Chromatic Change in Copper Oxide Layers Irradiated with Low Energy Ions
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Peña-Rodríguez, O.; Crespillo, M.L.; Díaz-Nuñez, P.; Perlado, J.M.; Rivera, A.; Olivares, J. In situ monitoring the optical properties of dielectric materials during ion irradiation. Opt. Mater. Express 2016, 6, 734–742. [Google Scholar] [CrossRef] [Green Version]
- Rivera, A.; Olivares, J.; Prada, A.; Crespillo, M.L.; Caturla, M.J.; Bringa, E.M.; Perlado, J.M.; Peña-Rodríguez, O. Permanent modifications in silica produced by ion-induced high electronic excitation: Experiments and atomistic simulations. Sci. Rep. 2017, 7, 10641. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.; Li, B. Origins of a damage-induced green photoluminescence band in fused silica revealed by time-resolved photoluminescence spectroscopy. Opt. Mater. Express 2017, 7, 2888–2898. [Google Scholar] [CrossRef]
- Crespillo, M.L.; Graham, J.T.; Zhang, Y.; Weber, W.J. In-situ luminescence monitoring of ion-induced damage evolution in SiO2 and Al2O3. J. Lumin. 2016, 172, 208–218. [Google Scholar] [CrossRef] [Green Version]
- Bandourko, V.; Umeda, N.; Plaksin, O.; Kishimoto, N. Heavy-ion-induced luminescence of amorphous SiO2 during nanoparticle formation. Nucl. Instrum. Meth. B 2005, 230, 471–475. [Google Scholar] [CrossRef]
- Forck, P.; Andre, C.; Becker, F.; Haseitl, R.; Reiter, A.; Walasek-Höhne, B.; Krishnakumar, R.; Ensinger, W. Scintillation Screen Investigations for High Energy Heavy Ion Beams at GSI. In Proceedings of the DIPAC2011, Hamburg, Germany, 16–18 May 2011; pp. 170–172. [Google Scholar]
- Johnson, C.D. The Development and Use of Alumina Ceramic Fluorescent; CERN/PS/90-12(AR); European Laboratory for Particle Physics: Geneva, Switzerland, 1990. [Google Scholar]
- Takahiro, K.; Terai, A.; Kawatsura, K.; Naramoto, H.; Yamam, S.; Tsuchiya, B.; Nagata, S.; Nishiyama, F. Rutherford backscattering spectrometry of electrically charged targets: Elegant technique for measuring charge-state distribution of backscattered Ions. Jpn. J. Appl. Phys. 2006, 45, 1823–1825. [Google Scholar] [CrossRef]
- Parajuli, R.K.; Kada, W.; Kawabata, S.; Matsubara, Y.; Sakai, M.; Miura, K.; Satoh, T.; Koka, M.; Yamada, N.; Kamiya, T.; et al. Ion-Beam-Induced Luminescence Analysis of β-SiAlON:Eu Scintillator under Focused Microbeam Irradiation. Sens. Mater. 2016, 28, 837–844. [Google Scholar]
- Warren, A.J.; Thomas, C.B.; Reehal, H.S.; Stevens, P.R.C. A study of the luminescent and electrical characteristics of films of ZnS doped with Mn. J. Lumin. 1983, 28, 147–162. [Google Scholar] [CrossRef]
- Calusi, S.; Colombo, E.; Giuntini, L.; Giudice, A.L.; Manfredotti, C.; Massi, M.; Pratesi, G.; Vittone, E. The ionoluminescence apparatus at the LABEC external microbeam facility. Nucl. Instrum. Meth. B 2008, 266, 2306–2310. [Google Scholar] [CrossRef]
- Feldmann, C.; Jüstel, T.; Ronda, C.R.; Schmidt, P.J. Inorganic Luminescent Materials: 100 Years of Research and Application. Adv. Funct. Mater. 2003, 13, 511–516. [Google Scholar] [CrossRef]
- Kleiman, J.; Iskanderova, Z.; Best, C.; Dennison, J.R.; Wood, B. Long-term stability of ion-beam treated space polymers in geo-simulated environment. In Proceeding of the 14th ISMSE $ 12th ICPMSE, Biarritz, France, 1–5 October 2018. [Google Scholar]
- Plis, E.A.; Engelhart, D.P.; Cooper, R.; Johnston, W.R.; Ferguson, D.; Hoffmann, R. Review of Radiation-Induced Effects in Polyimide. Appl. Sci. 2019, 9, 1999. [Google Scholar] [CrossRef] [Green Version]
- Matienzo, L.J.; Emmi, F.; Van Hart, D.C.; Gall, T.P. Interactions of high-energy ion beams with polyimide films. J. Vac. Sci. Technol. A 1989, 7, 1784–1789. [Google Scholar] [CrossRef]
- Computer Simulation of RBS, ERDA, NRA, MEIS and PIGE by Matej Mayer. Available online: https://home.mpcdf.mpg.de/~mam/Version6.html (accessed on 5 December 2020).
- Ziegler, J.F.; Ziegler, M.D.; Biersack, J.P. SRIM—The stopping and range of ions in matter (2010). Nucl. Instrum. Meth. B 2010, 268, 1818–1823. [Google Scholar] [CrossRef] [Green Version]
- Tanuma, S.; Powell, C.J.; Penn, D.R. Calculations of electron inelastic mean free paths. II. Data for 27 elements over the 50–2000 eV range. Surf. Interface Anal. 1991, 17, 911–926. [Google Scholar] [CrossRef]
- Kawai, J.; Tsuboyama, S.; Ishizu, K.; Miyamura, K.; Saburi, M. Covalency of copper complex determined by Cu 2p X-ray photoelectron spectrometry. Anal. Sci. 1991, 7, 337–340. [Google Scholar] [CrossRef] [Green Version]
- Pawar, S.M.; Kim, J.; Inamdar, A.I.; Woo, H.; Jo, Y.; Pawar, B.S.; Cho, S.; Kim, H.; Im, H. Multi-functional reactively-sputtered copper oxide electrodes for supercapacitor and electro-catalyst in direct methanol fuel cell applications. Sci. Rep. 2016, 6, 21310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Végh, J. The Shirley background revised. J. Electron Spectrosc. Relat. Phenom. 2006, 151, 159–164. [Google Scholar] [CrossRef]
- Anderson, C.R.; Lee, R.N. Comparison of APS and FRESCA core level binding energy measurements. J. Vac. Sci. Technol. 1982, 20, 617–621. [Google Scholar] [CrossRef]
- Seah, M.P.; Gilmore, I.S.; Beamson, G. XPS: Binding energy calibration of electron spectrometers 5—re-evaluation of the reference energies. Surf. Interface Anal. 1998, 26, 642–649. [Google Scholar] [CrossRef]
- Bird, R.J.; Swift, P. Energy calibration in electron spectroscopy and the re-determination of some reference electron binding energies. J. Electron Spectrosc. Relat. Phenom. 1980, 21, 227–240. [Google Scholar] [CrossRef]
- McIntyre, N.S.; Cook, M.G. X-ray photoelectron studies on some oxides and hydroxides of cobalt, nickel, and copper. Anal. Chem. 1975, 47, 2208–2213. [Google Scholar] [CrossRef]
- Schön, G. ESCA studies of Cu, Cu2O and CuO. Surf. Sci. 1973, 35, 96–108. [Google Scholar] [CrossRef]
- Losev, A.; Rostov, K.; Tyuliev, G. Electron beam induced reduction of CuO in the presence of a surface carbonaceous layer: An XPS/HREELS study. Surf. Sci. 1989, 213, 564–579. [Google Scholar] [CrossRef]
- Deroubaix, G.; Marcus, P. X-ray photoelectron spectroscopy analysis of copper and zinc oxides and sulphides. Surf. Interface Anal. 1992, 18, 39–46. [Google Scholar] [CrossRef]
- Haber, J.; Machej, T.; Ungier, L.; Ziółkowski, J. ESCA studies of copper oxides and copper molybdates. J. Solid State Chem. 1978, 25, 207–218. [Google Scholar] [CrossRef]
- Gaarenstroom, S.M.; Winograd, N. ESCA spectra of cadmium and silver oxides. J. Chem. Phys. 1977, 67, 3500–3506. [Google Scholar] [CrossRef]
- Tobin, J.P.; Hirschwald, W.; Cunningham, J. XPS and XAES studies of transient enhancement of Cu1 at CuO surfaces during vacuum outgassing. Appl. Surf. Sci. 1983, 16, 441–452. [Google Scholar] [CrossRef]
- Biesinger, M.C. Advanced analysis of copper X-ray photoelectron spectra. Surf. Interface Anal. 2017, 49, 1325–1334. [Google Scholar] [CrossRef]
- Panzner, G.; Egert, B.; Schmidt, H.P. The stability of CuO and Cu2O surfaces during argon sputtering studied by XPS and AES. Surf. Sci. 1985, 151, 400–408. [Google Scholar] [CrossRef]
- Dementyeva, M.M.; Prikhodko, K.E.; Gurovich, B.A.; Bukina, Z.V.; Komarov, D.A.; Kutuzov, L.V. Phase transitions in copper oxide thin films under proton irradiation. IOP Conf. Ser. Mater. Sci. Tecnol. 2017, 256, 012020. [Google Scholar] [CrossRef] [Green Version]
- Pan, J.; Yang, C.; Gao, Y. Investigations of Cuprous Oxide and Cupric Oxide Thin Films by Controlling the Deposition Atmosphere in the Reactive Sputtering Method. Sens. Mater. 2016, 28, 817–824. [Google Scholar]
- Gevorkyan, V.A.; Reymers, A.E.; Nersesyan, M.N.; Arzakantsyan, A.M. Characterization of Cu2O thin films prepared by evaporation of CuO powder. J. Phys. Conf. Ser. 2012, 350, 012027. [Google Scholar] [CrossRef]
- Goua, L.; Murphy, C.J. Controlling the size of Cu2O nanocubes from 200 to 25 nm. J. Mater. Chem. 2004, 14, 735–738. [Google Scholar] [CrossRef]
- Butte, S.M.; Waghuley, S.A. Optical properties of Cu2O and CuO. AIP Conf. Proc. 2020, 2220, 020093. [Google Scholar]
- Fredj, N.; Burleigh, T.D. Transpassive Dissolution of Copper and Rapid Formation of Brilliant Colored Copper Oxide Films. J. Electrochem. Soc. 2011, 158, C104–C110. [Google Scholar] [CrossRef] [Green Version]
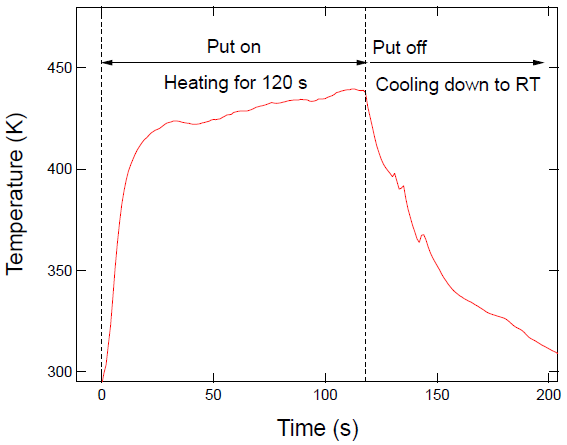

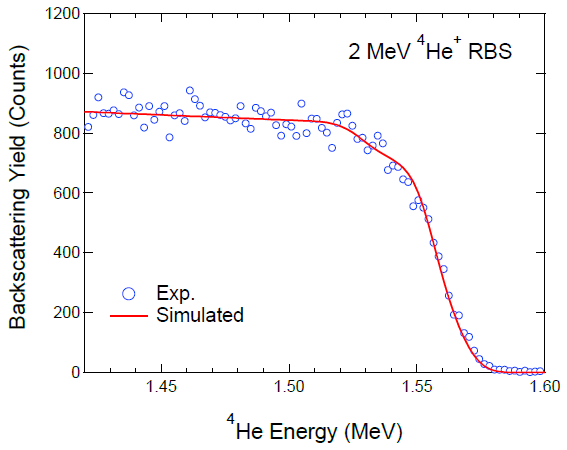



| Samples | Compositions (%) | |||
|---|---|---|---|---|
| Cu | Cu2O | CuO | Cu(OH)2 | |
| Unirradiated | 73.8 | 16.5 | 9.7 | |
| (65.7) 1 | (8.1) 1 | |||
| Irradiated | 94.1 | 5.5 | 0.4 | |
| (65.7) 1 | (28.4) 1 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kobayashi, T.; Nishiyama, F.; Takahiro, K. Chromatic Change in Copper Oxide Layers Irradiated with Low Energy Ions. Quantum Beam Sci. 2021, 5, 7. https://0-doi-org.brum.beds.ac.uk/10.3390/qubs5010007
Kobayashi T, Nishiyama F, Takahiro K. Chromatic Change in Copper Oxide Layers Irradiated with Low Energy Ions. Quantum Beam Science. 2021; 5(1):7. https://0-doi-org.brum.beds.ac.uk/10.3390/qubs5010007
Chicago/Turabian StyleKobayashi, Takuya, Fumitaka Nishiyama, and Katsumi Takahiro. 2021. "Chromatic Change in Copper Oxide Layers Irradiated with Low Energy Ions" Quantum Beam Science 5, no. 1: 7. https://0-doi-org.brum.beds.ac.uk/10.3390/qubs5010007





