Ethanol Production from Olive Stones through Liquid Hot Water Pre-Treatment, Enzymatic Hydrolysis and Fermentation. Influence of Enzyme Loading, and Pre-Treatment Temperature and Time
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Material
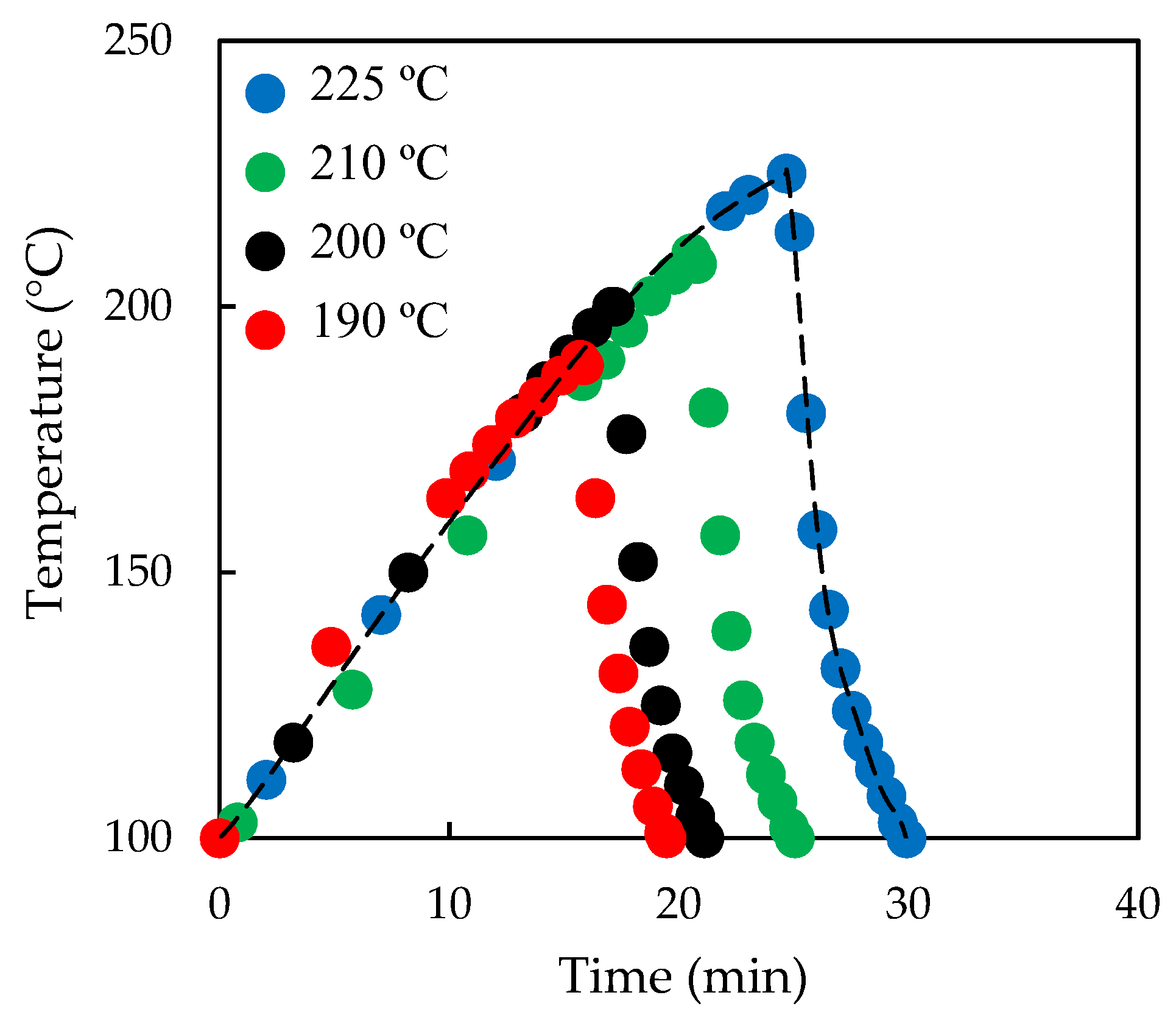
2.2. Liquid Hot Water (LHW) Pre-Treatment
2.3. Enzymatic Hydrolysis
2.4. Fermentation of Enzymatic Hydrolysates
2.5. Analytical Methods
3. Results and Discussion
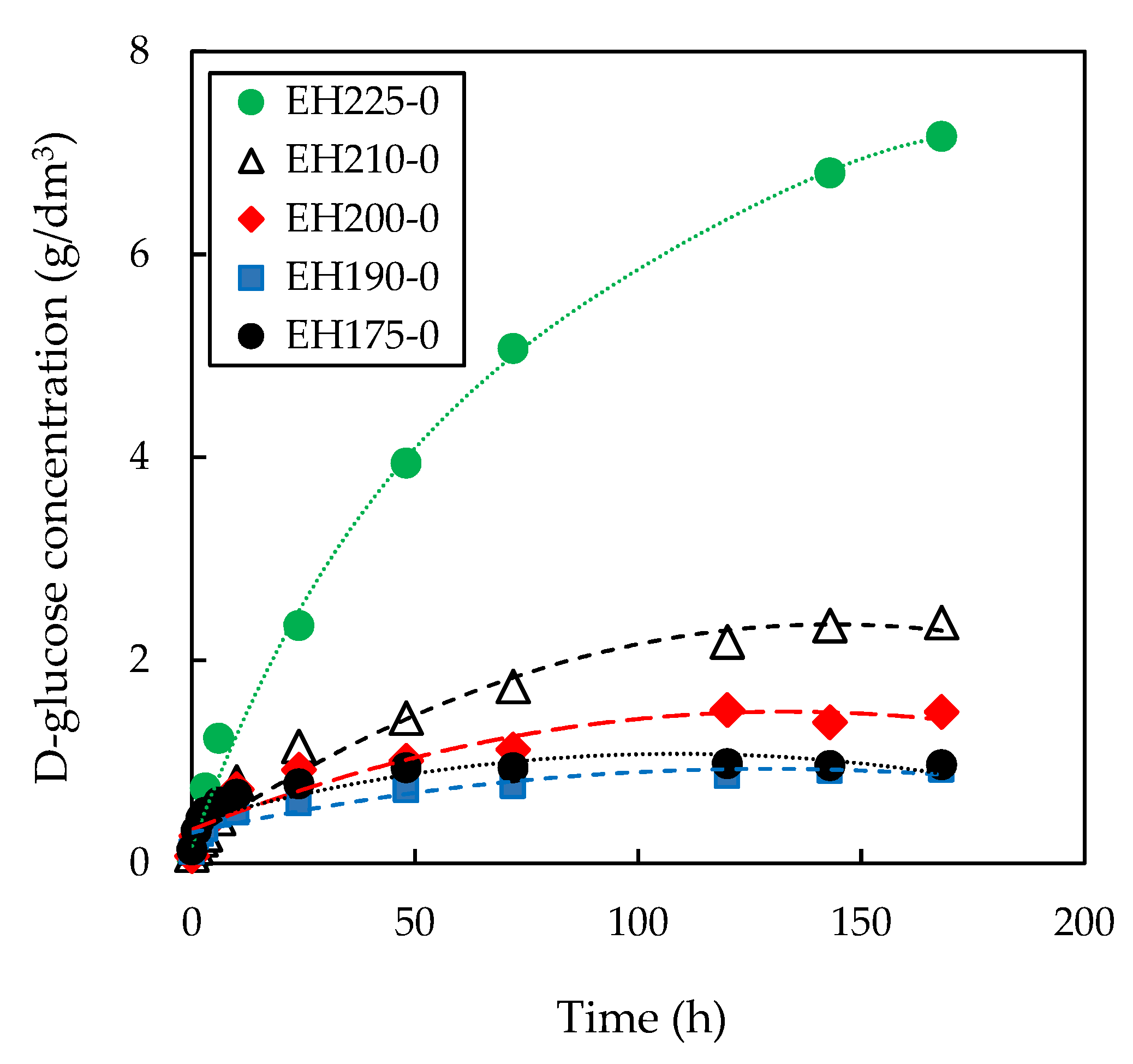
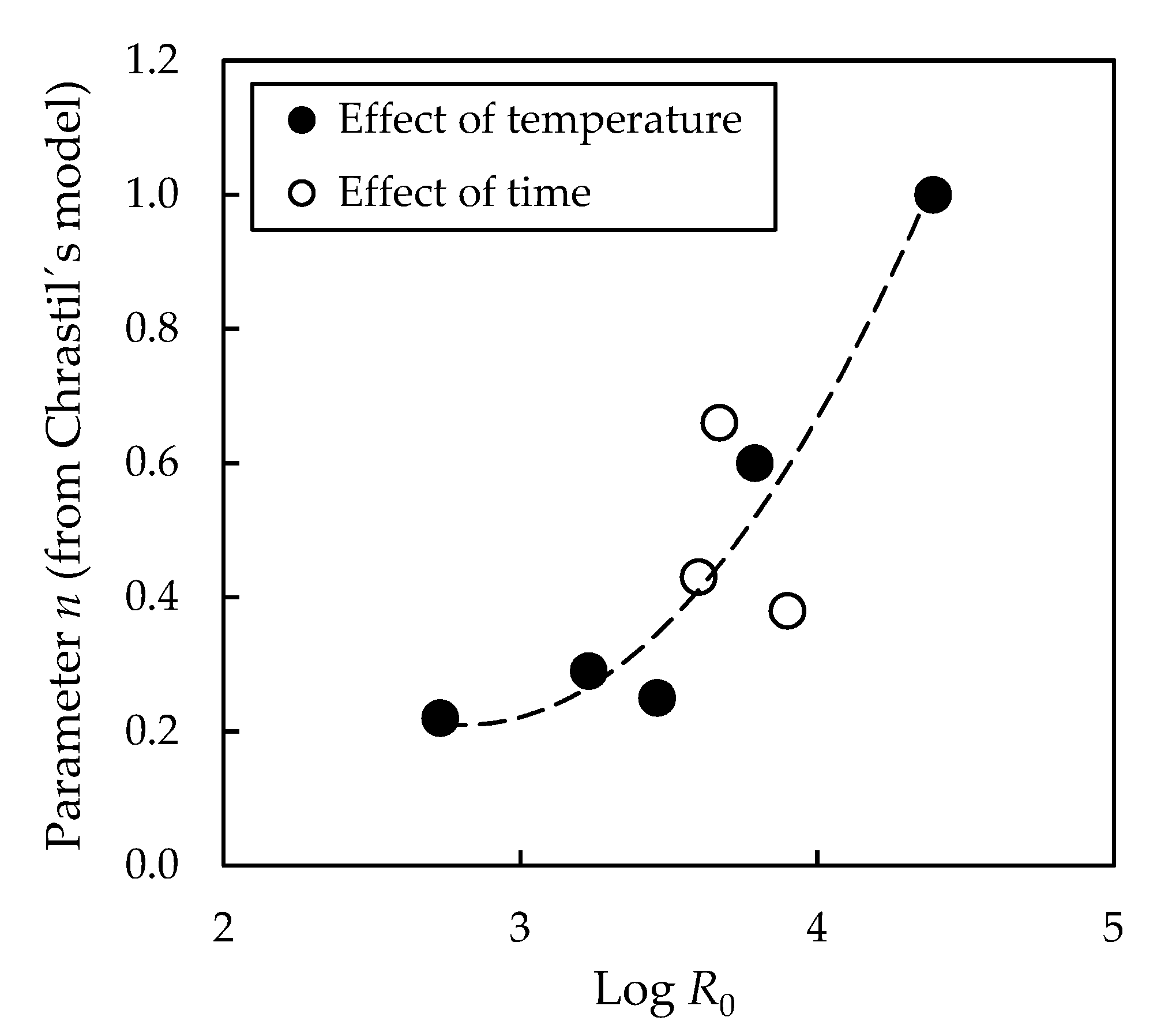
3.1. Effect of Pre-Treatment Temperature on Subsequent Enzymatic Hydrolysis
3.2. Effect of Pre-Treatment Maintenance Time on Subsequent Enzymatic Hydrolysis
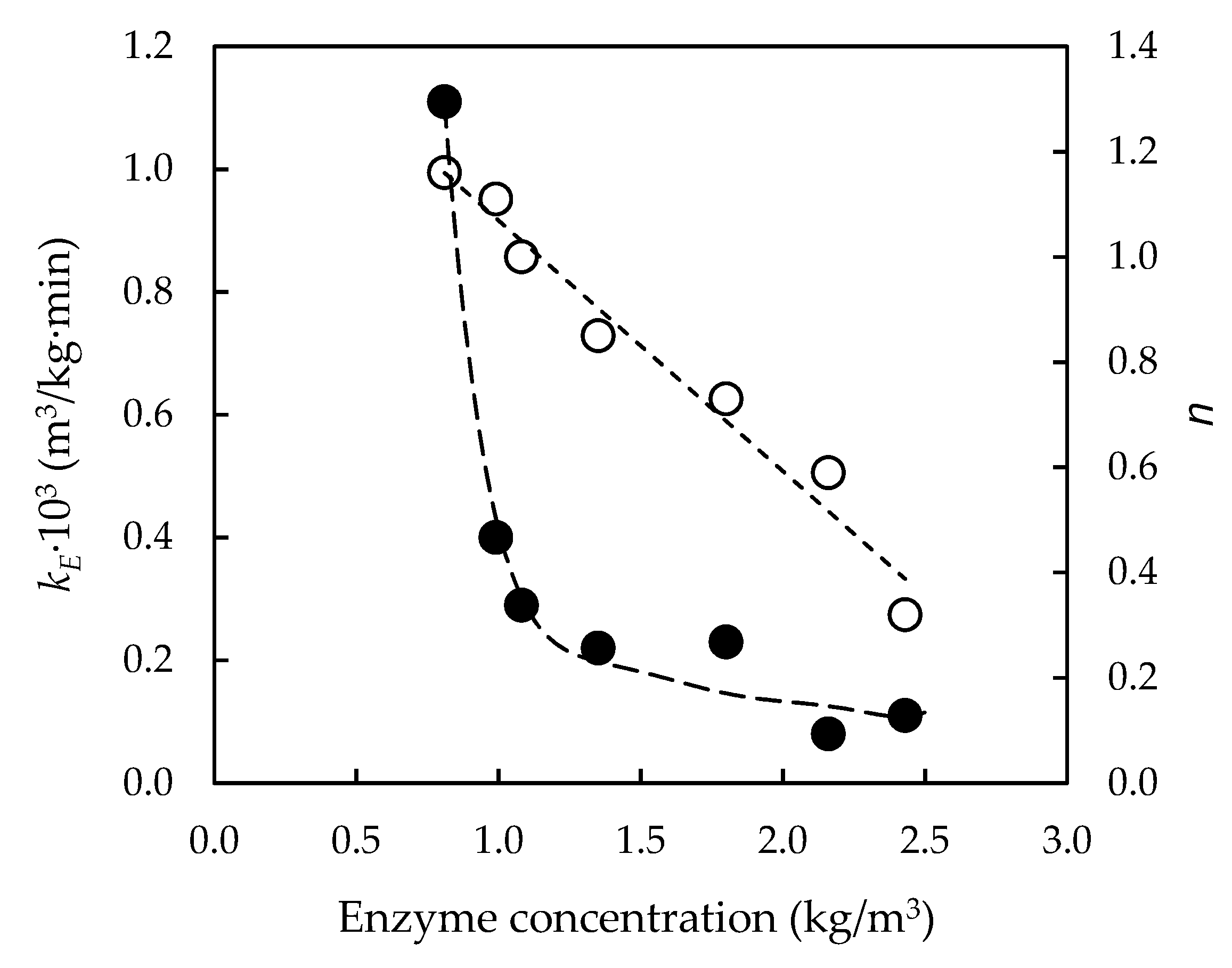
3.3. Effect of Initial Enzyme Concentration on Enzymatic Hydrolysis of Pre-Treated Slurries
3.4. Fermentation of Enzymatic Hydrolysates with P. tannophilus
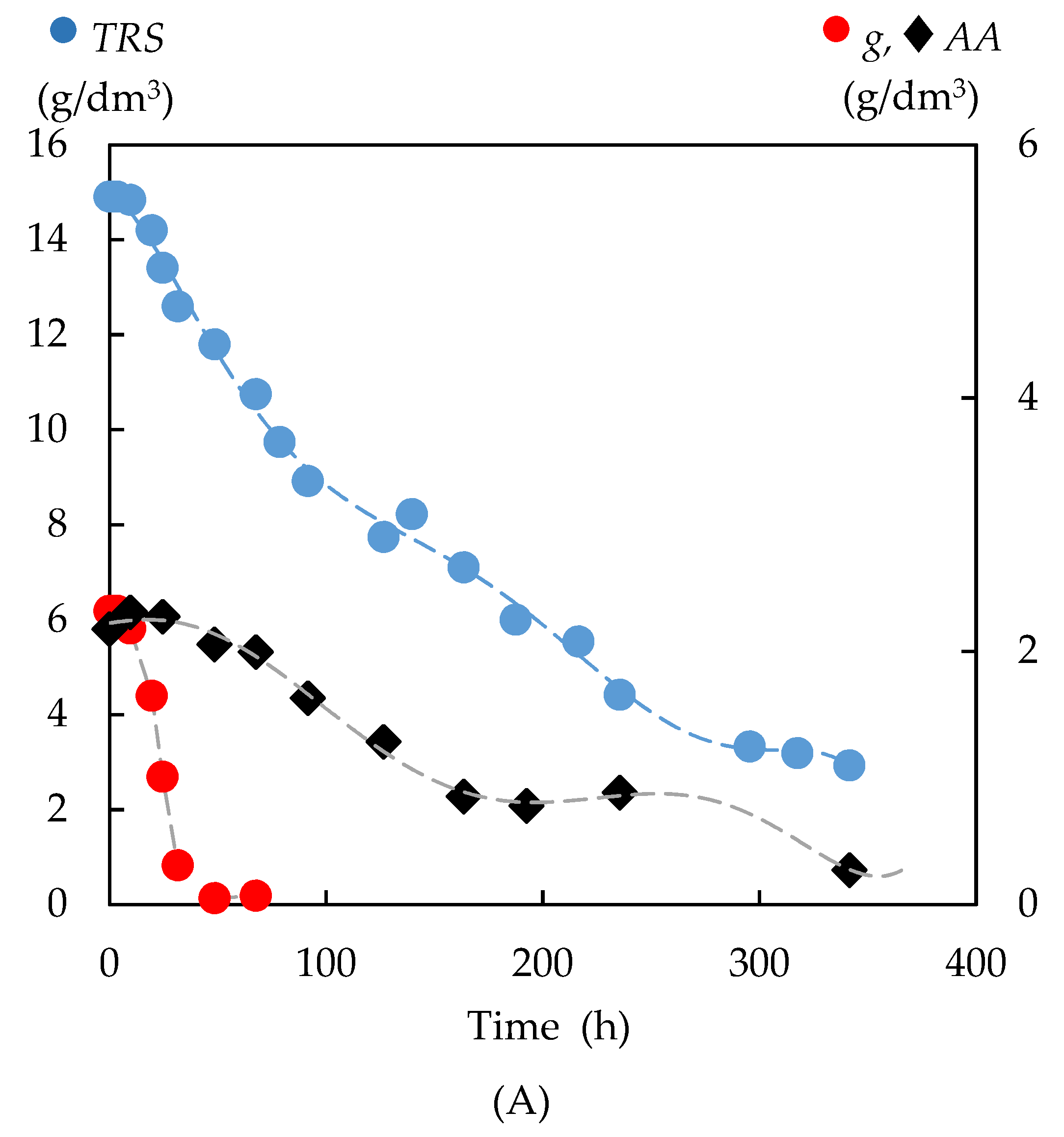
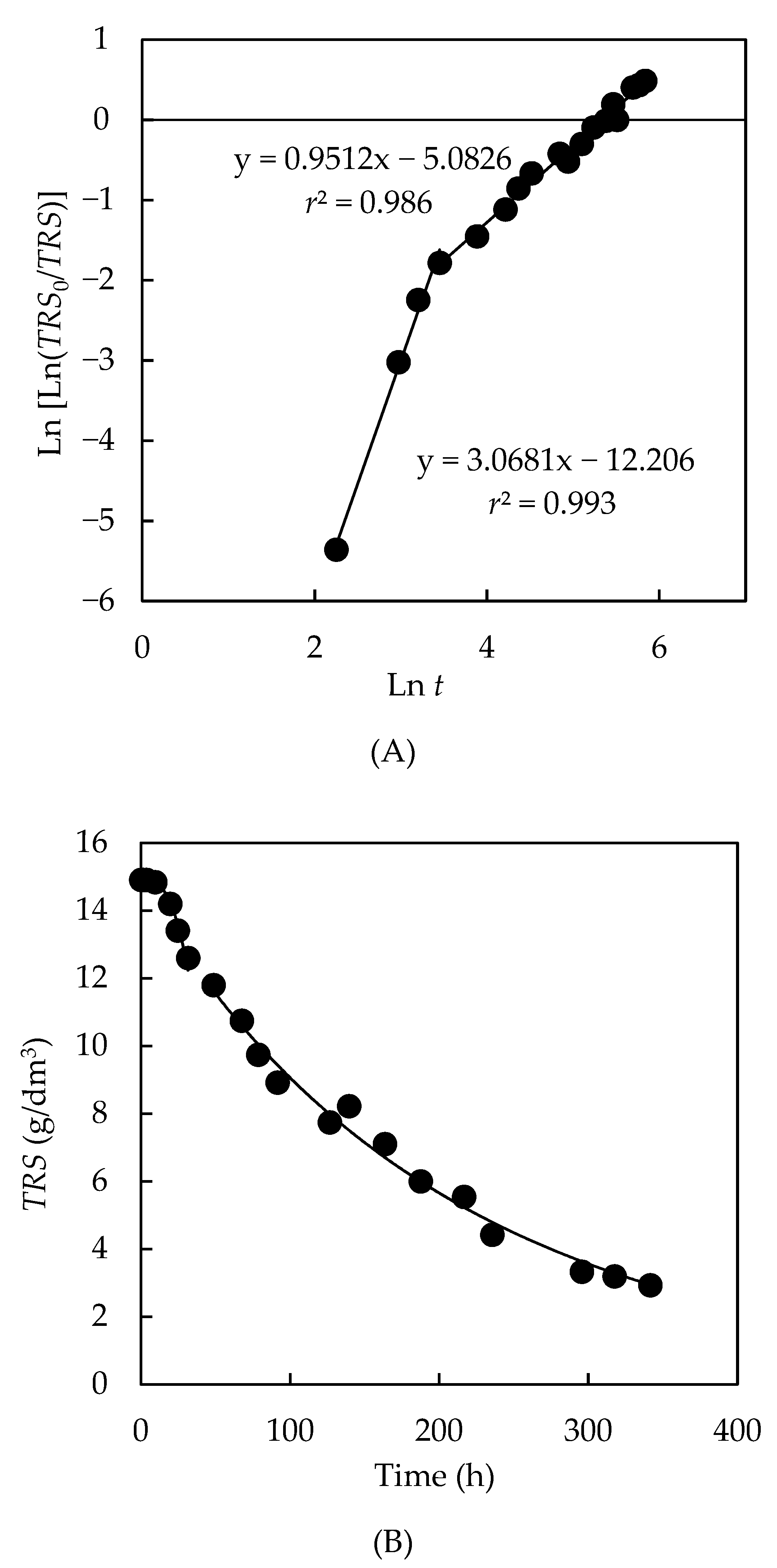
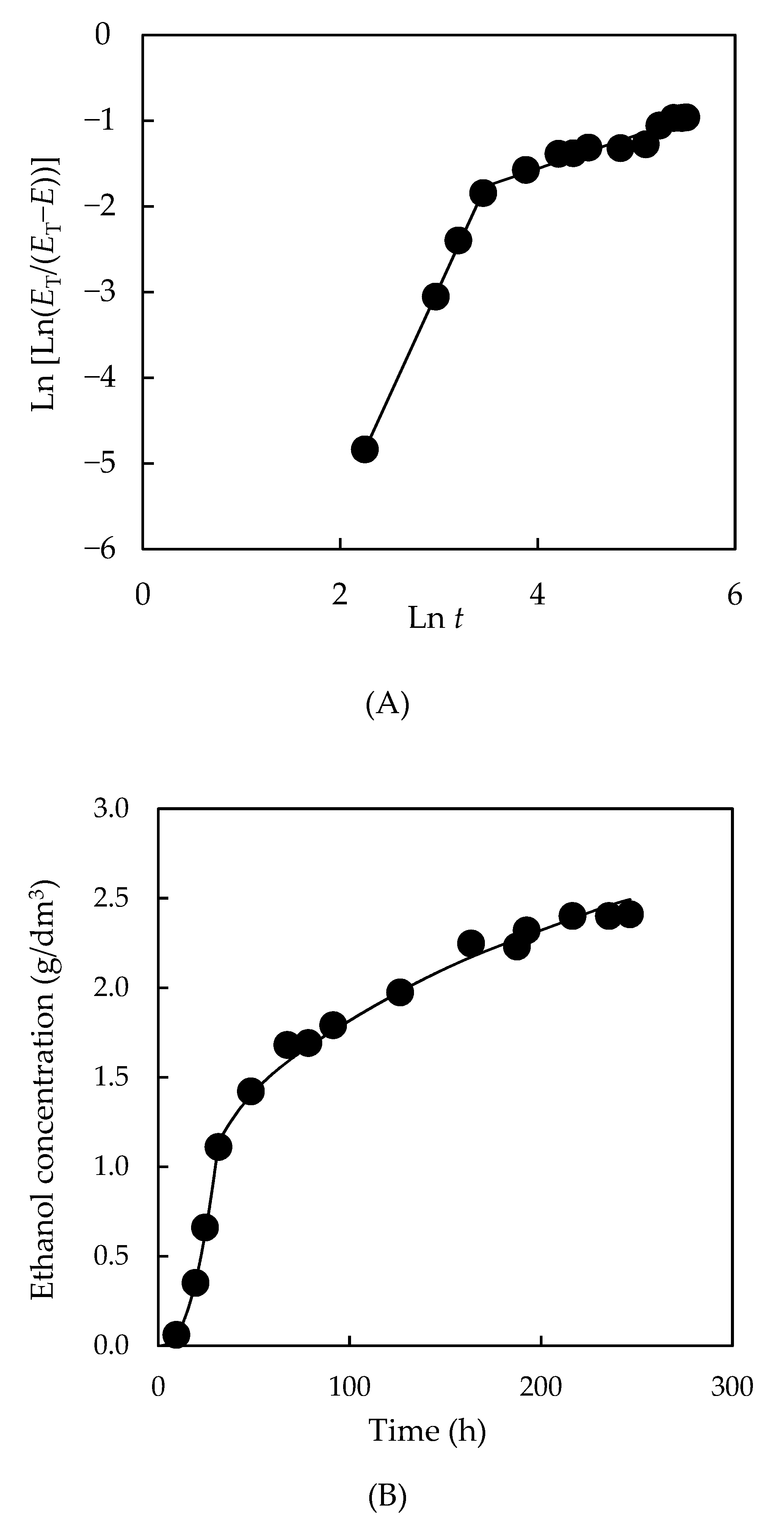
3.4.1. Kinetics of the Total Reducing Sugar Uptake
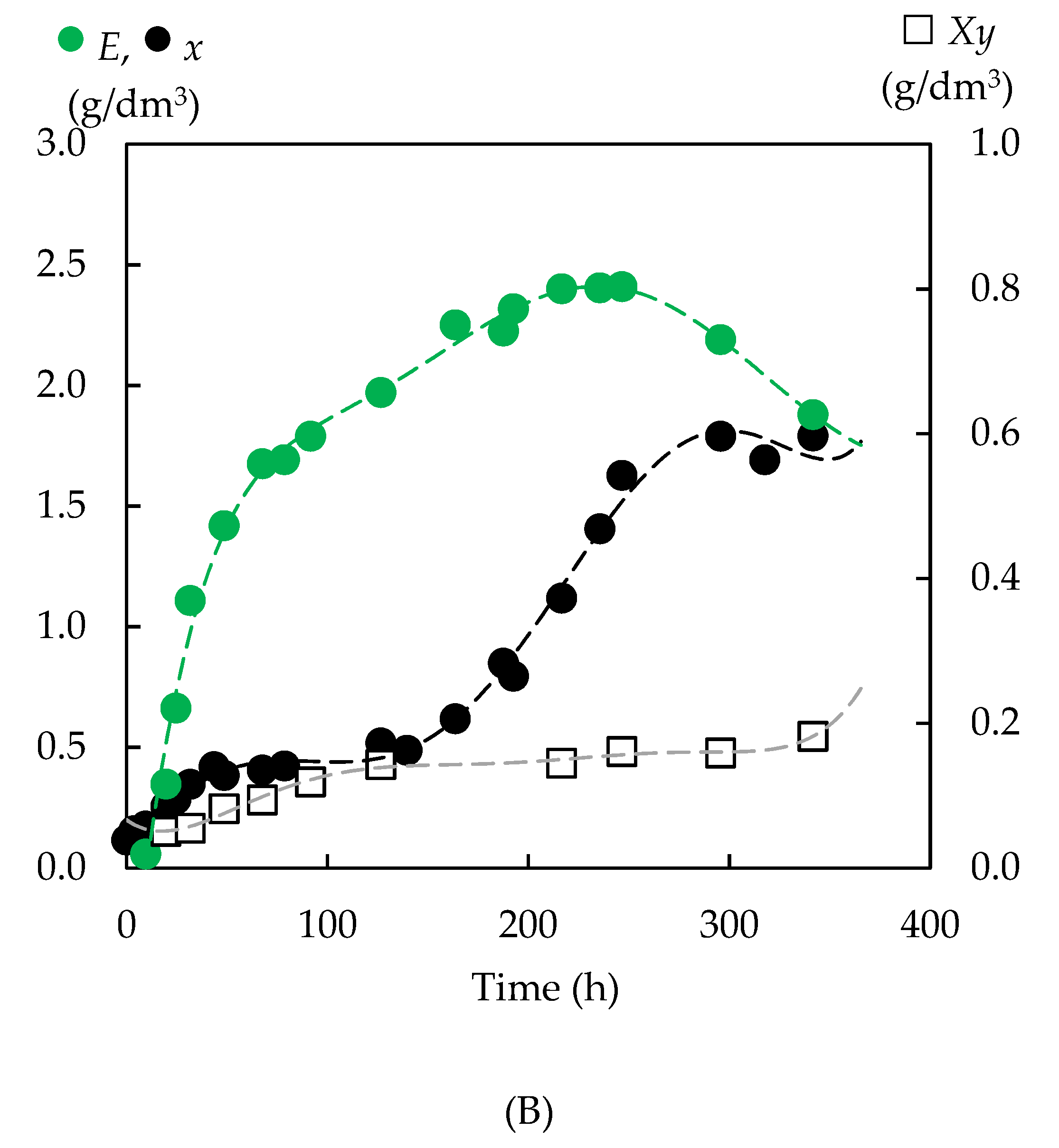
3.4.2. Biomass Yield
3.4.3. Formation of Bioproducts
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rodríguez, G.; Lama, A.; Rodríguez, R.; Jiménez, A.; Guillén, R.; Fernández-Bolaños, J. Olive stone an attractive source of bioactive and valuable compounds. Bioresour. Technol. 2008, 99, 5261–5269. [Google Scholar] [CrossRef] [PubMed]
- García Martín, J.F.; Cuevas, M.; Feng, C.H.; Mateos, P.Á.; García, M.T.; Sánchez, S. Energetic valorisation of olive biomass: Olive-tree pruning, olive stones and pomaces. Processes 2020, 8, 511. [Google Scholar] [CrossRef]
- Mata-Sánchez, J.; Pérez-Jiménez, J.A.; Díaz-Villanueva, M.J.; Serrano, A.; Núñez-Sánchez, N.; López-Giménez, F.J. Statistical evaluation of quality parameters of olive stone to predict its heating value. Fuel 2013, 113, 750–756. [Google Scholar] [CrossRef]
- Ministerio de Agricultura Pesca y Alimentación. Encuesta Sobre Superficies y Rendimientos Cultivos. Available online: https://servicio.mapama.gob.es/ (accessed on 15 January 2021).
- Cuevas, M.; Sánchez, S.; Bravo, V.; García, J.F.; Baeza, J.; Parra, C.; Freer, J. Determination of optimal pre-treatment conditions for ethanol production from olive-pruning debris by simultaneous saccharification and fermentation. Fuel 2010, 89, 2891–2896. [Google Scholar] [CrossRef]
- Cuevas, M.; Saleh, M.; García-Martín, J.F.; Sánchez, S. Acid and enzymatic fractionation of olive stones for ethanol production using Pachysolen tannophilus. Processes 2020, 8, 195. [Google Scholar] [CrossRef] [Green Version]
- Saleh, M.; Cuevas, M.; García, J.F.; Sánchez, S. Valorization of olive stones for xylitol and ethanol production from dilute acid pretreatment via enzymatic hydrolysis and fermentation by Pachysolen tannophilus. Biochem. Eng. J. 2014, 90, 286–293. [Google Scholar] [CrossRef]
- Cuevas, M.; Sánchez, S.; García, J.F.; Baeza, J.; Parra, C.; Freer, J. Enhanced ethanol production by simultaneous saccharification and fermentation of pretreated olive stones. Renew. Energy 2015, 74, 839–847. [Google Scholar] [CrossRef]
- Cuevas, M.; Sánchez, S.; Bravo, V.; Cruz, N.; García, J.F. Fermentation of enzymatic hydrolysates from olive stones by Pachysolen tannophilus. J. Chem. Technol. Biotechnol. 2009, 84, 461–467. [Google Scholar] [CrossRef]
- Cuevas, M.; García, J.F.; Cruz, N.; Sanchez, S. Generation of d-xylose by hydrothermal treatment of olives endocarps and enzymatic hydrolysis of oligosaccharides. Afinidad 2013, 70, 99–106. [Google Scholar]
- Cuevas, M.; García, J.F.; Hodaifa, G.; Sánchez, S. Oligosaccharides and sugars production from olive stones by autohydrolysis and enzymatic hydrolysis. Ind. Crops Prod. 2015, 70, 100–106. [Google Scholar] [CrossRef]
- Fernández-Bolaños, J.; Felizón, B.; Heredia, A.; Rodríguez, R.; Guillén, R.; Jiménez, A. Steam-explosion of olive stones: Hemicellulose solubilization and enhancement of enzymatic hydrolysis of cellulose. Bioresour. Technol. 2001, 79, 53–61. [Google Scholar] [CrossRef]
- Fernández-Bolaños, J.; Felizón, B.; Heredia, A.; Guillén, R.; Jiménez, A. Characterization of the lignin obtained by alkaline delignification and of the cellulose residue from steam-exploded olive stones. Bioresour. Technol. 1999, 68, 121–132. [Google Scholar] [CrossRef]
- Cano, M.E.; García-Martin, A.; Morales, P.C.; Wojtusik, M.; Santos, V.E.; Kovensky, J.; Ladero, M. Production of oligosaccharides from agrofood wastes. Fermentation 2020, 6, 31. [Google Scholar] [CrossRef] [Green Version]
- García, J.F.; Sánchez, S.; Bravo, V.; Rigal, L.; Cuevas, M. Acid hydrolysis of olive-pruning debris for d-xylose production. Collect. Czechoslov. Chem. Commun. 2008, 73, 637–648. [Google Scholar] [CrossRef]
- Overend, R.P.; Chornet, E. Fractionation of lignocellulosics by steam-aqueous pretreatments. Philos. Trans. R. Soc. London. Ser. A Math. Phys. Sci. 1987, 321, 523–536. [Google Scholar]
- Fillat, Ú.; Ibarra, D.; Eugenio, M.E.; Moreno, A.D.; Tomás-Pejó, E.; Martín-Sampedro, R. Laccases as a potential tool for the efficient conversion of lignocellulosic biomass: A review. Fermentation 2017, 3, 17. [Google Scholar] [CrossRef]
- Offei, F.; Mensah, M.; Thygesen, A.; Kemausuor, F. Seaweed bioethanol production: A process selection review on hydrolysis and fermentation. Fermentation 2018, 4, 99. [Google Scholar] [CrossRef] [Green Version]
- Chrastil, J. Enzymic product formation curves with the normal or diffusion limited reaction mechanism and in the presence of substrate receptors. Int. J. Biochem. 1988, 20, 683–693. [Google Scholar] [CrossRef]
- Carrillo, F.; Lis, M.J.; Colom, X.; López-Mesas, M.; Valldeperas, J. Effect of alkali pretreatment on cellulase hydrolysis of wheat straw: Kinetic study. Process Biochem. 2005, 40, 3360–3364. [Google Scholar] [CrossRef]
- Converti, A.; Del Borghi, M. Inhibition of the fermentation of oak hemicellulose acid-hydrolysate by minor sugars. J. Biotechnol. 1998, 64, 211–218. [Google Scholar] [CrossRef]
- García Martín, J.F.; Cuevas, M.; Bravo, V.; Sánchez, S. Ethanol production from olive prunings by autohydrolysis and fermentation with Candida tropicalis. Renew. Energy 2010, 35, 1602–1608. [Google Scholar] [CrossRef]
- Trinder, P. Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann. Clin. Biochem. Int. J. Lab. Med. 1969, 6, 24–27. [Google Scholar] [CrossRef]
- Bergmeyer, H.U.; Möllering, H. Acid acetic. In Methods of Enzymatic Analysis; Bergmeyer, H.U., Ed.; Verlag Chemie: Weinheim, Germany, 1974; pp. 1520–1528. [Google Scholar]
- Beutler, H.O. Ethanol. In Methods of Enzymatic Analysis; Bergmeyer, H.U., Ed.; Verlag Chemie: Weinheim, Germany, 1984; pp. 598–606. [Google Scholar]
- Beutler, H.O.; Becker, J. Enzymatische Bestimmung von D sorbit und Xylit in Lebensmitteln. Dtsch. Leb. 1977, 6, 182–187. [Google Scholar]
- Bravo, V.; Camacho, F.; Sánchez, S.; Castro, E. Influence of the concentrations of d-xylose and yeast extract on ethanol production by Pachysolen tannophilus. J. Ferment. Bioeng. 1995, 79, 566–571. [Google Scholar] [CrossRef]
- Yang, M.; Zhang, W.; Rosentrater, K.A. Anhydrous ammonia pretreatment of corn stover and enzymatic hydrolysis of glucan from pretreated corn stover. Fermentation 2017, 3, 9. [Google Scholar] [CrossRef] [Green Version]
- Oliva, J.M.; Negro, M.J.; Manzanares, P.; Ballesteros, I.; Chamorro, M.Á.; Sáez, F.; Ballesteros, M.; Moreno, A.D. A sequential steam explosion and reactive extrusion pretreatment for lignocellulosic biomass conversion within a fermentation-based biorefinery perspective. Fermentation 2017, 3, 15. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, M.L.; Sousa, R.; Rodríguez-Zúñiga, U.F.; Suarez, C.A.G.; Rodrigues, D.S.; Giordano, R.C.; Giordano, R.L.C. Kinetic study of the enzymatic hydrolysis of sugarcane bagasse. Braz. J. Chem. Eng. 2013, 30, 437–447. [Google Scholar] [CrossRef] [Green Version]
- Sánchez, S.; Bravo, V.; García, J.F.; Cruz, N.; Cuevas, M. Fermentation of d-glucose and d-xylose mixtures by Candida tropicalis NBRC 0618 for xylitol production. World J. Microbiol. Biotechnol. 2008, 24, 709–716. [Google Scholar] [CrossRef]
- García, J.F.; Sánchez, S.; Bravo, V.; Cuevas, M.; Rigal, L.; Gaset, A. Xylitol production from olive-pruning debris by sulphuric acid hydrolysis and fermentation with Candida tropicalis. Holzforschung 2011, 65, 59–65. [Google Scholar] [CrossRef]
- Romero, I.; Sánchez, S.; Moya, M.; Castro, E.; Ruiz, E.; Bravo, V. Fermentation of olive tree pruning acid-hydrolysates by Pachysolen tannophilus. Biochem. Eng. J. 2007, 36, 108–115. [Google Scholar] [CrossRef]
- Rorke, D.C.S.; Kana, E.B.G. Kinetics of bioethanol production from waste sorghum leaves using Saccharomyces cerevisiae BY4743. Fermentation 2017, 3, 19. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, G.; Pilcher, L.; Roslander, C.; Modig, T.; Galbe, M.; Liden, G. Dilute-acid hydrolysis for fermentation of the Bolivian straw material Paja Brava. Bioresour. Technol. 2004, 93, 249–256. [Google Scholar] [CrossRef] [PubMed]
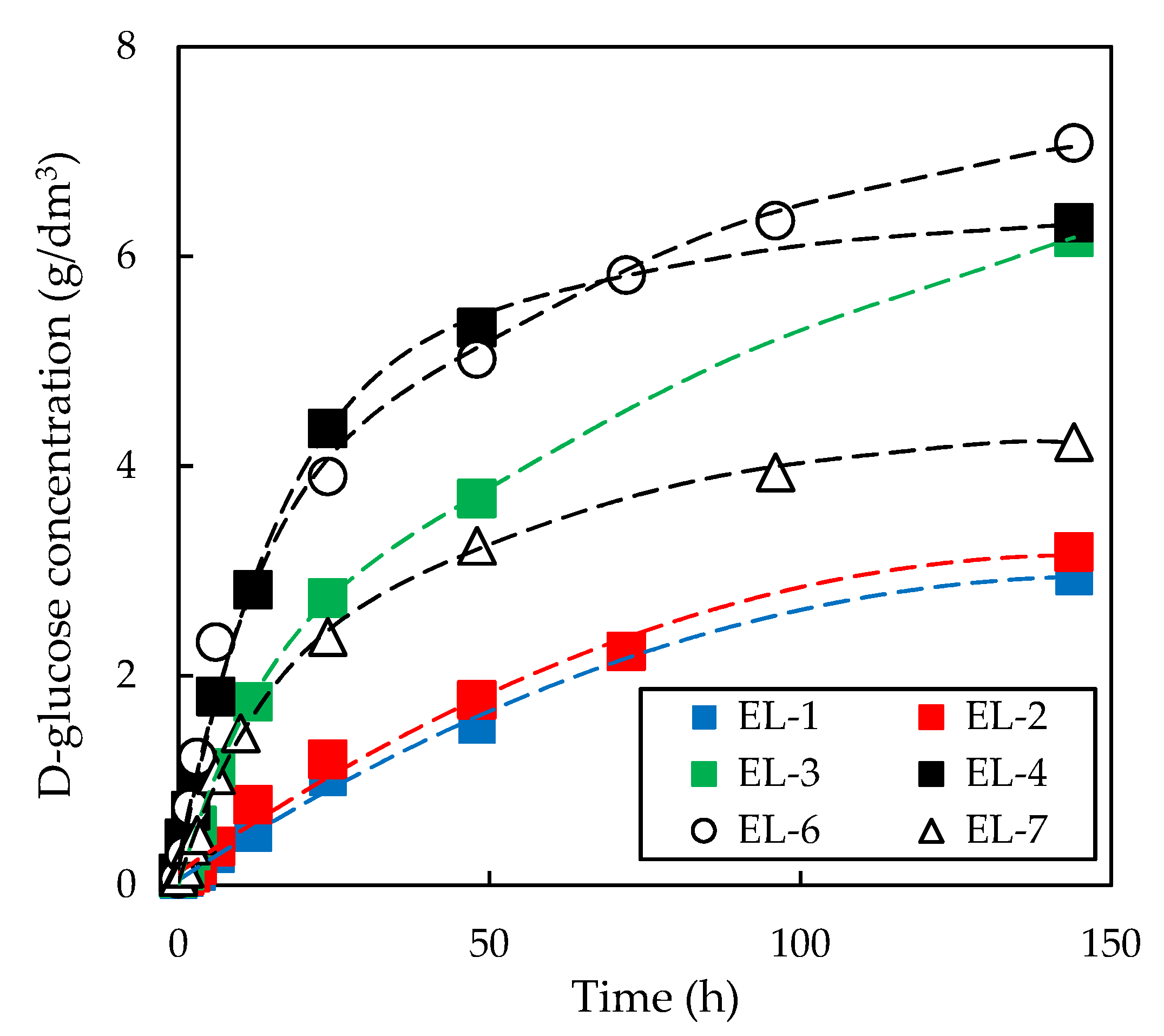



| Enzyme Loading | Relative Enzymatic Activity | ||
|---|---|---|---|
| Cellulases | β-Glucosidases | Hemicellulases | |
| EL-1 | 6 | 1 | 0 |
| EL-2 | 8 | 1 | 0 |
| EL-3 | 12 | 1 | 0 |
| EL-4 | 24 | 1 | 0 |
| EL-5 | 12 | 0 | 0 |
| EL-6 | 24 | 0 | 0 |
| EL-7 | 12 | 1 | 1 |
| Pre-Treated Solids’ Composition | |||||||
|---|---|---|---|---|---|---|---|
| LHW Pre-Treatment | Tpret (°C) | tpret (min) | Log R0 | CEL (%) | HEM (%) | AIL (%) | Enzymatic Hydrolysis |
| P175-0 | 175 | 0 | 2.73 | 26.5 | 35.4 | 34.7 | EH175-0 |
| P190-0 | 190 | 0 | 3.23 | 27.1 | 19.8 | 43.7 | EH190-0 |
| P200-0 | 200 | 0 | 3.46 | 24.1 | 26.0 | 39.6 | EH200-0 |
| P210-0 | 210 | 0 | 3.79 | 29.8 | 6.8 | 45.5 | EH210-0 |
| P225-0 | 225 | 0 | 4.39 | 38.2 | 0.0 | 42.3 | EH225-0 |
| Enzymatic Hydrolysis | CE0 (g/dm3) | kE 103 (dm3/g·min) | n | r2 |
|---|---|---|---|---|
| EH175-0 | 1.08 | 0.33 | 0.22 | 0.978 |
| EH190-0 | 1.08 | 0.20 | 0.29 | 0.995 |
| EH200-0 | 1.08 | 0.08 | 0.25 | 0.993 |
| EH210-0 | 1.09 | 0.20 | 0.60 | 0.995 |
| EH225-0 | 1.08 | 0.29 | 1.00 | 0.994 |
| Solid Composition | |||||||
|---|---|---|---|---|---|---|---|
| LHW Pre-Treatment | Tpret (°C) | tpret (min) | Log R0 | CEL (%) | HEM (%) | AIL (%) | Enzymatic Hydrolysis |
| P200-1 | 200 | 1 | 3.60 | 28.7 | 19.5 | 39.9 | EH200-1 |
| P200-2 | 200 | 2 | 3.67 | 32.2 | 13.8 | 40.7 | EH200-2 |
| P200-5 | 200 | 5 | 3.90 | 34.4 | 0.0 | 40.2 | EH200-5 |
| Enzymatic Hydrolysis | CE0 (g/dm3) | kE 103 (dm3/g·min) | n | r2 |
|---|---|---|---|---|
| EH200-1 | 1.07 | 0.22 | 0.43 | 0.991 |
| EH200-2 | 1.07 | 0.41 | 0.66 | 0.997 |
| EH200-5 | 1.08 | 0.16 | 0.38 | 0.995 |
| Enzymatic Hydrolysis | YG·102 (kg/kg) | CEL (%) | AIL (%) |
|---|---|---|---|
| EL-1 | 1.5 | 28.5 | 50.8 |
| EL-2 | 2.8 | 13.2 | 54.3 |
| EL-3 | 5.5 | 10.7 | 56.5 |
| EL-4 | 5.6 | 8.0 | 61.4 |
| EL-5 | 5.9 | 8.7 | 59.1 |
| EL-6 | 6.5 | 10.8 | 57.1 |
| EL-7 | 3.9 | 17.6 | 53.9 |
| Substrates Uptake | Ethanol Production | ||||||
|---|---|---|---|---|---|---|---|
| t (h) | (kg/kg·h) | (kg/kg·h) | (kg/kg·h) | (kg/kg·h) | t (h) | (kg/kg·h) | (kg/kg·h) |
| 10 | 0.237 | 0.319 | -- | -- | 10 | 0.100 | 0.254 |
| 20 | 0.403 | 0.319 | -- | -- | 15 | 0.153 | 0.254 |
| 100 | -- | -- | 0.091 | 0.090 | 25 | 0.218 | 0.254 |
| 150 | -- | -- | 0.062 | 0.042 | 50 | 0.028 | 0.034 |
| 200 | -- | -- | 0.027 | 0.042 | 75 | 0.019 | 0.013 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuevas, M.; García Martín, J.F.; Bravo, V.; Sánchez, S. Ethanol Production from Olive Stones through Liquid Hot Water Pre-Treatment, Enzymatic Hydrolysis and Fermentation. Influence of Enzyme Loading, and Pre-Treatment Temperature and Time. Fermentation 2021, 7, 25. https://0-doi-org.brum.beds.ac.uk/10.3390/fermentation7010025
Cuevas M, García Martín JF, Bravo V, Sánchez S. Ethanol Production from Olive Stones through Liquid Hot Water Pre-Treatment, Enzymatic Hydrolysis and Fermentation. Influence of Enzyme Loading, and Pre-Treatment Temperature and Time. Fermentation. 2021; 7(1):25. https://0-doi-org.brum.beds.ac.uk/10.3390/fermentation7010025
Chicago/Turabian StyleCuevas, Manuel, Juan F. García Martín, Vicente Bravo, and Sebastián Sánchez. 2021. "Ethanol Production from Olive Stones through Liquid Hot Water Pre-Treatment, Enzymatic Hydrolysis and Fermentation. Influence of Enzyme Loading, and Pre-Treatment Temperature and Time" Fermentation 7, no. 1: 25. https://0-doi-org.brum.beds.ac.uk/10.3390/fermentation7010025






