Hygienic Perspectives of Wood in Healthcare Buildings
Abstract
:1. Introduction
2. Wood as an Indoor Material for Healthcare Buildings
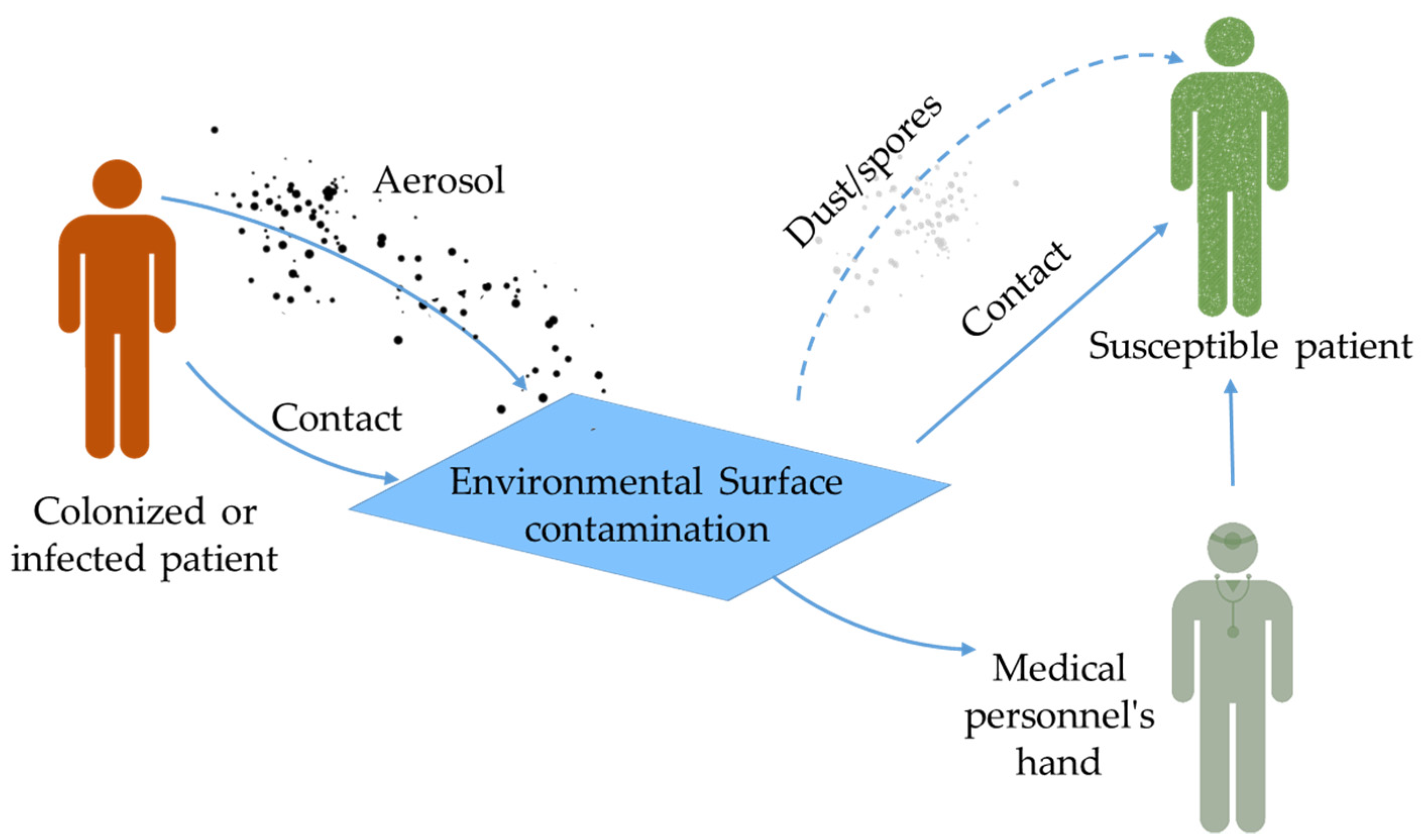
3. Healthcare-Associated Infections (HAI) and Role of Environmental Surfaces
4. Hygienic and Antimicrobial Behavior of Wooden Surfaces
4.1. Biochemical Composition and Antimicrobial Activity
4.2. Role of Physical Structure in the Antimicrobial Activity of Wood
4.2.1. Porosity of Wood
4.2.2. Hygroscopicity and Capillary Action
5. Conclusions and Future Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chías, P.; Abad, T. Green Hospitals, Green Healthcare. Int. J. Energy Prod. Manag. 2017, 2, 196–205. [Google Scholar] [CrossRef] [Green Version]
- Kumari, S.; Kumar, R. Green Hospital—A Necessity and Not Option. J. Manag. Res. Anal. 2020, 7, 46–51. [Google Scholar] [CrossRef]
- Kotradyová, V.; Kaliňáková, B. Wood as Material Suitable for Healthcare and Therapeutic Facilities. Adv. Mater. Res. 2014, 1041, 362–366. [Google Scholar] [CrossRef]
- Chou, L.-P.; Li, C.-Y.; Hu, S.C. Job Stress and Burnout in Hospital Employees: Comparisons of Different Medical Professions in a Regional Hospital in Taiwan. BMJ Open 2014, 4, e004185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade, C.C.; Devlin, A.S.; Pereira, C.R.; Lima, M.L. Do the Hospital Rooms Make a Difference for Patients’ Stress? A Multilevel Analysis of the Role of Perceived Control, Positive Distraction, and Social Support. J. Environ. Psychol. 2017, 53, 63–72. [Google Scholar] [CrossRef]
- Ulrich, R.S. View through a Window May Influence Recovery from Surgery. Science 1984, 224, 420–421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Lian, Z.; Ding, Q. Investigation Variance in Human Psychological Responses to Wooden Indoor Environments. Build. Environ. 2016, 109, 58–67. [Google Scholar] [CrossRef]
- Kotradyova, V.; Vavrinsky, E.; Kalinakova, B.; Petro, D.; Jansakova, K.; Boles, M.; Svobodova, H. Wood and Its Impact on Humans and Environment Quality in Healthcare Facilities. Int. J. Environ. Res. Public Health 2019, 16, 3496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munir, M.T.; Pailhories, H.; Eveillard, M.; Aviat, F.; Lepelletier, D.; Belloncle, C.; Federighi, M. Antimicrobial Characteristics of Untreated Wood: Towards a Hygienic Environment. Health 2019, 11, 152–170. [Google Scholar] [CrossRef] [Green Version]
- Pailhories, H.; Munir, M.T.; Aviat, F.; Federighi, M.; Belloncle, C.; Eveillard, M. Oak in Hospitals, the Worst Enemy of Staphylococcus aureus? Infect. Control Hosp. Epidemiol. 2017, 38, 382–384. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, S.; Murallidharan, J.S.; Agrawal, A.; Bhardwaj, R. Why Coronavirus Survives Longer on Impermeable than Porous Surfaces. Phys. Fluids 2021, 33, 021701. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-C.; Munir, M.T.; Aviat, F.; Lepelletier, D.; Le Pape, P.; Dubreil, L.; Irle, M.; Federighi, M.; Belloncle, C.; Eveillard, M.; et al. Survival of Bacterial Strains on Wood (Quercus petraea) Compared to Polycarbonate, Aluminum and Stainless Steel. Antibiotics 2020, 9, 804. [Google Scholar] [CrossRef]
- Corpet, D.E. Why Does SARS-CoV-2 Survive Longer on Plastic than on Paper? Med. Hypotheses 2021, 146, 110429. [Google Scholar] [CrossRef] [PubMed]
- Alapieti, T.; Mikkola, R.; Pasanen, P.; Salonen, H. The Influence of Wooden Interior Materials on Indoor Environment: A Review. Eur. J. Wood Wood Prod. 2020, 78, 617–634. [Google Scholar] [CrossRef]
- Hristozov, D.; Pizzol, L.; Basei, G.; Zabeo, A.; Mackevica, A.; Hansen, S.F.; Gosens, I.; Cassee, F.R.; De Jong, W.; Koivisto, A.J.; et al. Quantitative Human Health Risk Assessment along the Lifecycle of Nano-Scale Copper-Based Wood Preservatives. Nanotoxicology 2018, 12, 747–765. [Google Scholar] [CrossRef] [Green Version]
- Hedge, A. Survival of Escherichia coli, Pseudomona aeruginosa, Staphylococcus aureus on Wood and Plastic Surfaces. J. Microb. Biochem. Technol. 2015, 7. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhang, Y.; Li, X.; Sun, M.; Wei, Z.; Wang, Y.; Gao, A.; Chen, D.; Zhao, X.; Feng, X. Exploring the Effects of Different Types of Surfactants on Zebrafish Embryos and Larvae. Sci. Rep. 2015, 5, 10107. [Google Scholar] [CrossRef] [Green Version]
- EPA. Overview of Wood Preservative Chemicals. Available online: https://www.epa.gov/ingredients-used-pesticide-products/overview-wood-preservative-chemicals (accessed on 29 July 2020).
- Kandelbauer, A.; Widsten, P. Antibacterial Melamine Resin Surfaces for Wood-Based Furniture and Flooring. Prog. Org. Coat. 2009, 65, 305–313. [Google Scholar] [CrossRef]
- Papadopoulos, A.N.; Taghiyari, H.R. Innovative Wood Surface Treatments Based on Nanotechnology. Coatings 2019, 9, 866. [Google Scholar] [CrossRef] [Green Version]
- WHO. Report on the Burden of Endemic Health Care-Associated Infection Worldwide; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Khan, H.A.; Baig, F.K.; Mehboob, R. Nosocomial Infections: Epidemiology, Prevention, Control and Surveillance. Asian Pac. J. Trop. Biomed. 2017, 7, 478–482. [Google Scholar] [CrossRef]
- Cassini, A.; Plachouras, D.; Eckmanns, T.; Abu Sin, M.; Blank, H.-P.; Ducomble, T.; Haller, S.; Harder, T.; Klingeberg, A.; Sixtensson, M.; et al. Burden of Six Healthcare-Associated Infections on European Population Health: Estimating Incidence-Based Disability-Adjusted Life Years through a Population Prevalence-Based Modelling Study. PLoS Med. 2016, 13, e1002150. [Google Scholar] [CrossRef] [Green Version]
- Benenson, S.; Cohen, M.J.; Schwartz, C.; Revva, M.; Moses, A.E.; Levin, P.D. Is It Financially Beneficial for Hospitals to Prevent Nosocomial Infections? BMC Health Serv. Res. 2020, 20, 653. [Google Scholar] [CrossRef]
- CDC. Health Topics—HAI—POLARIS. Available online: https://www.cdc.gov/policy/polaris/healthtopics/hai.html (accessed on 28 July 2020).
- Haque, M.; Sartelli, M.; McKimm, J.; Abu Bakar, M. Health Care-Associated Infections—An Overview. Infect. Drug Resist. 2018, 11, 2321–2333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suleyman, G.; Alangaden, G.; Bardossy, A.C. The Role of Environmental Contamination in the Transmission of Nosocomial Pathogens and Healthcare-Associated Infections. Curr. Infect. Dis. Rep. 2018, 20, 12. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Hossain, M.A.; Khan, S.I.; Khan, M.N.; Sack, R.B.; Albert, M.J.; Huq, A.; Colwell, R.R. Survival of Shigella dysenteriae Type 1 on Fomites. J. Health Popul. Nutr. 2001, 19, 177–182. [Google Scholar] [PubMed]
- Choi, H.; Chatterjee, P.; Coppin, J.D.; Martel, J.A.; Hwang, M.; Jinadatha, C.; Sharma, V.K. Current Understanding of the Surface Contamination and Contact Transmission of SARS-CoV-2 in Healthcare Settings. Environ. Chem. Lett. 2021. [Google Scholar] [CrossRef]
- Schreck, J.H.; Lashaki, M.J.; Hashemi, J.; Dhanak, M.; Verma, S. Aerosol Generation in Public Restrooms. Phys. Fluids 2021, 33, 033320. [Google Scholar] [CrossRef]
- Kramer, A.; Assadian, O. Survival of microorganisms on inanimate surfaces. In Use of Biocidal Surfaces for Reduction of Healthcare Acquired Infections; Borkow, G., Ed.; Springer International Publishing: Cham, Switzerland, 2014; pp. 7–26. ISBN 978-3-319-08056-7. [Google Scholar]
- Otter, J.A.; Yezli, S.; Salkeld, J.A.G.; French, G.L. Evidence That Contaminated Surfaces Contribute to the Transmission of Hospital Pathogens and an Overview of Strategies to Address Contaminated Surfaces in Hospital Settings. Am. J. Infect. Control 2013, 41, S6–S11. [Google Scholar] [CrossRef]
- Lei, H.; Jones, R.M.; Li, Y. Exploring Surface Cleaning Strategies in Hospital to Prevent Contact Transmission of Methicillin-Resistant Staphylococcus aureus. BMC Infect. Dis. 2017, 17, 85. [Google Scholar] [CrossRef] [Green Version]
- Park, J.H.; Ryu, S.H.; Lee, J.Y.; Kim, H.J.; Kwak, S.H.; Jung, J.; Lee, J.; Sung, H.; Kim, S.-H. Airborne Fungal Spores and Invasive Aspergillosis in Hematologic Units in a Tertiary Hospital during Construction: A Prospective Cohort Study. Antimicrob. Resist. Infect. Control 2019, 8, 88. [Google Scholar] [CrossRef]
- Kanamori, H.; Rutala, W.A.; Sickbert-Bennett, E.E.; Weber, D.J. Review of Fungal Outbreaks and Infection Prevention in Healthcare Settings During Construction and Renovation. Clin. Infect. Dis. 2015, 61, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Bisesi, M.; Lee, J. Comparison of Survivability of Staphylococcus aureus and Spores of Aspergillus niger on Commonly Used Floor Materials. Am. J. Infect. Control 2017, 45, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Mensah-Attipoe, J.; Reponen, T.; Veijalainen, A.-M.; Rintala, H.; Täubel, M.; Rantakokko, P.; Ying, J.; Hyvärinen, A.; Pasanen, P. Comparison of Methods for Assessing Temporal Variation of Growth of Fungi on Building Materials. Microbiology 2016, 162, 1895–1903. [Google Scholar] [CrossRef]
- van Doremalen, N.; Bushmaker, T.; Morris, D.; Holbrook, M.; Gamble, A.; Williamson, B.; Tamin, A.; Harcourt, J.; Thornburg, N.; Gerber, S.; et al. Aerosol and Surface Stability of HCoV-19 (SARS-CoV-2) Compared to SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef] [PubMed]
- Chin, A.W.H.; Chu, J.T.S.; Perera, M.R.A.; Hui, K.P.Y.; Yen, H.-L.; Chan, M.C.W.; Peiris, M.; Poon, L.L.M. Stability of SARS-CoV-2 in Different Environmental Conditions. Lancet Microbe 2020, 1, e10. [Google Scholar] [CrossRef]
- Duan, S.-M.; Zhao, X.-S.; Wen, R.-F.; Huang, J.-J.; Pi, G.-H.; Zhang, S.-X.; Han, J.; Bi, S.-L.; Ruan, L.; Dong, X.-P.; et al. Stability of SARS Coronavirus in Human Specimens and Environment and Its Sensitivity to Heating and UV Irradiation. Biomed. Environ. Sci. 2003, 16, 246–255. [Google Scholar]
- Taylor, J.; Davies, M.; Canales, M.; Lai, K.M. The Persistence of Flood-Borne Pathogens on Building Surfaces under Drying Conditions. Int. J. Hyg. Environ. Health 2013, 216, 91–99. [Google Scholar] [CrossRef]
- Aviat, F.; Le Bayon, I.; Federighi, M.; Montibus, M. Comparative Study of Microbiological Transfer from Four Materials Used in Direct Contact with Apples. Int. J. Food Microbiol. 2020, 333, 108780. [Google Scholar] [CrossRef]
- Williams, A.P.; Avery, L.M.; Killham, K.; Jones, D.L. Persistence of Escherichia coli O157 on Farm Surfaces under Different Environmental Conditions. J. Appl. Microbiol. 2005, 98, 1075–1083. [Google Scholar] [CrossRef]
- Tiwari, A.; Patnayak, D.P.; Chander, Y.; Parsad, M.; Goyal, S.M. Survival of Two Avian Respiratory Viruses on Porous and Nonporous Surfaces. Avian Dis. 2006, 50, 284–287. [Google Scholar] [CrossRef]
- Sakaguchi, H.; Wada, K.; Kajioka, J.; Watanabe, M.; Nakano, R.; Hirose, T.; Ohta, H.; Aizawa, Y. Maintenance of Influenza Virus Infectivity on the Surfaces of Personal Protective Equipment and Clothing Used in Healthcare Settings. Environ. Health Prev. Med. 2010, 15, 344–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oxford, J.; Berezin, E.N.; Courvalin, P.; Dwyer, D.E.; Exner, M.; Jana, L.A.; Kaku, M.; Lee, C.; Letlape, K.; Low, D.E.; et al. The Survival of Influenza A(H1N1) Pdm09 Virus on 4 Household Surfaces. Am. J. Infect. Control 2014, 42, 423–425. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Chan, M.; VanderZaag, A. Inactivation of Avian Influenza Viruses on Porous and Non-porous Surfaces Is Enhanced by Elevating Absolute Humidity. Transbound. Emerg. Dis. 2017, 64, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Cosenza, B.J.; McCreary, M.; Buck, J.D.; Shigo, A.L. Bacteria Associated with Discolored and Decayed Tissues in Beech, Birch, and Maple. Phytopathology 1970, 60, 1547. [Google Scholar] [CrossRef]
- Dutkiewicz, J.; Sorenson, W.G.; Lewis, D.M.; Olenchock, S.A. Levels of Bacteria, Fungi and Endotoxin in Stored Timber. Int. Biodeterior. Biodegrad. 1992, 30, 29–46. [Google Scholar] [CrossRef]
- Górny, R.L.; Gołofit-Szymczak, M.; Cyprowski, M.; Stobnicka-Kupiec, A. Nasal Lavage as Analytical Tool in Assessment of Exposure to Particulate and Microbial Aerosols in Wood Pellet Production Facilities. Sci. Total Environ. 2019, 697, 134018. [Google Scholar] [CrossRef] [PubMed]
- Munir, M.T.; Pailhories, H.; Eveillard, M.; Irle, M.; Aviat, F.; Dubreil, L.; Federighi, M.; Belloncle, C. Testing the Antimicrobial Characteristics of Wood Materials: A Review of Methods. Antibiotics 2020, 9, 225. [Google Scholar] [CrossRef] [PubMed]
- Munir, M.T.; Aviat, F.; Pailhories, H.; Eveillard, M.; Irle, M.; Federighi, M.; Belloncle, C. Direct Screening Method to Assess Antimicrobial Behavior of Untreated Wood. Eur. J. Wood Wood Prod. 2019, 77, 319–322. [Google Scholar] [CrossRef]
- Ateş, S.; Gür, M.; Özkan, O.E.; Akça, M.; Olgun, Ç.; Güder, A. Chemical Contents and Antifungal Activity of Some Durable Wood Extractives vs. Pleurotus ostreatus. BioResources 2015, 10, 2433–2443. [Google Scholar] [CrossRef] [Green Version]
- Prida, A.; Puech, J.-L. Influence of Geographical Origin and Botanical Species on the Content of Extractives in American, French, and East European Oak Woods. J. Agric. Food Chem. 2006, 54, 8115–8126. [Google Scholar] [CrossRef]
- Prida, A.; Ducousso, A.; Petit, R.J.; Nepveu, G.; Puech, J.-L. Variation in Wood Volatile Compounds in a Mixed Oak Stand: Strong Species and Spatial Differentiation in Whisky-Lactone Content. Ann. For. Sci. 2007, 64, 313–320. [Google Scholar] [CrossRef]
- Munir, M.T.; Pailhories, H.; Eveillard, M.; Irle, M.; Aviat, F.; Federighi, M.; Belloncle, C. Experimental Parameters Influence the Observed Antimicrobial Response of Oak Wood (Quercus Petraea). Antibiotics 2020, 9, 535. [Google Scholar] [CrossRef] [PubMed]
- Laireiter, C.M.; Schnabel, T.; Köck, A.; Stalzer, P.; Petutschnigg, A.; Oostingh, G.J.; Hell, M. Active Anti-Microbial Effects of Larch and Pine Wood on Four Bacterial Strains. BioResources 2013, 9, 273–281. [Google Scholar] [CrossRef]
- Milling, A.; Kehr, R.; Wulf, A.; Smalla, K. Survival of Bacteria on Wood and Plastic Particles: Dependence on Wood Species and Environmental Conditions. Holzforschung 2005, 59, 72–81. [Google Scholar] [CrossRef]
- Koch, A.P.; Kofod, C.J.; Konova, D.; Kvist, K.E.; Lindergaard, B. Wood, Plastic and Steel—A Comparason of Hygenic Properties; Project P 99095 “wood in the food”; The Danish Technological Institute: Taastrup, Denmark, 2002. [Google Scholar]
- Milling, A.; Smalla, K.; Kehr, R.; Wulf, A. The Use of Wood in Practice—A Hygienic Risk? Holz Roh. Werkst. 2005, 63, 463–472. [Google Scholar] [CrossRef]
- Miller, A.J.; Brown, T.; Call, J.E. Comparison of Wooden and Polyethylene Cutting Boards: Potential for the Attachment and Removal of Bacteria from Ground Beef. J. Food Prot. 1996, 59, 854–858. [Google Scholar] [CrossRef] [PubMed]
- Schönwälder, A.; Kehr, R.; Wulf, A.; Smalla, K. Wooden Boards Affecting the Survival of Bacteria? Holz Roh. Werkst. 2002, 60, 249–257. [Google Scholar] [CrossRef]
- Munir, M.T.; Belloncle, C.; Pailhories, H.; Aviat, F.; Federighi, M.; Eveillard, M. Wood as a Safe Material for Indoor Surface Construction of Hygienically Sensitive Places; Northwestern Polytechnical University: Xi’an, China, 2017; pp. 79–80. [Google Scholar]
- Vainio-Kaila, T.; Rautkari, L.; Nordström, K.; Närhi, M.; Natri, O.; Kairi, M. Effect of Extractives and Thermal Modification on Antibacterial Properties of Scots Pine and Norway Spruce. Int. Wood Prod. J. 2013, 4, 248–252. [Google Scholar] [CrossRef]
- Vainio-Kaila, T.; Kyyhkynen, A.; Rautkari, L.; Siitonen, A. Antibacterial Effects of Extracts of Pinus sylvestris and Picea abies against Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, and Streptococcus pneumoniae. BioResources 2015, 10, 7763–7771. [Google Scholar] [CrossRef]
- Vainio-Kaila, T.; Zhang, X.; Hänninen, T.; Kyyhkynen, A.; Johansson, L.-S.; Willför, S.; Österberg, M.; Siitonen, A.; Rautkari, L. Antibacterial Effects of Wood Structural Components and Extractives from Pinus sylvestris and Picea abies on Methicillin-Resistant Staphylococcus aureus and Escherichia coli O157:H7. BioResources 2017, 12, 7601–7614. [Google Scholar] [CrossRef]
- Tomičić, R.; Tomičić, Z.; Thaler, N.; Humar, M.; Raspor, P. Factors Influencing Adhesion of Bacteria Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus and Yeast Pichia membranifaciens to Wooden Surfaces. Wood Sci. Technol. 2020, 54, 1663–1676. [Google Scholar] [CrossRef]
- Báder, M.; Németh, R. Moisture-Dependent Mechanical Properties of Longitudinally Compressed Wood. Eur. J. Wood Prod. 2019, 77, 1009–1019. [Google Scholar] [CrossRef] [Green Version]
- Vainio-Kaila, T.; Hänninen, T.; Kyyhkynen, A.; Ohlmeyer, M.; Siitonen, A.; Rautkari, L. Effect of Volatile Organic Compounds from Pinus sylvestris and Picea abies on Staphylococcus aureus, Escherichia coli, Streptococcus pneumoniae and Salmonella enterica Serovar Typhimurium. Holzforschung 2017, 71, 905–912. [Google Scholar] [CrossRef]
- Anouhe, J.-B.S.; Niamké, F.B.; Faustin, M.; Virieux, D.; Pirat, J.-L.; Adima, A.A.; Kati-Coulibaly, S.; Amusant, N. The Role of Extractives in the Natural Durability of the Heartwood of Dicorynia Guianensis Amsh: New Insights in Antioxydant and Antifungal Properties. Ann. For. Sci. 2018, 75, 15. [Google Scholar] [CrossRef] [Green Version]
- Shebani, A.N.; van Reenen, A.J.; Meincken, M. The Effect of Wood Extractives on the Thermal Stability of Different Wood Species. Thermochim. Acta 2008, 471, 43–50. [Google Scholar] [CrossRef]
- Geffert, A.; Geffertova, J.; Dudiak, M. Direct Method of Measuring the PH Value of Wood. Forests 2019, 10, 852. [Google Scholar] [CrossRef] [Green Version]
- Vainio-Kaila, T.; Kyyhkynen, A.; Viitaniemi, P.; Siitonen, A. Pine Heartwood and Glass Surfaces: Easy Method to Test the Fate of Bacterial Contamination. Eur. J. Wood Wood Prod. 2011, 69, 391–395. [Google Scholar] [CrossRef] [Green Version]
- Acuff, G.R.; Vanderzant, C.; Hanna, M.O.; Ehlers, J.G.; Gardner, F.A. Effects of Handling and Preparation of Turkey Products on the Survival of Campylobacter jejuni. J. Food Prot. 1986, 49, 627–631. [Google Scholar] [CrossRef]
- Ak, N.O.; Cliver, D.O.; Kaspar, C.W. Cutting Boards of Plastic and Wood Contaminated Experimentally with Bacteria. J. Food Prot. 1994, 57, 16–22. [Google Scholar] [CrossRef]
- Ak, N.O.; Cliver, D.O.; Kaspar, C.W. Decontamination of Plastic and Wooden Cutting Boards for Kitchen Use. J. Food Prot. 1994, 57, 23–30. [Google Scholar] [CrossRef]
- Zangerl, P.; Matlschweiger, C.; Dillinger, K.; Eliskases-Lechner, F. Survival of Listeria monocytogenes after Cleaning and Sanitation of Wooden Shelves Used for Cheese Ripening. Eur. J. Wood Wood Prod. 2010, 68, 415–419. [Google Scholar] [CrossRef] [Green Version]
- Lucke, F.-K.; Skowyrska, A. Hygienic Aspects of Using Wooden and Plastic Cutting Boards, Assessed in Laboratory and Small Gastronomy Units. J. Verbr. Lebensm. 2015, 10, 317–322. [Google Scholar] [CrossRef]
- Imhof, R.; Schwendimann, L.; Scettrini, P.R. Sanitising Wooden Boards Used for Cheese Maturation by Means of a Steam-Mediated Heating Process. J. Consum. Prot. Food Saf. 2017, 12, 255–263. [Google Scholar] [CrossRef] [Green Version]
- Cruciata, M.; Gaglio, R.; Scatassa, M.L.; Sala, G.; Cardamone, C.; Palmeri, M.; Moschetti, G.; Mantia, T.L.; Settanni, L. Formation and Characterization of Early Bacterial Biofilms on Different Wood Typologies Applied in Dairy Production. Appl. Environ. Microbiol. 2018, 84, e02107-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deza, M.A.; Araujo, M.; Garrido, M.J. Efficacy of Neutral Electrolyzed Water to Inactivate Escherichia coli, Listeria monocytogenes, Pseudomonas aeruginosa, and Staphylococcus aureus on Plastic and Wooden Kitchen Cutting Boards. J. Food Prot. 2007, 70, 102–108. [Google Scholar] [CrossRef] [PubMed]
- DeVere, E.; Purchase, D. Effectiveness of Domestic Antibacterial Products in Decontaminating Food Contact Surfaces. Food Microbiol. 2007, 24, 425–430. [Google Scholar] [CrossRef]
- Thormar, H.; Hilmarsson, H. Killing of Campylobacter on Contaminated Plastic and Wooden Cutting Boards by Glycerol Monocaprate (Monocaprin). Lett. Appl. Microbiol. 2010, 51, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Al-Qadiri, H.M.; Ovissipour, M.; Al-Alami, N.; Govindan, B.N.; Shiroodi, S.G.; Rasco, B. Efficacy of neutral electrolyzed water, quaternary ammonium and lactic acid-based solutions in controlling microbial contamination of food cutting boards using a manual spraying technique. J. Food Sci. 2016, 81, M1177–M1183. [Google Scholar] [CrossRef] [PubMed]
- Gehrig, M.; Schnell, G.; Zürcher, E.; Kucera, L.J. Hygienische Eigenschaften von Holz-und Kunststoffbrettern in der Nahrungsmittelverarbeitung und-präsentation: Ein Vergleich. Holz Roh. Werkst. 2000, 58, 265–269. [Google Scholar] [CrossRef]
- Chiu, T.-H.; Duan, J.; Liu, C.; Su, Y.-C. Efficacy of Electrolysed Oxidizing Water in Inactivating Vibrio parahaemolyticus on Kitchen Cutting Boards and Food Contact Surfaces. Lett. Appl. Microbiol. 2006, 43, 666–672. [Google Scholar] [CrossRef]
- Shi, Y.; Sun, R.; An, D.; Lu, W.; Zhang, C.; Wang, L.; Liu, Y.; Wang, Q. Mathematical Quantification of Inactivation of Vibrio parahaemolyticus on Two Types of Surface Soiled with Different Substrates. Food Control 2017, 74, 1–8. [Google Scholar] [CrossRef]
- Prechter, S.; Betz, M.; Cerny, G.; Wegener, G.; Windeisen, E. Hygienische Aspekte von Schneidebrettern aus Holz bzw. Kunststoff. Holz Roh. Werkst. 2002, 60, 239–248. [Google Scholar] [CrossRef]
- Dubreil, L.; Aviat, F.; Anthoine, V.; Ismail, R.; Rossero, A.; Federighi, M. Confocal Spectral Microscopy—An Innovative Tool for Tracking of Pathogen Agents on Contaminated Wooden Surfaces. Eur. J. Wood Wood Prod. 2018, 76, 1083–1085. [Google Scholar] [CrossRef]
- Carpentier, B. Sanitary Quality of Meat Chopping Board Surfaces: A Bibliographical Study. Food Microbiol. 1997, 14, 31–37. [Google Scholar] [CrossRef]
- Ismail, R.; Aviat, F.; Gay-Perret, P.; Le Bayon, I.; Federighi, M.; Michel, V. An Assessment of L. monocytogenes Transfer from Wooden Ripening Shelves to Cheeses: Comparison with Glass and Plastic Surfaces. Food Control 2017, 73, 273–280. [Google Scholar] [CrossRef]
- Montibus, M.; Ismaïl, R.; Michel, V.; Federighi, M.; Aviat, F.; Le Bayon, I. Assessment of Penicillium expansum and Escherichia coli Transfer from Poplar Crates to Apples. Food Control 2016, 60, 95–102. [Google Scholar] [CrossRef]
- Dantas, S.T.A.; Rossi, B.F.; Bonsaglia, E.C.R.; Castilho, I.G.; Hernandes, R.T.; Fernandes, A.; Rall, V.L.M. Cross-Contamination and Biofilm Formation by Salmonella enterica Serovar Enteritidis on Various Cutting Boards. Foodborne Pathog. Dis. 2018, 15, 81–85. [Google Scholar] [CrossRef] [Green Version]
- Munir, M.T.; Belloncle, C.; Irle, M.; Federighi, M. Wood-Based Bedding in Poultry Production: A Review. World Poult. Sci. J. 2019, 75, 5–16. [Google Scholar] [CrossRef]
- Cimiotti, W.; Glunder, G.; Hinz, K.H. Survival of the Bacterial Turkey Coryza Agent. Vet. Rec. 1982, 110, 304–306. [Google Scholar] [CrossRef]
- Coughenour, C.; Stevens, V.; Stetzenbach, L.D. An Evaluation of Methicillin-Resistant Staphylococcus aureus Survival on Five Environmental Surfaces. Microb. Drug Resist. 2011, 17, 457–461. [Google Scholar] [CrossRef]
- Xi, L.; Qin, D.; An, X.; Wang, G. Resistance of Natural Bamboo Fiber to Microorganisms and Factors That May Affect Such Resistance. BioResources 2013, 8, 6501–6509. [Google Scholar] [CrossRef]
- Tomičić, R.; Tomičić, Z.; Raspor, P. Adhesion of Candida Spp. and Pichia Spp. to Wooden Surfaces. Food Technol. Biotechnol. 2017, 55, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Munir, M.T. Wood and Hospital Hygiene: Investigating the Hygienic Safety and Antimicrobial Properties of Wood Materials. Ph.D. Thesis, Ecole Centrale de Nantes, Nantes, France, 2021. [Google Scholar]
| Microorganisms | Survival Time | |
|---|---|---|
| Various Substrates | Wood | |
| Acinetobacter baumannii | 3 days to 1 year (in vitro); 36 days within biofilm and <15 days for non-biofilm-forming strains [31]; <2 days plastic; <7 days aluminum and steel [12] | <24 h [12] |
| Aspergillus spp. | >30 days [31] | >28 days [36,37] |
| Candida spp. | >30 days [31] | >30 days |
| Clostridioides (Clostridium) difficile | 5 months for spores; 15 min (dry surface) and 6 h (moist surface) for vegetative form [31] | N/A |
| Coronavirus SARS CoV 1 and 2 | <5 min up to 24 h (on paper), 5–28 days (at room temp.), 28 days (at 4 °C) | ≤24 h SARS-Cov-2 [38,39] 8 to ≤96 h SARS-Cov-1 [40] |
| Enterococcus spp. | 5 days to 30 months [31] | >7 days (flood water) [41] |
| Escherichia coli | 1.5 h to 16 months [31] | 1h (food contact) [42], >7 days (flood water) [41], 28 days (farm surface) [43] |
| Influenza virus | 1–28 days (strain dependent), 1–3 days (on banknotes), up to 8 days (admixed in mucous) [31] | 1–2 days [44,45,46], 1–28 days (depending upon environmental conditions) [47] |
| Klebsiella spp. | >2 h to 30 months; 7 days in detergent solution [31]; > 15 days on plastic, steel and aluminum [12] | <2 days on untreated oak wood [12] |
| Pseudomonas aeruginosa | 6 h up to 16 months (5 weeks on dry floor and few hours in aerosol) [31] | N/A |
| Shigella dysenteriae | 2 h on plastic, aluminum and glass | 3 h [28] |
| Staphylococcus aureus | 7 days up to 1 year (in vitro) [31], >15 days (plastic, steel, aluminum) [12], 6 h (copper), 28 days (dry mops) and 14 days (in water) [31] | >28 days on hardwood floor [36] <7 days on untreated oak wood [12] |
| Target | Wood Chemicals |
|---|---|
| Cell wall and cell membrane | Flavonoids, tannins, aldehydes, phenolic acids, terpenoids, alkaloids, terpenes |
| Nucleic acid | Flavonoids, aldehydes, alkaloids |
| Metals metabolism | Tannins |
| Protein synthesis | Aldehydes, tannins |
| Energy metabolism | Flavonoids, phenolic acids |
| Adhesion and Biofilm formation | Phenolic acids, quinones |
| Bacteria | Ranking | Reference |
|---|---|---|
| S. aureus, P. aeruginosa, Enterobacter faecium, Bacillus subtilis | Pine > larch | [57] |
| E. coli E. faecium | Pine = oak = larch > maple > spruce > beech > polar Pine = oak > larch = maple = spruce = beech = poplar | [58] |
| B. subtilis, P. fluorescens | Oak > spruce | [59] |
| Poultry manure flora | Pine > larch = maple | [60] |
| E. coli 0157:H7 | White ash > red oak > black cherry > maple | [61] |
| E. coli, E. faecium | Pine > poplar = beech | [62] |
| S. aureus, P. aeruginosa, A. baumannii | Oak > Douglas fir = pine > poplar | [63] |
| S. aureus, E. coli, E. faecalis, Streptococcus pneumoniae | Pine > spruce | [64,65,66] |
| E. coli, Pichia membranifaciens, P. aeruginosa, S. aureus | Norway spruce > beech > poplar | [67] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munir, M.T.; Pailhoriès, H.; Aviat, F.; Lepelletier, D.; Pape, P.L.; Dubreil, L.; Irle, M.; Buchner, J.; Eveillard, M.; Federighi, M.; et al. Hygienic Perspectives of Wood in Healthcare Buildings. Hygiene 2021, 1, 12-23. https://0-doi-org.brum.beds.ac.uk/10.3390/hygiene1010002
Munir MT, Pailhoriès H, Aviat F, Lepelletier D, Pape PL, Dubreil L, Irle M, Buchner J, Eveillard M, Federighi M, et al. Hygienic Perspectives of Wood in Healthcare Buildings. Hygiene. 2021; 1(1):12-23. https://0-doi-org.brum.beds.ac.uk/10.3390/hygiene1010002
Chicago/Turabian StyleMunir, Muhammad Tanveer, Hélène Pailhoriès, Florence Aviat, Didier Lepelletier, Patrice Le Pape, Laurence Dubreil, Mark Irle, Julia Buchner, Matthieu Eveillard, Michel Federighi, and et al. 2021. "Hygienic Perspectives of Wood in Healthcare Buildings" Hygiene 1, no. 1: 12-23. https://0-doi-org.brum.beds.ac.uk/10.3390/hygiene1010002








