The Metastatic Capacity of Melanoma Reveals Alternative Pathways of Cancer Dissemination
Abstract
:1. Introduction
2. Non-Angiogenic Intratumoral Vascularization
3. Epithelial to Mesenchymal Transition
4. The Angiotropic Process: Walking on the Abluminal Side of Vessels
5. Role of the Extracellular Matrix
6. Approaches to Studying Angiotropism and EVMM
6.1. In Vitro 3D Models
6.2. In Vivo Models
7. Final Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target. Ther. 2020, 5, 28. [Google Scholar] [CrossRef]
- van Zijl, F.; Krupitza, G.; Mikulits, W. Initial steps of metastasis: Cell invasion and endothelial transmigration. Mutat. Res. 2011, 728, 23–34. [Google Scholar] [CrossRef]
- Barbolina, M.V. Molecular Mechanisms Regulating Organ-Specific Metastases in Epithelial Ovarian Carcinoma. Cancers 2018, 10, 444. [Google Scholar] [CrossRef] [Green Version]
- Wong, S.Y.; Hynes, R.O. Lymphatic or hematogenous dissemination: How does a metastatic tumor cell decide? Cell Cycle 2006, 5, 812–817. [Google Scholar] [CrossRef] [Green Version]
- Pennacchioli, E.; Tosti, G.; Barberis, M.; De Pas, T.M.; Verrecchia, F.; Menicanti, C.; Testori, A.; Mazzarol, G. Sarcoma spreads primarily through the vascular system: Are there biomarkers associated with vascular spread? Clin. Exp. Metastasis 2012, 29, 757–773. [Google Scholar] [CrossRef] [PubMed]
- Perissinotto, E.; Cavalloni, G.; Leone, F.; Fonsato, V.; Mitola, S.; Grignani, G.; Surrenti, N.; Sangiolo, D.; Bussolino, F.; Piacibello, W.; et al. Involvement of chemokine receptor 4/stromal cell-derived factor 1 system during osteosarcoma tumor progression. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2005, 11, 490–497. [Google Scholar]
- Rahman, M.; Mohammed, S. Breast cancer metastasis and the lymphatic system. Oncol. Lett. 2015, 10, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Riquet, M.; Rivera, C.; Gibault, L.; Pricopi, C.; Mordant, P.; Badia, A.; Arame, A.; Le Pimpec Barthes, F. Lymphatic spread of lung cancer: Anatomical lymph node chains unchained in zones. Rev. De Pneumol. Clin. 2014, 70, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Kotoulas, C.S.; Foroulis, C.N.; Kostikas, K.; Konstantinou, M.; Kalkandi, P.; Dimadi, M.; Bouros, D.; Lioulias, A. Involvement of lymphatic metastatic spread in non-small cell lung cancer accordingly to the primary cancer location. Lung Cancer 2004, 44, 183–191. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, L.; Xu, T.; Xue, R.; Yu, L.; Zhu, Y.; Wu, Y.; Zhang, Q.; Li, D.; Shen, S.; et al. Mapping the spreading routes of lymphatic metastases in human colorectal cancer. Nat. Commun. 2020, 11, 1993. [Google Scholar] [CrossRef]
- Romani, C.; Zizioli, V.; Silvestri, M.; Ardighieri, L.; Bugatti, M.; Corsini, M.; Todeschini, P.; Marchini, S.; D’Incalci, M.; Zanotti, L.; et al. Low Expression of Claudin-7 as Potential Predictor of Distant Metastases in High-Grade Serous Ovarian Carcinoma Patients. Front. Oncol. 2020, 10, 1287. [Google Scholar] [CrossRef]
- Nyangoga, H.; Mercier, P.; Libouban, H.; Basle, M.F.; Chappard, D. Three-dimensional characterization of the vascular bed in bone metastasis of the rat by microcomputed tomography (MicroCT). PLoS ONE 2011, 6, e17336. [Google Scholar] [CrossRef] [Green Version]
- Goel, S.; Duda, D.G.; Xu, L.; Munn, L.L.; Boucher, Y.; Fukumura, D.; Jain, R.K. Normalization of the vasculature for treatment of cancer and other diseases. Physiol. Rev. 2011, 91, 1071–1121. [Google Scholar] [CrossRef]
- Schaaf, M.B.; Garg, A.D.; Agostinis, P. Defining the role of the tumor vasculature in antitumor immunity and immunotherapy. Cell Death Dis. 2018, 9, 115. [Google Scholar] [CrossRef] [Green Version]
- Massague, J.; Obenauf, A.C. Metastatic colonization by circulating tumour cells. Nature 2016, 529, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Labelle, M.; Hynes, R.O. The initial hours of metastasis: The importance of cooperative host-tumor cell interactions during hematogenous dissemination. Cancer Discov. 2012, 2, 1091–1099. [Google Scholar] [CrossRef] [Green Version]
- Kienast, Y.; von Baumgarten, L.; Fuhrmann, M.; Klinkert, W.E.; Goldbrunner, R.; Herms, J.; Winkler, F. Real-time imaging reveals the single steps of brain metastasis formation. Nat. Med. 2010, 16, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Seano, G.; Jain, R.K. Vessel co-option in glioblastoma: Emerging insights and opportunities. Angiogenesis 2020, 23, 9–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandez-Cortes, M.; Delgado-Bellido, D.; Oliver, F.J. Vasculogenic Mimicry: Become an Endothelial Cell “But Not So Much”. Front. Oncol. 2019, 9, 803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Wang, S.; Dudley, A.C. Models and molecular mechanisms of blood vessel co-option by cancer cells. Angiogenesis 2020, 23, 17–25. [Google Scholar] [CrossRef]
- Frentzas, S.; Simoneau, E.; Bridgeman, V.L.; Vermeulen, P.B.; Foo, S.; Kostaras, E.; Nathan, M.; Wotherspoon, A.; Gao, Z.H.; Shi, Y.; et al. Vessel co-option mediates resistance to anti-angiogenic therapy in liver metastases. Nat. Med. 2016, 22, 1294–1302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pezzella, F.; Gatter, K. Non-angiogenic tumours unveil a new chapter in cancer biology. J. Pathol. 2015, 235, 381–383. [Google Scholar] [CrossRef] [Green Version]
- Ronca, R.; Benkheil, M.; Mitola, S.; Struyf, S.; Liekens, S. Tumor angiogenesis revisited: Regulators and clinical implications. Med. Res. Rev. 2017, 37, 1231–1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribatti, D.; Djonov, V. Intussusceptive microvascular growth in tumors. Cancer Lett. 2012, 316, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Flores, L.; Gutierrez, R.; Gayoso, S.; Garcia, M.P.; Gonzalez-Gomez, M.; Diaz-Flores, L., Jr.; Sanchez, R.; Carrasco, J.L.; Madrid, J.F. Intussusceptive angiogenesis and its counterpart intussusceptive lymphangiogenesis. Histol. Histopathol. 2020, 35, 1083–1103. [Google Scholar] [CrossRef]
- Diaz-Flores, L.; Gutierrez, R.; Pino Garcia, M.; Gonzalez-Gomez, M.; Diaz-Flores, L., Jr.; Carrasco, J.L. Intussusceptive lymphangiogenesis in the sinuses of developing human foetal lymph nodes. Ann. Anat. Anat. Anz. Off. Organ Anat. Ges. 2019, 226, 73–83. [Google Scholar] [CrossRef]
- Corsini, M.; Ravelli, C.; Grillo, E.; Dell’Era, P.; Presta, M.; Mitola, S. Simultaneously characterization of tumoral angiogenesis and vasculogenesis in stem cell-derived teratomas. Exp. Cell Res. 2021, 400, 112490. [Google Scholar] [CrossRef]
- Baker, G.J.; Yadav, V.N.; Motsch, S.; Koschmann, C.; Calinescu, A.A.; Mineharu, Y.; Camelo-Piragua, S.I.; Orringer, D.; Bannykh, S.; Nichols, W.S.; et al. Mechanisms of glioma formation: Iterative perivascular glioma growth and invasion leads to tumor progression, VEGF-independent vascularization, and resistance to antiangiogenic therapy. Neoplasia 2014, 16, 543–561. [Google Scholar] [CrossRef] [Green Version]
- Yagi, Y.; Aly, R.G.; Tabata, K.; Barlas, A.; Rekhtman, N.; Eguchi, T.; Montecalvo, J.; Hameed, M.; Manova-Todorova, K.; Adusumilli, P.S.; et al. Three-Dimensional Histologic, Immunohistochemical, and Multiplex Immunofluorescence Analyses of Dynamic Vessel Co-Option of Spread Through Air Spaces in Lung Adenocarcinoma. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer 2020, 15, 589–600. [Google Scholar] [CrossRef]
- Kuczynski, E.A.; Vermeulen, P.B.; Pezzella, F.; Kerbel, R.S.; Reynolds, A.R. Vessel co-option in cancer. Nat. Rev. Clin. Oncol. 2019, 16, 469–493. [Google Scholar] [CrossRef]
- Latacz, E.; Caspani, E.; Barnhill, R.; Lugassy, C.; Verhoef, C.; Grunhagen, D.; Van Laere, S.; Fernandez Moro, C.; Gerling, M.; Dirix, M.; et al. Pathological features of vessel co-option versus sprouting angiogenesis. Angiogenesis 2020, 23, 43–54. [Google Scholar] [CrossRef] [PubMed]
- Donnem, T.; Reynolds, A.R.; Kuczynski, E.A.; Gatter, K.; Vermeulen, P.B.; Kerbel, R.S.; Harris, A.L.; Pezzella, F. Non-angiogenic tumours and their influence on cancer biology. Nat. Rev. Cancer 2018, 18, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.; Roswall, P.; Cortez, E.; Hanahan, D.; Pietras, K. Pericytes promote endothelial cell survival through induction of autocrine VEGF-A signaling and Bcl-w expression. Blood 2011, 118, 2906–2917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caspani, E.M.; Crossley, P.H.; Redondo-Garcia, C.; Martinez, S. Glioblastoma: A pathogenic crosstalk between tumor cells and pericytes. PLoS ONE 2014, 9, e101402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Folberg, R.; Hendrix, M.J.; Maniotis, A.J. Vasculogenic mimicry and tumor angiogenesis. Am. J. Pathol. 2000, 156, 361–381. [Google Scholar] [CrossRef] [Green Version]
- Shirakawa, K.; Wakasugi, H.; Heike, Y.; Watanabe, I.; Yamada, S.; Saito, K.; Konishi, F. Vasculogenic mimicry and pseudo-comedo formation in breast cancer. Int. J. Cancer 2002, 99, 821–828. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, L.; Liu, W.X.; Wang, K. VEGF and SEMA4D have synergistic effects on the promotion of angiogenesis in epithelial ovarian cancer. Cell. Mol. Biol. Lett. 2018, 23, 2. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Chen, J.; Xie, H.; Liu, T.; Chen, Y.; Ma, Z.; Pei, X.; Yang, W.; Li, L. Androgen receptor suppresses prostate cancer metastasis but promotes bladder cancer metastasis via differentially altering miRNA525-5p/SLPI-mediated vasculogenic mimicry formation. Cancer Lett. 2020, 473, 118–129. [Google Scholar] [CrossRef]
- Zhang, Z.; Nong, L.; Chen, M.; Gu, X.; Zhao, W.; Liu, M.; Cheng, W. Baicalein suppresses vasculogenic mimicry through inhibiting RhoA/ROCK expression in lung cancer A549 cell line. Acta Biochim. Biophys. Sin. 2020, 52, 1007–1015. [Google Scholar] [CrossRef]
- Yeo, C.; Han, D.S.; Lee, H.J.; Lee, E.O. Epigallocatechin-3-Gallate Suppresses Vasculogenic Mimicry through Inhibiting the Twist/VE-Cadherin/AKT Pathway in Human Prostate Cancer PC-3 Cells. Int. J. Mol. Sci. 2020, 21, 439. [Google Scholar] [CrossRef] [Green Version]
- Mavrogenis, A.F.; Vottis, C.T.; Megaloikonomos, P.D.; Agrogiannis, G.D.; Theocharis, S. Neovascularization in Ewing’s sarcoma. Neoplasma 2018, 65, 317–325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Y.; Liu, X.; Zhao, P.; Zhao, H.; Gao, W.; Wang, L. Celastrol Suppresses Glioma Vasculogenic Mimicry Formation and Angiogenesis by Blocking the PI3K/Akt/mTOR Signaling Pathway. Front. Pharmacol. 2020, 11, 25. [Google Scholar] [CrossRef] [Green Version]
- Cao, W.; Xu, C.; Li, X.; Yang, X. Twist1 promotes astrocytoma development by stimulating vasculogenic mimicry. Oncol. Lett. 2019, 18, 846–855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delgado-Bellido, D.; Fernandez-Cortes, M.; Rodriguez, M.I.; Serrano-Saenz, S.; Carracedo, A.; Garcia-Diaz, A.; Oliver, F.J. VE-cadherin promotes vasculogenic mimicry by modulating kaiso-dependent gene expression. Cell Death Differ. 2019, 26, 348–361. [Google Scholar] [CrossRef] [Green Version]
- Hendrix, M.J.; Seftor, E.A.; Hess, A.R.; Seftor, R.E. Vasculogenic mimicry and tumour-cell plasticity: Lessons from melanoma. Nat. Rev. Cancer 2003, 3, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Andreucci, E.; Laurenzana, A.; Peppicelli, S.; Biagioni, A.; Margheri, F.; Ruzzolini, J.; Bianchini, F.; Fibbi, G.; Rosso, M.D.; Nediani, C.; et al. uPAR controls vasculogenic mimicry ability expressed by drug-resistant melanoma cells. Oncol. Res. 2021. (Online ahead of print). [Google Scholar] [CrossRef]
- Serova, M.; Tijeras-Raballand, A.; Dos Santos, C.; Martinet, M.; Neuzillet, C.; Lopez, A.; Mitchell, D.C.; Bryan, B.A.; Gapihan, G.; Janin, A.; et al. Everolimus affects vasculogenic mimicry in renal carcinoma resistant to sunitinib. Oncotarget 2016, 7, 38467–38486. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Sun, B.; Zhao, X.; Ma, Y.; Ji, R.; Gu, Q.; Dong, X.; Li, J.; Liu, F.; Jia, X.; et al. Twist1 expression induced by sunitinib accelerates tumor cell vasculogenic mimicry by increasing the population of CD133+ cells in triple-negative breast cancer. Mol. Cancer 2014, 13, 207. [Google Scholar] [CrossRef] [Green Version]
- Angara, K.; Rashid, M.H.; Shankar, A.; Ara, R.; Iskander, A.; Borin, T.F.; Jain, M.; Achyut, B.R.; Arbab, A.S. Vascular mimicry in glioblastoma following anti-angiogenic and anti-20-HETE therapies. Histol. Histopathol. 2017, 32, 917–928. [Google Scholar] [CrossRef]
- Angara, K.; Borin, T.F.; Rashid, M.H.; Lebedyeva, I.; Ara, R.; Lin, P.C.; Iskander, A.; Bollag, R.J.; Achyut, B.R.; Arbab, A.S. CXCR2-Expressing Tumor Cells Drive Vascular Mimicry in Antiangiogenic Therapy-Resistant Glioblastoma. Neoplasia 2018, 20, 1070–1082. [Google Scholar] [CrossRef]
- Mittal, V. Epithelial Mesenchymal Transition in Tumor Metastasis. Annu. Rev. Pathol. 2018, 13, 395–412. [Google Scholar] [CrossRef] [PubMed]
- Brabletz, T.; Jung, A.; Reu, S.; Porzner, M.; Hlubek, F.; Kunz-Schughart, L.A.; Knuechel, R.; Kirchner, T. Variable beta-catenin expression in colorectal cancers indicates tumor progression driven by the tumor environment. Proc. Natl. Acad. Sci. USA 2001, 98, 10356–10361. [Google Scholar] [CrossRef] [Green Version]
- Araki, K.; Shimura, T.; Suzuki, H.; Tsutsumi, S.; Wada, W.; Yajima, T.; Kobayahi, T.; Kubo, N.; Kuwano, H. E/N-cadherin switch mediates cancer progression via TGF-beta-induced epithelial-to-mesenchymal transition in extrahepatic cholangiocarcinoma. Br. J. Cancer 2011, 105, 1885–1893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loh, C.Y.; Chai, J.Y.; Tang, T.F.; Wong, W.F.; Sethi, G.; Shanmugam, M.K.; Chong, P.P.; Looi, C.Y. The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 2019, 8, 1118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vuoriluoto, K.; Haugen, H.; Kiviluoto, S.; Mpindi, J.P.; Nevo, J.; Gjerdrum, C.; Tiron, C.; Lorens, J.B.; Ivaska, J. Vimentin regulates EMT induction by Slug and oncogenic H-Ras and migration by governing Axl expression in breast cancer. Oncogene 2011, 30, 1436–1448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nieto, M.A.; Huang, R.Y.; Jackson, R.A.; Thiery, J.P. Emt: 2016. Cell 2016, 166, 21–45. [Google Scholar] [CrossRef] [Green Version]
- Pastushenko, I.; Brisebarre, A.; Sifrim, A.; Fioramonti, M.; Revenco, T.; Boumahdi, S.; Van Keymeulen, A.; Brown, D.; Moers, V.; Lemaire, S.; et al. Identification of the tumour transition states occurring during EMT. Nature 2018, 556, 463–468. [Google Scholar] [CrossRef]
- Landsberg, J.; Tuting, T.; Barnhill, R.L.; Lugassy, C. The Role of Neutrophilic Inflammation, Angiotropism, and Pericytic Mimicry in Melanoma Progression and Metastasis. J. Investig. Dermatol. 2016, 136, 372–377. [Google Scholar] [CrossRef] [Green Version]
- Barnhill, R.L.; Lugassy, C. Angiotropic malignant melanoma and extravascular migratory metastasis: Description of 36 cases with emphasis on a new mechanism of tumour spread. Pathology 2004, 36, 485–490. [Google Scholar] [CrossRef]
- Lugassy, C.; Kleinman, H.K.; Vermeulen, P.B.; Barnhill, R.L. Angiotropism, pericytic mimicry and extravascular migratory metastasis: An embryogenesis-derived program of tumor spread. Angiogenesis 2020, 23, 27–41. [Google Scholar] [CrossRef]
- Lugassy, C.; Vernon, S.E.; Warner, J.W.; Le, C.Q.; Manyak, M.; Patierno, S.R.; Barnhill, R.L. Angiotropism of human prostate cancer cells: Implications for extravascular migratory metastasis. BJU Int. 2005, 95, 1099–1103. [Google Scholar] [CrossRef]
- Cheng, L.; Huang, Z.; Zhou, W.; Wu, Q.; Donnola, S.; Liu, J.K.; Fang, X.; Sloan, A.E.; Mao, Y.; Lathia, J.D.; et al. Glioblastoma stem cells generate vascular pericytes to support vessel function and tumor growth. Cell 2013, 153, 139–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rustagi, T.; Gleeson, F.C.; Chari, S.T.; Lehrke, H.D.; Takahashi, N.; Malikowski, T.M.; Abu Dayyeh, B.K.; Chandrasekhara, V.; Iyer, P.G.; Kendrick, M.L.; et al. Safety, Diagnostic Accuracy, and Effects of Endoscopic Ultrasound Fine-Needle Aspiration on Detection of Extravascular Migratory Metastases. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2019, 17, 2533–2540.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lugassy, C.; Zadran, S.; Bentolila, L.A.; Wadehra, M.; Prakash, R.; Carmichael, S.T.; Kleinman, H.K.; Peault, B.; Larue, L.; Barnhill, R.L. Angiotropism, pericytic mimicry and extravascular migratory metastasis in melanoma: An alternative to intravascular cancer dissemination. Cancer Microenviron. Off. J. Int. Cancer Microenviron. Soc. 2014, 7, 139–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moy, A.P.; Duncan, L.M.; Muzikansky, A.; Kraft, S. Angiotropism in primary cutaneous melanoma is associated with disease progression and distant metastases: A retrospective study of 179 cases. J. Cutan. Pathol. 2019, 46, 498–507. [Google Scholar] [CrossRef]
- Lugassy, C.; Lazar, V.; Dessen, P.; van den Oord, J.J.; Winnepenninckx, V.; Spatz, A.; Bagot, M.; Bensussan, A.; Janin, A.; Eggermont, A.M.; et al. Gene expression profiling of human angiotropic primary melanoma: Selection of 15 differentially expressed genes potentially involved in extravascular migratory metastasis. Eur J Cancer 2011, 47, 1267–1275. [Google Scholar] [CrossRef]
- Lugassy, C.; Wadehra, M.; Li, X.; Corselli, M.; Akhavan, D.; Binder, S.W.; Peault, B.; Cochran, A.J.; Mischel, P.S.; Kleinman, H.K.; et al. Pilot study on “pericytic mimicry” and potential embryonic/stem cell properties of angiotropic melanoma cells interacting with the abluminal vascular surface. Cancer Microenviron. Off. J. Int. Cancer Microenviron. Soc. 2013, 6, 19–29. [Google Scholar] [CrossRef] [Green Version]
- Fornabaio, G.; Barnhill, R.L.; Lugassy, C.; Bentolila, L.A.; Cassoux, N.; Roman-Roman, S.; Alsafadi, S.; Del Bene, F. Angiotropism and extravascular migratory metastasis in cutaneous and uveal melanoma progression in a zebrafish model. Sci. Rep. 2018, 8, 10448. [Google Scholar] [CrossRef]
- Lugassy, C.; Kleinman, H.K.; Vernon, S.E.; Welch, D.R.; Barnhill, R.L. C16 laminin peptide increases angiotropic extravascular migration of human melanoma cells in a shell-less chick chorioallantoic membrane assay. Br. J. Dermatol. 2007, 157, 780–782. [Google Scholar] [CrossRef] [PubMed]
- Bentolila, L.A.; Prakash, R.; Mihic-Probst, D.; Wadehra, M.; Kleinman, H.K.; Carmichael, T.S.; Peault, B.; Barnhill, R.L.; Lugassy, C. Imaging of Angiotropism/Vascular Co-Option in a Murine Model of Brain Melanoma: Implications for Melanoma Progression along Extravascular Pathways. Sci. Rep. 2016, 6, 23834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodewald, A.K.; Rushing, E.J.; Kirschenbaum, D.; Mangana, J.; Mittmann, C.; Moch, H.; Lugassy, C.; Barnhill, R.L.; Mihic-Probst, D. Eight autopsy cases of melanoma brain metastases showing angiotropism and pericytic mimicry. Implications for extravascular migratory metastasis. J. Cutan. Pathol. 2019, 46, 570–578. [Google Scholar] [CrossRef]
- Montana, V.; Sontheimer, H. Bradykinin promotes the chemotactic invasion of primary brain tumors. J. Neurosci. Off. J. Soc. Neurosci. 2011, 31, 4858–4867. [Google Scholar] [CrossRef] [Green Version]
- Watkins, S.; Robel, S.; Kimbrough, I.F.; Robert, S.M.; Ellis-Davies, G.; Sontheimer, H. Disruption of astrocyte-vascular coupling and the blood-brain barrier by invading glioma cells. Nat. Commun. 2014, 5, 4196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winkler, F.; Kienast, Y.; Fuhrmann, M.; Von Baumgarten, L.; Burgold, S.; Mitteregger, G.; Kretzschmar, H.; Herms, J. Imaging glioma cell invasion in vivo reveals mechanisms of dissemination and peritumoral angiogenesis. Glia 2009, 57, 1306–1315. [Google Scholar] [CrossRef]
- Alieva, M.; Leidgens, V.; Riemenschneider, M.J.; Klein, C.A.; Hau, P.; van Rheenen, J. Intravital imaging of glioma border morphology reveals distinctive cellular dynamics and contribution to tumor cell invasion. Sci. Rep. 2019, 9, 2054. [Google Scholar] [CrossRef] [PubMed]
- Zagzag, D.; Esencay, M.; Mendez, O.; Yee, H.; Smirnova, I.; Huang, Y.; Chiriboga, L.; Lukyanov, E.; Liu, M.; Newcomb, E.W. Hypoxia- and vascular endothelial growth factor-induced stromal cell-derived factor-1alpha/CXCR4 expression in glioblastomas: One plausible explanation of Scherer’s structures. Am. J. Pathol. 2008, 173, 545–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koay, M.H.E.; Stewart, C.J.R. Extravascular Migratory Metastasis (Pericytic Mimicry) in Sarcomatoid Squamous Cell Carcinoma of the Vulva: A Report of 2 Cases. Int. J. Gynecol. Pathol. Off. J. Int. Soc. Gynecol. Pathol. 2019, 38, 27–31. [Google Scholar] [CrossRef]
- Fedda, F.; Migden, M.R.; Curry, J.L.; Torres-Cabala, C.A.; Tetzlaff, M.T.; Aung, P.P.; Prieto, V.G.; Ivan, D.; Myers, J.N.; Nagarajan, P. Angiotropism in recurrent cutaneous squamous cell carcinoma: Implications for regional tumor recurrence and extravascular migratory spread. J. Cutan. Pathol. 2019, 46, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Katayama, E.; Saruta, H.; Nanri, A.; Nakama, T.; Ohata, C. Angiotropic syringomatous carcinoma. J. Cutan. Pathol. 2017, 44, 397–400. [Google Scholar] [CrossRef]
- Dyke, J.M.; Crook, M.L.; Platten, M.; Stewart, C.J. Extravascular migratory metastasis in gynaecological carcinosarcoma. Histopathology 2014, 65, 363–370. [Google Scholar] [CrossRef]
- Shen, J.; Shrestha, S.; Rao, P.N.; Asatrian, G.; Scott, M.A.; Nguyen, V.; Giacomelli, P.; Soo, C.; Ting, K.; Eilber, F.C.; et al. Pericytic mimicry in well-differentiated liposarcoma/atypical lipomatous tumor. Hum. Pathol. 2016, 54, 92–99. [Google Scholar] [CrossRef] [Green Version]
- Levy, M.J.; Gleeson, F.C.; Zhang, L. Endoscopic ultrasound fine-needle aspiration detection of extravascular migratory metastasis from a remotely located pancreatic cancer. Clin. Gastroenterol. Hepatol. Off. Clin. Pract. J. Am. Gastroenterol. Assoc. 2009, 7, 246–248. [Google Scholar] [CrossRef]
- Lugassy, C.; Shahsafaei, A.; Bonitz, P.; Busam, K.J.; Barnhill, R.L. Tumor microvessels in melanoma express the beta-2 chain of laminin. Implications for melanoma metastasis. J. Cutan. Pathol. 1999, 26, 222–226. [Google Scholar] [CrossRef]
- Kuratomi, Y.; Nomizu, M.; Tanaka, K.; Ponce, M.L.; Komiyama, S.; Kleinman, H.K.; Yamada, Y. Laminin gamma 1 chain peptide, C-16 (KAFDITYVRLKF), promotes migration, MMP-9 secretion, and pulmonary metastasis of B16-F10 mouse melanoma cells. Br. J. Cancer 2002, 86, 1169–1173. [Google Scholar] [CrossRef]
- Lugassy, C.; Torres-Munoz, J.E.; Kleinman, H.K.; Ghanem, G.; Vernon, S.; Barnhill, R.L. Overexpression of malignancy-associated laminins and laminin receptors by angiotropic human melanoma cells in a chick chorioallantoic membrane model. J. Cutan. Pathol. 2009, 36, 1237–1243. [Google Scholar] [CrossRef]
- Martini, C.; DeNichilo, M.; King, D.P.; Cockshell, M.P.; Ebert, B.; Dale, B.; Ebert, L.M.; Woods, A.; Bonder, C.S. CD36 promotes vasculogenic mimicry in melanoma by mediating adhesion to the extracellular matrix. BMC Cancer 2021, 21, 765. [Google Scholar] [CrossRef] [PubMed]
- Barnhill, R.; Dy, K.; Lugassy, C. Angiotropism in cutaneous melanoma: A prognostic factor strongly predicting risk for metastasis. J. Investig. Dermatol. 2002, 119, 705–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, G.Y.; Kenny, P.A.; Lee, E.H.; Bissell, M.J. Three-dimensional culture models of normal and malignant breast epithelial cells. Nat. Methods 2007, 4, 359–365. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Wang, Y.; Cen, J.; Ma, X.; Cui, L.; Qiu, Z.; Zhang, Z.; Li, H.; Yang, R.Z.; Wang, C.; et al. Modelling liver cancer initiation with organoids derived from directly reprogrammed human hepatocytes. Nat. Cell Biol. 2019, 21, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Kopper, O.; de Witte, C.J.; Lohmussaar, K.; Valle-Inclan, J.E.; Hami, N.; Kester, L.; Balgobind, A.V.; Korving, J.; Proost, N.; Begthel, H.; et al. An organoid platform for ovarian cancer captures intra- and interpatient heterogeneity. Nat. Med. 2019, 25, 838–849. [Google Scholar] [CrossRef]
- Liu, C.; Qin, T.; Huang, Y.; Li, Y.; Chen, G.; Sun, C. Drug screening model meets cancer organoid technology. Transl. Oncol. 2020, 13, 100840. [Google Scholar] [CrossRef] [PubMed]
- Belleri, M.; Paganini, G.; Coltrini, D.; Ronca, R.; Zizioli, D.; Corsini, M.; Barbieri, A.; Grillo, E.; Calza, S.; Bresciani, R.; et al. beta-Galactosylceramidase Promotes Melanoma Growth via Modulation of Ceramide Metabolism. Cancer Res. 2020, 80, 5011–5023. [Google Scholar] [CrossRef] [PubMed]
- Prakash, R.; Thareja, N.S.; Carmichael, T.S.; Barnhill, R.L.; Lugassy, C.; Bentolila, L.A. Visualizing Pericyte Mimicry of Angiotropic Melanoma by Direct Labeling of the Angioarchitecture. Methods Mol. Biol. 2021, 2235, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Abdouh, M.; Arena, V.; Arena, M.; Arena, G.O. Reprogramming Malignant Cancer Cells toward a Benign Phenotype following Exposure to Human Embryonic Stem Cell Microenvironment. PLoS ONE 2017, 12, e0169899. [Google Scholar] [CrossRef] [Green Version]
- Hendrix, M.J.; Seftor, E.A.; Seftor, R.E.; Kasemeier-Kulesa, J.; Kulesa, P.M.; Postovit, L.M. Reprogramming metastatic tumour cells with embryonic microenvironments. Nat. Rev. Cancer 2007, 7, 246–255. [Google Scholar] [CrossRef]
- Bailey, C.M.; Kulesa, P.M. Dynamic interactions between cancer cells and the embryonic microenvironment regulate cell invasion and reveal EphB6 as a metastasis suppressor. Mol. Cancer Res. MCR 2014, 12, 1303–1313. [Google Scholar] [CrossRef] [Green Version]
- Giacobbe, A.; Abate-Shen, C. Modeling metastasis in mice: A closer look. Trends Cancer 2021, 7, 916–929. [Google Scholar] [CrossRef]
- Lee, M.W.; Miljanic, M.; Triplett, T.; Ramirez, C.; Aung, K.L.; Eckhardt, S.G.; Capasso, A. Current methods in translational cancer research. Cancer Metastasis Rev. 2021, 40, 7–30. [Google Scholar] [CrossRef]
- Cassidy, J.W.; Caldas, C.; Bruna, A. Maintaining Tumor Heterogeneity in Patient-Derived Tumor Xenografts. Cancer Res. 2015, 75, 2963–2968. [Google Scholar] [CrossRef] [Green Version]
- Garman, B.; Anastopoulos, I.N.; Krepler, C.; Brafford, P.; Sproesser, K.; Jiang, Y.; Wubbenhorst, B.; Amaravadi, R.; Bennett, J.; Beqiri, M.; et al. Genetic and Genomic Characterization of 462 Melanoma Patient-Derived Xenografts, Tumor Biopsies, and Cell Lines. Cell Rep. 2017, 21, 1936–1952. [Google Scholar] [CrossRef]
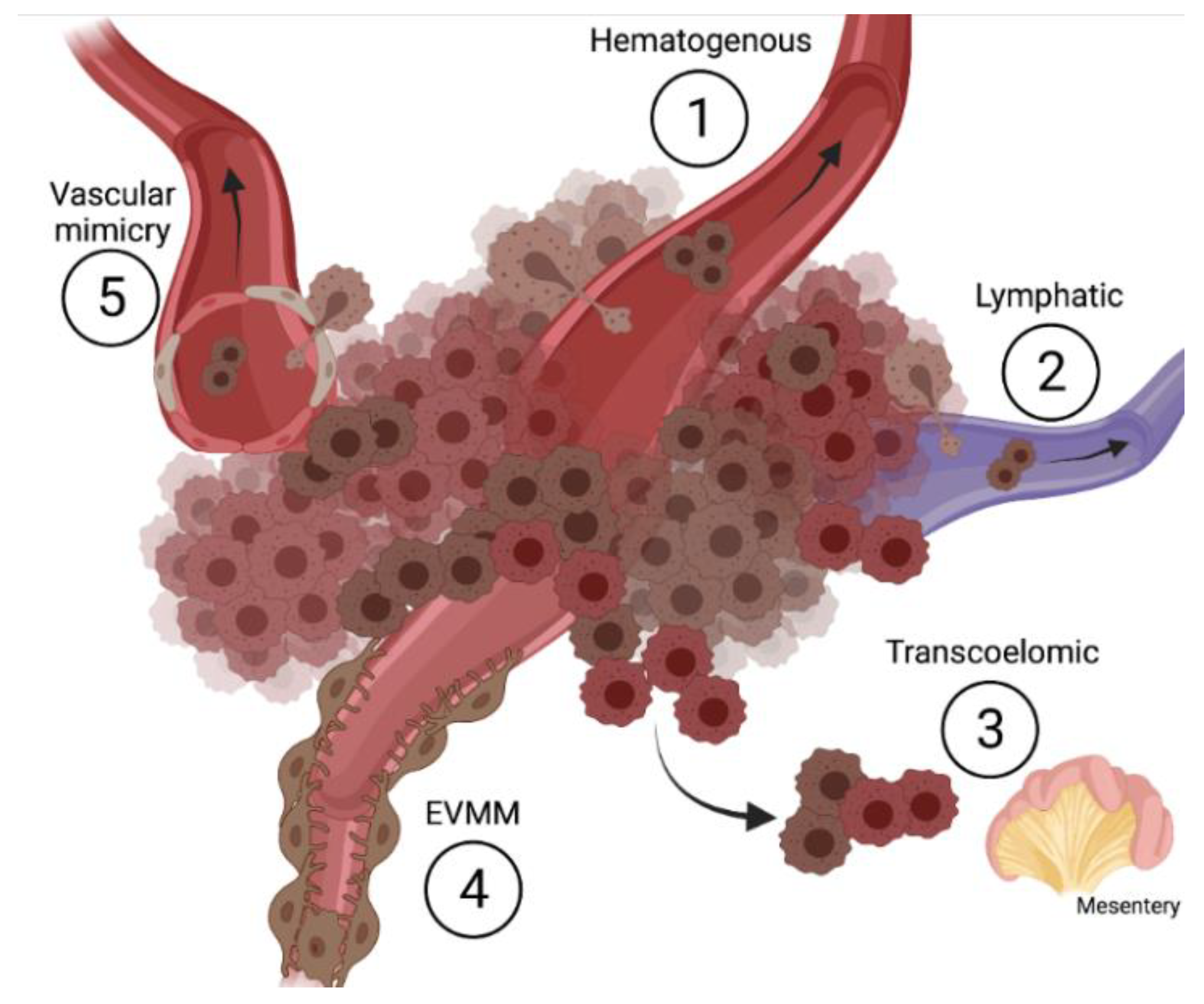

| Metastatic Process | Features |
|---|---|
| 1. Hematogenous | Metastatic cancer cells detaching from primary tumor approach capillaries or angiogenic blood vessels, degrade the basal lamina, invade the endothelium, and intravasate into the flow as single cells or small groups. Finally, they colonize receptive distant organs. |
| 2. Lymphatic | Metastatic cancer cells detaching from primary tumor degrade the basal lamina of lymphatic vessels and intravasate. Metastatic cancer cells enter into the lymphatic system by active movement, pass up the lymphatic flow, and colonize the lymph nodes and other organs. |
| 3. Transcoelomic | Metastatic cancer cells detached or exfoliated from the tumor remain as individual or groups of cells in the cavities. The spread of cancer cells into body cavities occurs via penetrating the surface of the peritoneal, pleural, pericardial, or subarachnoid spaces. In the cavities, metastatic cancer cells proliferate in suspension, generate ascites, and/or adhere to other tissues. |
| 4. Extra Vascular Migratory Metastasis (EVMM) | In EVMM, metastatic cancer cells detaching from the primary tumor approach capillaries or angiogenic blood vessels. Once in the vessel, cancer cells migrate along the abluminal side without intravasating, degrading the basal lamina or altering the structure of vessels. |
| 5. Vascular mimicry (VM) | Metastatic cancer cells, under hypoxic pressure, can form vascular channels interconnected with the tumor vasculature. These leaky structures give nutrients and oxygen to the tumor and support the spread of metastatic cells. |
| Cancer Type | In Vitro Observations | In Vivo Observations | Clinical Observations |
|---|---|---|---|
| Melanoma/Uveal melanoma | In vitro co-culture system: C.Lugassy et al. 2013 [67] G. Fornabaio et al. 2018 [68] | In CAM assay: C. Lugassy et al. 2007 [69] In zebrafish model: G. Fornabaio et al. 2018 [68] In murine model: L.A. Bentolila et al. 2016 [70] | R.L. Barnhill and C. Lugassy 2004 [59] A.K. Rodewald et al. 2019 [71] |
| Glioma | Organotypic slice tissues: V. Montana and H. Sontheimer 2011 [72] | In murine model: S. Watkins et al. 2014 [73] F. Winkler et al. 2009 [74] M. Alieva et al. 2019 [75] | D. Zagzag et al. 2008 [76] |
| Sarcomatoid squamous cell carcinoma | K.M.H. Eleanor and S. Colin 2019 [77] | ||
| Cutaneous squamous cell carcinoma | F. Fedda et al. 2018 [78] | ||
| Prostatic adenocarcinoma | 3D co-culture system: C. Lugassy et al. 2005 [61] | CAM model: C. Lugassy et al. 2005 [61] | C. Lugassy et al. 2005 [61] |
| Syringomatous carcinoma | E. Katayama et al. 2017 [79] | ||
| Gynaecological carcinosarcoma | J.M. Dyke et al. 2014 [80] | ||
| Well-differentiated liposarcoma | J. Shen et al. 2016 [81] | ||
| Pancreatic adenocarcinoma | M.J. Levy et al. 2009 [82] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corsini, M.; Ravelli, C.; Grillo, E.; Mitola, S. The Metastatic Capacity of Melanoma Reveals Alternative Pathways of Cancer Dissemination. Int. J. Transl. Med. 2021, 1, 163-174. https://0-doi-org.brum.beds.ac.uk/10.3390/ijtm1030012
Corsini M, Ravelli C, Grillo E, Mitola S. The Metastatic Capacity of Melanoma Reveals Alternative Pathways of Cancer Dissemination. International Journal of Translational Medicine. 2021; 1(3):163-174. https://0-doi-org.brum.beds.ac.uk/10.3390/ijtm1030012
Chicago/Turabian StyleCorsini, Michela, Cosetta Ravelli, Elisabetta Grillo, and Stefania Mitola. 2021. "The Metastatic Capacity of Melanoma Reveals Alternative Pathways of Cancer Dissemination" International Journal of Translational Medicine 1, no. 3: 163-174. https://0-doi-org.brum.beds.ac.uk/10.3390/ijtm1030012






