [Google Scholar] [CrossRef] [Green Version]
- Marlétaz, F.; Linda, Z.H.; Laudet, L.; Schubert, M. Retinoic acid signaling and the evolution of chordates. Int. J. Bio. Sci. 2006, 2, 38–47. [Google Scholar] [CrossRef]
- Das, B.C.; Thapa, P.; Verma, A.; Ray, S.K.; Evans, T.; Karki, R.; Das, S.; Mahapatra, S.; Liu, T.-C.; Torregroza, I.; et al. Retinoic acid signaling pathways in development and diseases. Bioorg. Med. Chem. 2014, 22, 673–683. [Google Scholar] [CrossRef] [Green Version]
- Al Tanoury, Z.; Piskunov, A.; Rochette-Egly, C. Vitamin A and retinoid signaling: Genomic and nongenomic effects thematic review series: Fat-soluble vitamins: Vitamin A. J. Lipid Res. 2013, 54, 1761–1775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khillan, J.S. Vitamin A/Retinol and Maintenance of Pluripotency of Stem Cells. Nutrients 2014, 6, 1209–1222. [Google Scholar] [CrossRef] [Green Version]
- Kane, M.A.; Folias, A.E.; Pingitore, A.; Perri, M.; Obrochta, K.M.; Krois, C.R.; Cione, E.; Ryu, J.Y.; Napoli, J.L. Identification of 9-cis-retinoic acid as a pancreas-specific autacoid that attenuates glucose-stimulated insulin secretion. Proc. Natl. Acad. Sci. USA 2010, 107, 21884–21889. [Google Scholar] [CrossRef] [Green Version]
- Simões-Costa, M.S.; Azambuja, A.P.; Xavier-Neto, J. The search for non-chordate retinoic acid signaling: Lessons from chordates. J. Exp. Zool. B Mol. Dev. Evol. 2008, 310, 54–72. [Google Scholar] [CrossRef]
- Harrison, E.H. MECHANISMS OF DIGESTION AND ABSORPTION OF DIETARY VITAMIN A. Annu. Rev. Nutr. 2005, 25, 87–103. [Google Scholar] [CrossRef]
- Kawaguchi, R.; Yu, J.; Honda, J.; Hu, J.; Whitelegge, J.; Ping, P.; Wiita, P.; Bok, D.; Sun, H. A Membrane Receptor for Retinol Binding Protein Mediates Cellular Uptake of Vitamin A. Sci. 2007, 315, 820–825. [Google Scholar] [CrossRef]
- Sandell, L.L.; Sanderson, B.W.; Moiseyev, G.; Johnson, T.; Mushegian, A.; Young, K.; Rey, J.-P.; Ma, J.-X.; Staehling-Hampton, K.; Trainor, P.A. RDH10 is essential for synthesis of embryonic retinoic acid and is required for limb, craniofacial, and organ development. Genes Dev. 2007, 21, 1113–1124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandell, L.L.; Lynn, M.L.; Inman, K.E.; McDowell, W.; Trainor, P.A. RDH10 oxidation of Vitamin A is a critical control step in synthesis of retinoic acid during mouse embryogenesis. PLoS ONE 2012, 7, e30698. [Google Scholar] [CrossRef]
- Belyaeva, O.V.; Adams, M.K.; Wu, L.; Kedishvili, N.Y. The antagonistically bifunctional retinoid oxidoreductase complex is required for maintenance of all-trans-retinoic acid homeostasis. J. Biol. Chem. 2017, 292, 5884–5897. [Google Scholar] [CrossRef] [Green Version]
- Duester, G. Involvement of Alcohol Dehydrogenase, Short-Chain Dehydrogenase/Reductase, Aldehyde Dehydrogenase, and Cytochrome P450 in the Control of Retinoid Signaling by Activation of Retinoic Acid Synthesis†. Biochemistry 1996, 35, 12221–12227. [Google Scholar] [CrossRef]
- Niederreither, K.; McCaffery, P.; Dräger, U.C.; Chambon, P.; Dollé, P. Restricted expression and retinoic acid-induced downregulation of the retinaldehyde dehydrogenase type 2 (RALDH-2) gene during mouse development. Mech. Dev. 1997, 62, 67–78. [Google Scholar] [CrossRef]
- Budhu, A.S.; Noy, N. Direct Channeling of Retinoic Acid between Cellular Retinoic Acid-Binding Protein II and Retinoic Acid Receptor Sensitizes Mammary Carcinoma Cells to Retinoic Acid-Induced Growth Arrest. Mol. Cell. Biol. 2002, 22, 2632–2641. [Google Scholar] [CrossRef] [Green Version]
- Delva, L.; Bastie, J.-N.; Rochette-Egly, C.; Kraïba, R.; Balitrand, N.; Despouy, G.; Chambon, P.; Chomienne, C. Physical and Functional Interactions between Cellular Retinoic Acid Binding Protein II and the Retinoic Acid-Dependent Nuclear Complex. Mol. Cell. Biol. 1999, 19, 7158–7167. [Google Scholar] [CrossRef] [Green Version]
- Dong, D.; Ruuska, S.E.; Levinthal, D.J.; Noy, N. Distinct Roles for Cellular Retinoic Acid-binding Proteins I and II in Regulating Signaling by Retinoic Acid. J. Biol. Chem. 1999, 274, 23695–23698. [Google Scholar] [CrossRef] [Green Version]
- Takase, S.; Ong, D.; Chytil, F. Transfer of retinoic acid from its complex with cellular retinoic acid-binding protein to the nucleus. Arch. Biochem. Biophys. 1986, 247, 328–334. [Google Scholar] [CrossRef]
- Venepally, P.; Reddy, L.G.; Sani, B.P. Analysis of the Effects of CRABP I Expression on the RA-Induced Transcription Mediated by Retinoid Receptors†. Biochemistry 1996, 35, 9974–9982. [Google Scholar] [CrossRef]
- Napoli, J.L. Cellular retinoid binding-proteins, CRBP, CRABP, FABP5: Effects on retinoid metabolism, function and related diseases. Pharmacol. Ther. 2017, 173, 19–33. [Google Scholar] [CrossRef] [Green Version]
- Duong, V.; Rochette-Egly, C. The molecular physiology of nuclear retinoic acid receptors. From health to disease. Biochim. et Biophys. Acta 2011, 1812, 1023–1031. [Google Scholar] [CrossRef] [Green Version]
- Samarut, E.; Rochette-Egly, C. Nuclear retinoic acid receptors: Conductors of the retinoic acid symphony during development. Mol. Cell. Endocrinol. 2012, 348, 348–360. [Google Scholar] [CrossRef] [PubMed]
- Rochette-Egly, C.; Germain, P. Dynamic and combinatorial control of gene expression by nuclear retinoic acid receptors (RARs). Nucl. Recept. Signal. 2009, 7, e005. [Google Scholar] [CrossRef] [PubMed]
- Le Maire, A.; Teyssier, C.; Balaguer, P.; Bourguet, W.; Germain, P. Regulation of RXR-RAR Heterodimers by RXR- and RAR-Specific Ligands and Their Combinations. Cells 2019, 8, 1392. [Google Scholar] [CrossRef] [Green Version]
- Glass, C.K. Differential recognition of target genes by nuclear receptor monomers, dimers, and heterodimers. Endocr. Rev. 1994, 15, 391–407. [Google Scholar] [CrossRef] [PubMed]
- Moutier, E.; Ye, T.; Choukrallah, M.-A.; Urban, S.; Osz, J.; Chatagnon, A.; Delacroix, L.; Langer, D.; Rochel, N.; Moras, D.; et al. Retinoic Acid Receptors Recognize the Mouse Genome through Binding Elements with Diverse Spacing and Topology. J. Biol. Chem. 2012, 287, 26328–26341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bastien, J.; Adam-Stitah, S.; Riedl, T.; Egly, J.M.; Chambon, P.; Rochette-Egly, C. TFIIH interacts with the retinoic acid receptor γ and phosphorylates its AF-1-activating domain through cdk7. J. Biol. Chem. 2000, 275, 21896–21904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bour, G.; Lalevee, S.; Rochette-Egly, C. Protein kinases and the proteasome join in the combinatorial control of transcription by nuclear retinoic acid receptors. Trends Cell Biol. 2007, 17, 302–309. [Google Scholar] [CrossRef]
- Rochette-Egly, C.; Adam, S.; Rossignol, M.; Egly, J.-M.; Chambon, P. Stimulation of RARα Activation Function AF-1 through Binding to the General Transcription Factor TFIIH and Phosphorylation by CDK7. Cell 1997, 90, 97–107. [Google Scholar] [CrossRef] [Green Version]
- Park, S.W.; Nhieu, J.; Persaud, S.D.; Miller, M.C.; Xia, Y.; Lin, Y.-W.; Lin, Y.-L.; Kagechika, H.; Mayo, K.H.; Wei, L.-N. A new regulatory mechanism for Raf kinase activation, retinoic acid-bound Crabp1. Sci. Rep. 2019, 9, 10929. [Google Scholar] [CrossRef] [Green Version]
- Reijntjes, S.; Blentic, A.; Gale, E.; Maden, M. The control of morphogen signalling: Regulation of the synthesis and catabolism of retinoic acid in the developing embryo. Dev. Biol. 2005, 285, 224–237. [Google Scholar] [CrossRef] [Green Version]
- Abu-Abed, S.; Dollé, P.; Metzger, D.; Beckett, B.; Chambon, P.; Petkovich, M. The retinoic acid-metabolizing enzyme, CYP26A1, is essential for normal hindbrain patterning, vertebral identity, and development of posterior structures. Genes Dev. 2001, 15, 226–240. [Google Scholar] [CrossRef] [Green Version]
- Emoto, Y.; Wada, H.; Okamoto, H.; Kudo, A.; Imai, Y. Retinoic acid-metabolizing enzyme Cyp26a1 is essential for determining territories of hindbrain and spinal cord in zebrafish. Dev. Biol. 2005, 278, 415–427. [Google Scholar] [CrossRef] [Green Version]
- Chithalen, J.V.; Luu, L.; Petkovich, M.; Jones, G. HPLC-MS/MS analysis of the products generated from all-trans-retinoic acid using recombinant human CYP26A. J. Lipid Res. 2002, 43, 1133–1142. [Google Scholar] [CrossRef] [Green Version]
- Idres, N.; Marill, J.; Flexor, M.A.; Chabot, G.G. Activation of Retinoic Acid Receptor-dependent Transcription by All-trans-retinoic Acid Metabolites and Isomers. J. Biol. Chem. 2002, 277, 31491–31498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pijnappel, W.W.M.; Hendriks, H.F.J.; Folkers, G.E.; Brink, C.E.V.D.; Dekker, E.J.; Edelenbosch, C.; Van Der Saag, P.T.; Durston, A.J. The retinoid ligand 4-oxo-retinoic acid is a highly active modulator of positional specification. Nat. Cell Biol. 1993, 366, 340–344. [Google Scholar] [CrossRef] [PubMed]
- White, R.J.; Nie, Q.; Lander, A.D.; Schilling, T.F. Complex Regulation of cyp26a1 Creates a Robust Retinoic Acid Gradient in the Zebrafish Embryo. PLoS Biol. 2007, 5, e304. [Google Scholar] [CrossRef] [PubMed]
- Shimozono, S.; Iimura, T.; Kitaguchi, T.; Higashijima, S.-I.; Miyawaki, A. Visualization of an endogenous retinoic acid gradient across embryonic development. Nat. Cell Biol. 2013, 496, 363–366. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Meno, C.; Fujii, H.; Nishino, J.; Shiratori, H.; Saijoh, Y.; Rossant, J.; Hamada, H. The retinoic acid-inactivating enzyme CYP26 is essential for establishing an uneven distribution of retinoic acid along the anterio-posterior axis within the mouse embryo. Genes Dev. 2001, 15, 213–225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gurdon, J.B.; Standley, H.; Dyson, S.; Butler, K.; Langon, T.; Ryan, K.; Stennard, F.; Shimizu, K.; Zorn, A. Single cells can sense their position in a morphogen gradient. Development 1999, 126, 5309–5317. [Google Scholar]
- Nagatomo, K.-I.; Fujiwara, S. Expression of Raldh2, Cyp26 and Hox-1 in normal and retinoic acid-treated Ciona intestinalis embryos. Gene Expr. Patterns 2003, 3, 273–277. [Google Scholar] [CrossRef]
- Hochgreb, T.; Linhares, V.L.; Menezes, D.C.; Sampaio, A.C.; Yan, C.Y.I.; Cardoso, W.V.; Rosenthal, N.; Xavier-Neto, J. A caudorostral wave of RALDH2 conveys anteroposterior information to the cardiac field. Development 2003, 130, 5363–5374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niederreither, K.; Vermot, J.; Le Roux, I.; Schuhbaur, B.; Chambon, P.; Dollé, P. The regional pattern of retinoic acid synthesis by RALDH2 is essential for the development of posterior pharyngeal arches and the enteric nervous system. Development 2003, 130, 2525–2534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Begemann, G.; Schilling, T.F.; Rauch, G.J.; Geisler, R.; Ingham, P.W. The zebrafish neckless mutation reveals a requirement for raldh2 in mesodermal signals that pattern the hindbrain. Development 2001, 128, 3081–3094. [Google Scholar]
- Carninci, P.; Kasukawa, T.; Katayama, S.; Gough, J.; Frith, M.C.; Maeda, N.; Oyama, R.; Ravasi, T.; Lenhard, B.; Wells, C.; et al. The transcriptional landscape of the mammalian genome. Science 2005, 309, 1559–1563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrow, J.; Frankish, A.; Gonzalez, J.M.; Tapanari, E.; Diekhans, M.; Kokocinski, F.; Aken, B.L.; Barrell, D.; Zadissa, A.; Searle, S.; et al. GENCODE: The reference human genome annotation for The ENCODE Project. Genome Res. 2012, 22, 1760–1774. [Google Scholar] [CrossRef] [Green Version]
- García-Padilla, C.; Aránega, A.; Franco, D. The role of long non-coding RNAs in cardiac development and disease. AIMS Genet. 2018, 5, 124–140. [Google Scholar] [CrossRef]
- Expósito-Villén, A.; Aránega, A.E.; Franco, D. Functional Role of Non-Coding RNAs during Epithelial-To-Mesenchymal Transition. Non-Coding RNA 2018, 4, 14. [Google Scholar] [CrossRef] [Green Version]
- Shi, X.; Sun, M.; Liu, H.; Yao, Y.; Song, Y. Long non-coding RNAs: A new frontier in the study of human diseases. Cancer Lett. 2013, 339, 159–166. [Google Scholar] [CrossRef]
- Statello, L.; Guo, C.-J.; Chen, L.-L.; Huarte, M. Gene regulation by long non-coding RNAs and its biological functions. Nat. Rev. Mol. Cell Biol. 2021, 22, 96–118. [Google Scholar] [CrossRef]
- Scott, M.S.; Ono, M. From snoRNA to miRNA: Dual function regulatory non-coding RNAs. Biochimie 2011, 93, 1987–1992. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Bajic, V.B.; Zhang, Z. On the classification of long non-coding RNAs. RNA Biol. 2013, 10, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.R.; Rameshwar, P. MicroRNA in Development and in the Progression of Cancer; Springer: New York, NY, USA, 2014. [Google Scholar]
- Lu, M.; Zhang, Q.; Deng, M.; Miao, J.; Guo, Y.; Gao, W.; Cui, Q. An Analysis of Human MicroRNA and Disease Associations. PLoS ONE 2008, 3, e3420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engreitz, J.M.; Haines, J.E.; Perez, E.M.; Munson, G.; Chen, J.; Kane, M.; McDonel, P.E.; Guttman, M.; Lander, E.S. Local regulation of gene expression by lncRNA promoters, transcription and splicing. Nat. Cell Biol. 2016, 539, 452–455. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, E.L.; Belhocine, M.; Dao, L.T.; Puthier, D.; Spicuglia, S. Functions of lncRNA in development and diseases. Med. Sci. 2014, 30, 790–796. (In French) [Google Scholar] [CrossRef] [Green Version]
- García-Padilla, C.; Domínguez, J.N.; Aránega, A.E.; Franco, D. Differential chamber-specific expression and regulation of long non-coding RNAs during cardiac development. Biochim. Biophys. Acta Gene Regul. Mech. 2019, 1862, 194435. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Lu, J.Y.; Liu, L.; Yin, Y.; Chen, C.; Han, X.; Wu, B.; Xu, R.; Liu, W.; Yuyang, L.J.; et al. Divergent lncRNAs Regulate Gene Expression and Lineage Differentiation in Pluripotent Cells. Cell Stem Cell 2016, 18, 637–652. [Google Scholar] [CrossRef]
- Zhu, L.; I Skoultchi, A. Coordinating cell proliferation and differentiation. Curr. Opin. Genet. Dev. 2001, 11, 91–97. [Google Scholar] [CrossRef]
- Yoshida, S.; Fujiwara, K.; Nishihara, H.; Kato, T.; Yashiro, T.; Kato, Y. Retinoic acid signalling is a candidate regulator of the expression of pituitary-specific transcription factor Prop1 in the developing rodent pituitary. J. Neuroendocr. 2018, 30, e12570. [Google Scholar] [CrossRef]
- Coyle, K.M.; Maxwell, S.; Thomas, M.L.; Marcato, P. Profiling of the transcriptional response to all-trans retinoic acid in breast cancer cells reveals RARE-independent mechanisms of gene expression. Sci. Rep. 2017, 7, 16684. [Google Scholar] [CrossRef] [Green Version]
- Li, A.; Zhu, X.; Brown, B.; Craft, C.M. Gene expression networks underlying retinoic acid-induced differentiation of human retinoblastoma cells. Investig. Opthalmol. Vis. Sci. 2003, 44, 996–1007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, R.; Wang, Y.; Li, R.; Chen, G. Transcriptional Factors Mediating Retinoic Acid Signals in the Control of Energy Metabolism. Int. J. Mol. Sci. 2015, 16, 14210–14244. [Google Scholar] [CrossRef] [Green Version]
- Jain, A.K.; Allton, K.; Iacovino, M.; Mahen, E.; Milczarek, R.J.; Zwaka, T.P.; Kyba, M.; Barton, M.C. p53 Regulates Cell Cycle and MicroRNAs to Promote Differentiation of Human Embryonic Stem Cells. PLoS Biol. 2012, 10, e1001268. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Xie, C.; Sun, X.; Ritchie, R.P.; Zhang, J.; Chen, Y.E. miR-10a Contributes to Retinoid Acid-induced Smooth Muscle Cell Differentiation. J. Biol. Chem. 2010, 285, 9383–9389. [Google Scholar] [CrossRef] [Green Version]
- Caretti, G.; Di Padova, M.; Micales, B.; Lyons, G.E.; Sartorelli, V. The Polycomb Ezh2 methyltransferase regulates muscle gene expression and skeletal muscle differentiation. Genes Dev. 2004, 18, 2627–2638. [Google Scholar] [CrossRef] [Green Version]
- Juan, A.H.; Kumar, R.M.; Marx, J.G.; Young, R.A.; Sartorelli, V. Mir-214-Dependent Regulation of the Polycomb Protein Ezh2 in Skeletal Muscle and Embryonic Stem Cells. Mol. Cell 2009, 36, 61–74. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Zhao, J.; Fu, B.; Yin, S.; Songna, Y.; Zhang, J.; Zhao, S.; Zhang, Y. Retinoic acid-induced upregulation of miR-219 promotes the differentiation of embryonic stem cells into neural cells. Cell Death Dis. 2017, 8, e2953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sempere, L.F.; Freemantle, S.; Pitha-Rowe, I.; Moss, E.; Dmitrovsky, E.; Ambros, V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 2004, 5, R13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janesick, A.; Wu, S.C.; Blumberg, B. Retinoic acid signaling and neuronal differentiation. Cell. Mol. Life Sci. 2015, 72, 1559–1576. [Google Scholar] [CrossRef] [Green Version]
- Su, X.; Gu, X.; Zhang, Z.; Li, W.; Wang, X. Retinoic acid receptor gamma is targeted by microRNA-124 and inhibits neurite outgrowth. Neuropharmacology 2020, 163, 107657. [Google Scholar] [CrossRef]
- Kageyama, R.; Ohtsuka, T.; Kobayashi, T. The Hes gene family: Repressors and oscillators that orchestrate embryogenesis. Development 2007, 134, 1243–1251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Yao, N.; Lu, C.-L.; Li, D.; Ma, X. Mouse microRNA-124 regulates the expression of Hes1 in P19 cells. Front. Biosci. 2010, 2, 127–132. [Google Scholar] [CrossRef] [Green Version]
- Ruzinova, M.B.; Benezra, R. Id proteins in development, cell cycle and cancer. Trends Cell Biol. 2003, 13, 410–418. [Google Scholar] [CrossRef]
- Martinsen, B.J.; Bronner-Fraser, M. Neural Crest Specification Regulated by the Helix-Loop-Helix Repressor Id2. Science 1998, 281, 988–991. [Google Scholar] [CrossRef]
- Annibali, D.; Gioia, U.; Savino, M.; Laneve, P.; Caffarelli, E.; Nasi, S. A New Module in Neural Differentiation Control: Two MicroRNAs Upregulated by Retinoic Acid, miR-9 and -103, Target the Differentiation Inhibitor ID2. PLoS ONE 2012, 7, e40269. [Google Scholar] [CrossRef] [Green Version]
- Walkley, C.R.; Yuan, Y.-D.; Chandraratna, R.A.S.; A McArthur, G. Retinoic acid receptor antagonism in vivo expands the numbers of precursor cells during granulopoiesis. Leukemia 2002, 16, 1763–1772. [Google Scholar] [CrossRef]
- Wang, X.-S.; Gong, J.-N.; Yu, J.; Wang, F.; Zhang, X.-H.; Yin, X.-L.; Tan, Z.-Q.; Luo, Z.-M.; Yang, G.-H.; Shen, C.; et al. MicroRNA-29a and microRNA-142-3p are regulators of myeloid differentiation and acute myeloid leukemia. Blood 2012, 119, 4992–5004. [Google Scholar] [CrossRef] [Green Version]
- Fazi, F.; Rosa, A.; Fatica, A.; Gelmetti, V.; De Marchis, M.L.; Nervi, C.; Bozzoni, I. A Minicircuitry Comprised of MicroRNA-223 and Transcription Factors NFI-A and C/EBPα Regulates Human Granulopoiesis. Cell 2005, 123, 819–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Haddad, M.; Notarnicola, C.; Evano, B.; El Khatib, N.; Blaquière, M.; Bonnieu, A.; Tajbakhsh, S.; Hugon, G.; Vernus, B.; Mercier, J.; et al. Retinoic acid maintains human skeletal muscle progenitor cells in an immature state. Cell. Mol. Life Sci. 2017, 74, 1923–1936. [Google Scholar] [CrossRef]
- Nakamori, M.; Takahashi, M.P. The Role of Alpha-Dystrobrevin in Striated Muscle. Int. J. Mol. Sci. 2011, 12, 1660–1671. [Google Scholar] [CrossRef] [Green Version]
- Li, N.; Tang, Y.; Liu, B.; Cong, W.; Liu, C.; Xiao, J. Retinoid acid-induced microRNA-27b-3p impairs C2C12 myoblast proliferation and differentiation by suppressing α-dystrobrevin. Exp. Cell Res. 2017, 350, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Oatley, J.M.; Brinster, R.L. Regulation of Spermatogonial Stem Cell Self-Renewal in Mammals. Annu. Rev. Cell Dev. Biol. 2008, 24, 263–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doyle, T.J.; Oudes, A.J.; Kim, K.H. Temporal profiling of rat transcriptomes in retinol-replenished vitamin A-deficient testis. Syst. Biol. Reprod. Med. 2009, 55, 145–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tong, M.-H.; Mitchell, D.; Evanoff, R.; Griswold, M.D. Expression of Mirlet7 Family MicroRNAs in Response to Retinoic Acid-Induced Spermatogonial Differentiation in Mice1. Biol. Reprod. 2011, 85, 189–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tong, M.-H.; Mitchell, D.A.; McGowan, S.D.; Evanoff, R.; Griswold, M.D. Two miRNA Clusters, Mir-17-92 (Mirc1) and Mir-106b-25 (Mirc3), Are Involved in the Regulation of Spermatogonial Differentiation in Mice1. Biol. Reprod. 2012, 86, 72. [Google Scholar] [CrossRef] [PubMed]
- Huszar, J.M.; Payne, C.J. MicroRNA 146 (Mir146) Modulates Spermatogonial Differentiation by Retinoic Acid in Mice1. Biol. Reprod. 2013, 88, 15. [Google Scholar] [CrossRef]
- Tang, X.-H.; Gudas, L.J. Retinoids, Retinoic Acid Receptors, and Cancer. Annu. Rev. Pathol. Mech. Dis. 2011, 6, 345–364. [Google Scholar] [CrossRef]
- Chen, M.-C.; Hsu, S.-L.; Lin, H.; Yang, T.-Y. Retinoic acid and cancer treatment. BioMedicine 2014, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Sakurai, T.; Kakudo, K. Retinoid, Retinoic Acid Receptor β and Breast Cancer. Breast Cancer Res. Treat. 2002, 76, 167–173. [Google Scholar] [CrossRef]
- Zeng, W.-G.; Hu, P.; Wang, J.-N.; Liu, R.-B. All-trans retinoic acid effectively inhibits breast cancer stem cells growth in vitro. Zhonghua Zhong Liu Za Zhi 2013, 35, 89–93. (In Chinese) [Google Scholar] [CrossRef]
- Wu, M.-J.; Kim, M.R.; Chen, Y.-S.; Yang, J.-Y.; Chang, C.-J. Retinoic acid directs breast cancer cell state changes through regulation of TET2-PKCζ pathway. Oncogene 2017, 36, 3193–3206. [Google Scholar] [CrossRef]
- Garattini, E.; Paroni, G.; Terao, M. Retinoids and breast cancer: New clues to increase their activity and selectivity. Breast Cancer Res. 2012, 14, 1–2. [Google Scholar] [CrossRef]
- Rubin, M.; Fenig, E.; Rosenauer, A.; Menendez-Botet, C.; Achkar, C.; Bentel, J.M.; Yahalom, J.; Mendelsohn, J.; Miller, W.H. 9-Cis retinoic acid inhibits growth of breast cancer cells and down-regulates estrogen receptor RNA and protein. Cancer Res. 1994, 54, 6549–6556. [Google Scholar] [PubMed]
- Terao, M.; Fratelli, M.; Kurosaki, M.; Zanetti, A.; Guarnaccia, V.; Paroni, G.; Tsykin, A.; Lupi, M.; Gianni, M.; Goodall, G.J.; et al. Induction of miR-21 by Retinoic Acid in Estrogen Receptor-positive Breast Carcinoma Cells. J. Biol. Chem. 2011, 286, 4027–4042. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saumet, A.; Vetter, G.; Bouttier, M.; Antoine, E.; Roubert, C.; Orsetti, B.; Theillet, C.; Lecellier, C.-H. Estrogen and retinoic acid antagonistically regulate several microRNA genes to control aerobic glycolysis in breast cancer cells. Mol. BioSyst. 2012, 8, 3242–3253. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Miao, L.; Mei, Y.; Wu, M. Retinoic acid-induced HOXA5 expression is co-regulated by HuR and miR-130a. Cell. Signal. 2013, 25, 1476–1485. [Google Scholar] [CrossRef]
- La Rocca, G.; Badin, M.; Shi, B.; Xu, S.Q.; DeAngelis, T.; Sepp-Lorenzino, L.; Baserga, R. Mechanism of growth inhibition by MicroRNA 145: The role of the IGF-I receptor signaling pathway. J. Cell. Physiol. 2009, 220, 485–491. [Google Scholar] [CrossRef]
- Shi, B.; Sepp-Lorenzino, L.; Prisco, M.; Linsley, P.; DeAngelis, T.; Baserga, R. Micro RNA 145 Targets the Insulin Receptor Substrate-1 and Inhibits the Growth of Colon Cancer Cells*. J. Biol. Chem. 2007, 282, 32582–32590. [Google Scholar] [CrossRef] [Green Version]
- Del Rincon, S.V.; Guo, Q.; Morelli, C.; Shiu, H.Y.; Surmacz, E.; Miller, W.H. Retinoic acid mediates degradation of IRS-1 by the ubiquitin–proteasome pathway, via a PKC-dependant mechanism. Oncogene 2004, 23, 9269–9279. [Google Scholar] [CrossRef] [Green Version]
- La Rocca, G.; Shi, B.; Audia, A.; Ferrari-Amorotti, G.; Mellert, H.S.; Calabretta, B.; McMahon, S.B.; Sepp-Lorenzino, L.; Baserga, R. Regulation of microRNA-145 by growth arrest and differentiation. Exp. Cell Res. 2011, 317, 488–495. [Google Scholar] [CrossRef]
- Sidell, N. Retinoic Acid-Induced Growth Inhibition and Morphologic Differentiation of Human Neuroblastoma Cells In Vitro2. J. Natl. Cancer Inst. 1982, 68, 589–596. [Google Scholar] [CrossRef]
- Reynolds, C.; Matthay, K.K.; Villablanca, J.G.; Maurer, B.J. Retinoid therapy of high-risk neuroblastoma. Cancer Lett. 2003, 197, 185–192. [Google Scholar] [CrossRef]
- Chen, Y.; Stallings, R.L. Differential Patterns of MicroRNA Expression in Neuroblastoma Are Correlated with Prognosis, Differentiation, and Apoptosis. Cancer Res. 2007, 67, 976–983. [Google Scholar] [CrossRef] [Green Version]
- Das, E.; Bhattacharyya, N.P. MicroRNA-432 contributes to dopamine cocktail and retinoic acid induced differentiation of human neuroblastoma cells by targeting NESTIN and RCOR1 genes. FEBS Lett. 2014, 588, 1706–1714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evangelisti, C.; Florian, M.C.; Massimi, I.; Dominici, C.; Giannini, G.; Galardi, S.; Buè, M.C.; Massalini, S.; McDowell, H.P.; Messi, E.; et al. MiR-128 up-regulation inhibits Reelin and DCX expression and reduces neuroblastoma cell motility and invasiveness. FASEB J. 2009, 23, 4276–4287. [Google Scholar] [CrossRef] [Green Version]
- Meseguer, S.; Mudduluru, G.; Escamilla, J.M.; Allgayer, H.; Barettino, D. MicroRNAs-10a and -10b Contribute to Retinoic Acid-induced Differentiation of Neuroblastoma Cells and Target the Alternative Splicing Regulatory Factor SFRS1 (SF2/ASF). J. Biol. Chem. 2011, 286, 4150–4164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonçalves, V.; Jordan, P. Posttranscriptional Regulation of Splicing Factor SRSF1 and Its Role in Cancer Cell Biology. BioMed Res. Int. 2015, 2015, 287048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foley, N.H.; Bray, I.; Watters, K.M.; Das, S.; Bryan, K.; Bernas, T.; Prehn, J.H.M.; Stallings, R.L. MicroRNAs 10a and 10b are potent inducers of neuroblastoma cell differentiation through targeting of nuclear receptor corepressor 2. Cell Death Differ. 2011, 18, 1089–1098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, S.; Foley, N.; Bryan, K.; Watters, K.M.; Bray, I.; Murphy, D.M.; Buckley, P.G.; Stallings, R.L. MicroRNA Mediates DNA Demethylation Events Triggered by Retinoic Acid during Neuroblastoma Cell Differentiation. Cancer Res. 2010, 70, 7874–7881. [Google Scholar] [CrossRef] [Green Version]
- Das, S.; Bryan, K.; Buckley, P.G.; Piskareva, O.; Bray, I.M.; Foley, N.H.; Ryan, J.; Lynch, J.; Creevey, L.; Fay, J.C.; et al. Modulation of neuroblastoma disease pathogenesis by an extensive network of epigenetically regulated microRNAs. Oncogene 2013, 32, 2927–2936. [Google Scholar] [CrossRef] [Green Version]
- Guan, H.; Zhang, P.; Liu, C.; Zhang, J.; Huang, Z.; Chen, W.; Chen, Z.; Ni, N.; Liu, Q.; Jiang, A. Characterization and functional analysis of the human microRNA let-7a2 promoter in lung cancer A549 cell lines. Mol. Biol. Rep. 2011, 38, 5327–5334. [Google Scholar] [CrossRef]
- Zhu, X.; Gao, G.; Chu, K.; Yang, X.; Ren, S.; Li, Y.; Wu, H.; Huang, Y.; Zhou, C. Inhibition of RAC1-GEF DOCK3 by miR-512-3p contributes to suppression of metastasis in non-small cell lung cancer. Int. J. Biochem. Cell Biol. 2015, 61, 103–114. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, R.; Wei, G.; Dai, S.; Zhang, X.; Yang, W.; Li, X.; Bai, C. Long Non-coding RNA Maternally Expressed 3 Increases the Expression of Neuron-Specific Genes by Targeting miR-128-3p in All-Trans Retinoic Acid-Induced Neurogenic Differentiation From Amniotic Epithelial Cells. Front. Cell Dev. Biol. 2019, 7, 342. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.-H.; Wang, W.-T.; Huang, W.; Fang, K.; Sun, Y.-M.; Liu, S.-R.; Luo, X.-Q.; Chen, Y.-Q. The lncRNA HOTAIRM1 regulates the degradation of PML-RARA oncoprotein and myeloid cell differentiation by enhancing the autophagy pathway. Cell Death Differ. 2017, 24, 212–224. [Google Scholar] [CrossRef]
- Zhang, X.; Weissman, S.M.; E Newburger, P. Long intergenic non-coding RNA HOTAIRM1 regulates cell cycle progression during myeloid maturation in NB4 human promyelocytic leukemia cells. RNA Biol. 2014, 11, 777–787. [Google Scholar] [CrossRef] [Green Version]
- Zhao, H.; Zhang, X.; Frazão, J.B.; Condino-Neto, A.; Newburger, P.E. HOX antisense lincRNA HOXA-AS2 is an apoptosis repressor in all Trans retinoic acid treated NB4 promyelocytic leukemia cells. J. Cell. Biochem. 2013, 114, 2375–2383. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Li, D.; Huang, D.; Song, H.; Mei, H.; Fang, E.; Wang, X.; Yang, F.; Zheng, L.; Huang, K.; et al. Risk-Associated Long Noncoding RNA FOXD3-AS1 Inhibits Neuroblastoma Progression by Repressing PARP1-Mediated Activation of CTCF. Mol. Ther. 2018, 26, 755–773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Hajj, J.; Nguyen, E.; Liu, Q.; Bouyer, C.; Adriaenssens, E.; Hilal, G.; Ségal-Bendirdjian, E. Telomerase regulation by the long non-coding RNA H19 in human acute promyelocytic leukemia cells. Mol. Cancer 2018, 17, 85. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Li, X.; Ren, Y.; Yin, Z.; Zhou, B. Long Noncoding RNA RAET1K Enhances CCNE1 Expression and Cell Cycle Arrest of Lung Adenocarcinoma Cell by Sponging miRNA-135a-5p. Front. Genet. 2020, 10, 1348. [Google Scholar] [CrossRef]
- Wang, Z.M.; Xia, S.W.; Zhang, T.; Wang, Z.Y.; Yang, X.; Kai, J.; Cheng, X.D.; Shao, J.J.; Tan, S.Z.; Chen, A.P.; et al. LncRNA-H19 induces hepatic stellate cell activation via upregulating alcohol dehydrogenase III-mediated retinoic acid signals. Int. Immunopharmacol. 2020, 84, 106470. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Padilla, C.; Lozano-Velasco, E.; López-Sánchez, C.; Garcia-Martínez, V.; Aranega, A.; Franco, D. Non-Coding RNAs in Retinoic Acid as Differentiation and Disease Drivers. Non-Coding RNA 2021, 7, 13. https://0-doi-org.brum.beds.ac.uk/10.3390/ncrna7010013
García-Padilla C, Lozano-Velasco E, López-Sánchez C, Garcia-Martínez V, Aranega A, Franco D. Non-Coding RNAs in Retinoic Acid as Differentiation and Disease Drivers. Non-Coding RNA. 2021; 7(1):13. https://0-doi-org.brum.beds.ac.uk/10.3390/ncrna7010013
Chicago/Turabian StyleGarcía-Padilla, Carlos, Estefanía Lozano-Velasco, Carmen López-Sánchez, Virginio Garcia-Martínez, Amelia Aranega, and Diego Franco. 2021. "Non-Coding RNAs in Retinoic Acid as Differentiation and Disease Drivers" Non-Coding RNA 7, no. 1: 13. https://0-doi-org.brum.beds.ac.uk/10.3390/ncrna7010013