Asymmetric Supercapacitors: Optical and Thermal Effects When Active Carbon Electrodes Are Embedded with Nano-Scale Semiconductor Dots
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
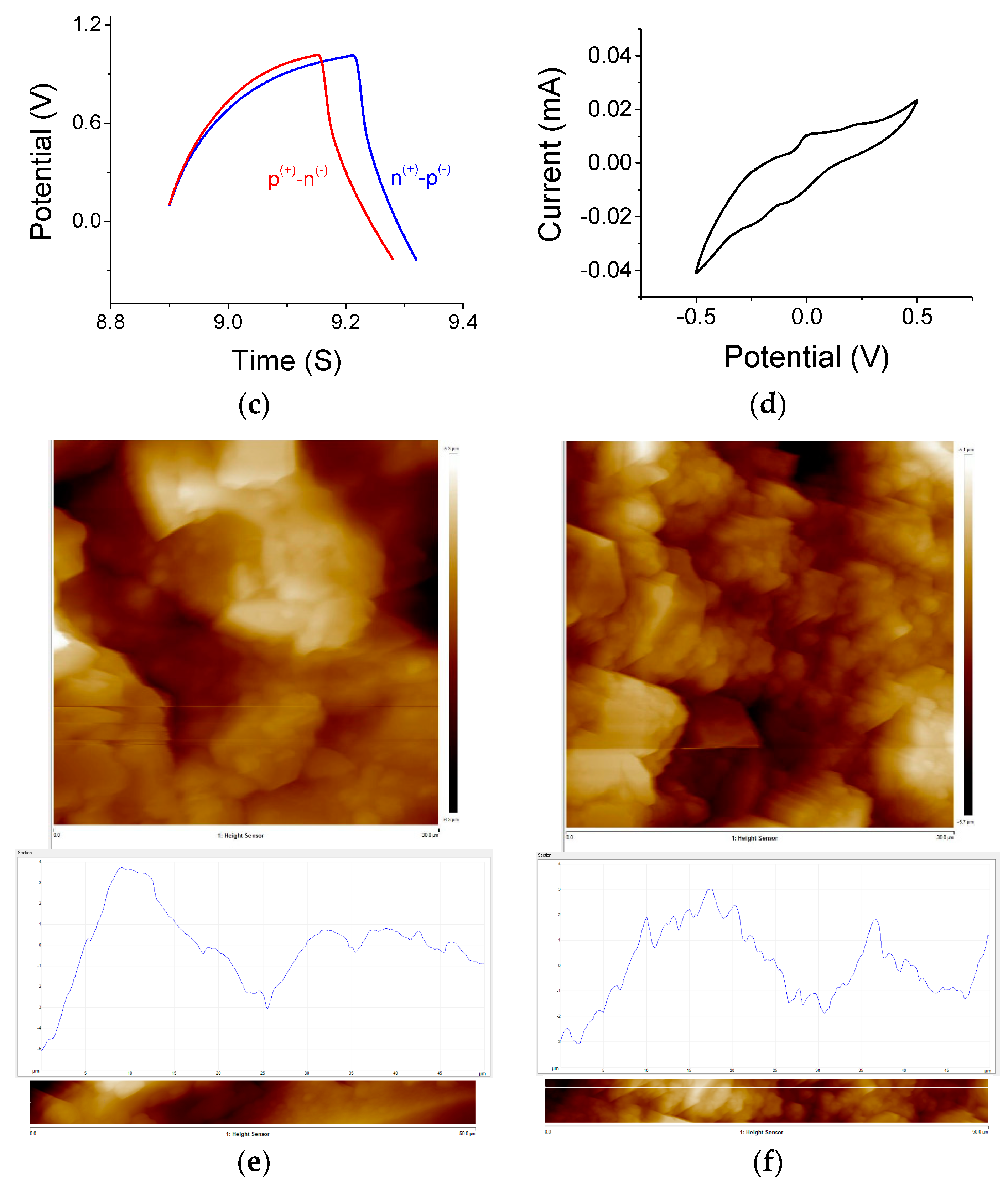
3.1. Film Characterizations
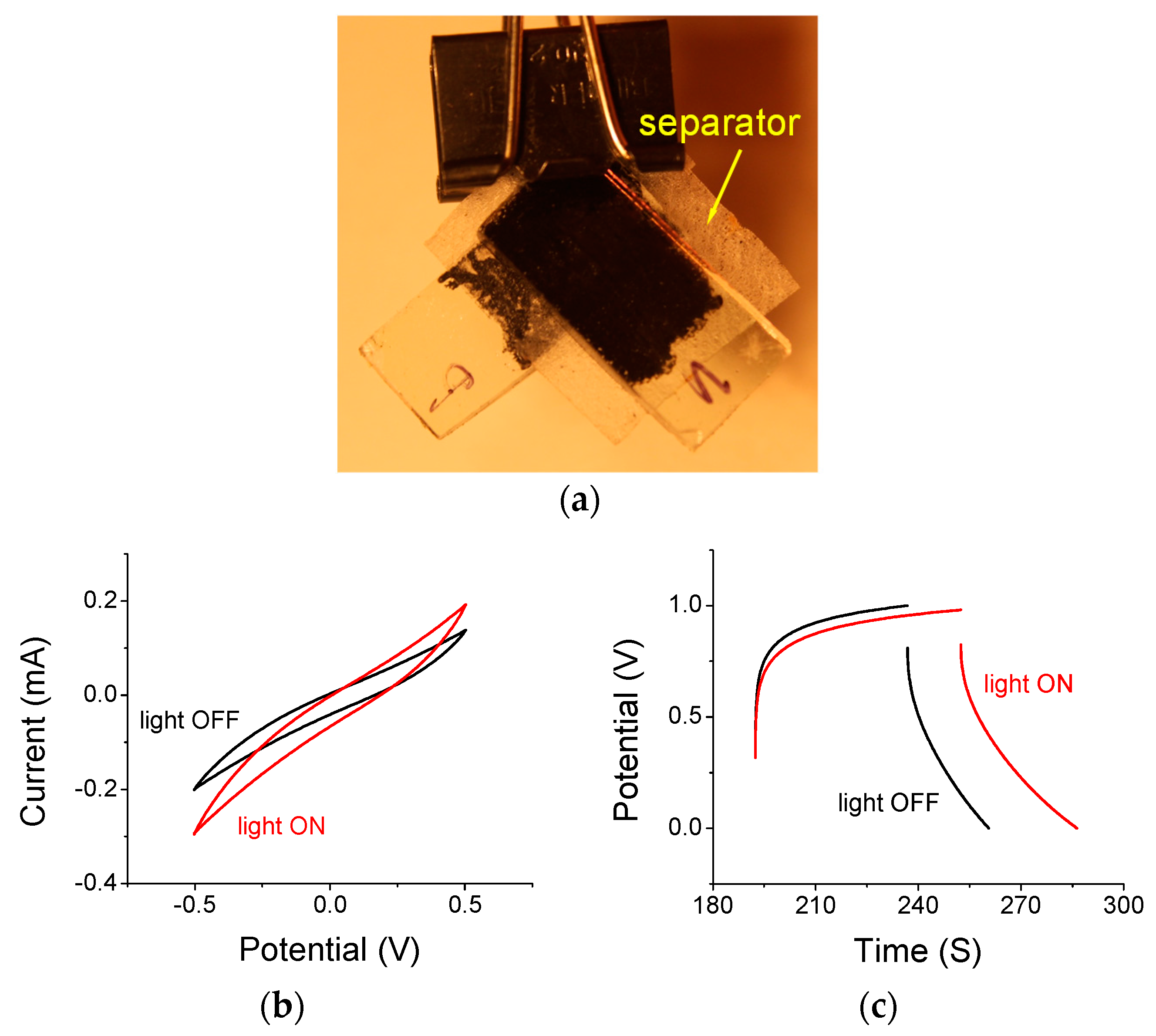
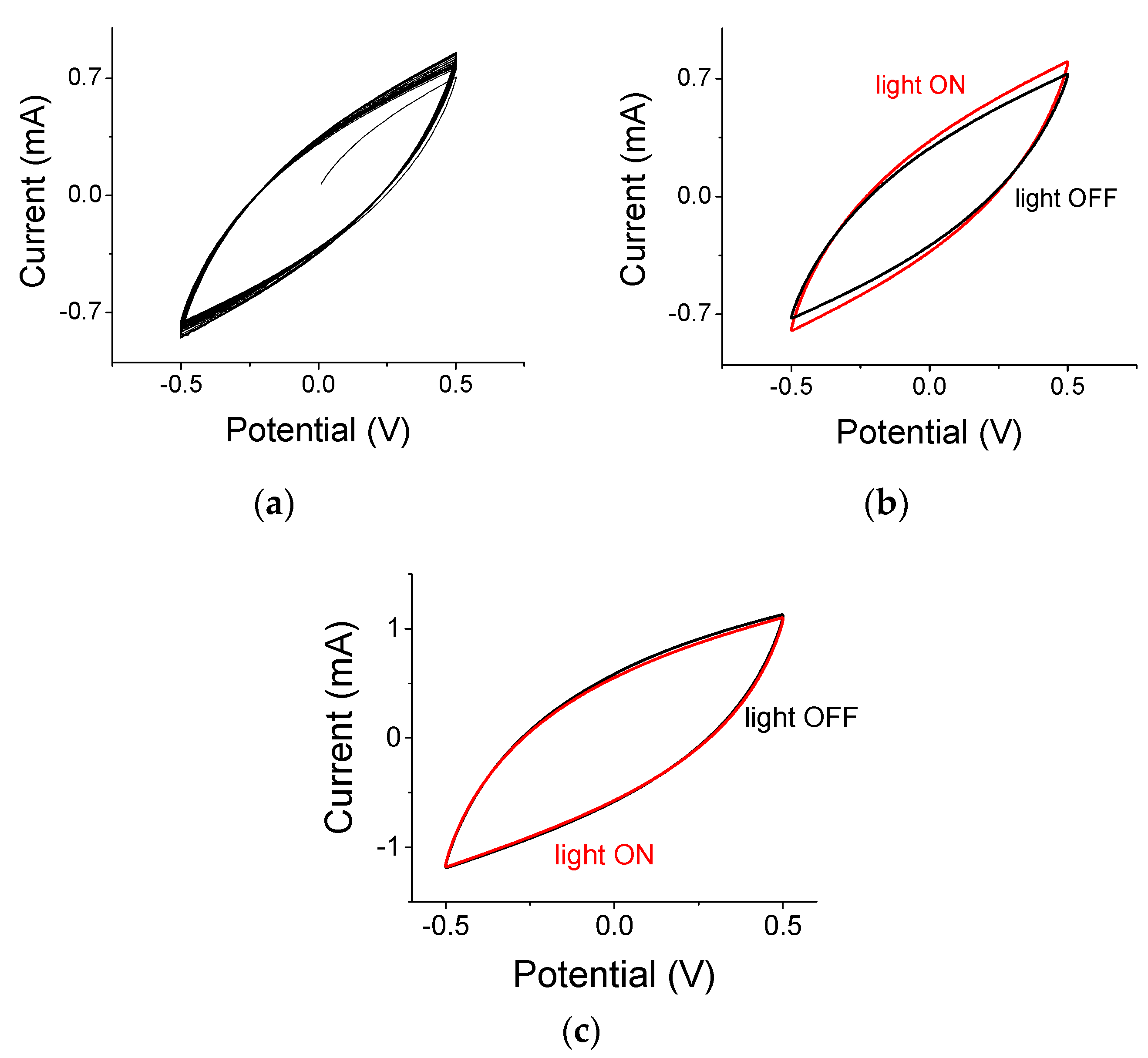
3.2. Supercapacitors under Light ON and Light OFF Conditions
3.3. Thermal Considerations during Light ON and Light OFF Conditions
3.3.1. PMMA Binder with Aqueous Solution
3.3.2. PMMA Binder with Ion Liquid Electrolyte
3.3.3. p-n Binders with Ion Liquid Electrolyte
3.3.4. p-n Binders with Aqueous Solution
3.4. The Effect of IR Absorption Band at 840 nm
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Simon, P.; Burke, A.F. Nanostructured Carbons: Double-Layer Capacitance and More. Electrochem. Soc. Interface 2008, 17, 38–43. [Google Scholar] [CrossRef]
- Inagaki, M.; Konno, H.; Tanaike, O. Carbon materials for electrochemical capacitors. J. Power Sources 2010, 195, 7880–7903. [Google Scholar] [CrossRef]
- Zhang, S.; Pan, N. Supercapacitors Performance Evaluation. Adv. Energy Mater. 2015, 5, 1401401. [Google Scholar] [CrossRef] [Green Version]
- Polat, E.O.; Kocabas, C. Broadband Optical Modulators Based on Graphene Supercapacitors. Nano Lett. 2013, 13, 5851–5857. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.; Rojas-Cessa, R.; Mohamed, A.; Grebel, H. The Digital Power Networks: Energy Dissemination Through a Micro-Grid. In Proceedings of the 2018 IEEE International Conference on Internet of Things (iThings) and IEEE Green Computing and Communications (GreenCom) and IEEE Cyber, Physical and Social Computing (CPSCom) and IEEE Smart Data (SmartData), Halifax, NS, Canada, 30 July–3 August 2018. [Google Scholar]
- Rojas-Cessa, R.; Grebel, H.; Jiang, Z.; Fukuda, C.; Pita, H.; Chowdhury, T.S.; Dong, Z.; Wan, Y. Integration of Alternative Energy Sources into Digital Micro-Grids. Environ. Prog. Sustain. Energy 2018, 37, 155–164. [Google Scholar] [CrossRef]
- Long, J.W.; Bélanger, D.; Brousse, T.; Sugimoto, W.; Sassin, M.B.; Crosnier, O. Asymmetric Electrochemical Capacitors-Stretching the Limits of Aqueous Electrolytes; Naval Research Lab.: Washington, DC, USA, 2011. [Google Scholar]
- Shao, Y.; El-Kady, M.F.; Sun, J.; Li, Y.; Zhang, Q.; Zhu, M.; Wang, H.; Dunn, B.; Kaner, R.B. Design and Mechanisms of Asymmetric Supercapacitors. Chem. Rev. 2018, 118, 9233–9280. [Google Scholar] [CrossRef]
- Rahmanifar, M.S.; Hemmati, M.; Noori, A.; El-Kady, M.F.; Mousavi, M.F.; Kaner, R.B. Asymmetric supercapacitors: An alternative to activated carbon negative electrodes based on earth abundant elements. Mater. Today Energy 2019, 12, 26–36. [Google Scholar] [CrossRef]
- Volkov, A.V.; Sun, H.; Kroon, R.; Ruoko, T.P.; Che, C.; Edberg, J.; Müller, C.; Fabiano, S.; Crispin, X. Asymmetric Aqueous Supercapacitor Based on p- and n-Type Conducting Polymers. ACS Appl. Energy Mater. 2019, 2, 5350–5355. [Google Scholar] [CrossRef]
- Shang, W.; Tan, Y.; Kong, L.B.; Ran, F. Fundamental Triangular Interaction of Electron Trajectory Deviation and P–N Junction to Promote Redox Reactions for the High-Energy Density Electrode. ACS Appl. Mater. Interfaces 2020, 12, 29404–29413. [Google Scholar] [CrossRef]
- Carbone, M.; Missori, M.; Micheli, L.; Tagliatesta, P.; Bauer, E.M. NiO Pseudocapacitance and Optical Properties: Does The Shape Win? Materials 2020, 13, 1417. [Google Scholar] [CrossRef] [Green Version]
- Wan, C.; Yuan, L.; Shen, H. Effects of Electrode Mass-loading on the Electrochemical Properties of Porous MnO2 for Electrochemical Supercapacitor. Int. J. Electrochem. Sci. 2014, 9, 1. [Google Scholar]
- Li, Y.; Xu, X.; He, Y.; Jiang, Y.; Lin, K. Nitrogen Doped Macroporous Carbon as Electrode Materials for High Capacity of Supercapacitor. Polymers 2017, 9, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sauvage, F.; Chhor, S.; Marchioro, A.; Moser, J.E.; Graetzel, M. Butyronitrile-Based Electrolyte for Dye-Sensitized Solar Cells. J. Am. Chem. Soc. 2011, 133, 13103–13109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, J.; Lan, Z.; Lin, J.; Huang, M.; Huang, Y.; Fan, L.; Luo, G. Electrolytes in Dye-Sensitized Solar Cells. Chem. Rev. 2015, 115, 2136–2173. [Google Scholar] [CrossRef]
- Sengottaiyan, C.; Jayavel, R.; Shrestha, R.G.; Subramani, T.; Maji, S.; Kim, J.H.; Hill, J.P.; Ariga, K.; Shrestha, L.K. Indium Oxide/Carbon Nanotube/Reduced Graphene Oxide Ternary Nanocomposite with Enhanced Electrochemical Supercapacitance. Bull. Chem. Soc. Jpn. 2019, 92, 521–528. [Google Scholar] [CrossRef]
- Zhu, F.; Liu, W.; Liu, Y.; Shi, W. Construction of porous interface on CNT@NiCo-LDH core-shell nanotube for supercapacitor applications. Chem. Eng. J. 2020, 383, 123150. [Google Scholar] [CrossRef]
- Wei, H.; Wang, H.; Li, A.; Li, H.; Cui, D.; Dong, M.; Lin, J.; Fan, J.; Zhang, J.; Hou, H.; et al. Advanced porous hierarchical activated carbon derived from agricultural wastes towards high performance supercapacitors. J. Alloy. Compunds 2020, 820, 153111. [Google Scholar] [CrossRef]
- O’Connell, M.J.; Boul, P.; Ericson, L.M.; Huffman, C.; Wang, Y.; Haroz, E.; Kuper, C.; Tour, J.; Ausman, K.D.; Smalley, R.E. Reversible water solubilization of single wall carbon-nanotubes by polymer wrapping. Chem. Phys. Lett. 2001, 342, 265. [Google Scholar] [CrossRef]
- Shim, M.; Javey, A.; Kam, N.W.S.; Dai, H. Polymer Functionalization for Air-Stable n-Type Carbon Nanotube Field-Effect Transistors. J. Am. Chem. Soc. 2001, 123, 11512. [Google Scholar] [CrossRef]
- Reddy, A.L.M.; Shaijumon, M.M.; Gowda, S.R.; Ajayan, P.M. Multisegmented Au-MnO2/Carbon Nanotube Hybrid Coaxial Arrays for High-Power Supercapacitor Applications. J. Phys. Chem. 2009, 114, 658. [Google Scholar] [CrossRef]
- Kaempgen, M.; Chan, C.K.; Ma, J.; Cui, Y.; Gruner, G. Printable thin film supercapacitors using single-walled carbon nanotubes. Nano Lett. 2009, 9, 1872–1876. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, T.S.; Grebel, H. Supercapacitors with electrical gates. Electrochim. Acta 2019, 307, 459–464. [Google Scholar] [CrossRef]
- Chowdhury, T.S.; Grebel, H. Ion-Liquid Based Supercapacitors with Inner Gate Diode-Like Separators. ChemEngineering 2019, 3, 39. [Google Scholar] [CrossRef] [Green Version]
- Balducci, A.; Dugas, R.; Taberna, P.L.; Simon, P.; Plee, D.; Mastragostino, M.; Passerini, S. High temperature carbon–carbon supercapacitor using ionic liquid as electrolyte. J. Power Sources 2007, 165, 922–927. [Google Scholar] [CrossRef] [Green Version]
- Ruiz, V.; Huynh, T.; Sivakkumar, S.R.; Pandolfo, A.G. Ionic liquid–solvent mixtures as supercapacitor electrolytes for extreme temperature operation. RSC Adv. 2012, 2, 5591–5598. [Google Scholar] [CrossRef]
- Tsay, K.C.; Zhang, L.; Zhang, J. Effects of electrode layer composition/thickness and electrolyte concentration on both specific capacitance and energy density of supercapacitor. Electrochim. Acta 2012, 60, 428–436. [Google Scholar] [CrossRef] [Green Version]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grebel, H. Asymmetric Supercapacitors: Optical and Thermal Effects When Active Carbon Electrodes Are Embedded with Nano-Scale Semiconductor Dots. C 2021, 7, 7. https://0-doi-org.brum.beds.ac.uk/10.3390/c7010007
Grebel H. Asymmetric Supercapacitors: Optical and Thermal Effects When Active Carbon Electrodes Are Embedded with Nano-Scale Semiconductor Dots. C. 2021; 7(1):7. https://0-doi-org.brum.beds.ac.uk/10.3390/c7010007
Chicago/Turabian StyleGrebel, Haim. 2021. "Asymmetric Supercapacitors: Optical and Thermal Effects When Active Carbon Electrodes Are Embedded with Nano-Scale Semiconductor Dots" C 7, no. 1: 7. https://0-doi-org.brum.beds.ac.uk/10.3390/c7010007




