Precision Medicine Treatment in Acute Myeloid Leukemia Is Not a Dream
Abstract
:1. Introduction
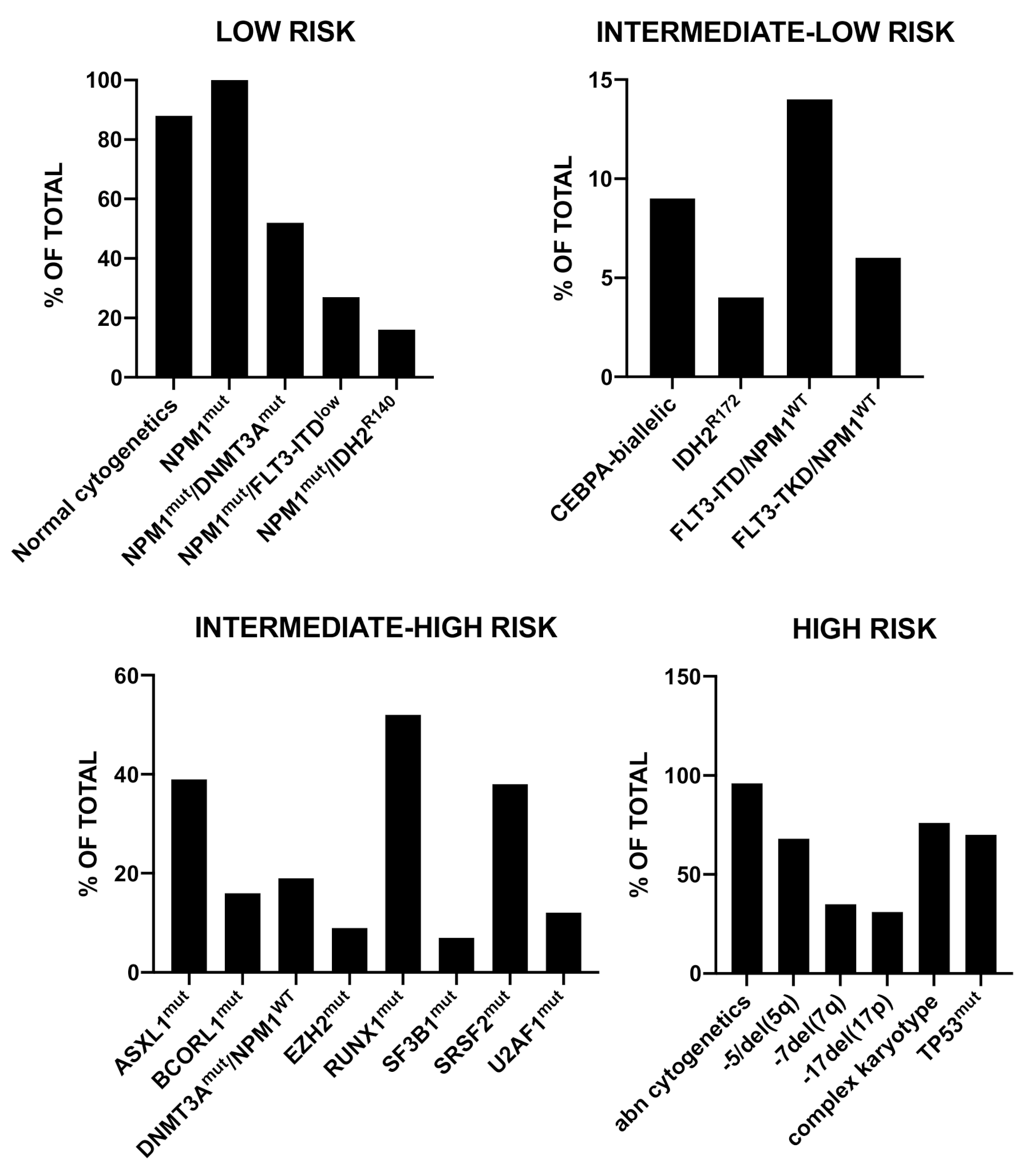
2. The Fundamental Contribution of Precision Medicine to a More Rational and Predictive Risk Stratification of AML Patients
3. The Contribution of the Machine Learning Approach to Improve the Assessment of AML Diagnosis and Prognosis
4. An Integrated Approach Is Required for the Development of Personalized Medicine in AML
5. Challenges in Clinical Development of Targeted Therapies for AML
6. Personalized Therapy for AML Patients Is Feasible
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cancer Genome Atlas Research Network; Ley, T.J.; Miller, C.; Ding, L.; Raphael, B.J.; Mingall, A.J.; Robertson, A.G.; Hoadley, A.S.K.; Triche, T.J.; Laird, P.W.; et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N. Engl. J. Med. 2013, 368, 20959–22074. [Google Scholar]
- Hou, H.A.; Lin, C.C.; Chou, W.C.; Liu, C.Y.; Chen, C.Y.; Tang, J.L.; Lai, Y.J.; Tseng, M.H.; Huang, C.F.; Chiang, Y.C.; et al. Integration of cytogenetic and molecular alterations in risk stratification of 318 patients with de novo non-M3 acute myeloid leukemia. Leukemia 2014, 28, 50–58. [Google Scholar] [CrossRef]
- Papaemmanuil, E.; Gerstung, M.; Bullinger, L.; Gaidzik, K.; Paschka, P.; Roberts, N.D.; Potter, N.E.; Hauser, M.; Thol, F.; Bolli, N.; et al. Genomic classification and prognosis in acute myeloid leukemia. N. Engl. J. Med. 2016, 374, 2209–2221. [Google Scholar] [CrossRef] [PubMed]
- Bullinger, L.; Dohner, K.; Dohner, H. Genomics of acute myeloid leukemia diagnosis and pathways. J. Clin. Oncol. 2017, 35, 934–946. [Google Scholar] [CrossRef]
- Moarii, M.; Papaemmanuil, E. Classification and risk assessment in AML: Integrating cytogenetics and molecular profiling. Hematol. Am. Soc. Hematol. Educ. Program. 2017, 8, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kishtagari, A.; Levine, R.L.; Viny, A.D. Driver mutations in acute myeloid leukemia. Curr. Opin. Hematol. 2020, 27, 9–57. [Google Scholar] [CrossRef]
- Short, N.J.; Konopleva, M.; Kadia, T.M.; Borthakur, G.; Ravandi, F.; DiNardo, C.D.; Daver, N. Advances in the treatment of acute myeloid leukemia: New drugs and new challenges. Cancer Discov. 2020, 10, 506–525. [Google Scholar] [CrossRef] [Green Version]
- Daver, N.; Wei, A.H.; Pollyea, D.A.; Fathi, A.T.; Vyas, P.; DiNardo, C.D. New directions for emerging therapies in acute myeloid leukemia: The next chapter. Blood Cancer J. 2020, 10, 107. [Google Scholar] [CrossRef]
- Yu, J.; Jiang, P.; Sun, H.; Zhang, X.; Jiang, Z.; Li, Y.; Song, Y. Advances in targeted therapy for acute myeloid leukemia. Biomarker Res. 2020, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Samra, B.; Knopleva, M.; Isidori, A.; Daver, N.; DiNardo, C. Venetoclax-based combinations in acute myeloid leukemia: Current evidence and future directions. Front. Oncol. 2020, 10, 562558. [Google Scholar] [CrossRef] [PubMed]
- Ganget, N.; Tefferi, A. Venetoclax-based chemotherapy in acute and chronic myeloid neoplasms: Literature survey and practice points. Blood Cancer J. 2020, in press. [Google Scholar] [CrossRef]
- Marando, L.; Huntly, B.J.P. Molecular landscape of acute myeloid leukemia: Prognostic and therapeutic implications. Curr. Oncol. Rep. 2020, 22, 61. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.A.; Tien, H.F. Genomic landscape in acute myeloid leukemia and its implications in risk classification and targeted therapies. J. Biomed. Sci. 2020, 27, 81. [Google Scholar] [CrossRef] [PubMed]
- Dohner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Buchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef] [Green Version]
- O’Donnell, M.R.; Tallman, M.S.; Abboud, C.N.; Altman, J.K.; Appelbaum, F.R.; Arber, D.A. Acute myeloid leukemia, version 3.2017, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2017, 15, 926–957. [Google Scholar] [CrossRef]
- Arber, D.A.; Orazi, A.; Hasserjian, R.; Thiede, J.; Borowitz, M.J.; Le Beau, M.M. The 2016 revision in the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 1391–2405. [Google Scholar] [CrossRef]
- Herold, T.; Rothenberg-Thurley, M.; Grumwald, V.V.; Janke, H.; Goerlich, D.; Sauerland, M.S.; Kostandin, N.P.; Dufour, A.; Schneider, S.; Neusser, M.; et al. Validation and refinement of the revised 2017 European Leukemia Net genetic risk stratification of acute myeloid leukemia. Leukemia 2020, in press. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, M.; Lindberg, J.; Kelvebring, D.; Nillsson, C.; Mer, A.S.; Rantalaien, M.; Lehmann, S.; Gronberg, H. Validation of risk stratification models in acute myeloid leukemia using sequencing-based molecular profiling. Leukemia 2017, 31, 2029–2036. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Lindberg, J.; Klevebring, D.; Nillsson, C.; Lehmann, S.; Gronberg, H.; Rantalainen, M. Development and validation of a novel RNA sequencing-based prognostic score for acute myeloid leukemia. J. Natl. Cancer Inst. 2018, 110, 1094–1101. [Google Scholar] [CrossRef]
- Ma, T.T.; Lin, X.J.; Cheng, W.Y.; Xue, Q.; Wang, S.Y.; Liu, F.J.; Yan, H.; Zhu, Y.M.; Shen, Y. Development and validation of a prognostic model for adult patients with acute myeloid leukemia. EBioMedicine 2020, 62, 103126. [Google Scholar] [CrossRef]
- Radakovich, N.; Nagy, M.; Nazha, A. Machine learning in haematological malignancies. Lancet Haematol. 2020, 7, e541–e550. [Google Scholar] [CrossRef]
- Radakovich, N.; Cortese, M.; Nazha, A. Acute myeloid leukemia and artificial intelligence, algorithms and new scores. Best Pract. Res. Clin. Hemat. 2020, 33, 101192. [Google Scholar] [CrossRef]
- Goecks, J.; Jalili, V.; Heiser, L.M.; Gray, J.W. How machine learning will transform biomedicine. Cell 2020, 181, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Eckardt, J.N.; Bornhauser, M.; Wendt, K.; Middeke, J.M. Application of machine learning in the management of acute myeloid leukemic: Current practice and future prospects. Blood Adv. 2020, 4, 6077–6085. [Google Scholar] [CrossRef] [PubMed]
- Beau Hilton, C.; Meggendorfer, M.; Sekeres, M.A.; Shreve, J.; Radakovich, N.; Rouphail, Y.; Walter, W.; Hutter, S.; Padron, E.; Savona, M.R.; et al. Geno-clinical model for the diagnosis of bone marrow myeloid neoplasms. Blood 2019, 134 (Suppl. S1), 4238. [Google Scholar] [CrossRef]
- Morita, K.; Wang, F.; Makishima, H.; Yan, Y.; Yoshizato, T.; Yoshida, K.; Przychodzen, B.P.; Patel, K.; Bueso-Ramos, C.E.; Gumbs, C.; et al. Pan-myeloid leukemia analysis: Machine learning-based approach to predict phenotype and clinical outcomes using mutation data. Blood 2018, 132 (Suppl. 1), 1801. [Google Scholar] [CrossRef]
- Radakovich, N.; Malcovati, L.; Meggendorfer, M.; Sekeres, M.A.; Shreve, J.; Beau Hilton, C.; Rouphail, Y.; Walter, W.; Hutter, S.; Gallì, A.; et al. Henotype-phenotype correlations in patients with myeloid malignancies using explainable artificial intelligence. Blood 2020, 138 (Suppl. S1), 31–32. [Google Scholar] [CrossRef]
- Gerstung, M.; Papaemmanuil, E.; Martincorena, I.; Bullinger, L.; Gaidzik, V.I.; Paschka, P.; Heuser, M.; Thol, F.; Bolli, N.; Ganly, P.; et al. Precision oncology for acute myeloid leukemia using a knowledge bank approach. Nat. Genet. 2017, 49, 332–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleming, S.; Tsai, C.H.; Dohner, H.; Dohner, K.; Papaemmanuil, E.; Tien, H.F.; Reynolds, J.; Wei, A.H.; Hou, H.A. Use of machine-learning in 2074 cases of acute myeloid leukemia for genetic risk profiling. Blood 2019, 134 (Suppl. S1), 1392. [Google Scholar] [CrossRef]
- Shreve, J.; Meggendorfer, M.; Awada, H.; Mukherjee, S.; Walter, W.; Hutter, S.; Makhoul, A.; Beau Hilton, C.; Radakovich, N.; Nagata, Y.; et al. A personalized prediction model to risk stratify patients with acute myeloid leukemia (AML) using artificial intelligence. Blood 2019, 134 (Suppl. S1), 2091. [Google Scholar] [CrossRef]
- Awada, H.; Durmaz, A.; Gurnari, C.; Kishtagari, A.; Meggendorfer, M.; Kerr, C.M.; Kuzmanoviuc, T.; Durrani, J.; Nagata, Y.; Rdivoyevitch, T.; et al. The application of machine learning to improve the subclassification and prognostication of acute myeloid leukemia. Blood 2020, 136 (Suppl. S1), 28. [Google Scholar] [CrossRef]
- Siddiqui, N.S.; Klein, A.; Godara, A.; Varga, C.; Buchsbaum, R.J.; Hughes, M.C. Supervised machine learning algorithms using patient related factors to predict in-hospital mortality following acute myeloid leukemia therapy. Blood 2019, 134 (Suppl. S1), 3435. [Google Scholar] [CrossRef]
- Shaikh, A.F.; Kakirde, C.; Dhamne, C.; Bhanshe, P.; Joshi, S.; Chaudhary, S.; Chatterjee, G.; Tembhare, P.; Prasad, M.; Roy Moulik, N.; et al. Machine learning derived genomics driven prognostication for acute myeloid leukemia with RUNX1-RUNX1T1. Leuk. Lymphoma 2020, 61, 3154–3160. [Google Scholar] [CrossRef]
- Patkar, N.; Shaikh, A.F.; Kakirde, C.; Nathany, S.; Ramesh, H.; Bhanshe, P.; Joshi, S.; Chaudhary, S.; Kannan, S.; Khizer, S.H.; et al. A novel machine-learning-derived genetic score correlates with measurable residual disease and is highly predictive of outcome in acute myeloid leukemia with mutated NPM1. Blood Cancer J. 2019, 9, 79. [Google Scholar] [CrossRef] [Green Version]
- Bullinger, L.; Dohner, K.; Bair, E.; Frohling, S.; Schlenk, R.F.; Tibshirani, R.; Dohner, H.; Pollack, J.R. Use of gene-expression profiling to identify prognostic subclones in adult acute myeloid leukemia. N. Engl. J. Med. 2004, 350, 1605–1616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Herold, T.; He, C.; Valk, P.; Chen, P.; Jurinovic, V.; Mansmann, U.; Radmacher, M.; Maharry, K.; Sun, M.; et al. Identification of a 24-gene prognostic signature that improves the European LeukemiaNet risk classification of acute myeloiud leukemia: An international collaborative study. J. Clin. Oncol. 2013, 31, 1172–1181. [Google Scholar] [CrossRef] [Green Version]
- Marcucci, G.; Yan, P.; Maharry, K.; Frankhouser, D.; Nicolet, D.E.; Metzler, K.H.; Kohlschmidt, J.; Mrozek, K.; Wu, Y.Z.; Bussi, D.; et al. Epigenetics meets genetics in acute myeloid leukemia: Clinical impact of a novel seven-gene score. J. Clin. Oncol. 2013, 32, 548–556. [Google Scholar] [CrossRef]
- Ng, S.; Mitchell, A.; Kennedy, J.; Chen, W.C.; McLeod, J.; Ibrahimova, N.; Arruda, A.; Popescu, A.; Gupta, V.; Schimmer, A.D.; et al. A 17-gene stemness score for rapid determination of risk in acute leukemia. Nature 2016, 540, 433–437. [Google Scholar] [CrossRef]
- Warnat-Herresthal, S.; Perrakis, K.; Taschler, B.; Becker, M.; Babler, K.; Beyer, M.; Gunther, P.; Schulte-Schrepping, J.; Seep, L.; Klee, K.; et al. Scalable prediction of acute myeloid leukemia using high-dimensional machine learning and blood transcriptomics. iScience 2020, 23, 100780. [Google Scholar] [CrossRef] [Green Version]
- Wagner, S.; Vadakekolathu, J.; Taisan, S.K.; Altmann, H.; Bornhauser, M.; Pockley, A.G.; Ball, G.R.; Rutella, S. A parsimonius 3-gene signature predicts clinical outcomes in an acute myeloid leukemia multicohort study. Blood Adv. 2019, 3, 1330–1340. [Google Scholar] [CrossRef] [Green Version]
- Rouphail, Y.; Radakovich, N.; Shreve, J.; Mukherejee, S.; Jha, B.K.; Maciejewski, J.P.; Sekeres, M.A.; Nazha, A. Personalized transcriptomic analyses identify unique signatures that correlate with genomic subtypes in acute myeloid leukemia (AML) using explainable artificial intelligence. Blood 2020, 136 (Suppl. S1), 33–34. [Google Scholar] [CrossRef]
- Gandelman, J.S.; Byrne, M.T.; Mistry, A.M.; Polikowsky, H.G.; Diggins, K.E.; Chen, H.; Lee, S.J.; Arora, M.; Cutler, C.; Flowers, M.; et al. Machine learning reveals chronic graft-versus host disease phenotypes and stratifies survival after stem cell transplant for hematologic malignancies. Haematologica 2019, 104, 189–196. [Google Scholar] [CrossRef] [Green Version]
- Choi, E.J.; Lee, J.H.; Park, J.H.; Park, H.S.; Lee, J.H.; Lee, Y.; Kang, Y.A.; Jeon, M.; Woo, J.M.; Kang, H.; et al. Machine learning-based approach to predict survival after allogeneic hematopoietic cell transplantation in hematologic malignancies. Blood 2020, 136 (Suppl. S1), 33–34. [Google Scholar]
- Nazha, A.; Hu, Z.H.; Wang, T.; Lindsley, R.C.; Abdel-Azim, H.; Aljurf, M.; Bacher, U.; Bashley, A.; Cahn, J.Y.; Cerny, J.; et al. A personalized prediction model for outcomes after allogeneic hematopoietic cell transplant in patients with myelodysplastic syndromes. Biol. Blood Marrow Transplant. 2020, 26, 2139–2146. [Google Scholar] [CrossRef]
- Lee, S.; Celik, S.; Logsdon, B.A.; Lundberg, S.M.; Martins, T.J.; Oehler, V.G.; Estey, E.H.; Miller, C.P.; Chien, S.; Dai, J.; et al. A machine learning approach to integrate big data for precision medicine in acute myeloid leukemia. Nat. Commun. 2018, 9, 42. [Google Scholar] [CrossRef]
- Chen, X.; Chen, H.Y.; Chen, Z.D.; Gong, J.N.; Chen, C.Y.N. A novel artificial intelligence protocol for finding potential inhibitors of acute myeloid leukemia. J. Mater. Chem. B Mater Biol. Med. 2020, 8, 2063–2081. [Google Scholar] [CrossRef]
- Cutler, G.; Fridman, J.S. A machine-learning analysis suggests that FLX925, a FLT3/CDK4/6 kinase inhibitor, is potent against FLT3-wild type tumors via its CDK4/6 activity. Blood 2016, 128, 3520. [Google Scholar] [CrossRef]
- Ko, B.S.; Wang, Y.F.; Li, J.L.; Weng, P.F.; Hsu, S.C.; Hou, H.A.; Huang, H.H.; Yao, M.; Lin, C.T.; Liu, J.H.; et al. Clinically validated machine learning algorithm for detecting residual diseases with multicolor flow cytometry analysis in acute myeloid leukemia and myelodysplastic syndrome. EBioMedicine 2018, 37, 91–100. [Google Scholar] [CrossRef] [Green Version]
- Haferlach, T.; Schmidts, I. The power and potential of integrated diagnostics in acute myeloid leukemia. Br. J. Haematol. 2020, 188, 36–48. [Google Scholar] [CrossRef]
- Letai, A. Functional precision cancer medicine -moving beyond pure genomic. Nat. Med. 2017, 23, 1028–1035. [Google Scholar] [CrossRef]
- Tyner, J.W.; Tognon, C.E.; Bottomly, D.; Wilmot, B.; Kurtz, S.E.; Savage, S.L.; Long, N.; Schultz, A.R.; Traer, E.; Abel, M.; et al. Functional genomic landscape of acute myeloid leukaemia. Nature 2018, 562, 526–531. [Google Scholar] [CrossRef]
- Kurtz, S.E.; Eide, C.A.; Kaempf, A.; Khanna, V.; Savage, S.L.; Rofelty, A.; English, I.; Ho, H.; Pandya, R.; Bolosky, W.J.; et al. Molecularly targeted drug combinations demonstrate selective effectiveness for myeloid- and lymphoid- derived hematologic malignancies. Proc. Natl. Acad. Sci. USA 2017, 114, E7554–E7563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, L.; Tong, Y.; Straube, J.; Zhao, J.; Gao, Y.; Bai, P.; Li, J.; Wang, J.; Wang, H.; Wang, X.; et al. Ex-vivo drug testing predicts chemosensitivity in acute myeloid leukemia. J. Leukoc. Biol. 2020, 107, 859–870. [Google Scholar] [CrossRef]
- Erkers, T.; Seashore-Ludlow, B.; Struyf, N.; Marabita, F.; James, T.; Malani, D.; Vesterlund, M.; Pawitan, Y.; Lehmann, S.; Ostling, P.; et al. High-throughput functional ex-vivo drug testing and multi-omics profiling in patients with acute myeloid leukemia. Blood 2019, 134 (Suppl. S1), 4641. [Google Scholar] [CrossRef]
- Martinez-Cuadròn, D.; Gil, C.; Serrano, J.; Rodriguez, G.; Perez-Oteyza, J.; Garcia-Boyero, R.; Jimenez-Bravo, S.; Vives, S.; Vidriales, M.B.; Lavilla, E.; et al. A precision medicine test predicts clinical response after idarubicin and cytarabine induction therapy in AML patients. Leukemia Res. 2019, 76, 1–10. [Google Scholar] [CrossRef]
- Simon, L.; Lavallée, V.P.; Bordeleau, M.E.; Krosl, J.; Baccelli, I.; Boucher, G.; Lenhertz, B.; Chagraoul, J.; MacRae, T.; Ruel, R.; et al. Chemogenomic landscape of RUNX1-mutated AML reveals importance of RUNX1 allele dosage in genetics and glucocorticoid sensitivity. Clin. Cancer Res. 2017, 23, 6969–6983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moison, C.; Lavallée, J.P.; Thiollier, C.; Spinella, J.F.; Boivin, I.; Lemiux, S.; Marinier, A.; Hébert, J.; Savageau, G. Chemogenomic profiling of complex karyotype AML reveals a novel susceptibility to G2/M checkpoint inhibition mediated by HMGA2 overexpression. Blood 2018, 132 (Suppl. S1), 3925. [Google Scholar] [CrossRef]
- Zhang, H.; Wilmot, B.; Bottomly, D.; Kurtz, S.E.; Eide, C.A.; Damnernsawad, A.; Romine, K.; Patel, S.; Druker, B.J.; Mcweeney, S.K.; et al. Biomarkers predicting venetoclax sensitivity and strategies for venetoclax combination treatment. Blood 2018, 132 (Suppl. S1), 175. [Google Scholar] [CrossRef]
- Zhang, H.; Nakauchi, Y.; Kohnkie, T.; Stafford, M.; Bottomly, D.; Thomas, R.; Wilmot, B.; McWeeney, S.K.; Majketi, R.; Tyner, J.W. Integrated analysis of patients samples identifies biomarkers for venetoclax efficacy and combination strategies in acute myeloid leukemia. Nat. Cancer 2020, 1, 826–839. [Google Scholar] [CrossRef]
- Stevens, B.M.; Jones, C.L.; Winters, A.; Gugan, J.; Abbott, D.; Savona, M.R.; Fesik, S.W.; Pollyea, D.A.; Jordan, C.T. PTPN11 mutations confer unique metabolic properties and increase resistance to venetoclax and azacytidine in acute myeloid leukemia. Blood 2018, 132 (Suppl. S1), 909. [Google Scholar] [CrossRef]
- Alfayez, M.; Issa, G.C.; Patel, K.P.; Wang, F.; Wang, X.; Short, N.J.; Cortes, J.E.; Kadia, T.; Ravandi, F.; Pierce, S.; et al. The clinical impact of PTPN11 mutations in adults with acute myeloid leukemia. Leukemia 2020, in press. [Google Scholar]
- Pei, S.; Pollyea, D.A.; Gustafson, A.; Stevens, B.M.; Minhajuddin, M.; Fu, R.; Riemondy, K.A.; Gillen, A.E.; Sheridan, R.M.; Kim, J.; et al. Monocytic subclones confer resistance to venetoclax-based therapy in patients with acute myeloid leukemia. Cancer Discov. 2020, 10, 536–551. [Google Scholar] [CrossRef]
- Spinner, M.A.; Alishin, A.; Santaguida, M.T.; Schaffert, S.A.; Zehnder, J.L.; Patterson, A.S.; Gekas, C.; Heiser, D.; Greenberg, P.L. Wx vivo drug screening defines novel drug sensitivity patterns informing personalized therapy in myeloid neoplasms. Blood Adv. 2020, 4, 2768–2778. [Google Scholar] [CrossRef]
- Snijder, B.; Vladimer, G.I.; Krall, N.; Miura, K.; Schmolke, A.S.; Kornauth, C.; de la Fuente, I.O.L.; Choi, H.S.; van der Kouwe, E.; Gultekin, S.; et al. Image-based ex-vivo drug screening for patients with aggressive haematological malignancies: Interim results from a single-arm, open-label, pilot study. Lancet Haematol. 2017, 4, e595–e606. [Google Scholar] [CrossRef] [Green Version]
- Collignon, A.; Hospital, M.A.; Montersino, C.; Courtier, F.; Charbonnier, A.; Saillard, C.; D’Incan, E.; Mohty, B.; Guille, A.; Adelaide, J.; et al. A chemogenomic approach to identify personalized therapy for patients with relapse or refractory acute myeloid leukemia: Results of a prospective feasibility study. Blood Cancer J. 2020, 10, 64. [Google Scholar] [CrossRef]
- Stone, R.M.; Mandrekar, S.J.; Sanford, B.L.; Laumann, K.; Geyer, S.; Bloomfield, C.D.; Thiede, C.; Prior, T.W.; Dohner, K.; Marcucci, G.; et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N. Engl. J. Med. 2017, 377, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Voso, M.T.; Larson, R.A.; Jones, D.; Marcucci, G.; Prior, T.; Kraufer, J.; Heuser, M.; Lavorgna, S.; Nomdedeu, J.; Geyer, S.M.; et al. Midostaurin in patients with acute myeloid leukemia and FLT3-TKD mutations: A subanalysis from the RATIFY trial. Blood Adv. 2020, 4, 4945–4954. [Google Scholar] [CrossRef]
- Rucker, F.G.; Du, L.; Blatte, T.J.; Benner, A.; Krzykalla, J.; Gathmann, I.; Voso, M.T.; Amadori, S.; Prior, T.W.; Brandwein, J.M.; et al. Molecular landscape and prognostic impact of FLT3 internal tandem duplication insertion site in acute myeloid leukemia (AML): Results from the Ratify study (Alliance 10603). Blood 2020, 136 (Suppl. S1), 391. [Google Scholar]
- Cortes, J.E.; Tallamn, M.S.; Schiller, G.J.; Trone, D.; Gammon, G.; Goldberg, S.L.; Perl, A.E.; Marie, J.P.; Martinelli, G.; Kantarjian, H.M.; et al. Phase 2b study of 2 dosing regimens of quizartinib monotherapy in FLT3-ITD-mutated, relapsed or refractory AML. Blood 2018, 132, 598–607. [Google Scholar] [CrossRef]
- Cortes, J.E.; Khaled, S.; Martinelli, G.; Perl, A.E.; Ganguly, S.; Russell, N.; Kramer, A.; Dombret, H.; Hogge, D.; Jonas, B.A.; et al. Quizartinib versus salvage chemotherapy in relapsed or refractory FLT3-ITD acute myeloid leukemia (QuANTUM-R): A multicenter, randomized, controlled, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 984–997. [Google Scholar] [CrossRef]
- Perl, A.E.; Altman, J.K.; Cortes, J.; Smith, C.; Litzow, M.; Baer, M.R.; Claxton, D.; Erba, H.P.; Gill, S.; Goldberg, S.; et al. Selective inhibition of FLT3 by gilteritinib in relapsed or refractory acute myeloid leukemia: A multicenter, first-in-human, open-label, phase 1–2 study. Lancet Oncol. 2019, 381, 1728–1740. [Google Scholar]
- Perl, A.E.; Martinelli, G.; Cortes, J.E.; Neubauer, A.; Berman, E.; Paolini, S.; Montesinos, P.; Baer, M.R.; Larson, R.A.; Ustun, C.; et al. Gilteritinib or chemotherapy for relapsed or refractory FLT3-mutated AML. N. Engl. J. Med. 2019, 381, 1728–1740. [Google Scholar] [CrossRef]
- Pandya, B.J.; Qi, C.Z.; Yang, H.; Garnham, A.; Shah, M.V.; Zeidan, A.M. Comparison of gilteritinib and salvage chemotherapy in FLT3-mutated acute myeloid leukemia on the number needed to treat for various clinical outcomes: A secondary analysis of the admiral trial. Blood 2020, 136 (Suppl. S1), 213. [Google Scholar] [CrossRef]
- Perl, A.E.; Altman, J.K.; Hosono, N.; Monteisnos, P.; Podoltsev, N.A.; Martinelli, G.; Smith, C.C.; Levis, M.; Rollig, C.; Grob-Langenhoff, M.; et al. Clinical outcomes in patients with relapsed/refractory acute myeloid leukemia treated with gilteritinib who received prior midostaurin or sorafenib. Blood 2020, 136 (Suppl. S1), 334. [Google Scholar] [CrossRef]
- Perl, A.E.; Lu, Q.; Fan, A.; Hasabou, N.; Berrak, E.; Tiu, R.V. Clinical outcomes following treatment with gilteritinib or quizartinib in patients with relapsed/refractory FLT3-ITD+ acute myeloid leukemia. Blood 2020, 136 (Suppl. S1), 995. [Google Scholar] [CrossRef]
- Abdelall, W.; Kantarjian, H.M.; Borthakur, G.; Garcia-Manero, G.; Patel, K.P.; Jabbour, E.J. The combination of quizartinib with azacitidine or low dose cytarabine is highly active in patients (Pts) with FLT3-ITD mutated myeloid leukemias: Interim report of a phase I/II trial. Blood 2016, 128 (Suppl. 1), 1642. [Google Scholar] [CrossRef]
- Wang, E.S.; Montesinos, P.; Minden, M.D.; Lee, J.H.; Heuser, M.; Naoe, T.; Chou, W.C.; Liu, S.; Wu, R.; Philipose, N.; et al. Phase 3, multicenter, open-label study of gilteritinib, gilteritinib plus azacitidine, or azacytidine alone in newly diagnosed FLT3 mutated (FLT3mut+) acute myeloid leukemia (AML) patients ineligible for intensive induction chemotherapy. Blood 2020, 136 (Suppl. 1), 27. [Google Scholar]
- Daver, N.; Altman, J.K.; Maly, J.; Levis, M.; Ritchie, E.; Litzow, M.; McCloskey, J.K.; Smith, C.C.; Schiller, G.J.; Bradley, T.; et al. Efficacy and safety of venetoclax in combination with gilteritinib for relapsed/refractory FLT3-mutated acute myeloid leukemia in the expansion cohort of a phase 1b study. Blood 2020, 136 (Suppl. 1), 333. [Google Scholar] [CrossRef]
- Maiti, A.; DiNardo, C.D.; Ravandi, F.; Pemmaraju, N.; Borthakur, G.; Bose, P.; Issa, G.C.; Kadia, T.M.; Short, N.J.; Yilmaz, M.; et al. Venetoclax, FLT3 inhibitor and decitabine in FLT3mut acute myeloid leukemia: Subgroup analysis of a phase II trial. Blood 2020, 136 (Suppl. S1), 1945. [Google Scholar] [CrossRef]
- Yilmaz, M.; Kantarjian, H.M.; Muftuoglu, M.; Kadia, T.M.; Konopleva, M.; Borthakur, G.; DiNardo, C.D.; Pemmaraju, N.; Short, N.J.; Alvarado, Y.; et al. Quizartinib with decitabine +/− venetoclax is highly active in patients (pts) with FLT3-ITD mutated (mut) acute myeloid leukemia (AML): Clinical report and signaling cytof from a phase IB/II trial. Blood 2020, 136 (Suppl. S1), 26. [Google Scholar] [CrossRef]
- Pratz, K.W.; Cherry, M.; Altman, J.K.; Cooper, B.W.; Cruz, J.C.; Jurcic, J.G.; Levis, M.; Lin, T.; Perl, A.E.; Podoltsev, N.A.; et al. A phase 1 study of gilteritinib in combination with induction and consolidation chemotherapy in patients with newly diagnosed AML: Final report. Blood 2020, 136 (Suppl. S1), 24. [Google Scholar] [CrossRef]
- Altman, J.K.; Foran, J.M.; Pratz, K.W.; Trone, D.; Cortes, J.E.; Talmman, M.S. Phase 1 study of quizartinib in combination with induction and consolidation chemotherapy in patients with newly diagnosed acute myeloid leukemia. Am. J. Hematol. 2018, 93, 213–221. [Google Scholar] [CrossRef]
- Galanis, A.; Ma, H.; Rajkhowa, T.; Ramachandran, A.; Small, D.; Cortes, J.; Levis, M. Crenolanib is a potent inhibitor of FLT3 with activity against resistance-conferring point mutatnts. Blood 2014, 123, 94–100. [Google Scholar] [CrossRef] [Green Version]
- Cortes, J.E.; Kantarzian, H.M.; Kadia, T.M.; Borthakur, G.; Konopleva, M.; Garcia-Manero, G.; Daver, N.G.; Pemmaraju, N.; Jabbour, E.; Ramachandran, A.; et al. Crenolanib besylate, a type I pan-FLT3 inhibitor, to demonstrate clinical activity in multiply relapsed FLT3-ITD and D835 AML. J. Clin. Oncol. 2016, 14 (Suppl. 15), 7008. [Google Scholar] [CrossRef]
- Randhawa, J.K.; Kantarjian, H.M.; Borthakur, G.; Thompson, P.A.; Konopleva, M.; Daver, N.; Pemmaraju, N.; Jabbour, E.; Kadia, T.M.; Estrov, Z.; et al. Results of a phase II study of crenolanib in relapsed/refractory acute myeloid leukemia patients (Pts) with activating FLT3 mutations. Blood 2014, 124 (Suppl. 1), 389. [Google Scholar] [CrossRef]
- Goldberg, A.D.; Coombs, C.; Wang, E.S.; Walter, R.B.; Karanes, C.; Vigil, C.E.; Messahel, B.; Stone, R.M.; Collins, R.H. Younger patients with newly diagnosed FLT3-mutant AML treated with crenolanib plus chemotherapy achieve adequate free crenolanib levels and durable remissions. Blood 2019, 134 (Suppl. 1), 1326. [Google Scholar] [CrossRef]
- Wang, E.S.; Griffiths, E.A.; Walter, R.B.; Tallman, M.S.; Goldberg, A.D.; Messahekl, B. Tolerability and efficacy of crenolanib and cytarabine/Anthracycline chemotherapy in older patients (aged 61 to 75) with newly diagnosed FLT3-mutated acute myeloid leukemia (AML). Blood 2019, 134 (Suppl. 1), 134. [Google Scholar] [CrossRef]
- Tarlock, K.; Meshinchi, S.; Rubnitz, J.E.; Karol, S.E.; Spitzer, B.; Sabnis, A.J.; Pathan, A.; Messahel, B. Clinical benefit and tolerability in children with relapsed acute myeloid leukemia harboring treatment resistant FLT3-ITD and variant FLT3-TKD mutations treated on compassionate access. Blood 2020, 136 (Suppl. 1), 1973. [Google Scholar] [CrossRef]
- Goldberg, A.D.; Geyer, M.D.; Kell, J.; Di Bona, E.; Pardee, T.S.; Bhave, R.; Grumwald, M.R.; Marconi, G.; Wang, Y.; Pathan, A.; et al. Clinical benefit of crenolanib, with or without salvage chemotherapy, in multiply relapsed, FLT3 mutant AML patients after prior treatment with gilteritinib. Blood 2020, 136 (Suppl. 1), 1057. [Google Scholar] [CrossRef]
- Di Nardo, C.D.; Stein, E.M.; De Botton, S.; Roboz, G.J.; Altman, J.K.; Mims, A.S.; Swords, R.; Collins, R.A.; Mamis, G.N.; Pollyea, D.A.; et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N. Engl. J. Med. 2018, 378, 2386–2398. [Google Scholar] [CrossRef]
- Stein, E.M.; Di Nardo, C.D.; Pollyea, D.A.; Fathi, A.T.; Roboz, G.J.; Altman, J.K.; Stone, R.M.; De Angelo, D.J.; Levine, R.L.; Finn, J.W.; et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood 2017, 130, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Stein, E.M.; Di Nardo, G.D.; Faithi, A.T.; Pollyea, D.A.; Stone, R.M.; Altman, J.K.; Roboz, G.J.; Patel, M.R.; Collins, R.; Flinn, I.W.; et al. Molecular remission and response patterns in patients with mutant-IDH2 acute myeloid leukemia treated with enasidenib. Blood 2019, 133, 676–687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roboz, G.J.; Di Nardo, C.D.; Stein, E.M.; De Botton, S.; Mims, A.S.; Prince, G.T.; Altman, J.K.; Arellano, M.L.; Donellan, W.; Erba, H.P.; et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood 2020, 135, 463–471. [Google Scholar] [CrossRef] [Green Version]
- Pollyea, D.A.; Tallman, M.S.; De Botton, S.; Komborjian, A.M.; Collins, R.; Stein, A.S.; Frattini, M.G.; Xu, Q.; Tosolini, A.; See, W.L.; et al. Enasidenib, an inhibitor of mutant IDH2 proteins, induces durable remissions in older patients with newly diagnosed acute myeloid leukemia. Leukemia 2019, 33, 2575–2584. [Google Scholar] [CrossRef] [Green Version]
- Paschka, P.; Dombret, H.; Thomas, X.; Recher, C.; Chantepie, S.; Fernandez, P.M.; Acuna-Cruz, E.; Vyas, P.; Kreuzer, K.A.; Heuser, M.; et al. Ivosidenib improves overall survival relative to standard therapies in relapsed or refractory mutant IDH1: Results from matched comparisons to historical controls. Blood 2020, 136 (Suppl. S1), 625. [Google Scholar] [CrossRef]
- Quek, L.; David, M.D.; Kennedy, A.; Metzner, M.; Amatangelo, M.; Shih, A.; Stoilova, B.; Quivoron, C.; Heiblig, M.; Willekens, C.; et al. Clonal heterogeneity of acute myeloid leukemia treated with the IDH2 inhibitor enasidenib. Nat. Med. 2018, 14, 1167–1177. [Google Scholar] [CrossRef]
- Harding, J.J.; Lowery, M.A.; Shih, A.H.; Schvartzman, J.M.; Hou, S.; Famulare, C.; Patel, M.; Roshal, M.; Do, R.K.; Zehir, A. Isoform switching as a mechanism of acquired resistance to mutant isocitrate dehydrogenase inhibition. Cancer Discov. 2018, 8, 1540–1547. [Google Scholar] [CrossRef] [Green Version]
- Intlekofer, A.M.; Shih, A.H.; Wang, B.; Nazer, A.; Rustenbug, A.S.; Albanese, S.K.; Patel, M.; Famulare, C.; Correa, F.M.; Takamoto, N.; et al. Acquired resistance to IDH inhibition through trans or cis dimer-interface mutations. Nature 2018, 559, 125–129. [Google Scholar] [CrossRef]
- Di Nardo, C.D.; Schuh, A.C.; Stein, E.M.; Montesinos Fernandez, P.; Wei, A.H.; De Botton, S.; Zeidan, A.M.; Fathi, A.T.; Quek, L.; Kantarjian, H.M.; et al. Enasidenib plus azacitidine significantly improves complete remission and overall response compared with azacytidine alone in patients with newly diagnosed acute myeloid leukemia (AML) with isocitrate dehydrogenase (IDH2) mutations: Interim phase II results from an ongoing, randomized study. Blood 2019, 134 (Suppl. S1), 643. [Google Scholar]
- DiNardo, C.D.; Stein, A.S.; Stein, E.M.; Fathi, A.T.; Frankfurt, O.; Schuh, A.C.; Dohner, H.; Martinelli, G.; Patel, P.A.; Raffoux, E.; et al. Mutant isocitrate dehydrogenase 1 inhibitor ivosidenib in combination with azacytidine for newly diagnosed acute myeloid leukemia. J. Clin. Oncol. 2021, 39, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, C.D.; Schuh, A.C.; Stein, E.M.; Montesinos Fernandez, P.; Wei, A.H.; De Botton, S.; Zeidan, A.M.; Fathi, A.T.; Quek, L.; Kantarjian, H.M.; et al. Effect of enasidenib (ENA) plus azacytidine monotherapy in mutant-IDH2 (mIDH2) newly diagnosed acute myeloid leukemia (ND-AML). J. Clin. Oncol. 2020, 38 (Suppl. S1), 7501. [Google Scholar]
- Ok, C.Y.; Loghavi, S.; Sui, D.; Wei, P.; Kanagal-Shamanna, R.; Yin, C.C.; Zuo, Z.; Routbort, M.J.; Tang, G.; Tang, Z.; et al. Persistent IDH1/2 mutations in remission can predict relapse in patients with acute myeloid leukemia. Haematologica 2019, 104, 305–311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stein, E.M.; DiNardo, C.; Fathi, A.T.; Mims, A.S.; Pritz, K.W.; Savona, M.R.; Stein, A.S.; Stone, R.M.; Winer, E.S.; Seet, C.S.; et al. Ivosidenib or enasidenib combined with intensive chemotherapy in patients with newly diagnosed AML: A phase 1 study. Blood 2020, in press. [Google Scholar] [CrossRef]
- Stein, E.M.; DiNardo, C.; Fathi, A.T.; Mims, A.S.; Pratz, K.W.; Savona, M.R.; Stein, A.S.; Stone, R.M.; Winer, E.S.; Seet, C.S.; et al. Ivosidenib or enasidenib with induction and consolidation chemotherapy in patients with newly diagnosed AML with an IDH1 or IDH2 mutation is safe, effective, and leads to MRD-negative complete remissions. Blood 2018, 132 (Suppl. 1), 560. [Google Scholar] [CrossRef]
- Di Nardo, C.D.; Pratz, K.W.; Letai, A.; Jonas, B.A.; Wei, A.H.; Thirman, M.; Arellano, M.; Frattini, M.G.; Kantarjian, H.; Popovic, R.; et al. Safety and preliminary efficacy of venetoclax with decitabine or azacytidine in elderly patients with previously untreated acute myeloid leukaemia: A non-randomised, open-label, phase 1b study. Lancet Oncol. 2018, 19, 216–228. [Google Scholar] [CrossRef]
- Wei, A.H.; Strickland, S.A.; Hou, J.Z.; Fiedler, W.; Lin, T.L.; Walter, R.B.; Enjeti, A.; Tiong, I.S.; Savona, M.; Lee, S.; et al. Venetoclax combined with low-dose cytarabine for previously untreated patients with acute myeloid leukemia: Results from a phase Ib/II study. J. Clin. Oncol. 2019, 37, 1277–1284. [Google Scholar] [CrossRef] [PubMed]
- Di Nardo, C.D.; Jonas, B.A.; Pullarkat, M.J.; Thirman, M.J.; Garcia, J.S.; Wei, A.H.; Konopleva, M.; Dohner, H.; Letai, A.; Fenaux, P.; et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N. Engl. J. Med. 2020, 383, 617–629. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Tiong, I.S.; Quaglieri, A.; MacRaild, S.; Loghavi, S.; Brown, F.C.; Thijssen, R.; Pomilio, G.; Ivey, A.; Salmon, J.M.; et al. Molecular patterns of response and treatment failure after frontline venetoclax combinations in older patients with AML. Blood 2020, 135, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Pollyea, D.A.; DiNardo, C.D.; Arellano, M.L.; Pigneux, A.; Fiedler, W.; Konopleva, M.; Rizzieri, D.A.; Smith, D.; Shinagawa, A.; Lemoli, R.M.; et al. Results of venetoclax and azacytidine combination in chemotherapy ineligible untreated patients with acute myeloid leukemia with IDH1/2 mutations. Blood 2020, 136 (Suppl. S1), 5–7. [Google Scholar] [CrossRef]
- Maiti, A.; Rausch, C.R.; Cortes, J.E.; Pemmaraju, N.; Daver, N.G.; Ravandi, F.; Garcia-Manero, G.; Borthakur, G.M.; Naqvi, K.; Ohanian, M.; et al. Outcomes of relapsed or refractory acute myeloid leukemia after frontline hypomethylating agent with venetoclax regimens. Blood 2019, 134 (Suppl. S1), 738. [Google Scholar] [CrossRef]
- DiNardo, C.D.; Maiti, A.; Rausch, C.R.; Pemmanaiu, N.; Naqvi, K.; Daver, N.G.; Kadia, T.M.; Borthakur, G.; Gohaninan, N.; Alvarado, Y.; et al. 10-day decitabine with venetoclax for newly diagnosed intensive chemotherapy ineligible, and relapsed or refractory acute myeloid leukemia: A single-centre, phase 2 trial. Lancet Hematol. 2020, 7, e724–e736. [Google Scholar] [CrossRef]
- Hammond, D.; Loghavi, S.; Konopleva, M.; Kadia, T.M.; Daver, N.; Ohanina, M.; Issa, G.C.; Alavarado, Y.; Montalban-Bravo, G.; Garcia-Manero, G.; et al. Response patterns and MRD by flow cytometry and NGS in patients with mutant-IDH acute myeloid leukemia treated with venetoclax in combination with hypomethylating agents. Blood 2020, 136 (Suppl. S), 35–36. [Google Scholar] [CrossRef]
- Wei, A.H.; Montesinos, P.; Ivanov, V.; DiNardo, C.D.; Novak, J.; Laribi, K.; Kim, I.; Stevens, D.A.; Fiedler, W.; Pagoni, M.; et al. Venetoclax plus LDAC for newly diagnosed AML ineligible for intensive chemotherapy: A phase 3 randomized placebo-controlled trial. Blood 2020, 135, 2137–2146. [Google Scholar] [CrossRef]
- Chua, C.C.; Roberts, A.W.; Reynolds, J.; Fong, C.Y.; Ting, S.B.; Salmon, J.B.; MacRaild, S.; Ivey, A.; Tiong, I.S.; Fleming, S.; et al. Chemotherapy and venetoclax in elderly acute myeloid leukemia trial (CAVEAT): A phase Ib dose-escalation study of venetoclax combined with modified intensive chemotherapy. J. Clin. Oncol. 2020, 38, 3506–3517. [Google Scholar] [CrossRef]
- Lachowiez, C.A.; Borthakur, G.; Loghavi, S.; Zhihong, Z.; Kadia, T.M.; Masarova, L. Phase Ib/II study of the IDH1-mutant inhibitor ivosidenib with the BCL2 inhibitor venetoclax ± azacytidine in IDH1-mutated hematologic malignancies. J. Clin. Oncol. 2020, 38, 7500. [Google Scholar] [CrossRef]
- Richard-Charpentier, G.; Di Nardo, C.D. Venetoclax for the treatment of newly diagnosed acute myeloid leukemia in patients who are ineligible for intensive chemotherapy. Ther. Adv. Hematol. 2019, 10, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Collins, F.S.; Varmus, H. A new initiative on precision medicine. N. Engl. J. Med. 2015, 372, 793–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunter, D.J. Uncertainty in the era of precision medicine. N. Engl. J. Med. 2016, 375, 711–713. [Google Scholar] [CrossRef]
- Bertoli, S.; Bérard, E.; Huguet, F.; Huynh, A.; Tavitan, S.; Vergez, F.; Dobbelstein, S.; Dastugue, N.; Mansat-De Mas, V.; Delabesse, E.; et al. Time from diagnosis to intensive chemotherapy initiation does not adversely impact the outcome of patients with acute myeloid leukemia. Blood 2013, 121, 2618–2624. [Google Scholar] [CrossRef] [Green Version]
- Burd, A.; Levine, R.L.; Shoben, A.; Mims, A.S.; Borate, U.; Stein, E.M.; Patel, P.A.; Baer, M.R.; Stock, W.; Deininger, M.W.; et al. Initial report of the Beat AML umbrella study for previously untreated AML: Evidence of feasibility and early success in molecularly driven phase 1 and 2 studies. Blood 2018, 132 (Suppl. S1), 559. [Google Scholar] [CrossRef]
- Burd, A.; Levine, R.L.; Ruppert, A.S.; Mims, A.S.; Borate, U.; Stein, E.M.; Patel, P.; Baer, M.R.; Stock, W.; Deininger, M.; et al. Precision medicine treatment in acute myeloid leukemia using prospective genomic profiling: Feasibility and preliminary efficacy of the Beat AML Master Trial. Nat. Med. 2020, 26, 1852–1858. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Testa, U.; Pelosi, E.; Castelli, G. Precision Medicine Treatment in Acute Myeloid Leukemia Is Not a Dream. Hemato 2021, 2, 131-153. https://0-doi-org.brum.beds.ac.uk/10.3390/hemato2010008
Testa U, Pelosi E, Castelli G. Precision Medicine Treatment in Acute Myeloid Leukemia Is Not a Dream. Hemato. 2021; 2(1):131-153. https://0-doi-org.brum.beds.ac.uk/10.3390/hemato2010008
Chicago/Turabian StyleTesta, Ugo, Elvira Pelosi, and Germana Castelli. 2021. "Precision Medicine Treatment in Acute Myeloid Leukemia Is Not a Dream" Hemato 2, no. 1: 131-153. https://0-doi-org.brum.beds.ac.uk/10.3390/hemato2010008





